Abstract
Hematological malignancies encompass a wide variety of severe diseases that pose a serious threat to human health. Given the fact that hematological malignancies are difficult to treat due to their unpredictable and rapid deterioration and high rates of recurrence, growing attention has been paid to their early screening and diagnosis. However, developing a rapid and effective diagnostic tool featuring a noninvasive sampling technique is still extremely challenging. In recent years, novel nanomaterials-based electrochemical biosensors have attracted great interest because of such advantages as simple operation, low cost, fast response, etc. As a kind of rising nanomaterials, two-dimensional materials have excellent electronic and chemical properties, which have been proven to improve the performance of electrochemical biosensors. This review summarizes the applications of different types of electrochemical biosensors (nucleic acid sensors, immunosensors, aptamer biosensors, and cytosensors) based on two-dimensional materials in the detection of biological molecules related to hematological malignancies. Two-dimensional materials-based electrochemical biosensors designed for the diagnosis of leukemia could rapidly detect the target biomolecules at a trace level and show great merits such as wide linear range, low detection limit, high sensitivity, excellent selectivity, and cost-effectiveness. In addition, these biosensors have also achieved satisfactory results in the diagnosis of lymphoma and multiple myeloma. Thus, two-dimensional materials-based electrochemical biosensors are attractive for the early diagnosis of hematological malignancies in clinical practice. Nevertheless, more efforts are still required to further improve the performance of electrochemical biosensors. In this review, we propose the possible main concerns in the design of future two-dimensional materials-based electrochemical biosensors, involving the development of sensors for synchronous detection of diverse target biomolecules, the exploration of other superior two-dimensional materials, the simplification of the sensors fabrication process, the construction of new hybrid structures and how to avoid possible environmental issues.
Keywords
Introduction
Cancer, as a severe global problem, is reported to be one of the biggest obstacles to social development. Not only does it impose a huge financial burden on the numerous affected families, but also reduces global life expectancy. 1 Hematological malignancy (HM) is a general term for a series of heterogeneous diseases in the hematologic system which poses a serious threat to human health. 2 According to the global cancer statistics in 2018, HMs accounted for 7.2% of total mortality and 6.5% of new cancers. 3 HMs are challenging to treat due to their unpredictable and rapid deterioration as well as high rates of recurrence. Therefore, early detection and diagnosis are of great significance in terms of reducing the risk of such diseases and alleviating the economic and emotional burden of patients.4,5 Morphology, immunology, cytogenetics, and molecular biology (MICM) are the most important diagnostic methods currently used in clinical practice for HMs. 6 In addition, biopsy, and clinical imaging techniques or laboratory testing technologies like flow cytometry, real-time quantity polymerase chain reaction (RTQ-PCR), or DNA/RNA sequencing, etc are also used as the main detection techniques.2,7,8 However, the practical applications of these techniques are often limited due to their radiotoxicity, invasiveness, complex operation, high cost, application limitations, difficulty in achieving standardization or high requirements for operators, etc.9,10 Therefore, it is highly desired to develop a novel cost-effective, and easy-to-use technique that causes small incisions but yields rapid and accurate responses to assist in the early detection and diagnosis of HMs.
A biosensor, a device capable of converting the concentrations of analytes to analysis signals, can be classified into various types according to different standards, such as optical biosensors, mass-based biosensors, calorimetric biosensors, electrochemical biosensors, colorimetric biosensors, fluorescent biosensors, surface plasmon biosensors, etc.11,12 Among them, the electrochemical biosensors have been considered to be promising analytical equipment, which consists of 3 components: a biometric unit or identification system, a converter or transduction system, and a detector or electronic system. The identification system selectively interacts with the target analytes and converts the generated parameters into certain signals, and then the transduction system receives the signals and transmits them to the electronic system in the forms of electrochemical signals, such as current, voltage, resistance, etc. Finally, the electronic system further amplifies and outputs the electrochemical signals to quantify target analytes.13,14 Voltammetry, amperometry, and impedance methods are the most common analytical strategies in electrochemical biosensors according to the different input and output electrochemical signals. 15 Owning to their merits of low cost, simple operation, good selectivity, high sensitivity, and fast response, electrochemical biosensors have been widely applied in clinical medicine,16–19 drug screening,20,21 food detection,22,23 environmental monitoring24,25 and other fields.26,27
With the increasing demands for the identification and detection of biological molecules associated with various diseases, the design of functional biosensors aimed to analyze target substances at a trace level economically and rapidly has become the focus of clinical research. It has been proved that nanomaterials can significantly enhance the important indicators of biosensors with small-size effects, quantum-size effects, and surface interface effects. 13 Nowadays, benefiting from the continuous exploration of functional nanomaterials and the development of advanced nanotechnologies, great achievements have been achieved in nanomaterials-based electrochemical biosensors.28–30 The performance of the electrochemical biosensors in the detection of biological molecules can be further improved through the use of nanomaterials. These sensing devices may provide a new solution for ultrasensitive diagnosis and effective treatment of diseases in the future.
Two-dimensional (2D) materials, a new type of nanomaterials, have become the research focus of biosensing and chemical sensing.31,32 With transverse dimensions ranging from a few nanometers to hundreds of nanometers and above, 2D materials show atomic-thick sheet-like structures.13,33 Moreover, 2D materials exhibit great anisotropic physical and chemical properties, and the electrons are confined in two dimensions that endow them with brilliant electronic properties compared to other dimensional (0D, 1D, and 3D) materials. Also, the atomic-thick structure offers them a high specific surface area and the maximum mechanical flexibility. Thus, 2D materials can provide numerous anchoring sites, facilitating the effective immobilization of analytes and increasing the contact and interaction between materials and analytes.34,35 Besides, 2D materials have better compatibility with metal electrodes, which is crucial for improving the performance of sensor systems.36,37 2D materials overcome the shortcomings of other dimensional materials (0D, 1D, and 3D) regarding device integration, electrical contact establishment, and device miniaturization, respectively. Thus, 2D materials are more suitable for sensor applications. 37
Graphene, as the first discovered 2D material, 38 is a hexagonal carbon mesh with a large specific surface area, good electrical conductivity, excellent mechanical strength, and flexibility, etc. 39 Moreover, in light of graphene's extraordinary properties, such as high sensitivity, excellent selectivity, and high stability, the potential of designing graphene-based electrochemical biosensing devices is staggering.40,41 Besides, transition metal dichalcogenides (TMDs), MXenes, graphitic C3N4 (g-C3N4), metalorganic frameworks (MOFs), and black phosphorous (BP) are also popular 2D materials that have received wide attention.13,32 TMDs are semiconductors of the MX2, in which a layer of metal atoms (M) is sandwiched between 2 hexagonal sulfide planes (X). Thus, TMDs differ from other nanomaterials with their tunable electronic properties from semiconductors to semimetals and from real metals to superconductors.42,43 In addition to typical molybdenum sulfide (MoS2), other TMDs such as molybdenum selenide (MoSe2), tungsten sulfide (WS2), tungsten selenide (WSe2), and vanadium sulfide (VS2) have also been used in the field of electrochemical biosensors.44,45 MXenes are one of the most frequently reported materials in the 2D materials family recently, where M stands for early transition metals and X for carbon (C), nitrogen (N), or both. The abundant surface functional groups, high metal conductivity, hydrophilicity, and biocompatibility make them attractive candidates for electrochemical biosensors.33,42,46 g-C3N4 is a novel nitrogen-doped carbon-based material with a large specific surface area and good biocompatibility. The rich defect structure of g-C3N4 makes it a good electron donor and the doping of N further increases the electron transport rate in the electrocatalytic process, 47 which is important in electrochemical sensors, biosensors, and catalysis. 48 MOFs are crystalline coordination polymers with regular porous structures and abundant unique functional groups. 49 Due to their high porosity, functional sites, and properties of high regeneration, storage, and separation capabilities, MOFs have been used in electrochemical biosensors to capture different analytic species. 50 BP has a 2D layered structure similar to other 2D materials. Given excellent molecular energy adsorption efficiency, semiconductor properties, anisotropic conductivity, high specific surface area, good biocompatibility, and low cytotoxicity, BP has become a feasible candidate for a wide range of electrochemical sensing applications.40,51
To date, applications of electrochemical biosensors based on 2D materials and their hybrid composites in clinical detection have acquired satisfactory results. For instance, in the field of endocrinology, electrochemical biosensors have been applied in blood glucose monitoring in clinical and family diabetics 8 or in other hormone monitoring, 18 which benefits the patients considerably. Besides, the detection of important biomarkers of various diseases such as central nervous system disorders,52,53 cardiovascular diseases,54,55 colorectal cancers,56,57 breast cancers,58–60 lung cancers,61,62 or hepatic diseases63,64 has also achieved good results. Inspired by these, some relevant biomolecules associated with severe HMs have been investigated by many research teams based on electrochemical biosensors.2,8,65–68Hence, we present a brief review of the state-of-the-art electrochemical biosensors based on 2D materials (graphene, TMDs, MXenes, g-C3N4, MOFs, or BP, etc) and their hybrid composites in the detection of HMs related biomolecules (Scheme 1). In addition, current limitations and the prospect of using 2D materials-based materials electrochemical biosensors in HMs diagnosis are discussed, which may bring new opportunities for the design and development of high-performance 2D materials-based electrochemical biosensors in the HMs diagnosis field.
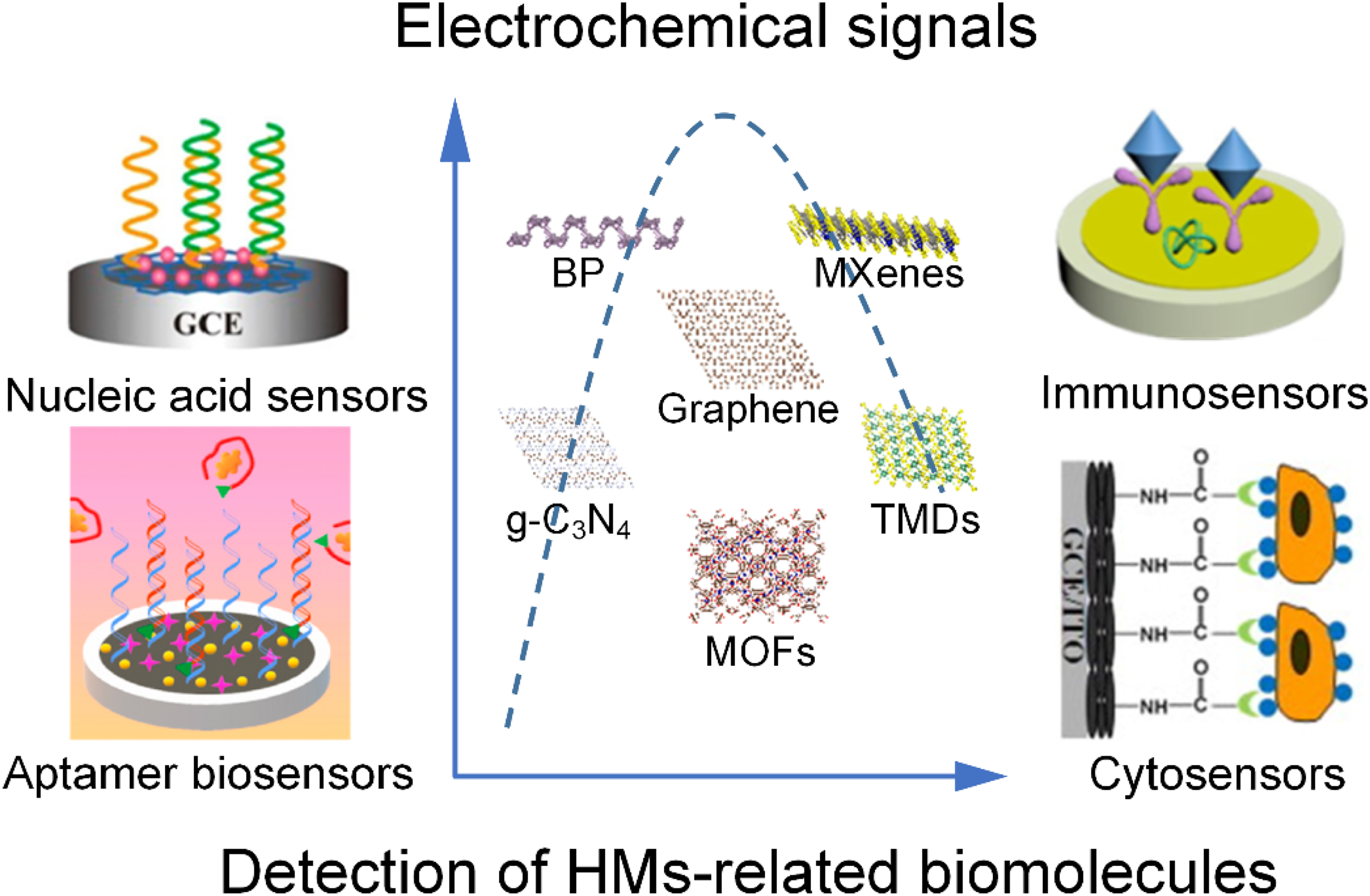
A general scheme of the applications of 2D materials-based electrochemical biosensors in the detection of biomolecules of HMs.
Electrochemical Biosensors
Electrochemical biosensors can be classified into nucleic acid sensors, immunosensors, aptamer biosensors, and cytosensors according to the recognition unit.26,48 For the nucleic acid sensors, the sensing principle is to utilize nucleic acid molecules as recognition elements and fix an oligonucleotide chain on the electrode for the hybridization of target DNA, and then analyze the target substances such as DNA, RNA, or other biological molecules by comparing the changes of electrochemical parameters before and after hybridization. 14 Immunosensors are mainly based on the specific reactions between antigens and antibodies. 14 Antibodies or antigens can be immobilized on the electrode surface to specifically recognize the corresponding antigens or antibodies. The immunoreactions in the electrochemical sensors can quickly cause changes in the electrochemical signals such as current, resistance, etc. 69 Aptamers are functional oligonucleic acids (DNA or RNA) with high specificity or affinity for small molecules, proteins, and cells. 70 Regarding aptamers as recognition units, the detection principle of aptamer biosensors depends on the specific interaction between aptamers and target molecules. 71 Cytosensors are devices that take cells or cell surface substances like proteins, glycans, and receptors as the original recognition elements. The changes in electrochemical signals are caused by the combination of receptors and ligands or nucleic acid sequences. 26 Using these biosensors, various HM-related biomolecules have been investigated as summarized in Table 1.
Summary of the Applications of 2D Materials-Based Electrochemical Biosensors in HMs Detection.

Applications of Electrochemical Biosensors in Leukemia
Leukemia, lymphoma, and multiple myeloma (MM) are the 3 main types of HMs. 2 Among them, leukemia, the most common HM of all age groups, can be divided into 2 main types, acute and chronic leukemia. 88 According to the origin, acute leukemia can be further divided into 2 types, described as acute lymphocytic leukemia (ALL) and acute myeloid leukemia (AML). As the most prevalent type of leukemia and the most common type of leukemia in children, ALL usually spreads or transfers rapidly to multiple organs and affects the functions of the invaded organs. 89 AML is a heterogeneous malignant disease characterized by uncontrolled proliferation and maturation of leukemia cells. It accounts for about 90% of all acute leukemia cases among adults. 90 For chronic leukemias, chronic lymphocytic leukemia (CLL) is characterized by the proliferation and accumulation of mature CD5+ B lymphocytes in disease-related tissues. What is worse, different patients suffering from CLL may have different clinical symptoms, which makes the treatment even more complicated. Chronic myelogenous leukemia (CML) is a chronic myeloproliferative disease, and the abnormal fusion gene on the Philadelphia chromosome (BCR/ABL) is considered an important marker of it. 91 As measurable indicators, specific biological molecules can define or identify whether a disease is in a normal or abnormal state. Similarly, monitoring some biological molecules called cancer biomarkers related to leukemia can be used to assess the risks of occurrence and progression or the responses of patients to treatment. Here, we provide an overview of current advances in using electrochemical biosensors for the detection of leukemia-related biomarkers.
Nucleic Acid Sensors
The balanced translocation between chromosome 9 and chromosome 22 results in the formation of the BCR/ABL fusion gene in all CML and a few ALL patients. Therefore, the rapid detection of BCR/ABL is very important for the early diagnosis of CML and ALL. Mazloum-Ardakani et al 72 reported a sensitive DNA sensor by utilizing the designed DNA sequence based on the BCR/ABL of childhood ALL. The glassy carbon electrode (GCE) was modified by the poly(catechol), graphene (G) nanosheets, and biosynthesized gold nanoparticles (AuNPs). In this work, G nanosheets presented a thin wrinkled, and paper-like appearance under scanning electron microscopy (SEM), providing a large specific surface area for the immobilization of DNA-AuNPs and enhancing the conductivity of the DNA biosensor owing to its great electrical properties. Besides, the AuNPs had a strong affinity to thiol-modified 5′-SH probe DNA. Based on the synergistic effect as discussed above, the redox peak current gradually increased and the voltage tended to decrease in the cyclic voltammetry (CV) analysis when G and AuNPs were loaded on the electrode in turn. With the nucleic acids loading and binding to analytes, the radius of the resistance curve would gradually increase in the electrochemical impedance spectroscopy (EIS) test due to the larger resistance of biological molecules themselves. According to the signal changes of current or resistance, the sensor of Bio AuNPs/Pol/G/GCE linearly detected target DNA in the range of 100.0 μM to 10.0 pM with the detection limit of 1.0 pM. What was more, this DNA sensor was also successfully applied to clinical samples of PCR amplification. Similar to this study, Shamsipur et al 41 combined G and AuNPs to immobilize target DNA and added hemoglobin-capped gold nanoclusters (Hb@AuNCs) as a protective agent to solve the issue of instability and agglomeration of G nanosheets in aqueous media, which greatly boosted G's conductive performance and reduced biological toxicity. By further introducing signal probes called methylene blue (MB) trihydrate and Fe(CN6)3−/4−, a dual-mode label-free DNA sensor for electrochemical detection of BCR/ABL in CML patients was successfully developed. By using EIS and CV methods, the linear detection ranges of this sensing system were from 0.1 fM to 10 pM with a limit of detection (LOD) of 0.037 fM and from 0.1 fM to 10 pM with an LOD of 0.030 fM, respectively. MoS2, one type of TMDs material, also has been regarded as a type of promising nanomaterial that can provide large active surface areas for DNA immobilization. However, the restacking and electrical conductivity of MoS2 are still big challenges. To solve these problems, Soni et al 82 incorporated conducting polymer polyaniline (PANI) with MoS2 to construct a DNA biosensor for the detection of BCR/ABL. Firstly, the target DNA was loaded on MoS2 and then the electrostatic interaction between MoS2 and PANI offered abundant sites for efficient electron transfer to improve the conductivity of the biosensor. As a result, an increased in peak current value could be observed within tens of seconds for PANI-MoS2 decorated electrode. And the target DNA could be quantified according to the corresponding relationship between the peak current values and different concentrations of the target DNA. The constructed sensor had a wide linear range of 10−6 to 10−17 M with an LOD of 3 × 10−18 M, accompanied with good regeneration ability and stability. Besides, Yu et al 83 reported a cascade catalytic amplification strategy based on 2D Ti3C2Tx MXene for the detection of BCR/ABL. According to their study, Ti3C2Tx MXene could provide large surface areas for the immobilization of alkaline phosphatase (ALP) and DNA probes, which showed specific area-dependent nanozyme activity for 1-naphthol. They concluded that the catalysis activities of MXene may be originated from the 2D basal planes, and the 1-naphthol generated from ALP hydrolyzed 1-naphthyl phosphate (1-NPP) could be further oxidized by Ti3C2Tx MXene. Then obvious redox current was used for the quantitative detection of the DNA sequence of BCR/ABL. This strategy realized highly sensitive detection of BCR/ABL with a linear range from 0.2 fM to 20 nM and the detection limit was low as 0.05 fM.
As a characteristic gene of acute promyelocytic leukemia (APL), the promyelocytic leukemia protein and retinoic acid receptor alpha (PML/RARα) fusion gene is caused by the translocation of chromosome 15 and chromosome 17. In clinical practice, effective identification of the PML/RARα fusion gene is the key to early diagnosis, prognosis, and monitoring of APL. 92 In the electrochemical sensing field, Zhang et al 73 prepared an electrochemical DNA sensor for the detection of the PML/RARα fusion gene based on a nanocomposite of graphene oxide (GO) and carbon quantum dots (CDs) (Figure 1a). GO, one of the most important derivatives of G retains the large specific surface area of G while is much easier to synthesize. It also shows good biocompatibility and contains various oxygen functional groups to provide binding sites for the covalent immobilization of DNA probes. Nevertheless, problems such as large resistance and worse conductivity of GO still exist. Thus, Zhang et al introduced CDs to improve the performance of the sensor due to their strong conductivity. In addition, MB was applied as a signal probe on the surface of the CDs-GO decorated electrode to capture the target DNA. MB had a strong affinity for the free guanine on DNA molecules, generating different binding capacities with ssDNA or double-stranded DNA (dsDNA) on the electrode. Different current values could be investigated in the different pulse voltammetry (DPV) methods and the hybridization between DNA probes and target DNA could be further monitored and evaluated in 40 min. The linear range of this DNA sensor was 2.50 × 10−10 to 2.25 × 10−9 M and the detection limit was 8.3 × 10−11 M.

The applications of nucleic acid sensors based on 2D materials for the detection of biomolecules. (A) Schematic diagram and detection principle of DNA biosensor based on MB-CDs-GO. Reprinted with permission from Zhang et al 73 (Zhang et al, 2021). Copyright 2021 International Journal of Nanomedicine. (B) Schematic fabrication of electrochemical platform for RNA based on MoS2-Thi-AuNPs. (C-D) TEM imaging of MoS2 nanosheets and MoS2-Thi-AuNPs nanocomposite. (E) Nyquist plots (a-e) of bare GCE, MoS2-Thi-AuNPs/GCE, DNA/MoS2-Thi-AuNPs/GCE, MCH/DNA/MoS2-Thi-AuNPs/GCE, and miRNA-21/MCH/DNA/MoS2-Thi-AuNPs/GCE, respectively. Reprinted with permission from Zhu et al. 65 Copyright 2017 American Chemical Society.
miRNAs, a class of noncoding single-stranded RNA molecules with a length of 18 to 25 nucleotides, are also considered potential biomarkers of CML and AML. 93 Some electrochemical biosensors have been applied in the analysis of miRNAs recently. For example, Zhu et al 65 designed a nucleic acid sensor based on MoS2 for the detection of miRNA-21 (Figure 1b). To better fix DNA probes and enhance the conductivity of the sensor, they functionalized MoS2 with thionine (Thi) and AuNPs. As shown in Figure 1c and d, a typical thin-layered nanostructure of MoS2 and the MoS2-Thi-AuNPs composite was observed under the transmission electron microscope (TEM). In the sensing process, the resistance of the biosensor increased gradually with the increase of the substances on the surface of the electrode (Figure 1e). The combination of DNA probes and miRNA-21 would hinder electron transfer and result in the decrease of the current signal of Thi. Then, it could oppositely quantify miRNA-21 concentrations according to the decline degree of the signal. Similar to the work of Zhu et al, 65 a paper-based electrochemical biosensor based on AuNPs decorated reduced graphene oxide (RGO) or MoS2 nanosheets for the detection of miRNA-21 and miRNA-155 was reported by Torul et al. 80 With the assistance of AuNPs, the AuNPs/RGO, or AuNPs/MoS2-based electrochemical biosensor in this work took only 35 min to achieve a rapid detection. A more exciting finding was that the sensor exhibited good stability and obtained great economic benefits.
Immunosensors
It has been reported that CD66 is abnormally expressed in ALL cell lines and childhood ALL. 94 Carcinoembryonic antigen (CEA) is an important biomarker for various cancers and antibodies against CD66 can recognize CEA family antigens according to the latest research.47,85,94 Therefore, the detection of CEA may be useful for the diagnosis of leukemia. Liao et al 47 proposed an enzyme-free sandwich-structure electrochemical immunosensor based on 2D g-C3N4 nanosheets for detecting CEA. Defect-rich structures made 2D g-C3N4 a good electron donor and the loading of Ag nanoparticles (AgNPs) not only achieved more effective antibody 1 (Ab1) loading but also further enhanced the conductivity of the sensor. To construct a typical sandwich structure, Ab1/g-C3N4/AgNPs composites were fixed on the electrode to recognize the CEA. After that, the Au@SiO2/Cu2O composites with excellent peroxidase-like activity were utilized to combine with antibody 2 (Ab2) to target the other end of CEA. In the presence of hydrogen peroxide and CEA with different concentrations, Au@SiO2/Cu2O-Ab2 composites contacted with CEA and catalyzed the redox reaction between hydrogen peroxide and Cu+ to generate redox current. At a specific ratio of Au@SiO2/Cu2O-Ab2 composites to CEA, rapid detection of CEA with unknown concentrations could be achieved by establishing a linear relationship between the redox current and different concentrations of CEA as mentioned above. As a result, the detection range of the sensor was 0.01 pg/mL to 80 ng/mL with an LOD as low as 0.0038 pg/mL. For the same purpose of CEA detection, Zhang et al 85 fabricated a dual-mode electrochemical immunosensor based on Ti3C2 MXene anchored CuAu-laminated double hydroxides (LDH). The assembly process of the immunosensor and the morphology structures of Ti3C2 and Ti3C2@CuAu-LDH are shown in Figure 2a and b, respectively. The 2D sheet-like Ti3C2 and absorbed Cu, AuNPs could be clearly observed in the SEM and TEM images. The introduction of Ti3C2 with high conductivity could compensate for the insulation defects of LDH and promote electron transfer between different valence copper ions to generate electrochemical signals. What was more, it could also enhance the stability of antibodies on the electrode due to the abundant functional groups on the surfaces of Ti3C2. By analyzing the electrochemical signals generated by the 2 electrochemical analytical techniques such as square wave voltammetry (SWV) and amperometry current (i–t), highly sensitive and accurate detection of CEA in standard samples and clinical samples was achieved with low system errors.

The applications of immunosensors based on 2D materials for the detection of biomolecules. (A) Assembly process of the immunosensor based on AuNPs-Ti3C2@CuAu-LDH. (B) TEM and SEM of Ti3C2 (a, d); TEM of Ti3C2@CuAu-LDH (b, c); SEM of Ti3C2@CuAu-LDH (e, f). Reprinted with permission from Zhang et al. 85 Copyright 2021 Bioelectrochemistry. (C) The ASV responses for Bcl-2 and Bax proteins with different concentrations on the immunosensor based on Ag@RGO-CdSeTe@CdS. (D-E) The calibration curves between the oxidation peak current of Ag and the concentration of Bax and between the oxidation peak current of Cd and the concentration of Bcl-2, respectively. Reprinted with permission from Zhou et al. 66 Copyright 2016 American Chemical Society.
Studies have shown that receptor tyrosine kinase-like orphan receptor 1 (ROR-1), an antigen on the surface of tumor-specific cells, can be expressed or even overexpressed in leukemia cells of CLL patients resulting in leukemia chemotaxis and proliferation.95,96 Thus, to minimize the risk of disease progression, the early detection of ROR-1 is particularly important for the early diagnosis of CLL. Abolhasan et al 81 attempted to construct a highly sensitive label-free electrochemical immunosensor for the determination of ROR-1. In their research, NiFe-LDH composites were chosen because of their large specific surface area and adjustable structures for the absorption of antibodies. RGO was used to inhibit the aggregation of LDH and enhance the conductivity of the sensor, while AuNPs were prepared to further fix the ROR-1 antibodies. The resistance would gradually increase when ROR-1 was bound to the related antibody, and the current peak gradually decreased in DPV or CV electrochemical technology. Finally, the concentration of ROR-1 could be estimated according to the declined magnitude of the current value. The sensing platform exhibited a wide linear range of 0.01 to 1 pg/mL and a detection limit of 10 pg/mL. The sensor also had been applied to the analysis of the real samples of CLL patients.
With functions of regulating the cell cycle and inhibiting oxidation, etc, B-cell lymphoma-2 (Bcl-2) can interact with Bcl2-related X protein (Bax) to form heterodimers (Bcl2-Bax). Bcl2-Bax can effectively reflect the apoptosis of tumor cells and is often used to evaluate the effect of cancer drugs based on their role as a pair of apoptosis-related genes and apoptosis regulators. In light of this, Zhou et al 66 introduced a novel dual-signal-marked electrochemical immunosensor for the detection of Bcl2 and Bax proteins. RGO layers were utilized as a substrate to increase surface area and fix anti-Bcl2 and anti-Bax. Ag nanoclusters modified CdSeTe@CdS quantum dots were further adorned with secondary antibodies, and the concentrations of Ag+ and Cd2+ were proportional to the concentrations of Bcl-2 and Bax. Therefore, the increased concentrations of Bcl-2 and Bax could be quantified by measuring the depending peak current of Ag+ and Cd2+ respectively via the CV method (Figure 2c to e). With a detection range of 1 to 250 ng/mL, the immunosensor was applied to analyze the expressions of Bcl-2 and Bax in K562 cells of CML treated with nilotinib and it may serve as a promising technique for clinical determination.
Aptamer Biosensors
Compared with antibodies, aptamers are known as “chemical antibodies” with a synthetic nucleic acid sequence and possess advantages such as lower cost, less toxicity, and easier chemical modification or labeling. 51 In recent decades, many types of aptamers have been applied to electrochemical biosensors to detect biomolecules. Due to the merits of large specific surface area, good reducibility, and biocompatibility, MXenes have been combined with other materials to improve electrochemical sensing performances. Yang et al 67 constructed an aptamer sensor based on MXene to detect CEA with different concentrations. The construction steps of the sensor are shown in Figure 3a. In this strategy, apart from generating Fe3O4 in situ, Ti3AlC2 MXene could further introduce ruthenium hexamine ([Ru(NH3)6]3+) covalently to form MXC-Fe3O4-Ru nanocomposite. It can be seen from the SEM images in Figure 3b that Ti3AlC2 is etched into layered MXene, where Fe3O4 nanoparticles are densely distributed on the surfaces. With high conductivity and large surface area, MXC-Fe3O4-Ru nanocomposite was modified on the magnetic gold electrode (MGE) to load a ferrocene (Fc)-modified aptamer probe. In the process of sensing, the aptamer probe could be bonded to the target CEA and leave the electrode surface in the form of a probe-CEA complex. Then a signal ratio proportional to the concentration of CEA was established showing that the peak current signal of Fc was significantly decreased while the peak current signal of [Ru(NH3)6]3+ almost remained unchanged (Figure 3c). The linear range of the aptamer biosensor was 1 pg/mL to 1 μg/mL with an LOD of 0.62 pg/mL (Figure 3d).

The applications of aptamer biosensors based on 2D materials for the detection of biomolecules. (A) The schematic diagram of the aptamer biosensor based on MXC-Fe3O4-Ru. (B) (a-c): SEM images of Ti3AlC2, MXene, and MXC-Fe3O4. (C) DPV responses of the different concentrations of CEA. (D) The linear range for the detection of CEA. Reprinted with permission from Yang et al. 67 Copyright 2022 Biosensors and Bioelectronics.
For other cancer-related biomolecules, Hong et al 97 reported that AML patients had higher levels of exosomes in serum than healthy controls. Exosomes are endogenous membrane vesicles of 30 to 150 nm produced by all cells under physiological and pathological conditions. In recent years, exosomes have been regarded as promising biomarkers and potential therapeutic targets for HMs. Therefore, some aptamer biosensors were designed for the detection of exosomes. For instance, Zhang et al 84 proposed an aptamer biosensor by loading Prussian blue (PB) on the Ti3C2 MXene as a nanozyme for exosomes detection. CD63 aptamers were designed and immobilized on polyamide-modified AuNPs electrodes and PB-Ti3C2 MXene hybrid was used as a nanoprobe. During the detection, Ti3C2 MXene played the main role in enhancing the current signal at low potential and avoiding the interference of possible electrochemical active substances. With a linear range of 5 to 5 × 105 particles/μL and a detection limit of 229 particles/μL, the sensor realized an efficient amplification of electrical signals and an effective measurement of exosomes or CD63 proteins. Sun et al 86 constructed an automatic platform based on BP nanosheets (BPNSs) and MOFs for exosomes detection with dual signals and internal self-calibration. In their work, they prepared BPNSs with sizes ranging from tens of nanometers to hundreds of nanometers and ferrocene (Fc) doped MOF (ZIF-67) films for the immobilization of MB-labeled ssDNA aptamers. Dual redox-signal responses of MB and Fc could be observed on the constructed aptamer-BPNSs/Fc/ZIF-67/ITO sensing platform. As the main detection signal, the redox current of MB would gradually decrease because the composite would release from the surface of ITO when ssDNA aptamers contacted with the specific exosomes. On the other hand, the current signal of Fc was used as a reference and changed slightly. Hence, with this kind of internal self-calibration, the aptamer sensor realized sensitive detection of exosomes. This successful sensing platform suggested that it would provide a new useful strategy for future clinical testing.
Cytosensors
The detection of disease-related cells is also conducive to the early diagnosis of diseases. In the field of biosensors, the number of cells is often indirectly counted by targeting cell surface biomolecules, especially protein receptors. Folate receptor (FR) is a protein receptor on the surface of the cell membrane that usually overexpresses in some tumor cells like the human leukemic cells (HL-60). 68 Therefore, HL-60 cells can be detected by testing folic acid (FA). Yang et al 68 constructed an effective FA-decorated cytosensor based on the carboxymethyl chitosan-functionalized graphene (CMC-G) for the detection of HL-60. The schematic diagram for the synthesis process of CMC-G and the establishment steps of the cytosensor based on CMC-G are shown in Figure 4a and the sheet-like structure of CMC-G is shown in Figure 4b. The main purpose of using CMC was to improve the poor dispersion of G in water. As a result, the CMC-G remained stable for several months. To make the sensor-specific for HL-60 cells, the CMC-G modified electrode was further functionalized by covering polyethyleneimine (PEI) and FA layer upon layer assembly. Different current signals of different decorated electrodes are shown in Figure 4c. Compared with the bare electrode, CMC-G decorated electrode exhibited a stronger peak current, but with more and more biomolecules adsorbed on the electrode, the resistance of the electrode would gradually increase, resulting in weakened currents. The proposed cytosensor exhibited a great cell capture ability for HL-60 cells with a detection limit of 500 cells/mL. Khoshfetrat and Mehrgardi 74 presented an excellent cytosensor based on nitrogen-doped graphene (N-G) for the detection of human acute lymphoblastic leukemia cells (CCRF-CEM). As a substrate, N-G fixed sulfated-sgc8c aptamer on the GCE was used to recognize protein tyrosine kinase 7 (PTK7), a transmembrane receptor overexpressed in CCRF-CEM. The current of the sensor would decrease after the connection between the sgc8c aptamer and CCRF-CEM. The liner range of the cytosensor was from 10 to 1 × 106 cells/mL with a detection limit of 10 cells/mL, showing a great improvement in cell detection.

The applications of cytosensors based on 2D materials for the detection of biomolecules. (A) The schematic diagram for the synthesis process of nanomaterials composite (CMC-G) and the establishment steps of the cytosensor based on CMC-G. (B) TEM image of CMC-G, where the inset was the SAED pattern. (C) (a-d): CV of bare, CMC-G, FA/PEI/CMC-G, and 3.0 × 105 cells/mL HL-60 cells/FA/PEI/CMC-G modified GCE, respectively. Reprinted with permission from Yang et al. 68 Copyright 2013 Carbon.
Besides, as one of the most uncontrolled and destructive human CML cell lines, K562 cells are usually selected to detect CML. For example, Wang et al 75 constructed a new GO-based cytosensor for the detection of K562 cells. They used PANI to modify GO to improve electron transfer rates and enhance the conductivity of the sensor. Attributing to the large specific surface area, the assembled GO-PANI composites further absorbed a large amount of concanavalin A (Con A) to act as the first probe, and PS-(CdS)n-Con A conjugates were regarded as the second probe for capturing K562 cells. In the presence of nitric acid, CdS quantum dots were dissolved and Cd2+ would be released into the electrochemical cell. The concentration of Cd2+ was proportional to the amount of K562 cells. In other words, the currents in the anodic stripping voltammetry (ASV) method varied with the concentrations of Cd2+ and it could indirectly quantify K562 cells by analyzing the peak current values. A broad linear range for the detection of K562 cells was achieved from 10 to 1.0 × 107 cells/mL with a low detection limit of 3 cells/mL. Liu et al 71 developed a cytosensor for capturing K562 cells based on the gold nanoflowers (AuNFs) modified graphene oxide-hemin (H-GO) composite. Their research revealed that the H-GO-Au composite exhibited excellent peroxidase-like activity for the detection of K562 cells. With DNA aptamer, this peroxidase mimic cytosensor could achieve a lower detection limit of 2 cells/mL through the recognition reaction between DNA aptamer and K562 cells. These breakthroughs in the field of cytosensors would open up new research opportunities in terms of the detection of leukemia cells in the future.
Applications of Electrochemical Biosensors in Lymphoma and MM
Lymphoma includes Hodgkin lymphoma (HL) and non-Hodgkin lymphoma (NHL). As the first recognized lymphocytic tumor, HL has a special age distribution in the elderly and young adults. It can be divided into 2 forms, namely classical HL (cHL) based on Hodgkin and Reed-Sternberg (HRS) cells, and nodular lymphocyte predominant HL (NLPHL) based on lymphocyte predominant (LP) cells. 98 Composed of multiple subtypes, NHL is the most common HM that originates from B lymphocytes, T lymphocytes, and natural killer cells. 99 According to the latest GLOBOCAN data, NHL accounted for 2.8% of global cancers and 2.6% of cancer mortality in 2018. 100 MM is one type of HM characterized by monoclonal proliferation of plasma cells with unknown etiology. 101 This kind of disease can result in severe complications such as anemia, destructive bone lesions, acute kidney injury, hypercalcemia or amyloidosis, and central nervous system lesions, etc. 102 Several research teams have made great efforts to detect lymphoma and MM based on the electrochemical biosensors.76–79,87
Acid phosphatase (ACP) has been proven that its abnormal concentration would result in a prompt effect on MM. Gan et al 76 proposed a label-free and simple electrochemical DNA sensor for ACP by using 3′-phosphate DNA-coupled RGO as a nanoprobe. The electrochemical signal could be amplified with the induction of terminal deoxynucleotidyl transferase (TDT). As a result, the electrochemical detection range for ACP was 5 to 100 U/L with a low detection limit of 0.52 U/L. Leptin receptors have been found to express in various hematopoietic cells, including MM cells, HL, and NHL cells. 103 The imbalance of leptins and leptin receptors may be involved in the pathogenesis of various cancers by activating multiple signaling pathways. 104 Cai et al 87 fabricated a label-free immunosensor for the detection of leptin based on porous graphene functionalized BP (PG-BP) composites. With the synergistic effect of PG and BP and the covalent interaction between AuNPs and leptin antibodies, the conductivity of the immunosensor was improved and the protein loading capacity was enhanced. Under optimal conditions, the linear range of the immunosensor was 0.150 – to 2500 pg/mL with an LOD of 0.036 pg/mL, indicating the potential for early screening and diagnosis of lymphoma and MM. Interleukin-6 (IL-6), a small inflammatory cytokine, has been a clinical trial target for MM, lymphoma, and other solid carcinomas. 105 Besides, it has been found that FR 106 and heat shock proteins 70 (HSP70) 107 are also overexpressed in MM. Aiming at these biological molecules, Lou et al 77 constructed a competitive electrochemical immunosensor for the detection of IL-6 based on ERGO-AuPdNPs-Ab1 and AgNPs-Ab2 probes. Moreover, Geetha et al 78 developed a novel electrochemical immunosensor for FR detection, adopting FA-modified RGO nanosheets as the substrate. Through the covalent immobilization of HSP70 antibodies on GO as a bio-recognition element, Ozcan and Sezginturk 79 proposed a label-free immunosensor to analyze HSP70 via electrochemical methods such as CV and AC impedance. All of these sensors used for the detection of biomolecules related to lymphoma or MM have achieved relatively satisfactory results, suggesting their great potentiality in the early evaluation of tumor diseases.
Discussion
Different types of electrochemical biosensors based on 2D materials have been used for the detection of biological molecules related to leukemia, lymphoma, or MM according to the latest research. Most of the 2D materials mentioned in this review such as G, MoS2, Ti3C2 MXene, BPs or MOFs, etc have sheet-like structures with single or several layers. Their large specific surface area and good biocompatibility are conducive to loading nucleic acids and antibodies, aptamers, or other biological molecules. GO or Ti3C2 MXene also contain abundant surface functional groups such as hydroxyl, oxygen ions, etc, leading to better absorption of some surface biological molecules and thereby improving the stability of biosensors and reducing detection errors as well. Moreover, the excellent electrical conductivity of materials 2D materials such as G and BPs can promote electron transfer rates and enhance the electrical properties of biosensors. What is more, attributing to the intrinsic peroxidase-like activity, G or some TMDs such as MoS2 can be used as substitutes for natural enzymes to carry out rapid catalysis and amplification of signals, achieving lower detection limits and higher sensitivity. However, problems of some 2D materials such as easy oxidation, poor dispersion, and easy agglomeration in water are yet to be addressed. Although the development of the derivatives of G such as GO can improve the dispersion and reduce the agglomeration in water and exhibits larger specific surface area and better biocompatibility or lower cytotoxicity compared to G, the conductivity becomes worse. Furthermore, some 2D materials, such as Ti3C2 MXene, MoS2, etc exhibit poor conductivity themselves. In these cases, doping with nitrogen atoms and hybridizing with conductive materials, such as PANI, CMC, Au, or Ag nanoparticles are great choices to improve these deficiencies. The hybrid composites bring an incredible synergistic effect to improve the catalytic activity and conductivity, so as to optimize the performances of electrochemical biosensors and achieve the purpose of rapid detection of trace biological molecules. But at the same time, it also increases the difficulty of material preparation and complicates the construction of the electrochemical biosensors if a multistep process is needed. Besides, in the process of electrochemical sensing, biomolecules such as nucleic acids, antibodies, and aptamers modified 2D materials or their composites are associated with the concentrations of target analytes. The resistance or currents in electrochemical methods such as DPV, EIS, CV, ASV, or i–t, etc will change when the specific biomolecules identify and combine with the analytes. Thus, redox reactions catalyzed by 2D materials and the conversion between different valence states of some ions are general designing principles. Depending on the relationship between the changes in electrochemical signals and different concentrations of analytes, it can establish a linear range for the detection of target analytes with unknown concentrations. Although the rapid detection of extremely trace biological molecules can be achieved in tens of seconds or minutes at present, before that, the antibodies, aptamers, or cells incubation time should not be ignored, especially some immunosensors with typical sandwich structures that require double antibodies. Therefore, the preparation steps of both the selection of 2D materials and the construction of electrochemical biosensors still need to be further simplified to take full advantage of the sensors.
All in all, these 2D materials-based electrochemical biosensors have obtained great results with a wide linear range, low detection limit, high sensitivity, excellent selectivity, and cost-effectiveness. Nevertheless, most electrochemical biosensors still focus on a common single target analyte. Therefore, the combined detection of multiple important and correlated biomarkers should be carried out to improve the screening rate of biomolecules and the diagnostic rate of diseases in the future. In addition, research on electrochemical biosensors currently prefers typical 2D materials such as graphene and MoS2. Considering the huge 2D materials family, increasing attention toward other 2D materials should be received. Discovering novel 2D materials and exploring their unique properties may contribute to the construction of high-performance electrochemical biosensors. It is also advised to simplify the hybridization of nanomaterials while ensuring detection efficiency or to hybridize new structures/nanomaterials and introduce cascade catalytic reactions. Recently, liquid metal with unique interfacial chemistry and physics has been widely explored in different physical and chemical sensors, and emerged as multipurpose functional materials.108,109 Utilizing liquid metal as raw material to construct a 2D functional hybrid structure may be a good choice to improve the sensitivity of electrochemical biosensors and enhance the detection selectivity. 109 Owing to the combined metallic and liquid properties, the liquid metal can also serve as electrodes with flexible shapes, thereby simplifying electrode cleaning and sensor fabrication steps.108,109 Beyond that, researchers seem to be more inclined to develop disposable electrochemical biosensors. However, since improper disposal of nondegradable inorganic nanomaterials is likely to cause serious environmental problems, how to deal with discarded sensors is also worthy of further consideration. Perhaps the development of biodegradable materials with excellent electrochemical properties might be the focus of future research in this area. From another perspective, designing sensors that can be reused is also potentially attractive. Green nanotechnologies and nanomaterials with self-cleaning functions are encouraged to modify electrodes, thereby eliminating electrode passivation and removing contaminants from the electrode surface. Renewable electrochemical biosensor enables continuous detection of multiple analytes with a single sensor and minimizes the differences between different sensors. Applying such renewable biosensors to clinical disease-related molecular detection not only significantly reduces production costs, but also greatly reduces detection errors for accurate diagnosis of diseases in the future.110–113
Conclusion
This review outlines the applications of 2D materials-based electrochemical biosensors in the detection of biomolecules related to 3 kinds of HMs. We mainly focus on the applications of 4 different types of electrochemical biosensors in the detection of leukemia-related biomarkers and the applications in some biomolecules of lymphoma and multiple myeloma. Although various types of existing electrochemical biosensors have achieved satisfactory results, we point out some limitations of electrochemical sensors in practical applications. In summary, 2D materials-based electrochemical sensors may be a great candidate for clinical diagnosis in the future.
Footnotes
Abbreviations
Acknowledgements
This work was financially supported by the Natural Science Foundation of Guangdong Province (Nos. 2020A1515010639, 2021A1515010708, and 2022A1515011959), National Natural Science Foundation of China (No. 21975280), Science and technology special competitive allocation project of Zhanjiang City (No. 2021A05147), PhD Start-up Program of Central People's Hospital of Zhanjiang (No. 2020A07), Science and Technology Plan Project of Zhanjiang city (Nos. 2021A05153, 2020A0611 and 2020A06001).
Ethical Approval
This work does not involve animal and human studies.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangdong Province, National Natural Science Foundation of China (grant nos. 2020A1515010639, 2021A1515010708, 2022A1515011959, and 21975280).
