Abstract
Introduction
Currently, the standard treatment of locally advanced cervical cancer (LACC) is pelvic concurrent chemoradiotherapy (CCRT). International Federation of Gynecology and Obstetrics (FIGO) IIIB cervical cancer accounted for approximately 25% of LACC. 1 It had long been recognized that many patients with LACC harbored occult para-aortic lymph node (PALN) metastases,2,3 and the incidence of PALN metastasis ranged from 10% to 25% in LACC.4–6 In addition, pelvic wall involvement, which was included in stage IIIB, was associated with an increased rate of PALN metastasis.7–9 Prophylactic extended-field radiation (EFRT), including the para-aortic region, might be considered to reduce PALN recurrence rates in CCRT treatment of IIIB patients without PALN metastasis.10–14 However, the benefit of prophylactic EFRT is controversial. Yap et al 15 showed that no improvement in para-aortic relapse rates or survival benefit was noted in patients with prophylactic para-aortic irradiation compared with patients without prophylactic para-aortic irradiation.
To date, information on other risk factors had been relatively limited; however, prophylactic EFRT was recommended when the common iliac lymph node was positive.16,17 Regarding the decision to use prophylactic EFRT during CCRT treatment of stage IIIB cervical cancer, each institution had its own guidelines, and treatment was selected based on the physician's preference. Therefore, it remains unclear who might benefit from prophylactic EFRT among stage IIIB cervical cancer patients without PALN and/or common iliac lymph node metastasis during pelvic CCRT. Moreover, few studies have assessed the risk factors for PALN recurrence after pelvic CCRT,9,18 which has resulted in either too much or too little treatment for some stage IIIB cervical cancer patients.
The nutritional and immunological status of patients was reported to be associated with cancer invasion, treatment tolerability, cancer progression and prognosis.19–21 Previous studies have devised some systems to evaluate nutritional status and/or immune status and demonstrated their efficiency in predicting the prognosis of cancer patients.19,21–23 As a recently emerging scoring system of nutritional status, the controlling nutritional status (CONUT) score was composed of serum albumin levels, total cholesterol levels, and total peripheral lymphocyte counts 24 and was demonstrated to be a predictive or prognostic factor in malignant tumors.20,25,26 However, whether the CONUT score could be a predictor of PALN recurrence and guide the use of prophylactic EFRT in FIGO stage IIIB cervical cancer patients treated with CCRT remains unknown. In our study, we tried to assess the effectiveness of the CONUT score for predicting PALN recurrence and the value of the CONUT score in the selection of radiotherapy fields in the treatment of FIGO stage IIIB cervical cancer patients.
Materials and Methods
Patients
The retrospective study was approved by the Ethics Committee of Shandong First Medical University (Shandong Cancer Hospital and Institute, SDTHEC201904002). All medical information was anonymous, and the institutional research ethics board indicated that informed consent was unnecessary. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline. 27 From 2010 to 2015, the medical records of patients with FIGO stage IIIB disease who were treated by definitive pelvic radiotherapy or CCRT were reviewed at our institutes. The following patient inclusion criteria were employed: cervical squamous cell carcinoma, adenocarcinoma or adenosquamous carcinoma, FIGO clinical stages IIIB (2009), negative PALN and/or common iliac lymph node determined by positron emission tomography-computed tomography (PET-CT) scan and/or magnetic resonance imaging (MRI), and treatment with definitive pelvic radiotherapy or CCRT. In total, 116 consecutive patients were included in this analysis. Thirty-eight patients were excluded from the study, including 7 patients who had not completed pelvic radiotherapy and 31 patients treated with prophylactic EFRT (Supplemental fig 1).
Treatment
All patients underwent computed tomography (CT) simulation. External pelvic irradiation was performed by intensity-modulated radiotherapy (IMRT). The IMRT technique included conventional fixed-field IMRT (FF-IMRT) or helical tomotherapy (HT). The clinical target volume (CTV) for pelvic radiotherapy included the gross tumor, cervix, uterus, parametrium, upper half of the vagina, and regional lymph node regions (obturator, common, internal and external iliacs, and presacral region). The gross tumor volume of lymph nodes (GTVnd) covered pelvic metastatic lymph nodes (MLNs). The CTV in the pelvis consisted of a 2 cm margin around the cervix, uterus, parametria, presacral space, and vagina. The external, internal, and common iliac nodal volumes were based on contrast-enhanced vessels with a 0.7- to 1-cm circumferential margin. Accounting for patient motion and setup uncertainty in our institutions, the CTV was expanded 0.8 to 1 cm nonuniformly to create the planned target volume (PTV) with FF-IMRT, and a margin of 0.6 to 0.8 cm to produce the PTV with HT. We provided a margin of 0.5 cm to form the planning gross tumor volume of lymph nodes (PGTVnd). A dose of 50.4 Gy in 28 fractions was prescribed to the PTV, and 57.6 to 59.4 Gy was simultaneously boosted to the PGTVnd. All patients underwent high-dose-rate (HDR) intracavitary brachytherapy once or twice a week after 3 weeks of external pelvic irradiation. Five to seven fractions of 6 Gy each were delivered to point A. Combining the external beam radiation doses with brachytherapy doses, the cumulative linear quadratic equivalent doses (EQD2) delivered to point A were 99 GyEQD2 (α/β = 10 Gy)-116 GyEQD2 (α/β = 10 Gy). No external irradiation treatment was provided on the day of HDR brachytherapy treatment.
The common concurrent chemotherapy regimen was 40 mg/m2 cisplatin weekly for 4 to 6 cycles. Some patients with renal comorbidities received 60 mg/m2/week paclitaxel.
CONUT Score and Other Factors
CONUT scores were calculated based on 3 parameters: the serum albumin concentration, total blood cholesterol level, and total peripheral lymphocyte count 24 (Table 1). Blood samples of enrolled patients were collected and assayed within 1 week before curative treatment. According to our previous studies showing the optimal cutoff CONUT value of 3, patients were divided into 2 groups: the low CONUT group (score ˂ 3) and the high CONUT group (score≥3). 28 Pelvic MLNs were defined as a short axis of the lymph node of >1 cm on CT, MRI, or PET-CT. Gynecologists determined tumor extension to the pelvic wall by gynecological examination.
The Details of Controlling Nutritional Status (CONUT) Score.

Follow-up
All patients were evaluated 1 month after completion of all treatments followed by evaluations at 3-month intervals for 2 years, every 6 months within the next 3 years, and every year thereafter. The follow-up included gynecological examination, blood biochemistry, squamous cell carcinoma antigen (SCC-Ag), pelvic MRI, and chest and abdomen enhanced CT. When tumor recurrence was suspected, a biopsy or PET-CT was performed.
Statistical Analysis
The overall survival (OS), disease-free survival (DFS), and para-aortic lymph node metastasis-free survival (PALNMFS) rates were estimated using the Kaplan-Meier method. Statistical Package for Social Sciences version 26.0 (SPSS, Chicago, IL) software was used for all statistical analyses. PALNMFS was defined from the date of treatment initiation to the date of the first PALN metastasis or related death with an upper border at the level of the left renal vein and a lower border at the aortic bifurcation. Univariate analysis was performed by the log-rank method, and when the factor was found to be significant (P < .05), the Cox regression model was used to perform multivariate analysis. A P-value < .05 was considered statistically significant.
Results
The clinical characteristics of the patients were summarized in Table 2. Thirty-three patients (28.5%) underwent PET-CT staging, 73 patients (62.9%) underwent MRI staging, and 10 patients (8.6%) underwent PET-CT and MRI staging. All patients were divided into 4 groups based on the pretreatment CONUT score: 57 patients (49.1%) were classified as normal; 44 (37.9%) were classified as light; 14 (12.1%) were classified as moderate; and 1 (0.9%) were classified as severe. Finally, 83 (71.6%) patients with a score < 3 were included in the low CONUT group, and 33 (28.4%) patients with a score≥3 were included in the high CONUT group. The median follow-up period was 42.2 months (range, 3.5-104.2 months). Eleven patients (9.5%) experienced tumor PALN recurrence. Furthermore, of the 11 patients with recurrence, 5 patients were diagnosed with PALN recurrence alone and the other 6 patients experienced tumor relapse in combination with other sites simultaneously, including 2 patients with pelvic lymph node recurrence, 2 patient with parametrial recurrence, 1 patient with inguinal lymph node metastases, and 1 patient with supraclavicular metastasis. For all patients, the 3-year DFS, OS, and PALNMFS were 65.5%, 74.1%, and 91.2%, respectively (Figure 1).
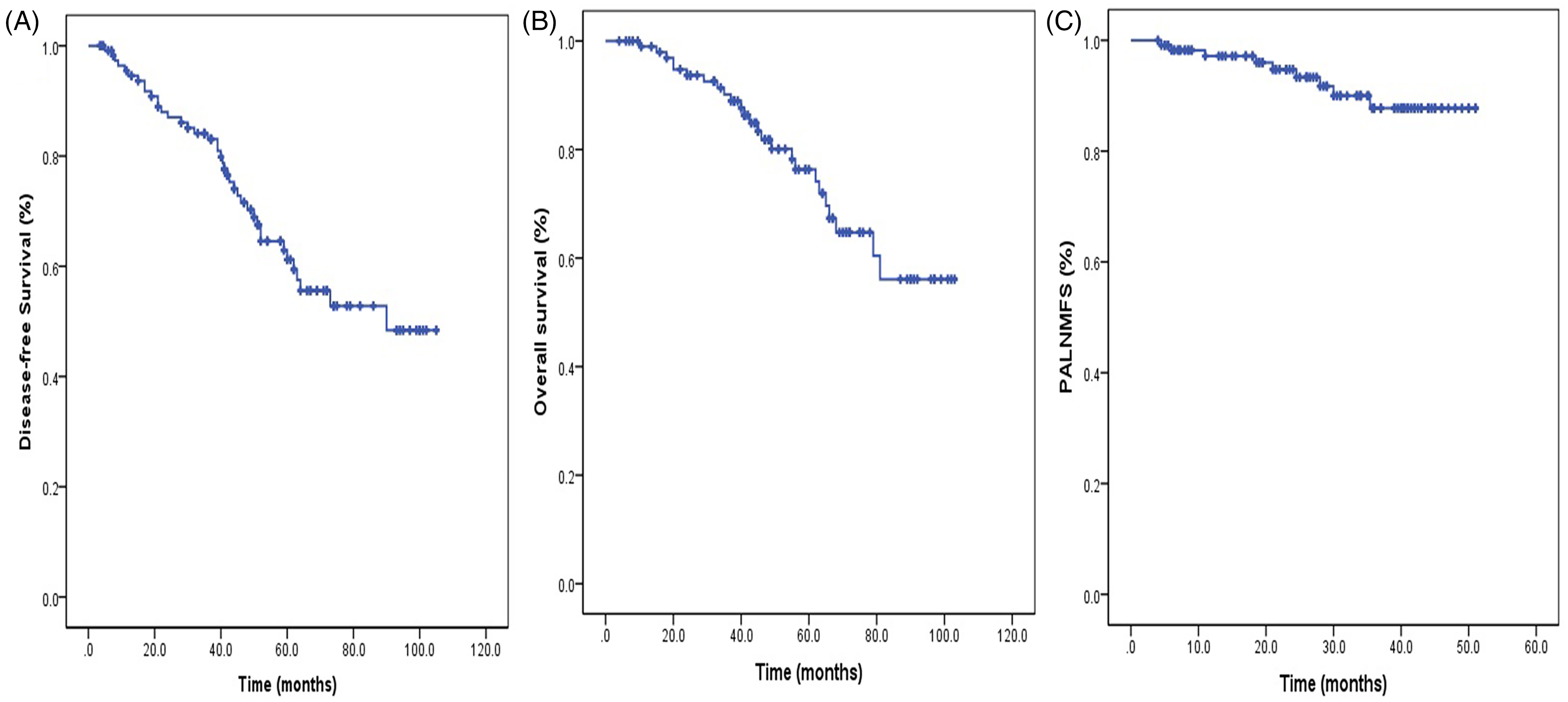
Disease-free survival (A), overall survival (B), and para-aortic lymph node metastasis-free survival (C) for all FIGO stage IIIB cervical cancer patients treated by definitive pelvic radiotherapy.
The Basic Patient and Tumor Clinical Features Classified.

Abbreviations: FIGO, International Federation of Gynecology and Obstetrics (2009); SCC-Ag, squamous cell carcinoma antigen; CCRT, concurrent chemoradiotherapy; RT, radiotherapy; CONUT, controlling nutritional status.
Univariate analysis was performed to determine the predictive factors for PALN recurrence. The results showed that tumor invasion to the pelvic wall (P = .024), the number of pelvic lymph nodes metastases (P = .001), and the CONUT score (P < .001) were associated with PALN failure. Multivariate analysis revealed that the CONUT score (HR: 3.141; 95% CI: 2.321-5.436; P < .001) and ≥3 pelvic lymph node metastases (HR: 2.235; 95% CI: 1.428-11.242; P < .001) were independent risk predictors of PALNMFS (Table 3).
Results of Univariate and Multivariate Analyses of Factors Associated With PALN Metastasis-free Survival.

Abbreviations: HR, hazard ratio; CI, confidence interval; PALNMFS, para-aortic lymph node metastasis-free survival; SCC, squamous cell carcinoma; Non-SCC, adenocarcinoma and adenosquamous carcinoma; SCC-Ag, squamous cell carcinoma antigen; CONUT, controlling nutritional status.
P ~.05.
Therefore, the CONUT score and the number of pelvic lymph node metastases were clearly key risk factors for PALN failure. Twenty-eight patients had ≥3 pelvic lymph node metastases. Sixty-four patients had no risk factors, 43 patients had one defined risk factor, and 9 patients had both defined risk factors. The 3-year DFS rates for patients with 0-2 pelvic MLNs and ≥3 pelvic MLNs were 68.2% and 35.7% (P < .001), the 3-year OS rates were 81.2% and 60.7% (P < .001), and the 3-year PALNMFS rates were 96.7% and 71.4% (P < .001), respectively (Figure 2). For patients in the low CONUT group and the high CONUT group, the 3-year DFS rates were 69.5% and 46.9% (P = .001), the 3-year OS rates were 79.7% and 68.5% (P = .016), and the 3-year PALNMFS rates were 96.4% and 74.8% (P < .001), respectively (Figure 3). The 3-year DFS rates for patients without and with risk factors were 70.5% and 48.8% (P < .001), the 3-year OS rates were 79.8% and 63.5% (P = .014), and the 3-year PALNMFS rates were 97.3% and 75.9% (P < .001), respectively (Figure 4).

Disease-free survival (A, P < .001), overall survival (B, P < .001), and para-aortic lymph node metastasis-free survival (C, P < .001) for patients with 0-2 pelvic metastatic lymph nodes and ≥3 pelvic metastatic lymph nodes.
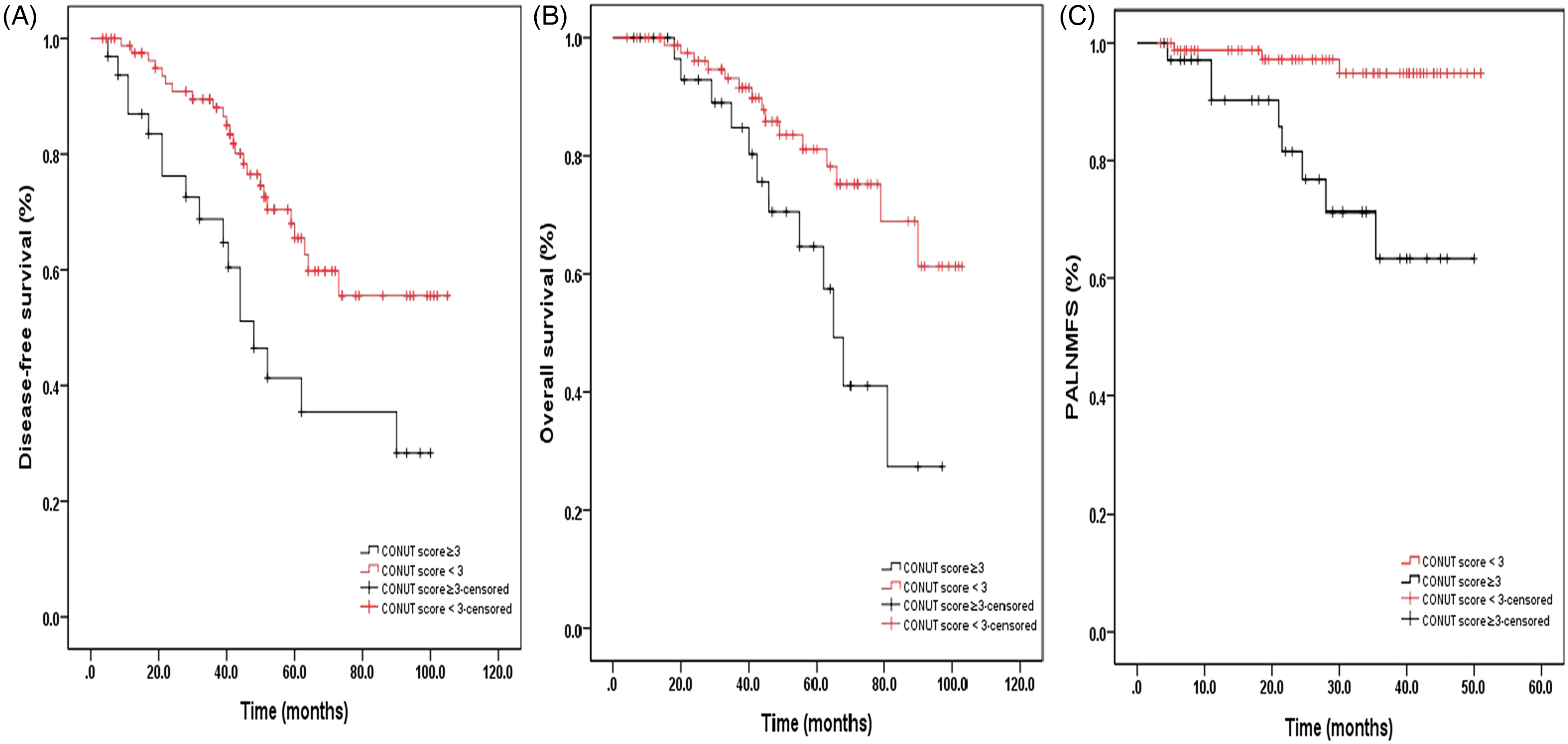
Disease-free survival (A, P = .001), overall survival (B, P = .016), and para-aortic lymph node metastasis-free survival (C, P < .001) based on the CONUT score in all patients.

Disease-free survival (A, P < .001), overall survival (B, P = .014), and para-aortic lymph node metastasis-free survival (C, P < .001) for patients without and with risk factors.
Discussion
Some previous studies demonstrated that patients with FIGO stage IIIB cervical cancer could benefit from prophylactic EFRT which could improve OS and DFS.10,13,14 Furthermore, studies to determine which stage IIIB cervical cancer patients would benefit from prophylactic EFRT were limited. Huang et al 18 found that SCC-Ag, advanced parametrial involvement, and pelvic MLNs were positively associated with PALN failure. In another study, prophylactic EFRT significantly improved 5-year cancer-specific survival rate in LACC patients with common iliac MLNs or ≥3 pelvic MLNs. 17 In the present study, we not only established that ≥3 pelvic MLNs was a risk factor for PALN failure but also found for the first time that a CONUT score ≥3 was a risk factor for PALN recurrence.
Recently, the immune-nutritional status, as indicated by the CONUT score, had been reported to independently predict prognosis and progression in many malignancies.20,25,26,29 In the study of Takagi et al, 30 meta-analysis found a significant association of the CONUT score with outcomes including OS (HR: 2.51; 95% CI: 1.75-3.60; P < .001), cancer-specific survival (HR: 2.60; 95% CI: 1.53-4.41; P < .001), and recurrence-free survival (HR: 2.08; 95% CI: 1.39-3.12; P < .001) for esophageal cancer. In another study of Harimoto et al, 31 2461 patients with hepatocellular carcinoma, a higher CONUT score was significantly associated with poor OS and recurrence-free survival using multivariate analysis. In our present study, we found a positive relationship between the CONUT score and PALN failure. Multivariate analysis revealed that the CONUT score was an independent predictor related to the PALNMFS, DFS, and OS in FIGO stage IIIB cervical cancer after pelvic radiation. A high CONUT score (score≥3) was significantly associated with a lower PALNMFS (P < .001) and worse DFS (P = .001) and OS (P = .016) compared with a low CONUT score (score < 3) in FIGO stage IIIB cervical cancer. Patients in the high CONUT group might particularly benefit from prophylactic EFRT. Therefore, the present study is the first to note that the CONUT score is an independent biomarker associated with PALN failure in FIGO stage IIIB cervical cancer patients after pelvic radiation. To date, this factor has not been recognized to predict the treatment success of prophylactic EFRT in LACC.
We hypothesized that the predictive effect of the CONUT score was related to its constituent parameters. Serum albumin reflected nutritional status and systematic inflammatory responses and was related to cancer prognosis.32–34 At the same time, the total cholesterol level had been found to be related to survival in cancer. 35 The total lymphocyte count was known as an indicator reflecting the immune-nutritional status, and lymphocytopenia was also associated with poor survival in several malignancies.36,37 The prognostic nutritional index (PNI, combined albumin (g/L) × total lymphocyte count × 109/L) score had been reported to predict poor prognosis in cervical cancer patients. 38 Some studies had demonstrated that the pretreatment CONUT score was an effective independent predictor of prognosis and was a more precise marker of prognosis in tumor patients than the PNI.20,30,39 In part, this was due to the fact that the total number of lymphocytes was more emphasized in the CONUT score. Furthermore, total cholesterol concentration which was not assessed in PNI might play an important role in the CONUT composite measure. Therefore, CONUT was a reliable marker of nutrition and immunization and exhibited a remarkable predictive value, as shown in the study.
Our study showed that tumor invasion to the pelvic wall was not significantly associated with PALNMFS in FIGO stage IIIB cervical cancer. However, some previous studies had reported that tumor invasion to the pelvic wall was associated with tumor PALN metastasis.7–9 The data in our study could not support this conclusion.
There were some limitations in the present study. This was a retrospective study and there might be potential selection bias. Second, some patients with common iliac lymph node metastases had already received prophylactic EFRT and were excluded from the study. Therefore, the number of samples and events were limited in the present study, which potentially influenced the conclusions of our study. Third, in this study, we did not verify whether the 2 risk factors for PALN failure could indeed predict the treatment success of prophylactic EFRT in patients with FIGO stage IIIB cervical cancer. Fourth, the few different treatment options may bias the results. Finally, some patients inevitably received nutritional support after curative treatment, and we did not collect posttreatment CONUT scores. These situations potentially complicated our results. We agree that further prospective randomized controlled studies should be performed to better understand whether this hypothesized predictive potential is accurate.
Conclusions
In conclusion, ≥3 pelvic MLNs and a high CONUT score (score≥3) were significant predictors of PALNMFS after pelvic radiation in FIGO stage IIIB cervical cancer patients. Patients with these risk factors might benefit from prophylactic EFRT.
Supplemental Material
sj-tif-1-tct-10.1177_15330338221141541 - Supplemental material for A Novel Risk Factor for Para-Aortic Lymph Node Recurrence After Definite Pelvic Radiotherapy in Stage IIIB Cervical Cancer
Supplemental material, sj-tif-1-tct-10.1177_15330338221141541 for A Novel Risk Factor for Para-Aortic Lymph Node Recurrence After Definite Pelvic Radiotherapy in Stage IIIB Cervical Cancer by Guangyu Zhang, Cong Wang, Changdong Ma, Li Miao, Fangfang He and Chunli Fu in Technology in Cancer Research & Treatment
Supplemental Material
sj-doc-2-tct-10.1177_15330338221141541 - Supplemental material for A Novel Risk Factor for Para-Aortic Lymph Node Recurrence After Definite Pelvic Radiotherapy in Stage IIIB Cervical Cancer
Supplemental material, sj-doc-2-tct-10.1177_15330338221141541 for A Novel Risk Factor for Para-Aortic Lymph Node Recurrence After Definite Pelvic Radiotherapy in Stage IIIB Cervical Cancer by Guangyu Zhang, Cong Wang, Changdong Ma, Li Miao, Fangfang He and Chunli Fu in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the Ethics Committee of Shandong First Medical University (Shandong Cancer Hospital and Institute, SDTHEC201904002).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
