Abstract
Importance:
Our understanding of the utility of circulating tumor cells (CTCs) and disseminated tumor cells (DTCs) as clinical biomarkers continues to evolve.
Objective:
This study evaluated (1) clinicopathologic factors associated with the presence of CTCs or DTCs, (2) the prognostic value of CTCs or DTCs by disease stage, 3), the value of these biomarkers in predicting the benefit of chemotherapy.
Design:
This is a retrospective analysis of patients with breast cancer (BC) diagnosed between 2004 and 2016 using the National Cancer Database (NCDB). To evaluate variables associated with the presence of CTCs or DTCs at the univariate level, we used chi-squared and Wilcoxon rank-sum tests. Multivariate logistic regression models were then constructed using significant variables. Consequently, we included CTC or DTC status (i.e. positive or negative) in multivariate, stage-by-stage Cox regression analyses for overall survival (OS). After stratifying by receptor status and staging, we used the Kaplan-Meier method to explore chemotherapy efficacy in CTC- or DTC-positive vs. CTC- or DTC-negative subsets.
Results:
Factors significantly associated with CTCs were race, progesterone receptor status, HER2 status, histology and AJCC N- and M-staging. Factors associated with DTCs were race, HER2 status, histology and AJCC N-staging. CTCs were associated with poor OS in late-stage (III and IV) but not early-stage (0-II) BC. DTCs were not significantly associated with OS in either context. In hormone receptor (HR)-positive disease, chemotherapy was associated with better OS when CTC status was positive, both in early-stage and late-stage disease. In a subset of patients without CTCs, however, chemotherapy conferred no survival benefit. DTC status was not a significant predictor of chemotherapy efficacy in early or late-stage, HR+ disease.
Conclusions:
This study suggests that CTC-status is a significant prognostic factor at later stages of BC; yet it can also help guide management of early-stage disease as it appears predictive for chemotherapy benefit.
Introduction
Cells shed by a solid tumor into the bloodstream are known as “circulating tumor cells” (CTCs). Alongside other biomarkers, including circulating tumor DNA (ctDNA), CTCs are an important component of the increasingly-researched “liquid biopsy” in patients with solid tumors. 1
Several studies have investigated the potential use of the liquid biopsy as a diagnostic tool in breast cancer (BC). 2,3 However, conclusions derived from these studies have been limited by low sensitivity, 4,5 particularly in the diagnosis of early-stage disease. 6,7 The presence of CTCs has been associated with treatment failure, 8,9 as well as poor survival in both non-metastatic 8,10 and metastatic disease 11 -13 ; even in the absence of radiologically-identified disease progression. 14 Our understanding of the utility of liquid biopsies in solid tumors, however, is still evolving. At the time of this study, the recommended use of the liquid biopsy in National Comprehensive Cancer Network (NCCN) BC guidelines was limited to diagnosing PIK3CA mutations prior to alpelisib/fulvestrant administration in hormone receptor-positive/HER2-negative metastatic disease. 15
In addition to CTCs, the presence of disseminated tumor cells (DTCs) has also emerged as a potential biomarker in the evaluation of solid tumors. DTCs are tumor cells detectable in non-primary site tissue or bone marrow. When tumor cells migrate to the bone marrow, they can enter a quiescent state and survive there for years. 16 When malignant cells spread through the circulation to distant sites, this represents a disease state commonly referred to as “minimal residual disease” (MRD). 17 This is thought to contribute to cases of disease recurrence after a period of extended remission for many years. 16 As with CTCs, the presence of DTCs has also been shown to be associated with poorer survival. 18 -20
The value of biomarkers in oncology practice extends beyond diagnosis and prognostication. Multigene sequencing, for example, allows clinicians to optimize therapeutic decision-making based on predictions of disease outcomes, including metastasis and recurrence. The 21-gene assay (Oncotype DX) generates a recurrence score (RS) that is used to predict the benefit of adding chemotherapy to adjuvant endocrine therapy in hormone receptor positive, HER2-negative, node-negative breast cancer [15].
The aim of this study was to utilize a large repository of data from the National Cancer Database (NCDB) to evaluate: 1) clinicopathologic factors associated with the presence of CTCs or DTCs, 2) the prognostic value of CTCs or DTCs by disease stage, and 3) the association between CTC or DTC status and systemic therapy treatment outcomes.
Materials and Methods
Patient Data
We analyzed data from patients with breast cancer, diagnosed between 2004-2016 as captured by the NCDB, based on a Participant User File (PUF) award granted to the principal investigator (Z.N.). The NCDB is a large, outcomes-based cancer registry estimated to include 70% of all cancer patients in the United States of America. 21 Supported jointly by the Commission on Cancer (CoC) and the American College of Surgeons (ACS), the information in this database is collected from over 1500 medical institutions (21). All NCDB cases included in this analysis contained data on CTC or DTC status (i.e. positive vs. negative),
Statistical Analysis
Data analysis was conducted using SPSS version 26.0 (IBM Corp, Armonk, NY). Descriptive statistics, namely frequency and proportion, were performed to describe patient clinicopathologic factors in either a cohort of cases that included documented CTC status, or a cohort with documented DTC status. Univariate chi-squared analyses were then used to evaluate for a significant association between these clinicopathologic factors and the presence of either CTCs or DTCs. Significant variables (p < 0.05) were subsequently included in multivariate logistic regression models, one predicting the presence of CTCs and another predicting the presence of DTCs. Overall AJCC clinical staging (i.e. 0, I, II, III, IV) was used to describe each cohort, however individualized T-, N- and M-staging were used in our multivariable models predicting the presence of these biomarkers. This was done because the variable for overall staging in the NCDB is not always highly-specific (e.g. reporting I vs. IA or IB). Including the TNM components separately allows us to understand with greater detail how each contributes independently to the likelihood of having CTCs or DTCs.
Survival analyses were performed using multivariate Cox regression for overall survivorship (OS). We conducted models on the following groups: 1) all BC patients with documented CTC status; 2) all BC patients with documented DTC status. Additional survival models were run after stratifying by disease stage (early-stage vs. late-stage). These multivariate survival models controlled for known predictors of survival that are available in the NCDB database, including: age, race, Charlson/Deyo comorbidity index, use of surgery, use of radiotherapy, use of chemotherapy, use of hormonal therapy and the use of immunotherapy. Finally, the potential benefit of additional chemotherapy to endocrine therapy was assessed using Kaplan-Meier survival analysis in a subset of hormone receptor-positive (ER+ or PR+) patients with stratifications for CTC status or DTC status (positive vs. negative) and stage of disease (early-stage vs. late-stage).
Results
Description of CTC Cohort
Descriptive Statistics of this Group are Presented in Table 1.
Clinicopathologic characteristics of patients and univariate association with circulating tumor cell (CTC) or disseminated tumor cell (DTC) status
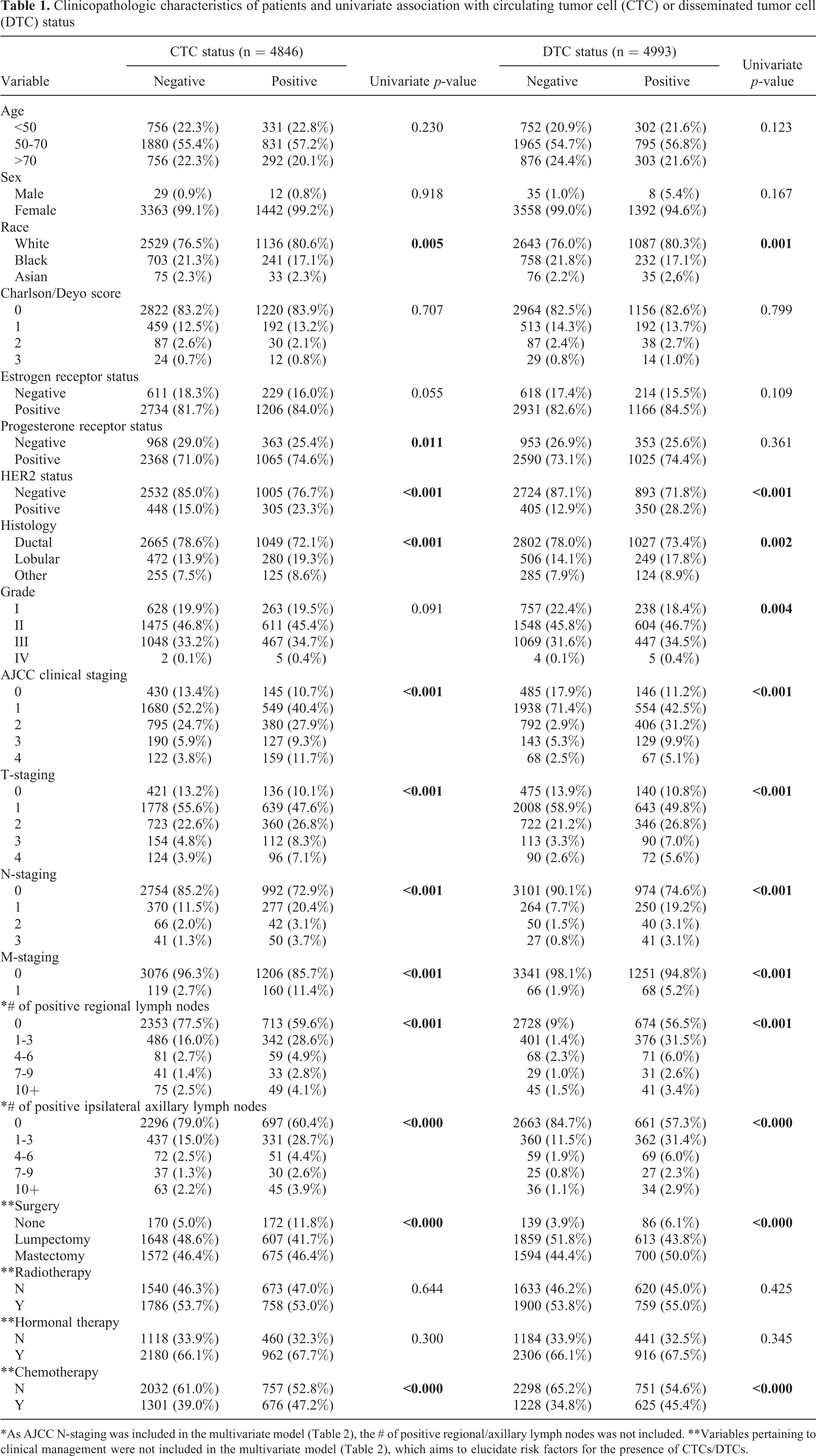
*As AJCC N-staging was included in the multivariate model (Table 2), the # of positive regional/axillary lymph nodes was not included. **Variables pertaining to clinical management were not included in the multivariate model (Table 2), which aims to elucidate risk factors for the presence of CTCs/DTCs.
CTC status was collected on a total of n = 4846 cases. Of these, n = 3392 (70.0%) were negative and n = 1454 (30.0%) were positive. Most patients were between 50-70 years old (n = 2711, 55.9%), White (n = 3665, 73.4%), and had a Charlson/Deyo score of 0 (n = 4042, 81.0%). The predominant histological subtype was ductal carcinoma (n = 3714, 74.4%). Most cases were diagnosed at stage I (n = 2229, 48.7%), followed by stage II (n = 1175, 25.7%), stage 0 (n = 575, 12.6%), stage III (n = 317, 6.9%) and stage IV (n = 281, 6.1%). Regarding management: the majority of patients in the CTC cohort received surgery (n = 2225, 45.2% undergoing lumpectomy, and n = 2247, 45.0% undergoing mastectomy), radiotherapy (n = 2544, 51.0%), and hormonal therapy (n = 3142, 62.9%), but a minority received chemotherapy (n = 1977, 39.5%).
Our multivariable model predicting positive CTC status is presented in Table 2. Statistically significant predictors of positive CTC status were as follows:
Multiple Logistic Regression for Clinicopathologic Characteristics Associated With Circulating Tumor Cell (CTC) or Disseminated Tumor Cell (DTC) Status.

Description of DTC Cohort
Descriptive Statistics of this Group are Presented in Table 1
DTC status was available for a total of n = 4993 patients. Of these, n = 3593 (72.0%) were negative and n = 1400 (28.0%) were positive. As with the CTC cohort, most patients were between 50-70 years old (n = 2760, 55.3%), White (n = 3730, 74.7%), and had a Charlson/Deyo score of 0 (n = 4120, 82.5%). The predominant histological subtype was ductal (n = 3829, 76.7%). Most cases were diagnosed at stage I (n = 1938, 41.0%), followed by stage II (n = 792, 16.8%), stage 0 (n = 485, 10.3%), stage III (n = 143, 3.0%) and stage IV (n = 122, 2.7%). As with the CTC cohort: the majority received surgery (n = 2472, 49.5% undergoing lumpectomy, and n = 2294, 45.9% undergoing mastectomy), radiotherapy (n = 2659, 53.3%), and hormonal therapy (n = 3222, 64.5%), but a minority received chemotherapy (n = 1853, 37.1%).
Our multivariable model predicting positive DTC status is presented in Table 2. Statistically significant predictors of positive DTC status were:
Survival Analysis
The results of our survival analyses can be found in Table 3.
Results From Survival Analyses in (1) Cohort Overall, (2) Early-Stage Sub-Group, and (3) Late-Stage Subgroup.
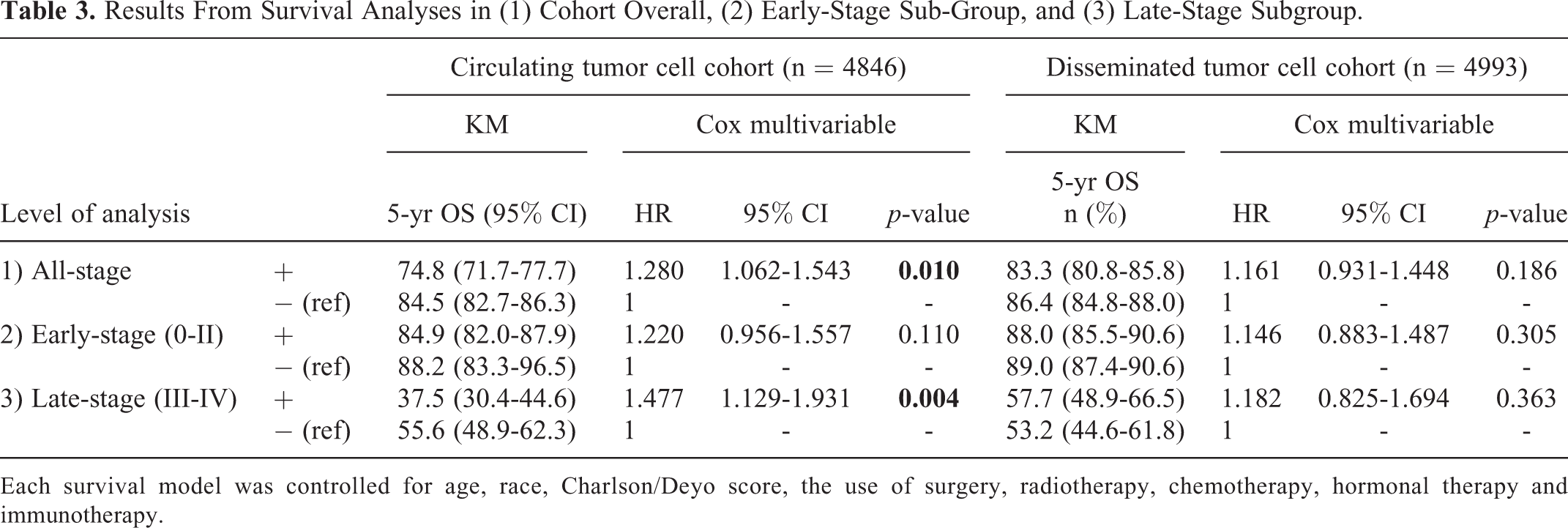
Each survival model was controlled for age, race, Charlson/Deyo score, the use of surgery, radiotherapy, chemotherapy, hormonal therapy and immunotherapy.
CTC cohort (n = 4846)
Positive CTC status was significantly associated with an increased risk of mortality (HR: 1.280, 95% CI: 1.062-1.543, p = 0.010) in a model controlling for age, race, Charlson/Deyo score, AJCC clinical staging, receptor status, use of surgery, radiotherapy, chemotherapy, hormonal therapy and immunotherapy. In a subgroup analysis of patients with early-stage breast cancer (0, I and II), CTC status was not a significant predictor of OS (HR: 1.220, 95% CI: 0.956-1.557, p = 0.110). Conversely, CTC-positivity was a significant predictor of mortality (HR: 1.477, 95% CI: 1.129-1.931, p = 0.004) in advanced-stage disease (III and IV), with a 5-year OS of 37.5% (95% CI 30.4-44.6) for those with CTCs compared to 55.6% (95% 48.9-62.3%) for those without.
DTC cohort (n = 4993)
Positive DTC status was not a significant predictor of mortality (HR: 1.161, 95% CI: 0.931-1.448, p = 0.186) in the cohort overall after controlling for the effect of age, race, Charlson/Deyo score, AJCC clinical staging, receptor status, use of surgery, radiotherapy, chemotherapy, hormonal therapy and immunotherapy. Subgroup analyses of early-stage BC (HR: 1.146, 95% CI: 0.883-1.487, p = 0.305) and late-stage BC (HR: 1.182, 95% CI: 0.825-1.694, p = 0.363) did not reveal a significant association between DTC status and OS.
Benefit of Chemotherapy in Hormone-Receptor Positive Disease
In a cohort of hormone receptor-positive BC, CTC status was predictive of the efficacy of chemotherapy in improving OS. Figure 1 demonstrates no difference in survival for early-stage BC patients between patients treated with endocrine therapy alone vs. endocrine therapy with chemotherapy when CTC was negative (p = 0.638), but a survival benefit for those receiving chemotherapy when CTC status was positive (p = 0.003). Similarly, Figure 2 demonstrates there was no difference in survival between late-stage BC patients treated with endocrine therapy alone vs. endocrine therapy with chemotherapy with negative CTC status (p = 0.501), but improved OS in patients receiving chemotherapy when CTC status was positive (p = 0.023). DTC status was not a significant predictor of chemotherapy efficacy in early or late-stage, HR+ disease.
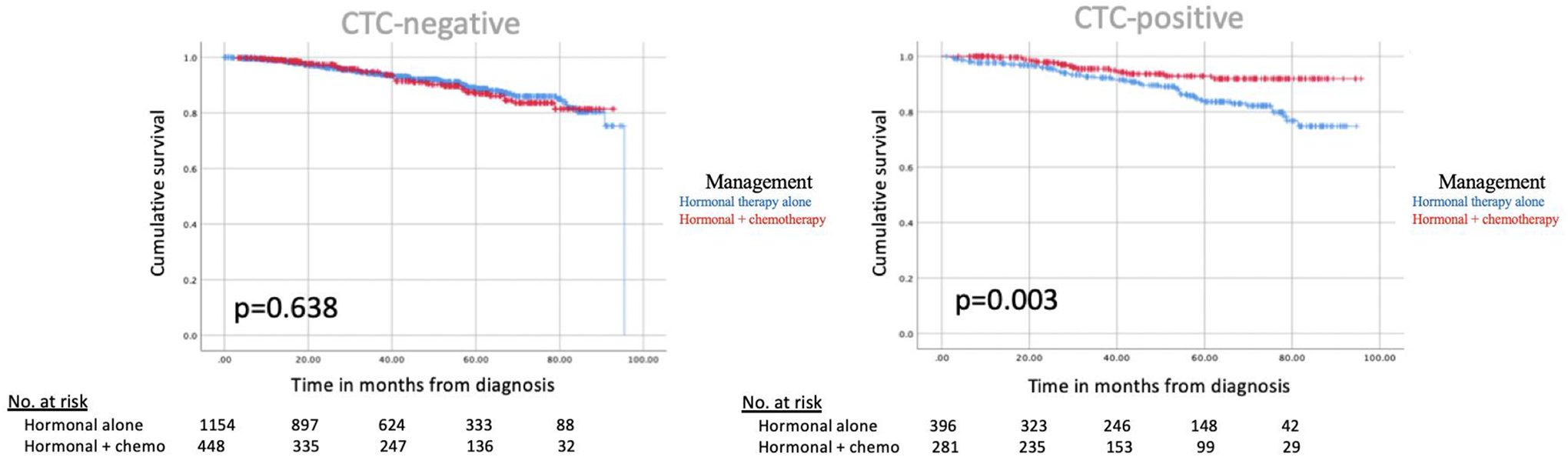
Kaplan-Meier curves for hormone receptor positive, early-stage (I and II) breast cancer.
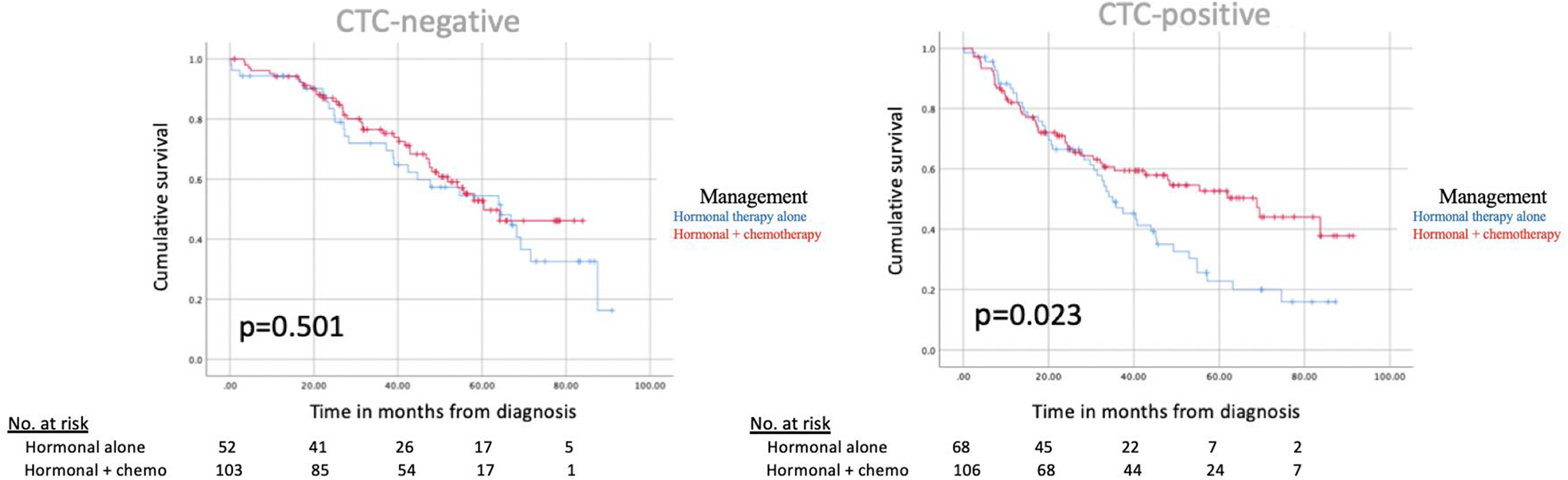
Kaplan-Meier curves for hormone receptor positive, late-stage (III and IV) breast cancer.
Discussion
In this large study, we found that that CTC status is a significant predictor of OS in BC, but DTC status is not. Patients with CTCs exhibited shorter OS, specifically in advanced disease stages (5-year OS 37.5% compared to 55.6%). This corroborates literature emphasizing the potential of CTC status as a prognostic cancer biomarker. 8 -10
Of all patients tested and included in this study, 30% were positive for CTCs. Positive PR or HER2 status (but not ER status), lobular histology, and higher AJCC clinical N-staging and M-staging were all significantly associated with the presence of CTCs. The association between receptor status and CTC-positivity substantiates literature already outlining a role of HER2 in early breast cancer metastasis. 22 Using data from this study and others, we may be able to synthesize a risk profile of patients likely to have circulating tumor cells, which may be used by clinicians to efficaciously triage BC patients to liquid biopsy, expanding upon already-existing precision medicine practices.
CTC status may also be useful in the optimization of systemic therapy, in late-stage as well as early-stage BC. We found that additional chemotherapy, versus endocrine therapy alone, was associated with better OS in patients with early-stage and late-stage, HR+ BC patients when CTC status was positive, but not when it was negative. Current clinical practice uses a 21-gene recurrence score assay (Oncotype DX, Genomic Health Inc., Redwood City, CA) for a 10-year risk for recurrence score (RS) in patients with early-stage, ER+ BC. 23 High-risk individuals may benefit most from the addition of chemotherapy to endocrine therapy, helping clinicians make an evidence-based decision before escalating therapy in certain patients. 15 In one study, a high RS was not significantly associated with the presence of CTCs or DTCs, 24 suggesting that the predictive value of CTCs may be independent of this 21-gene assay. Goodman et al. also used the NCDB to show that there was a significant association between CTC status with a benefit of radiotherapy in early-stage BC, 25 further emphasizing the potential utility of this biomarker in clinical decision-making. More research is needed to clarify the potential role of liquid biopsy components across disease stage and subtype.
The strengths of this study include its large sample size compared to other published literature on this topic, as well as the consistency and reliability of NCDB data, which undergoes a rigorous internal verification process. The limitations include those expected of a retrospective database. The NCDB does not report temporal data pertaining to disease progression. Of the total BC dataset (over 2 million BC patients), a small minority were assessed for CTC or DTC status. Additionally, the NCDB does not document the specific assay used to assess circulating or disseminated tumor cell status, the timing of the assay relative to diagnosis, nor the quantity of cells measured in each case. Data inconsistencies based on the assay used has been documented in the literature. 1
The promising results from this study highlight the need for more research to clarify the potential role of liquid biopsy components across BC disease stage and subtype.
Footnotes
Authors’ Note
N.B. is a major contributor in study design, conduction of statistics and data interpretation, and manuscript writing. Z.N. participated in design, interpretation of the data, and writing the manuscript. H.L. participated in data interpretation. L.E., R.A. and E.E. participated in writing and editing. All authors read and approved the final manuscript. The data that support the findings of this study are available from the American College of Surgeons/American Cancer Society but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of the American College of Surgeons/American Cancer Society. No individual person’s data included; all data is reported in an aggregate manner. Ethical approval was obtained from the Cleveland Clinic Institutional Review Board (IRB) prior to conducting this study. All patient data was strictly de-identified and provided, with approval, from the American College of Surgeons as part of the National Cancer Database.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
