Abstract
Introduction
In the past 20 years, axillary staging for breast cancer has been a standard part of initial surgical treatment. Sentinel lymph node biopsy (SLNB) has become the standard of care for axillary staging over the past decade. Currently, the Choosing Wisely statement by the Society of Surgical Oncology, Ontario Health (Cancer Care Ontario) and ASCO guideline recommend against the routine use of sentinel node biopsy in clinically node-negative women ≥70 years old with early-stage hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative invasive breast cancer.1, 2 However, other consensus favored the procedure in women even those aged in their 80s who were undergoing surgery for breast cancer.3, 4 There is still controversy regarding whether to perform SLNB in patients aged ≥70 years with early-stage invasive breast cancer. In our practice, SLNB is performed for clinically node-negative patients irrespective of age. This study focused on patients treated with SLNB in their 70s and 80s to figure out whether sentinel lymph node (SLN) status affects prognosis and treatment.
Materials and Methods
This study conforms to the STROBE guidelines. 5 The research protocol was examined and approved by the Ethics Committee of the Beijing Cancer Hospital (2022YJZ30) on April 18, 2022.
Patient Characteristics
Data from patients ≥70 years old with primary breast cancer between 2010 and 2019 were retrospectively obtained from the database of Peking University Cancer Hospital. The inclusion criteria for this study were as follows: patients aged ≥70 years with newly diagnosed unilateral invasive breast cancers, with clinically negative nodes, who were treated with SLNB followed by mastectomy or lumpectomy. Patients with previous breast cancer or other cancers, bilateral breast cancers, or newly diagnosed breast carcinoma in situ were excluded. A total of consecutive 399 patients were initially included in this study. The overall success rate for SLN localization was 94.2% (376 / 399). Because SLN mapping was unsuccessful, 23 patients were not included in the final analysis. The need for specified informed consent was waived owing to the retrospective nature of the study. All patient details have been de-identified.
Sentinel Lymph Node Biopsy
Axillary lymph nodes were evaluated using ultrasound combined with fine-needle aspiration. Clinically negative nodes were defined as normal nodes under ultrasound or abnormal nodes that were confirmed negative by fine-needle aspiration. For SLNB, 99mTc-rituximab was used as the single tracer. Two of the three sites were chosen for tracer injection: the peritumoral breast parenchyma, subcutaneous, and subareolar tissues. SLNB was performed under local anesthesia. The samples were formalin-fixed and embedded in paraffin, with at least one cut in 5-mm intervals, and stained with hematoxylin and eosin. Cytokeratin immunohistochemistry was routinely performed. Isolated tumor cells in SLNs were defined as negative nodes. Macrometastatic and micrometastatic lesions were considered pathologically positive.
Follow-Up and Outcomes
The duration of follow-up was calculated from the date of surgery. The endpoints of this study were disease-free survival (DFS), distant disease-free survival (DDFS), breast cancer-specific survival (BCSS), and overall survival (OS). DFS events included local recurrence, regional recurrence, distant metastasis, contralateral breast cancer, non-breast secondary primary cancer, and any death. DDFS was measured from surgery until distant metastases were observed. BCSS was defined as the time to breast cancer-related death with patients censored at the date of the last follow-up or death from other causes. OS was defined as the time to any death.
Statistical Analysis
Statistical power calculations were not performed before the study. Patient characteristics were compared between subtypes using Pearson's chi-square test and Fisher's exact test. Survival probabilities were estimated using the Kaplan–Meier method. Cox proportional hazards regression was performed to assess the independent associations of several variables with DFS, DDFS, BCSS, and OS. Hazard ratios and their 95% confidence intervals (CIs) were estimated using a Cox model with a backward stepwise method. All reported p-values were two-sided with a value of <0.05 considered as statistically significant. All statistical analyses were performed using SPSS 23.0 (IBM, Armonk, NY, USA).
Results
A total of 376 patients ≥70 years old who underwent SLNB were included in our study (Figure 1): of these patients, 311 (82.7%) had a negative SLN and 65 (17.3%) had at least one positive node.

Flowchart of patient selection in this study. SLNB, sentinel lymph node biopsy.
Table 1 shows the clinicopathological characteristics of patients stratified according to SLN status. One hundred and eighteen patients received adjuvant radiation therapy: 6/207 (2.9%) mastectomy patients underwent postmastectomy radiation therapy and 112/169 (66.3%) lumpectomy patients received whole breast (86/112, 76.8%) or intraoperative breast (26/112, 23.2%) radiation. A total of 321/324 (99.1%) patients who were estrogen receptor-positive and/or progesterone receptor-positive (>1% positive cells by immunohistochemistry staining) received endocrine treatment. Only 9/44 (20.5%) patients with HER2-positive tumors received trastuzumab.
Characteristics of Patients and SLNB Results.
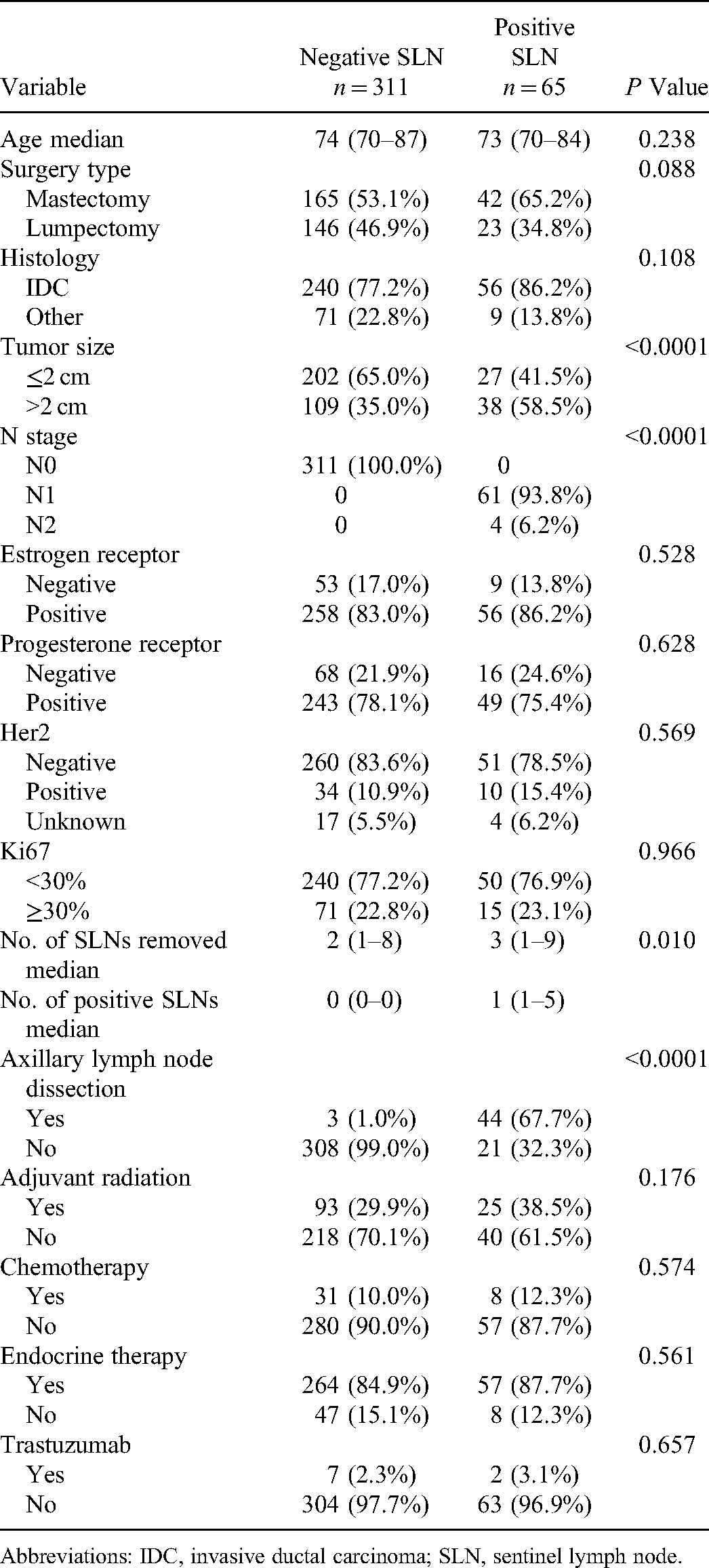
Abbreviations: IDC, invasive ductal carcinoma; SLN, sentinel lymph node.
The median age at diagnosis was 74 and 73 years in the SLN-negative and SLN-positive groups, respectively. SLN-positive patients were more likely to undergo axillary lymph node dissection (ALND) (P < 0.0001) and had larger tumors (P < 0.0001) compared to SLN-negative patients. Estrogen receptor, progesterone receptor, ki67, and HER2 status were not significantly different between groups. In terms of treatment, there were no statistically significant differences in surgery type, adjuvant radiation, chemotherapy, or endocrine treatment between the two groups (Table 1).
After a median follow-up of 70 (range 3–143) months, 30 (8.0%) patients experienced recurrence. Twenty-four SLN-negative patients experienced locoregional (n = 6) or distant (n = 11) recurrence, contralateral breast tumor (n = 1), or other primary cancers (n = 6). Six SLN-positive patients experienced locoregional (n = 1) or distant recurrence (n = 4) or other primary cancers (n = 1) (Table 2). The 5-year DFS rates were 88.2% (95% CI, 84.3–92.1%) in the SLN-negative group and 87.6% (95% CI, 79.8–95.4%) in the SLN-positive group. The 5-year DDFS was 96.7% (95% CI, 94.3–99.1%) in the SLN-negative group and 94.8% (95% CI, 88.9–>99.9%) in the SLN-positive group. The 5-year BCSS was 96.2% (95% CI, 93.8–98.6%) in the SLN-negative group and 93.6% (95% CI, 86.6–>99.9%) in the SLN-positive group. The 5-year OS was 93.5% (95% CI, 90.6–96.4%) in the SLN-negative group and 90.0% (95% CI, 81.6–98.4%) in the SLN-positive group. Kaplan–Meier survival analyses showed no significant differences in DFS, DDFS, BCSS, or OS between the two groups (Table 3). The variables of SLN status, tumor size, chemotherapy, endocrine therapy, and adjuvant radiation were included in the Cox multivariate analysis. None of the variables were found to significantly affect DFS, DDFS, BCSS, or OS. Multivariable analysis showed that SLN positivity was not significantly associated with DFS, DDFS, BCSS, or OS (Table 4).
Description of Events.

Abbreviation: SLN, sentinel lymph node.
Comparisons of Survival Outcomes Between SLN-Negative and SLN-Positive Groups in the Entire Cohort.

Abbreviations: BCSS, breast cancer-specific survival; CI, confidence interval; DDFS, distant disease-free survival; DFS, disease-free survival; SLN, sentinel lymph node; OS, overall survival.
Multivariate Analyses for the Effect of Clinicopathologic Characteristics and SLN Status on Survival in the Entire Cohort.

Abbreviations: BCSS, breast cancer-specific survival; DDFS, distant disease-free survival; DFS, disease-free survival; SLN, sentinel lymph node; OS, overall survival.
Discussion
Our study found that the treatment of patients ≥70 years old was similar between the SLN-negative and SLN-positive groups. SLN status affected ALND but not the use of radiation, endocrine, or chemotherapy. There were no significant differences in DFS, DDFS, BCSS, or OS according to SLN status. The results of our study support the recommendation of omitting SLNB in patients ≥70 years old with early-stage invasive breast cancer.
The positive rate of SLNB in our study was 17.3%. It was lower than the positive rate of 25.6% in the entire population who underwent SLNB in our center, which was reported in another study. 6 Meanwhile, only 6.2% of patients in the SLN-positive group had ≥4 lymph node metastases. Our data showed that elderly patients had a low tumor burden in the axillary lymph nodes, regardless of the positivity rate of SLNB or N2 stage. The indolent biological characteristics of tumors in elderly patients have also been reported by other studies.7–9
In our study, 66.3% of lumpectomy patients received radiation and 20.5% of patients with HER2-positive tumor received trastuzumab. These data suggest that elderly patients are often treated with de-escalating adjuvant therapy. First, competing comorbid conditions, limited life expectancy, and concerns regarding the quality of life after treatment are important factors when making decisions about treatment plans for elderly patients. Second, adjuvant radiation therapy has not been proven to improve survival in this patient population.8, 10 Furthermore, a retrospective study involving 492 patients showed that elderly patients with positive nodes were significantly less likely to receive chemotherapy than younger patients. 11 Adjuvant treatment recommendations are increasingly based on biology rather than prognostic information coming from nodal statuses, such as triple-negative and HER2-positive breast cancer. 12
There is an ongoing debate regarding whether SLNB should be performed in patients ≥70 years old with early-stage invasive breast cancer. On one hand, two prospective studies and one meta-analysis found that omission of axillary staging by ALND in women ≥70 years, with clinically negative axilla, resulted in an increased risk of regional recurrence, but did not affect overall and breast cancer-specific mortality.13–15 A recent study from the European Institute of Oncology using a matched analysis based on 1748 patients ≥70 years old found that axillary staging in elderly women did not impact long-term survival. 16 Based on these evidence, the Choosing Wisely statement by the Society of Surgical Oncology, Ontario Health (Cancer Care Ontario) and ASCO guideline recommend that SLNB is not required for patients aged ≥70 years with clinically node-negative (T1N0) early-stage invasive breast cancer, which is hormone receptor-positive and HER2-negative.1, 2 On the other hand, no solid evidence is available at this time for staging by SLNB compared with observation, but some retrospective studies concluded that SLNB may still provide important information affecting treatment and survival.17,18 The 2021 St Gallen consensus, European Society of Breast Cancer Specialists (EUSOMA), and International Society of Geriatric Oncology (SIOG) still recommend that patients aged ≥70 years should be offered the same surgery as younger patients.3, 4
Currently, there are three ongoing trials focused on the comparison between SLNB and observation. One trial is examining the outcomes of lumpectomy without sentinel node biopsy in women aged ≥65 years with clinically node-negative breast cancer (NCT02564848). Another trial in the Netherlands is determining whether omitting the SLN procedure is not inferior to the current axillary staging regimen in clinically node-negative breast cancer patients undergoing breast-conserving therapy in terms of regional recurrence rate (NCT02271828). The SOUND trial is comparing SLNB with no surgery at all in the axilla when axilla ultrasonography findings are negative (NCT02167490).
Our study has several limitations. First, this study was retrospective in design, and power calculations were not conducted to estimate the required sample size. Second, studies with small sample populations are usually too underpowered to show any statistical significance. Third, selection bias was unavoidable due to the retrospective nature of the study.
Conclusions
In our study, SLN status may not affect systemic treatment decisions or survival in patients aged ≥70 years with breast cancer. Our results support the omission of SLNB in patients ≥ 70 years with clinically negative axillary lymph nodes. Several ongoing prospective trials will provide more evidence on this issue.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221137216 - Supplemental material for Impact of Sentinel Lymph Node Biopsy on Treatment Decision and Survival in Patients Aged ≥70 Years with Breast Cancer: A Retrospective Study
Supplemental material, sj-docx-1-tct-10.1177_15330338221137216 for Impact of Sentinel Lymph Node Biopsy on Treatment Decision and Survival in Patients Aged ≥70 Years with Breast Cancer: A Retrospective Study by Chongshan Gu, Xue Chen, Lize Wang, Yingjian He, Tao Ouyang, Jinfeng Li, Tianfeng Wang, Tie Fan and Zhaoqing Fan in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
The authors acknowledge Xue Chen, who managed the database. We appreciate Yingjian He, who worked on the statistics.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by capital's funds for health improvement and research (grant number 2018-2-2152).
Ethical Approval
The research protocol was examined and approved by the Ethics Committee of Beijing Cancer Hospital (2022YJZ30) on April 18, 2022.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
