Abstract
MicroRNA-378a (miR-378a), including miR-378a-3p and miR-378a-5p, are encoded in PPARGC1B gene. miR-378a is essential for tumorigenesis and is an independent prognostic biomarker for various malignant tumors. Aberrant expression of miR-378a affects several physiological and pathological processes, including proliferation, apoptosis, tumorigenesis, cancer invasion, metastasis, and therapeutic resistance. Interestingly, miR-378a has a dual functional role in either promoting or inhibiting tumorigenesis, independent of the cancer type. In this review, we comprehensively summarized the role and regulatory mechanisms of miR-378a in cancer development, hoping to provide a direction for its potential use in cancer therapy.
Introduction
Micro RNAs (miRNAs) are small non-coding RNAs (19-24 nucleotides) produced from an endogenous transcript having a local hairpin structure with the help of RNase III-type enzyme. 1 miRNAs can repress the translation of target proteins by cleaving or clinging to the 3′ untranslated region (UTR) of the corresponding messenger RNAs (mRNAs). 2 The first miRNA was discovered over 20 years ago in mammals. 3 Based on their genomic origin, miRNAs are divided into intergenic, intronic, and exonic. 4 They are essential regulators of various critical biological processes, such as cell differentiation,5,6 apoptosis,7,8 proliferation, 9 cell division, 10 protein secretion, 11 and viral infection. 12 Recently, several studies have shown that the aberrant expression of miRNA is tightly associated with the progression of human diseases, especially cancers, 13 as they can suppress or promote tumor growth by regulating the target gene mRNA.14-16
The miRNA miR-378a is one such essential tumor-regulating miRNA. It is located on chromosome 5q32 and embedded within the first intron of the PPARGC1β gene, which encodes the PGC1β protein. miR-378a is divided into miR-378a-3p (usually identified as miR-378) and miR-378a-5p (usually identified as miR-378*), which are the guide and passenger strands of miR-378a, respectively. 17 Both miR-378a-3p and miR-378a-5p are synchronously transcribed with PGC1β,18-20 although there is an exception. Recently, it has been demonstrated that the over-expression (Table 1) or under-expression (Table 2) of miR-378a regulates the expression of its target genes and affects the proliferation, 21 invasion, and metastasis 22 of cancers. For example, low miR-378a expression is usually correlated to a significantly poor overall survival (OS) 23 in colorectal cancer patients and associated with significantly decreased OS and disease-free survival (DFS) in gastric cancer patients. 24 However, its expression and role in different cancers, such as lung 25 and cervical cancers, 26 is still controversial. miR-378a was identified as an onco-RNA in various tumors, including lung cancer,22,25,27-29 ovarian cancer,30-32 cervical cancer, 33 nasopharyngeal carcinoma, 34 melanoma, 35 osteosarcoma, 36 cholangiocarcinoma, 37 acute myeloid leukemia, 38 chronic myeloid leukemia, 21 and Burkitt lymphoma. 39 In contrast, it acted as an tumor suppressor in gastric cancer, 40 colorectal cancer, 41 liver cancer, 42 glioblastoma, 43 prostate cancer, 44 breast cancer (BC), 45 medulloblastoma (MB), 46 pituitary adenoma (PA), 47 oral squamous cell carcinomas, 48 bladder cancer, 49 esophageal carcinoma, 50 rhabdomyosarcoma (RMS), 51 and retinoblastoma (RB). 52 Compared with normal tissues and no lymph node metastasis cancer tissues, miR-378a was upregulated in cervical cancer tissues, especially in CIN III and lymph node metastasis cervical cancer. 33 Additionally, a study of 120 cholangiocarcinoma tissues and adjacent noncancerous tissues found that miR-378a expression increased with the development of the TNM (T-tumor, N-nodes, M-metastasis) stages. 37 Positive miR-378a expression is also tightly linked to a low OS in cholangiocarcinoma patients. 37 However, in colorectal cancer patients, miR-378a is downregulated and low miR-378a correlated with poor OS, 4 suggesting an inhibiting effect of miR-378a. 24 Thus, miR-378a can drive cancer cell proliferation,29,53 invasion, and migration27,32 in some tumors, while inhibiting these processes in other ftumors.23,24,32,54,55 Further, a study on the radiation response in a glioblastoma xenograft model found that the overexpression of miR-378a was associated with enhanced local tumor response to radiotherapy by increasing the vascular density and perfusion, thereby prolonging the survival of tumor-bearing hosts and acting as a novel therapeutic intervention. 43 To summarize, the abnormal expression of miR-378a is crucial for the occurrence and development of malignant tumors. Therefore, it is important to understand the functions of miR-378a in tumor biology to enhance the efficiency of early screening and diagnosis of cancer, strengthen anticancer therapy, and develop novel targeted methods for tumor therapy. Hence, this review focuses on the roles and targets of miR-378a in different cancers to elucidate the specific molecular mechanisms of miR-378a during tumor progression.
miR-378a Plays Tumorigenic Roles as an Oncogene.

↑indicates upregulation (in case of expression level) or promoting (in case of functional consequences).
↓indicates downregulation (in case of expression level) or suppressing (in case of functional consequences).
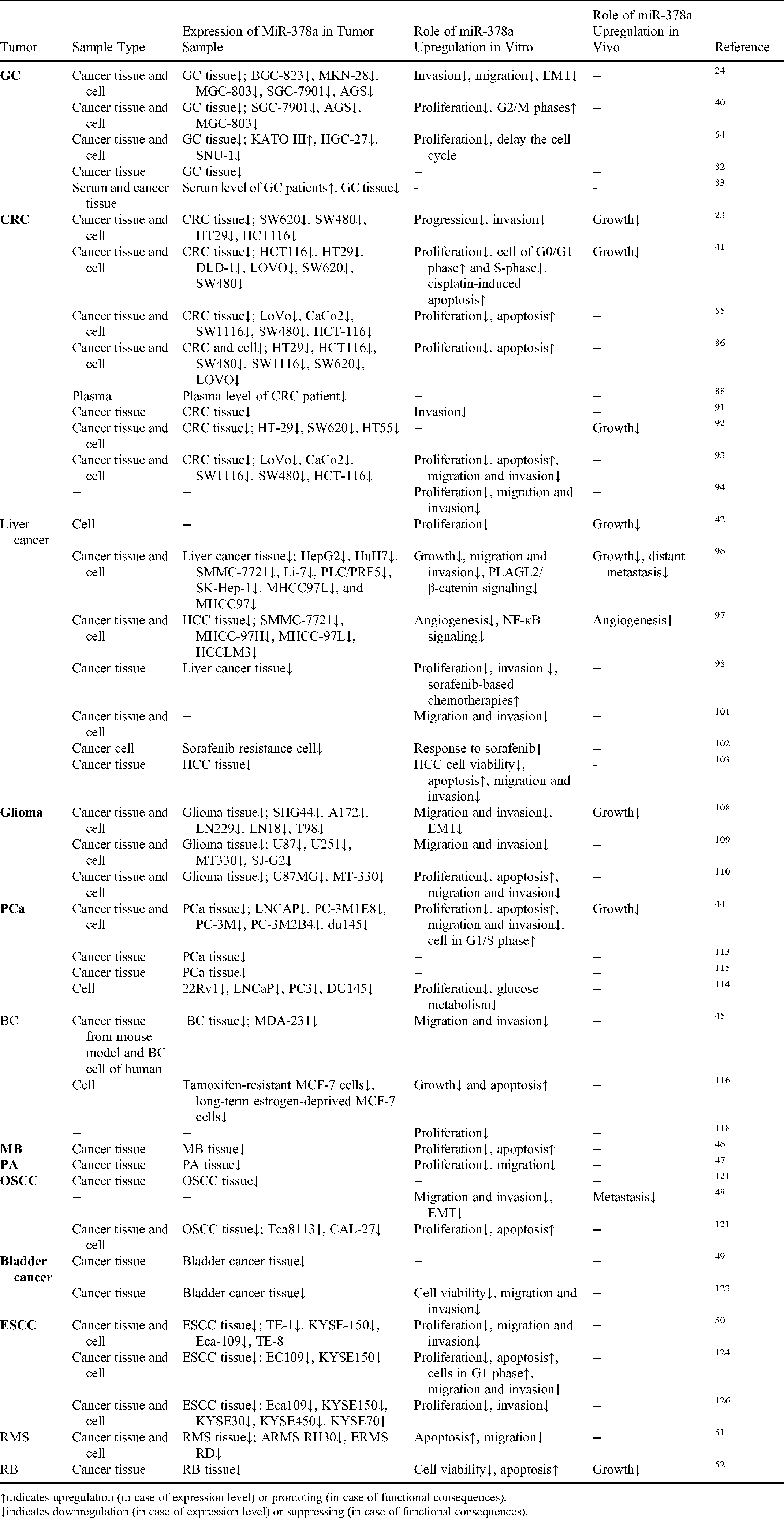
miR-378a Plays Inhibiting Roles by Acting as a Tumor Suppressor.
↑indicates upregulation (in case of expression level) or promoting (in case of functional consequences).
↓indicates downregulation (in case of expression level) or suppressing (in case of functional consequences).
Dual Function of miR-378a in Cancer Development
Growing research studies have elucidated the critical role of miR-378a in cancer progression, and most studies focused on the specific targets of miR-378a in tumor cell proliferation, migration, invasion, and regulatory pathways. Here, we summarize the roles and targets of miR-378a in tumorigenesis. Interestingly, both oncogenic (Table 1) and tumor-suppressive (Table 2) roles have been reported for miR-378a, implicating its dual function in cancer progression. By regulating downstream target genes, miR-378a promotes (Table 3) or inhibits tumors (Table 4), while it is regulated by its upstream regulators (Figure 1).

The regulatory factors upstream of miR-378a and the downstream target genes regulated by miR-378a in human cancer. miR-378a can be targeted and regulated by lncRNAs and circRNAs, thereby affecting its downstream gene expression and playing an important role in tumors.
miR-378a Functions as an Onco-miRNA in Cancers by Regulating the Downstream Targets.

↑indicates upregulation; ↓indicates downregulation.
miR-378a Functions as a Suppressor in Various Cancers by Regulating the Downstream Targets.

↑indicates upregulation; ↓indicates downregulation
Upregulation of miR-378a Promotes Malignant Transformation and Progression
Lung Cancer
Lung cancer is a common malignant tumor with a high mortality rate. 56 Despite advances in cancer therapy, the OS rate of patients with advanced lung cancer is low. The expression of miR-378a both in tumor tissues and cell lines was significantly higher than in adjacent normal tissues or cells, and upregulation of miR-378a promoted the tumor progression.22,25,27-29 Further, miR-378a was significantly overexpressed in serum exosomes of non-small cell lung cancer (NSCLC) patients. The aberrant expression of miR-378a was closely associated with an advanced TNM stage, positive metastasis, negative therapeutic response, and poor OS.25,57 miR-378a can promote NSCLC cell migration, invasion, and tumor angiogenesis.22,27 Moreover, the circular RNA has-circ-0007059 was identified as an upstream regulatory factor of miR-378a, which can inhibit the proliferation and epithelial-mesenchymal transition (EMT) of A549 and H1975 cells via inhibiting miR-378a. 58 The lncRNA ACTA2-AS1 suppressed malignancy by decoying miR-378a-3p, thus enhancing SOX7 expression. 59 As for the downstream regulation, miR-378a works on different targets and pathways to produce a plethora of effects on lung cancer progression. Studies on stimulating miR-378a for promoting lung cancer progression and constructing miR-378a overexpression cell lines have shown that miR-378a can strengthen metastasis by regulating EMT. 27 Further, miR-378a targets RBX1, and miR-378 upregulation boosts NSCLC cell invasion by downregulating RBX1, but it did not have any major effect on the angiogenesis signaling pathway. Likewise, HMOX1 was another target of miR-378a, and the interplay between HMOX1 and miR-378a 29 modulated the miRNA transcriptome and the 3′ UTR of HMOX1 in tumors. Further experiments in NCI-H292 cells with stable overexpression of miR-378a indicated that miR-378a enhanced the proliferation, migration, and angiogenic capabilities of the cells both in vitro and in vivo by downregulating HMOX1 mRNA and protein expression. However, when HMOX1 was diminished, miR-378a could not modulate the cell's resistance to chemotherapeutic drugs and oxidative stress. Another study noted a reverse correlation between miR-378a and FOXG1. 28 Silencing FOXG1 led to tumor suppression in NSCLC cells by downregulating miR-378a, thereby inhibiting NSCLC cell proliferation, promoting apoptosis, and increasing the length of the G0/G1 phase in the cell cycle. Hence, all the above studies show a positive relation between miR-378a and tumorigenesis.
However, some studies have shown the opposite result. Cisplatin (CDDP)-sensitive patients have high expression of miR-378a and low expression of small cell lung cancer (SCLU). 25 Forced upregulation of miR-378a inhibits neoplasm growth and SCLU expression and makes cells more sensitive to CDDP. Overexpression of miR-378a-3p can inhibit cell proliferation and decrease the expression of proliferation-related proteins CDK4 and CDK6. Since lncRNA OIP5-AS1 functioned as a competing endogenous RNA of miR-378a-3p, overexpressed wild-type OIP5-AS1 increased CDK4 and CDK6 expression, thereby promoting tumor growth. 60 In addition, SCP inhibits tumorigenesis by upregulating TUSC2 by targeting miR-378a-5p. 61 Thus, the aberrant miR-378a expression in lung cancer significantly affects tumors. However, the controversy over whether it is the low or high expression of miR-378a that increases lung cancer progression is yet to be resolved.
Gynecological Cancer
Ovarian cancer (OC) and cervical cancer (CC) are the most common gynecological tumors. In OC and CC, miR-378a is overexpressed both in the tumor tissues and cancer cell lines.31,33 Among recurrent OC patients treated with bevacizumab, higher miR-378a correlated to longer PFS. miR-378a and its downstream targets ALCAM and EHD1 can be used as markers for anti-angiogenic therapy. 62 In OC tissues and cells, it has been shown that circATRNL1 is under-expressed while miR-378a is overexpressed. 32 Further study revealed that circATRNL1 could adhere to miR-378a while miR-378a directly acts on SMAD4. High circATRNL1 expression also reduces miR-378a, which, in turn, inhibits SMAD4 expression, resulting in the suppression of angiogenesis, cell proliferation, invasion, and migration, both in vitro and in vivo. Hence, in OC, circATRNL1 acts as a miR-378a sponge, thereby accelerating SMAD4 signaling and attenuating angiogenesis and metastasis. 32
miR-378a is also a target of circ-LOPD2. circ-LOPD2 expression is high in both OC tissue and cells and is responsible for promoting OC cell proliferation. 63 Several studies have revealed that circ-RNAs worked as molecule sponges of miR-378a to regulate cancer development.
As for CC, miR-378a acts as an oncogene since high miR-378a expression significantly enhances cancer migration and invasion in vitro, and metastasis in vivo, while downregulating miR-378a produces the opposite effect in vitro. 33 The lncRNA LINC00641 inhibits CC progression by diminishing miR-378a-3p. Further, knockout of CPEB3, a downstream target of miR-378a-3p, can reverse the effects induced by LINC00641 overexpression. 64 Likewise, ATG12 targets miR-378a and their expression is inversely correlated, thereby promoting tumor cell progression. 33 Moreover, overexpressed miR-378a facilitates cell cycle progression, reduces apoptosis, and promotes cell growth by attenuating ST7L expression. miR-378a upregulation also activates the Wnt/β-catenin pathway to regulate tumor progression. 65
However, miR-378a-3p is lower in the serum and tissues of CC patients than in the serum and tissues of healthy control subjects and normal tissues, leading to the poor prognosis of patients with CC. 26 In this study, high miR-378a-3p expression reduced the proliferation and migration of CC cells both in vitro and in vivo. In short, miR-378a functions as an onco-miRNA by being regulated by circ-RNAs and then regulating its downstream targets and pathways, which is crucial in the malignancy of gynecological tumors. However, due to some inconsistent results, more studies are needed to elucidate the exact function of miR-378a in gynecological tumors.
Melanoma
Melanoma is the most aggressive type of skin cancer with high mortality. 66 The miRNA miR-378a shows the highest upregulation in advanced melanoma patients and those with lymph node metastasis 35 and miR-378a-5p is upregulated in metastatic melanoma patients, especially those resistant to targeted therapy. High miR-378a-5p expression enhances cell invasion, migration, and activates EMT in melanoma cells, and promotes angiogenesis. Further, several studies implementing various approaches have demonstrated that miR-378a-5p plays a carcinogenic role by regulating downstream targets such as SUFU, FUS-1, KLF9, STAMBP, and HOXD10. 67 However, when miR-378a targets the 3′ UTR of FOXN3, it mitigates the stimulation of miR-378a for proliferation, migration, and invasion. Moreover, FOXN3 interacts with β-catenin to downregulate the Wnt/β-catenin signaling proteins, 53 and this pathway is crucial for cell proliferation, self-regeneration, differentiation, and tissue homeostasis.68-70
Other Cancers
Apart from the positive function of miR-378a in the tumors mentioned above, similar results have been reported in nasopharyngeal carcinoma (NPC), osteosarcoma, cholangiocarcinoma, renal carcinoma, Burkitt lymphoma (BL), acute lymphoblastic leukemia (AML), and chronic lymphoblastic leukemia (CML). In NPC, miR-378a is upregulated in NPC tissues and cell lines. 34 Elevating the expression of miR-378a dramatically promotes the capability of NPC to proliferate, migrate, and invade in vitro, as well as grow in vivo by restraining the expression of the TOB2 transducer. TOB2 is a potential tumor suppressor which inhibits cell proliferation by arresting the progression of the G0/G1 phase cells to the S phase. 71 In osteosarcoma cells and patient-derived tumor specimens, overexpression of miR-378a promotes osteosarcoma cell proliferation by diminishing the levels of KLF9. Forced upregulation of KLF9 extraneously reverses the tumor proliferation-promoting effect of miR-378a. 36 miR-378a-3p upregulation can repress cell invasion and suppress EMT by suppressing BYSL. 72
Elevated miR-378a exerts a similar promoting effect in cholangiocarcinoma, which is significantly associated with an advanced TNM stage, positive lymph node metastasis, and short OS, and a miR-378a knockout inhibits cell proliferation, migration, and invasion. 37 Similarly, miR-378a was identified as a diagnostic biomarker when renal carcinoma cancer tissues and cells showed increased proliferation,73-76 and increased miR-378a inhibited POLR2A and RUNX2 expression and subsequently promoted cell apoptosis. 77
In BL, miR-378a-3p can facilitate tumor growth by targeting IRAK4 and MNT. 39 miR-378a dysregulation is also detected in non-solid tumors, such as AML and CML. The upregulation of miR-378a is positively correlated with poor survival in AML patients. 38 The 5′ flanking region of miR-378a is hypomethylated in AML. Thus, miR-378a can be reactivated by demethylation after 5-aza-dC treatment, but this is unlikely to provide helpful prognostic information in AML patients. 78 Knockdown of LINC00641 inhibited cell malignancy and promoted apoptosis by downregulating miR-378a by promoting ZBTB20 expression in AML. 79 Besides, lower FUS-1 expression, whose expression inversely correlates with miR-378a, is linked to the poor prognosis of AML patients. 80 In CML, enhanced expression of miR-378a in leukemia cell line K562 facilitates cell proliferation, clonality, and drug resistance to 5-FU, boosts expression of stem cell self-renewal markers OCT4 and c-Myc, and suppresses apoptosis. 21 As in AML, FUS-1 was also identified as a target of miR-378a in CML, but its role has not been elucidated yet. 21 Together, miR-378a overexpression exerts a stimulative effect on tumors mentioned above by regulating its different targets.
Downregulation of miR-378a in Several Malignancies Suggestive of its Antitumorigenic Roles
Unlike the cancers overexpressing miR-378a, it is worth noting that miR-378a is downregulated in many cancers. An inverse relationship between miR-378a expression and tumors progression has been reported in various carcinomas, including gastric cancer (GC), colorectal cancer (CRC), liver cancer, glioma, prostate cancer (PCa), BC, MB, pituitary cancer, oral squamous carcinoma (OSCC), bladder cancer, esophageal carcinoma (ESCC), RMS, and RB.
Gastric Cancer
GC has a high incidence worldwide and severely affects the quality of life. 81 Several studies have explored the underlying molecular alterations of miR-378a mediating tumorigenesis and GC development. A marked reduction of miR-378a was detected in GC tissues and cells compared to their controls. 24 Underexpression of miR-378a led to poor prognosis and malignant clinicopathologic features in GC patients. Circulating miR-378a level is considered the best biomarker for GC detection since it has a higher sensitivity and specificity than that of the serum levels of CEA and CA199.82,83 Low miR-378a expression in GC patients is also associated with an advanced TNM stage, poor tumor differentiation, high lymph node metastasis rate, and poor OS and DFS. Further, GC cells are arrested in the G0/G1 or G2/M phase after miR-378 overexpression. 40
miR-378a regulates various targets to exert its function as a tumor suppressor. For example, miR-378a targets BMP2, which has a high expression in GC patients with a shorter OS and DFS. Reverse-validation experiment confirmed that miR-378a overexpression inhibits invasion, migration, and EMT of GC by modulating BMP2, suggesting that components of the miR-378a/BMP2 axis might be potential therapeutic targets of GC. 24 Additionally, two other targets of miR-378a in GC—MAPK1 and VEGF—are closely associated with cancer malignancy. miR-378 inhibits MAPK1 in GC. 84 Depletion of MAPK1 by RNA interference in MC-803 cells reduces cell growth, increases apoptosis, and suppresses cell migration and invasion, which corresponded to the tumor-inhibiting effects caused by miR-378a restoration. Furthermore, miR-378a can directly bind to GAPLINC and reduce its expression, thereby repressing MAPK1 expression. 54 Upregulated miR-378a suppresses MAPK1 expression, cell proliferation, and cell cycle progression in GC cells, while these effects are reversed by increasing the expression of the lncRNA GAPLINC. Another miR-378 target in GC is VEGF, 85 as confirmed by the evidence that VEGF is downregulated by exogenously overexpressed miR-378. 40 Together, these findings suggest that miR-378 regulates GC development and can act as a promising biomarker for the treatment of GC patients.
Colorectal Cancer
Decreased miR-378a expression significantly correlates with shorter OS in CRC patients.23,86 Hence, miR-378a is a potential biomarker for the early detection and diagnosis of patients with CRC.23,87,88 Interestingly, miR-378a might also function as a novel biomarker to predict the efficacy of vaccines against CRC.89,90 The overexpression of miR-378a in CRC repressed cell growth, 23 induced apoptosis, 86 and inhibited migration and invasion by restraining EMT. 55 Studies also showed that miR-378a mitigated the malignant phenotypes of CRC cells by inhibiting the Wnt/ β-catenin pathway. 91
Two lncRNAs were found to regulate miR-378a-3p. SP1-activated LINC00339 decreased miR-378a-3p but enhanced MED19 expression, contributing to cell proliferation, cell cycle progression, migration, and invasion in CRC cells. 91 Further, the lncRNA CYTOR decreased miR-378a-5p and led to the increase in SERPINE1, which inhibited L-OHP resistance and EMT. 92
Several other genes are the downstream targets of miR-378a, including IGF1R, CDC40, vimentin, BRAF, SDAD1, and KISS1. IGF1R protein expression significantly negatively correlated with miR-378a in CRC tissues. 86 Further, luciferase reporter assays confirmed CDC40 as a direct target of miR-378a. 41 miR-378a represses cell growth and G1/S transition in CRC cells, and consistent with the inhibitory effect of miR-378a on tumor growth, CDC40 knockdown reduces proliferation and represses cell cycle progression by regulating G1/S and G2/M phases and pre-mRNA splicing.
High miR-378a expression restrains CRC cell growth and invasion and downregulates vimentin while silencing miR-378a produces the inverse effects. 23 miR-378 diametrically targets the 3′ UTR of vimentin to inhibit tumor progression. Similarly, BRAF 55 and SDAD1 93 were significantly reduced by miR-378a-5p overexpression in CRC. SDAD1 expression increased upon blocking miR-378a, suggesting that miR-378a and SDAD1 have a direct but negative regulatory relationship. Further, the expression of KiSS1 is enhanced in CRC cells with miR-378a elevation, leading to decreased proliferation, migration, and invasion abilities of the cancer cells. 94
Liver Cancer
Low expression of miR-378a95-97 is related to a poor prognosis of liver cancer. 97 Enhanced miR-378a expression inhibits the proliferation and invasion 98 but increases the apoptosis 99 in liver cancer. Moreover, miR-378a elevated the sensitivity of sorafenib treatment by targeting VEGFR, PDGFRβ, and c-Raf. 98
miR-378a also enhances hepatocellular carcinoma (HCC) development by downregulating FUS expression, 100 while miR-378a-5p targets VEGF. 101 Moreover, miR-378a targets TRAF1 and weakens NF-κB signaling, consequently downregulating VEGF. 97 IGF1R was also identified as a novel target of miR-378a in sorafenib-resistant HCC cells, with high miR-378a expression activating the PI3K/AKT and the Ras/Raf/MAP kinase pathways to mediate cell survival 102 and IGF1R knockout significantly restrained the miR-378a-mediated sorafenib resistance. miR-378a also suppresses HCC tumorigenesis by downregulating PD-L1 and STAT3 expression. 103 Moreover, the overexpressed miR-378a suppressed HCC cell proliferation by arresting G2/M and inhibited tumor growth in vivo. 42 Interestingly, in HCC, circCRIM1 acts as an upstream regulator of miR-378a that upregulates SKP2 by acting as a sponge for miR-378a and facilitates cell proliferation, angiogenesis, and the transition from the G1 to the S phase in the cell cycle. 104
Glioma
Even with the advancement of therapeutic strategies, glioma still shows a poor prognosis due to its high tumor aggressiveness and unlimited proliferation.105,106 Serval clinical studies in glioma have shown that miR-378a is remarkably decreased in cancer tissue and can potentially act as a diagnostic biomarker.107-109 Glioma patients with lower miR-378a expression showed poorer OS, and miR-378a overexpression restrained cell migration and invasion. 109 Downregulated miR-378a increased the target IRG1 expression, which enhanced glioma cell growth, invasion, migration, and EMT. 108 Another study showed that TSPAN17 was a direct target of miR-378a and correlated with poor prognosis in glioblastoma patients. Overexpressed miR-378a weakened TSPAN17 expression, consequently promoting apoptosis and reducing proliferation, migration, and invasion. These effects were attenuated in rescue experiments increasing TSPAN17 expression. 110 Interestingly, interfilamentous vimentin can also mediate miR-378a self-renewal by regulating SOX2 transcription factor expression. 111 Further, miR-378a promotes cell survival, tumor growth, and angiogenesis by targeting SUFU and FUS-1 expression. 112
Prostate Cancer
miR-378a is notably underexpressed in PCa tissues. 44 The reduction of miR-378a levels resulted in higher Gleason score, larger diameter tumors, and elevated serum prostate-specific antigen in PCa.113,114 miR-378a improved risk stratification based on the Gleason score or tumor stage and led to a higher risk of recurrence in patients with low miR-378a levels. Thus, miR-378a is a promising independent predictor of short-term recurrence in patients at high and very high risk of recurrence. 113
CircRNA PDHX boosts PCa progression by sponging miR-378a. 115 MiR-378a also inhibits prostate cell proliferation and glucose metabolism by repressing GLUT1. 114 Forced miR-378a overexpression represses prostate cancer cell migration and invasion but promotes cell apoptosis in vitro. 44 Further, studies have shown that KLK2, KLK4, 113 and MAPK1 are the targets of miR-378a in PCa, and the ectopic expression of MAPK1 rescues miR-378a-inhibited cell migration and invasion capacity. 44
Breast Cancer
Even in BC, miR-378a shows a low expression and correlates with the unfavorable prognosis of BC patients upon tamoxifen treatment. 116 GOLT1A is regulated by miR-378a and is critical for the mechanisms underlying BC endocrine resistance. 116 The m6A-mediated increase in LINC00958 expression enhanced tumorigenesis through the miR-378a/YY1 pathway. 117 Further, DCTPP1 is modulated by miR-378a, and decreased miR-378a expression elevates the DCTPP1 level, which facilitates cell proliferation by activating DNA repair signaling. 118
RUNX1 is significantly increased upon inhibition of miR-378a expression, which also enhances the invasion and migration of the triple-negative breast cancer (TNBC) cells. Further, the regulation of RUNX1 is mediated by the PPARGC1Β/miR-378a/RUNX1 regulatory pathway. 45 It has also been revealed that lncRNA GAS5 and miR-378a bind to each other, and the target of miR-378a-5p, SUFU, promotes GAS5-induced apoptosis of TNBC cells. This implicates that GAS5 stimulates apoptosis in TNBC cells by regulating miR-378a-5p/SUFU signaling. 119 Moreover, although no significant effect of miR-378a was found on KLK4 expression, both miR-378a and KLK4 represent unfavorable prognostic markers in TNBC patients. 120
Other Cancers
In addition to the above-mentioned tumors, decreased miR-378a expression has also been reported in other cancers. miR-378a is significantly reduced in MB. miR-378a negatively regulates UHRF1 expression in MB by binding to the 3′ UTR of its transcript. 46 Therefore, miR-378a overexpression inhibits UHRF1, but increased expression of UHRF1 reverses the miR-378a-induced suppression of cell proliferation and promotion of apoptosis.
In PA, RNF31 is highly expressed, and its expression is negatively regulated by miR-378a. 47 Knocking out RNF31 inhibits the proliferation and migration of the pituitary tumor cell line GH3. Similarly, miR-378a plays an inhibitory role in oral squamous cell carcinoma (OSCC). miR-378a is downregulated 121 in OSCC and also targets KLK4. 48 Overexpressed KLK4 reverses the miR-378a-induced suppression of migration and invasion by enhancing MMP-9, MMP-2, and N-cadherin expression while decreasing E-cadherin levels. CircRNA-100290 can also rescue the miR-378a-induced suppression of GLUT1 by functioning as a competing endogenous RNA (ceRNA), thereby accelerating cell proliferation and glycolysis in OSCC. 122 NCAPG, a target of miR-378a in OSCC, promotes cell proliferation and cell cycle progression but inhibits apoptosis by activating the GSK-3β/β-catenin signaling in OSCC. 123
In bladder cancer, miR-378a is downregulated and can act as an independent prognostic factor for the OS and recurrence. 49 LINC00958, an upstream regulator of miR-378, promotes the proliferation and metastasis of bladder cancer cells by increasing IGF1R, a downstream target of miR-378a, by sponging miR-378a. 124 miR-378a is lower in esophageal squamous cell carcinoma (ESCC) cells and tissues than in normal cells and adjacent tissues. Low miR-378a expression leads to an unfavorable prognosis and shorter OS in ESCC patients. Further, low miR-378a levels elevate cell proliferation, migration, and invasion. 50 However, miR-378a overexpression exerts the opposite effect by inhibiting Rab10. 125 Mechanistically, the lncRNAs SLC2A1-AS1 and LINC00514 are essential in ESCC progression by acting as miR-378a sponges. SLC2A1-AS1 inhibits miR-378a-5p by regulating glycolysis, thereby restoring GLUT1 expression and enhancing ESCC. 126 LINC00514, acting as a ceRNA, upregulates SPHK1 by sponging miR-378a-5p and activates adipogenesis-related pathways, thereby promoting the proliferation and invasion of ESCC cells. 127
In a RMS-derived cell line, miR-378a upregulation inhibited IGF1R expression and impacted phosphorylated-Akt protein levels. 51 Moreover, ectopic expression of miR-378a modulated apoptosis, cell migration, cytoskeleton organization, and the expression of the muscle markers MyoD1, MyoR, desmin, and MyHC in RMS. DNA demethylation by 5-aza-2 -deoxycytidine (5-aza-dC) also elevates miR-378a levels which correlate with increased apoptosis, cell viability reduction, and cell cycle arrest in G2-phase. 51 In RB, upregulated miR-378a-3p inhibits cell proliferation by targeting FOXG1. 52
The Role of miR-378a in Anti-Cancer Therapeutic Resistance
Unfortunately, cancer cells cumulatively get resistant to miscellaneous anti-tumor therapies, leading to disease recurrence and metastasis. Hence, understanding the internal molecular mechanism of cancer drug resistance is essential for finding new therapeutic approaches. Given that miR-378 is involved in various biological processes, the abnormal expression of miR-378a might be responsible for the anti-cancer resistance. For instance, miR-378a expression is preternatural in chemotherapy resistance in esophageal cancer cells. 128 Many studies have also shown miR-378a as a promising candidate for anti-tumor treatment. In lung cancer, elevated miR-378a recovered CDDP chemosensitivity by targeting SCLU and downregulating Bcl-2, pCas-3, p-Erk1/2, and p-Akt. 25 In ovarian cancer, low miR-378a expression can elevate the efficacy of bevacizumab treatment in patients with recurrent ovarian cancer, thereby lengthening PFS. Further, multivariate analysis showed miR-378a level as an efficient predictor of PFS after anti-angiogenic therapy. 62
In CRC cells with upregulated miR-378a, sensitivity to cetuximab was restored in all BRAF mutants and half of KRAS mutants, and lauric acid improved the sensitization of Cetuximab in KRAS/BRAF mutated CRC cells by regaining miR-378a expression. 129 Moreover, EPA-induced upregulation of miR-378 led to the significant restoration of sensitivity to cetuximab in the KRAS-mutant cells. 130 In glioma, the inhibitory effect of curcumin was enhanced in miR-378a-expressing stable U87 cells. 43 Likewise, increased miR-378a expression enhances radiation response by promoting radiation-induced TGD in glioblastoma cells. 107 However, reduced miR-378a expression was found in sorafenib-resistant HCC cells than in the sorafenib-sensitive group. Further, LXR stimulated miRNA-378a-3p transcription and hence can be used as a potential combinable treatment strategy with sorafenib to suppress HCC progression. 102 Metformin also induces miR-378a to downregulate CDK1, leading to suppression of cell proliferation and induction of the G2/M cell cycle arrest in HCC. 42 Moreover, the biologically active component isolated from the fungus Ganoderma lucidum can also overcome the drug resistance conferred by miR-378. 131
Conclusion
Cancer is the leading cause of death worldwide. The high mortality rate of cancers can be ascribed to its inefficient early detection, inherent or acquired therapeutic resistance, and poor predictive power of conventional screening techniques and clinicopathological parameters. Hence, searching for novel molecular targets is necessary to solve this problem.
The miRNA miR-378a is a promising biomarker and therapeutic target for cancers. As discussed in this paper, miR-378a expression is aberrant in more than 23 cancers and is responsible for affecting the cell's capability to proliferate, invade, metastasize, and resist multiple anti-tumor treatment regimens. However, there is still some controversy and ambiguity over its expression and roles in some cancers. miR-378a plays a dual role in tumors and can produce different impacts on different tumors based on whether it is overexpressed or under-expressed. The fact that it acts as an onco-miRNA makes it an attractive therapeutic target in lung cancer,22,25,27-29 CC, 33 OC,30-32 NPC, 34 melanoma, 35 osteosarcoma, 36 cholangiocarcinoma, 37 AML, 38 CML, 21 renal carcinoma,73-76 and BL. 39 However, it acts as a tumor suppressor in GC, 84 CRC , 41 liver cancer, 42 glioma, 43 PCa, 44 BC, 45 MB, 46 PA, 47 OSCC, 48 bladder cancer, 49 ESCC, 50 RMS, 51 and RB. 52 Notably, the expression and role of miR-378a are still controversial in malignant tumors, including lung cancer 60 and CC. 26 All these effects may be due to the different expression of the two strands of miR-378a and it is also due to the different specific expression of miR-378a in tissues and cells of different tumors. Further, the differences and limitations in the source of cell and tissue samples might be the key influencing factors.
The expression of circulating miR-378a can be used for cancer screening and diagnosis.22-24,31,33 The identification of the signaling pathways involved in cell proliferation, 34 migration, 35 invasion, 33 and apoptosis 41 also provides evidence for the different functions of miR-378a. miR-378a has also shown value as a therapy sensitizer 25 and drug target.25,131 All the results mentioned above might be due to the regulation of miR-378a by various factors, such as circRNA and lncRNA, and the interaction between miR-378a and its targets that constitute complex regulatory networks. Depending on the cellular environment, miR-378a might have various biological functions and clinical applications and can even be expressed differently and have varied roles in different tumors.
Because of the dual function of miR-378a in cancer development and whether targeting miR-378a is an effective and feasible approach for individual tumor patient-based treatment, more detailed investigations and understanding of the mechanisms underlying how miR-378a regulates tumor progression and therapy resistance are required in the future. Different analysis methods and intervention strategies for the role of the miRNA in different cancers are essential. Further, considering that miR-378a can effectively regulate the response of cancer cells to chemotherapy25,128 and radiotherapy, 43 a combination of miR-378a targeted therapy and chemotherapy or radiotherapy might achieve better therapeutic effects in some cancer types.
Thus, in this review, we provide insights into the rationale underlying the dual functions of miRNAs in tumor onset and progression. We show that miR-378 is critical for key biological and pathological processes via its complex network regulation mechanism in human cancers. In the future, miR-378 has great potential to become a clinically effective approach for cancer diagnosis and prognosis by refining cancer types and subtypes and an effective therapeutic strategy against cancer.
Footnotes
Abbreviations
Author Contributions
YQ and RL conceived the idea, YQ and LL performed literature retrieval, YQ conducted literature reading and sorting, YQ wrote initial article and revised the manuscript, RL provided helpful ideas and comments on this article, PL played an essential role in manuscript revision and responding to comments, XZ supervised the study. All the authors have read and approved the final version of this article. YQ and RL have contributed equally to this work as co-first author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Independent Project of Key Laboratory of Early Prevention and Treatment for Regional High-Incidence Tumor (grant number GKE-22202120).
