Abstract
Keywords
Introduction
Lung cancer is one of the most common types of cancer, with non-small cell lung cancer (NSCLC) accounting for 85% of lung malignancies. Approximately 25% of patients with NSCLC are diagnosed as locally advanced, typically defined as disease involving regional lymph nodes, and 20% of these patients are stage III. 1 According to guidelines, thoracic radiation with chemotherapy (CCRT) is recommended as the mainstay of treatment in patients with lung cancer. 2 Although survival has been prolonged through these treatments, 5-year local recurrences still occur in 28.9% of patients.3,4 This remains a major problem because recurrent tumors often cannot be resected. 5 In this case, although chemotherapy is recommended as the standard treatment for relapse, some patients cannot receive chemotherapy due to performance reasons. Unfortunately, response rates to salvage chemotherapy at relapse are still disappointing, with a median overall survival (OS) of only 10 to 12 months reported in the literature. 6
In very few series, second-series radiotherapy was used for recurrence.6–9 Second-series radiotherapy is challenging because of the adverse effects of the first series on the lungs and organs at risk (OAR).10–13 However, with the development of radiotherapy techniques such as stereotactic body radiotherapy (SABR), favorable distribution of an ablative dose to the tumor volume, optimal sparing of healthy tissues, and improved local control with low toxicity can be achieved.14,15 Despite these advances, the management of patients with recurrent lung cancer remains a major challenge, especially given the lack of data on toxicity. Adverse effects such as bleeding, radiation pneumonitis, esophagitis, and radiation-induced myelopathy are still concerns. In terms of radiation pneumonitis, SABR after the first series of external beam radiation therapy (EBRT) was investigated in the literature, particularly because these patients are prone to treatment complications due to preexisting comorbidities. 16 Despite all its possible adverse effects, reirradiation may play a more important role in the treatment of patients with recurrent lung cancer.
Although quite promising results in intrapulmonary metastases of lung cancer have been described, and even neighboring the radiotherapy area, 17 there are few analyses of reirradiation of extended volumes in the literature because patients with initial locally advanced lung cancer generally present with extensive tumor burden in the case of recurrent disease. This retrospective study analysis aimed to evaluate local control and disease-free survival (DFS), OS, and toxicity after curative doses of thoracic reirradiation with SABR for patients with locally recurrent lung cancer, and to identify the feasibility and tolerance of this treatment.
Materials and Method
Patient Eligibility
In our department, radiosurgery treatment techniques have been performed in various malignant and benign tumors using CyberKnife® and 4D respiratory tracking systems since 2011 and Varian® Linac devices since 2012. Of the patients who previously received simultaneous chemoradiotherapy due to stage III lung cancer or only radiotherapy due to poor performance and were not candidates for postrecurrence surgery, salvage therapies with SABR were administered. In this report, we included patients presenting with isolated primary lung recurrence within at least the 95% isodose of the previous SABR treatment.
The exclusion criteria included the presence of other malignancies and radiosurgery applications for primary disease outside the locoregional region. The initial treatments of all our patients were performed using 3D conformal, intensity-modulated radiation therapy (IMRT), or modulated arc therapy (VMAT) techniques. Treatment plans were evaluated by tumor boards, and radiosurgery was performed due to poor performance. Recurrences of patients were determined using [18F] fluorodeoxyglucose positron emission tomography–computed tomography (PET-CT) and/or computed tomography (CT) with a high predictive value (95%). 18 The Response Evaluation Criteria In Solid Tumors (RECISTs) were used as a general guideline, and recurrence was considered when the standardized uptake value (SUV) was greater than or equal to the pretreatment values with the use of PET/CT. 19
According to our tumor board, all 15 patients in the specified date range were determined as having recurrence through PET-CT and CT evaluations as described above. Of the 15 patients, 2 had a mediastinal recurrence, 4 had a hilar recurrence, 4 patients had an apical recurrence, and 5 patients’ recurrence was located far from all critical structures. As a result, our ultracentral radiotherapy rate is 40%. Primary tumor formations or intrapulmonary metastases were excluded; only patients who had previously received radiotherapy in the same field were included. Recurrences in which the geometric center of the mass was in the 80% to 90% isodose line were considered in-field enlarged tumor recurrence.
Disease in all patients was restaged according to the eighth (2018) edition of the American Joint Committee on Cancer (AJCC) Cancer Staging Manual. 20
Follow-up and Primary and Secondary Endpoints
Tumor response was evaluated using the RECIST system. 21 Acute and late toxicity was evaluated using the Common Terminology Criteria for Adverse Events (CTCAE) (Version 4.0). 22 According to our clinical protocol, if possible, all our patients were evaluated in the third month after SABR, and treatment evaluations were performed using PET-CT. The patients were first assessed through PET-CT or CT 3 months after the treatment. Until January 2020, for as long as they lived, all patients were evaluated using CT at least every 6 months and with PET-CT in cases in which a decision could not be made with CT.
The local control rate was accepted as the primary endpoint. Recurrence was accepted in cases in which the tumor diameter and its viable tumor in PET-CT SUV increased by 20% or more.
As secondary endpoints, DFS, OS, and toxicity were determined.
Radiation Therapy Specifications
Our patients were treated using the CyberKnife® (Accuracy Inc., Sunnyvale, CA, USA) radiosurgery system with 6-MV X-rays under a respiratory gating system and Xsight spine tracking system or treated on a Varian Trilogy linear accelerator platform under a perform gating with Respiratory Gating System (RPM) in patients treated with CyberKnife®, images in inhalation and exhalation were used to create gross tumor volume (GTV), and in 9 patients who underwent tumor monitoring using the Xsight spine system, the internal target volume (ITV) was created. It was supported using PET-CT fusion support for the creation of GTVs in all patients. GTVs accounting for respiration (IGTV) were created with the Xsight lung system in 2 patients and with a 4D RPM respiratory monitoring system in 4 patients, and they were treated with the Varian® Trilogy device. The planning target volume (PTV) was created as a 3 to 5-mm expansion of the ITV. Following our clinical protocol, we do not create the clinical target volume in any patients with recurrence or metastasis. The dose and fractionation were selected depending on the location of the tumor and the initial radiotherapy dose. Treatment plans were designed to ensure that 95% of the PTV received at least 100% of the prescribed dose, and 99% of the PTV was prescribed to receive 90% of the dose. The median SABR dose was 30.0 (R:22.5-45.0) Gy, and the median biologically effective dose at an α/β ratio of 10 (BED10) dose was 60 (R:39.38-85.0) Gy. The median fraction number was 3 (R:3-5). Cumulative dosimetry had to comply with the constraints of a 3- to 5-fraction plan for OAR according to the Quantitative Analyses of Normal Tissue Effects in the Clinic (QUANTEC): dose constraints. Accordingly, cumulative dosimetry had to obey spinal canal constraints (V23 < 0.03 cc), lung tissue constraints V5 < 65%, and lung V12.5 < 1500 cc and V13.5 < 1000 cc. Moreover, there was no maximal dose limit in the lungs, and the maximal dose had to be localized in the PTV of the reirradiated target volume. 23 Deformable image registration was used in all patients. When planning, OARs were prioritized compared with target volumes in the majority of cases. ALARA should be the prevailing principle. We used all available techniques to minimize the irradiated volume.
Although the biologically effective dose (BED) is suitable for converting doses to each other and ensuring equality, in practice we have achieved equality with the formula equivalent dose in 2 Gy (EQD2). The cumulative EQD2 was 116 Gy. If one year has passed since the first treatment (EBRT), cumulative OAR doses are calculated by reducing the EQD2 doses on the OAR by 50% and adding them over the SABR doses.
Statistical Design
All time-related events (failure or death) were calculated from the date of the first SABR to the date of death or censored at the last clinical follow-up and analyzed using Kaplan–Meier and Cox proportional hazard methods. The completion of SABR was used as time zero. Significance was considered at P < .05 and all significance levels were 2-sided. The IBM® SPSS® Statistics Version 25 software package was used for all statistical analyses. Univariate Cox regression analysis was conducted to identify clinical variables associated with end-points of interest, followed by multivariate models including all variables. In this study, local control and OS, whether the tumor diameter affected these, and the statistical significance of the PET-CT/CT responses in the third month was tested.
Results
Patients who were previously able to receive concomitant chemoradiotherapy due to stage III lung cancer (12 patients, 80%) or had poor Karnofsky Performance Scale (KPS) scores (3 patients, 20%) and local–regional recurrence were included in our study. One of these patients received radiotherapy in October 2013, the other 14 patients received radiotherapy between 2016 and 2017. All patients had tumor recurrence in the pretreated area (100%), and 3 also had mediastinal recurrence. Four patients who received chemotherapy but whose disease progressed under chemotherapy were given SABR treatment by the tumor board, and 6 patients (40%) had metastasis at the time of recurrence. The most common comorbid diseases were chronic obstructive pulmonary disease (COPD) in 10 patients (66.6%) and renal failure (6.6%). One patient had histologically verified renal cells as a second primary.
The median time between previous radiotherapy and SABR was 11 (IQR: 7-17) months. Local control was 8 (4-12) months in those with <12 months between radiotherapy and SABR, and 10 (0-24) in those with >12 months (P = .228). The OS was 14 (range: 8-20) months in those with <12 months between radiotherapy and SABR, and 13 (range: 0-27) (a patient died after the first fraction) months in those with > 12 months between radiotherapy and SABR (P = .763).
The median EBRT dose was 66 Gy/33 fraction (40 Gy/16 frx [only 1 patient] – all others were 60 Gy/30 frx-66 Gy/33 frx). The median reirradiation radiosurgery dose of BED10 60.0 Gy (IQR: 39.38-85.0) was given in 3 to 5 fractions. The pretreatment patient and tumor characteristics are presented in Table 1. Detailed patient doses, adverse events, and OS characteristics are given in Table 2. At the time of tumor recurrence, because our patients were generally those who were not found suitable for chemotherapy due to poor performance, only 4 patients received chemotherapy after treatment, one of whom had small cell cancer, and one received additional targeted therapy. One out of 4 patients receiving systemic chemotherapy had small cell lung cancer histology.
Patient and Treatment Characteristics.

Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography; NSCLC, non-small cell lung cancer; pathologies of patients whose adeno- or squamous subgroup differentiation was not pathologically performed were reported as general NSCLC. SCLC, small-cell lung cancer; BED10, biologically effective dose at an α/β ratio of 10; KPS, Karnofsky Performance Scale. As a result, 14 of the 15 patients had NSCLC and 1 had SCLC.
Dose, Adverse Events, and Overall Survival.
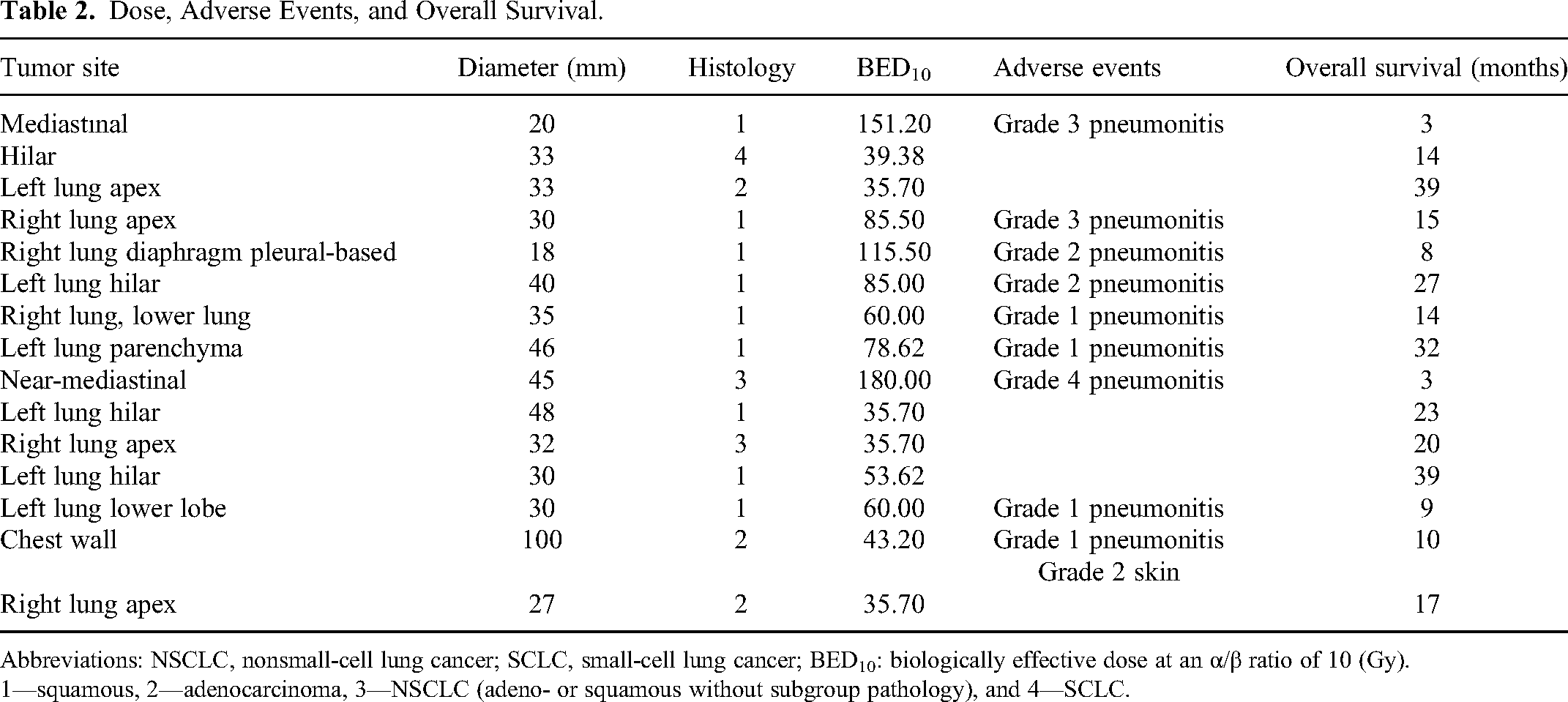
Abbreviations: NSCLC, nonsmall-cell lung cancer; SCLC, small-cell lung cancer; BED10: biologically effective dose at an α/β ratio of 10 (Gy).
1—squamous, 2—adenocarcinoma, 3—NSCLC (adeno- or squamous without subgroup pathology), and 4—SCLC.
The median follow-up period was 9 (IQR: 10-23) months. The median DFS was 8 (IQR: 6-20) months. The median OS was 14 (IQR: 10-23) months.
Treatment Information
Our median 60.0 Gy BED10 radiosurgery doses that we administered to our patients were between 35.70 and 180 Gy and in 3 to 5 fractions (IQR: 39.38-85.0). Our treatment doses were determined according to the previous doses of our patients, the location of the recurrence, and the time elapsed between both radiotherapy sessions.
Local Control
Patients were examined in detail in terms of local control, survival rates, and causes of death. Local control was achieved in 13 (86.6%) patients as detected with PET-CT in the third month, but could not be achieved in 2 patients. One patient died of local progression (10 cm chest wall recurrence), and the other had small-cell lung cancer histology and developed local regional and distant metastasis after salvage reirradiation.
However, 2 patients who could not be locally controlled were those receiving below BED10 60 Gy (median dose), and 2 patients who developed local progression later (one patient with small-cell brain metastasis) were given a dose below BED10 60 Gy. Considering a BED10 of 60 Gy, the local control rate was 100% during the follow-up period in 8 patients. On the other hand, among 7 patients who received a dose below 60 Gy BED10, there were a total of 4 local progressions (57% local failure), including 2 patients for whom local control could not be achieved at the beginning and 2 patients who subsequently developed progression (43% local control in those receiving below 60 Gy). In our study, almost all local failure-related events were observed at doses below 50 Gy.
Disease-Free Survival and Overall Survival
The median DFS was 8 (95% CI: 6.6-1.38) months, the mean DFS was 14.93 (95% CI: 7.8-21.9) months, and the median OS was 14 (95% CI: 11.47-16.5) months (Figures 1 and 2). The median survival of patients receiving chemotherapy was 14 (95% CI: 8.9-19.1) months and that of patients not receiving chemotherapy was 17 (95% CI: 6.2-27.8) months (P = .239). BED10 and 50 Gy were evaluated in terms of local control and were considered statistically significant (P = .044). Nine patients who received doses of BED10 ≥ 50 Gy developed no local recurrence and 4 of 6 patients (66.6%) who received doses below this developed local recurrence (P = .044).

Disease-free survival (months).
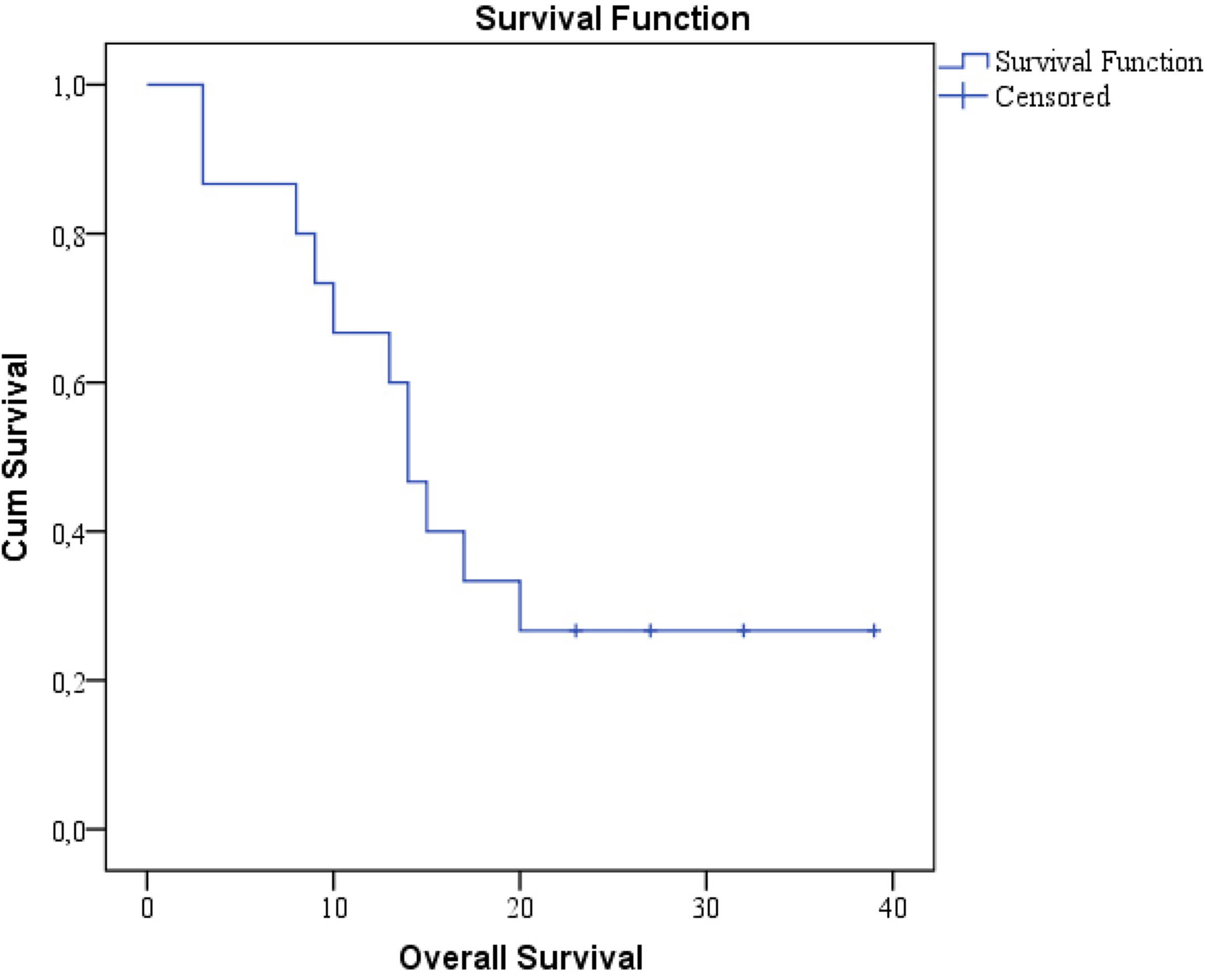
Overall survival (months).
The median OS was 17 (95% CI: 9.3-24.7) months in patients receiving BED10 below 60 Gy and 9 months (95% CI: 0.7-17.3) in those receiving BED10 over 60 Gy (P = .438). The median DFS was found as 8 months in patients both below and over BED10, 60 Gy. In a patient with SCLC histology, disease-free survival was calculated as 2 months and OS was calculated as 14 months. On the basis of this patient, these results showed parallelism with the patients we included in our study, and did not make any difference in our results.
Latest Status of Patients and Causes of Death of Recurrence After SABR Including Other Therapies
Two patients who received BED10 ≥ 60 Gy and 3 patients who received doses below are still alive (26%). The first and second-year survival rates were 66.6% and 33.3%, respectively. One of these patients received a dose below 60 Gy and developed local recurrence. Two patients who received a BED10 ≥ 60 Gy dose died, 1 in the second month (lung pneumonia findings after brain metastasectomy) and the other in the third month of brain metastasis that developed 1 and 2 months after the treatment.
Two patients died in the eighth and 15th months due to complications of chemotherapy administered because of the progression of disease in the seventh and eighth months. In one patient (40 Gy/2.5 Gy/16 frx first doses), local control was achieved with PET-CT in the third month, but pleural effusion associated with local progression developed in the sixth month and was taken under control through chemotherapy. Currently, this patient is still alive, and 1 of the 4 patients with local progression is alive. The other patient regressed in the second month, but progression was observed in this patient in the sixth month. This patient progressed even under chemotherapy and died. The third patient, who had small-cell lung cancer, developed concurrent local–regional and distant metastases. This patient died due to local and distant metastases. The patient with recurrence in the chest wall died of local progression. Two patients died due to COPD that existed before the treatment. Two patients died due to the continuation of their metastatic condition before SABR. One patient is still alive with the disease, and 4 patients are alive without the disease. In univariate analyses, the presence of tumors below 2 cm was found to have a significant effect on local control and overall survival (P < .05), but it was statistically borderline meaningless for regional control. In multivariate analyses, it was found that only tumors < 2 cm were effective for OS (P < .05). Local control, regional control, and survival uni-multivariate analyses are presented in Table 3. All patients are summarized in Table 4.
Local Control, Regional Control, and Survival Analysis.

Abbreviations: BED10, biologically effective dose at an α/β ratio of 10; SCC; squamous cell carcinoma; adeno, adenocarcinoma; PET/CT, positron emission tomography/computed tomography.
Summary of All Patients.

Brain metastasis developed in 3 patients who were not initially metastatic in the first, second, and seventh months.
Adverse Events
SABR was generally well tolerated by our patients and was easily applied. In terms of radiation pneumonia, 6 patients had grade 0 radiologic and clinical findings, 4 patients had grade 1, 2 patients had grade 2, and 2 patients had grade 3 dyspnea, hypoxia, cough, and radiologic pneumonitis findings, one of whom required oxygen support. Development of grade 4 pneumonitis requiring serious intervention in one patient.
Grade 2 skin findings (radiodermatitis) developed in one patient who had a 10-cm diameter tumor and underwent irradiation of the chest wall (Table 2
Discussion
Our main aim in this study was to investigate the limited treatment possibilities that can be used in cases of residual and recurrence in locally advanced lung cancers that have previously undergone thoracic radiotherapy, due to the general condition of these patients and the treatments they have received before. Typically the mechanism of death in NSCLC is a local failure or distant disease. In-field recurrence was found associated with the development of regional and distant failure, and finally poor OS. 24 The present retrospective cohort analysis investigated the effect of SABR on local control and survival outcomes of 15 patients with infield recurrent lung cancer who were previously treated with CCRT, and the results demonstrated that SABR was a useful and effective treatment that might represent a new salvage option for these inoperable patients with significant comorbidities.
In addition, a BED10 value of above 50 Gy was found to have prognostic significance in patients with lung cancer who had recurrence after CCRT and underwent SABR. In univariate analyses, tumor size below 2 cm was found to be effective for both local control and OS, and tumor diameter contributed to OS in multivariate analyses. We believe that this shows how important follow-up is in lung cancer and that early treatment has an effect on local control and even OS.
Despite the improved OS of the RTOG 0617 trial, local, regional, and locoregional failures remained common in standard dose (60 Gy) treatment (38.2%, 35.7%, and 49.7%, respectively). 25 For patients who have received definitive radiation therapy or chemoradiation, local recurrence is a therapeutic challenge because patients are often nonsurgical candidates and have limited other treatment options. However, in patients developing local recurrence, the only standard treatment remains chemotherapy or immunotherapy alone, with an overall poor prognosis. In this status, systemic chemotherapy is generally given with palliative intent, with a median OS of ≤ 10 months; < 30% of patients obtain an objective response. 26 Similarly, a second series of radiotherapies with low BED10 values can be given for palliation for these patients, who are locally uncontrollable and have a very low quality of life for the remainder of their lives. 27 In our study, we included all patients with in-field recurrence for whom it was attempted to achieve local control by using the dosimetric advantages of radiosurgery techniques. Despite being a small group, very good local control over a certain dose was provided for these patients, the vast majority of whom had poor KPS to receive chemotherapy. Although there was no significant difference in the OS of the patients who received doses above BED10 > 60 Gy, BED10 ≥ 50 Gy was found to be the critical dose threshold to provide prognostic significance in patients with lung cancer who developed recurrence after CCRT and underwent SABR (P < .044). It was thought that this could be due to adverse effects related to reirradition, and BED10 ≥ 50 Gy might be a confident dose threshold. Because in our study, almost all local failure-related events were observed at doses below 50 Gy. Although our local control rate is 100% in patients with BED10 > 60, the reason why our best survival results are not in 50 to 60 Gy patients and the reason why we lose our survival advantage over BED10 > 60 Gy: perhaps our rate of patients with ultracentral tumors (40%) is quite high compared to other high-dose studies. 28
Time from the first treatment to SABR shorter than or longer than 12 months did not achieve a statistically significant change in local control and OS in our study. After primary radiotherapy, our knowledge about the formation of radionecrosis and some other complications in radiosurgery applications is mostly based on second-series cranial irradiation studies. Depending on radiobiologic parameters, it is recommended that the interval between these studies be at least 1 year as a result of radionecrosis and treatment response evaluations after these studies.29,30 In the lung, although there are no specific data, 31 it is thought to be related to the radiation resistance of relapses developing before 1 year, but this was not supported in our study. This may be due to the small number of patients. This may not explain the difference between the results, however, because the fractionation/biology is changed as much as possible in patients with early relapse who decisively resisted the initial treatment plan. As it is known, conventional radiotherapy mostly creates DNA breaks indirectly, and radiosurgery treatments kill radioresistant hypoxic cells directly. 32
Considering all treated patients, our local control rates were lower than those of Kelly et al 7 because our mean local doses were relatively low. However, in their study, more adverse effects were detected after SABR than in our study, such as dyspnea in 50% (19% of patients required oxygen supplementation), chest wall pain in 30%, and grade 3 esophagitis in 8%. We detected only 2 patients who had grade 3 hypoxia, cough, and radiologic pneumonitis findings; one required oxygen support, and the other developed grade 4 pneumonitis requiring serious intervention. Different from other studies, in our study, the recurrence of all of our patients was within the first-series radiotherapy areas and had a relatively large volume, and we treated mediastinal (central and ultracentral) recurrent tumors neighboring the bronchial tree of recurrences, so a relatively low dose was used. Our patients experienced quite tolerable adverse effects with the second-series radiotherapy because we could not go up to high doses like for the primary; due to the short OS time, late adverse effects and local failures might not have occurred.
We found no difference in survival benefit between doses of BED10 < 60 Gy and BED10 > 60 Gy. The reason for this was thought to be the death of patients due to distant metastasis and chemotherapy toxicity. No patients experienced local recurrence at the time of death. In univariate analysis, BED10 ≥ 50 Gy was a significant factor; however, over BED10 60 Gy was not significant. There was no survival advantage with doses > BED10 60 Gy; this may be attributed to the fact that patients who had a relatively smaller tumor volume and better KPS in this patient group received higher doses of reirradiation dose, and some, mostly in this group, even received additional chemotherapy. Although our patient population is limited, the decrease in OS in our patients who were dosed over 60 Gy might also have been an adverse effect related to the dose increase; however, we do not have sufficient data to confirm this.
In univariate analyses, tumor size below 2 cm was found to be effective for both local control and OS, and tumor diameter contributed to OS in multivariate analyses. We believe that this shows how important follow-up is in lung cancer and that early treatment has an effect on local control and even OS.
Hearn et al used salvage treatment with SABR in 10 patients previously treated for early-stage NSCLC who underwent lung SABR. However, all our patients had locally advanced-stage disease, and 66 Gy (2 Gy × 33 standard fractionation) chemoradiotherapy was administered to the patients in our series. Recently, John et al 33 reported a retrospective multicenter study on patients (n = 27, 12 patients with NSCLC) treated with 2 courses of lung SABR. The published one-year local control rate was 78.3%, and no grade > 2 toxicity was observed. However, the reported applied doses were lower than those of our cohort.
The recommendation of radiotherapy treatment in central tumors, especially in central relapses, is avoided in the literature. 31 Grade 5 toxicity rates in this region have been reported to be up to 21%. However, in our study, although the majority of our cases were central region and even ultra-central region recurrences, we did not encounter any fatal toxicity. The main reason why large vessel rupture, airway toxicity (necrosis, rupture, and fistula), or fatal cardiac toxicities did not occur in our study may be considered related to the lower doses used compared with other studies.28,34 For this reason, dose constraints should be strictly adhered to. Even in the first radiotherapy application, there is a possibility that the patient may relapse. The ALARA principle should be adhered to.
This study has several limitations. First, it is a retrospective study of a varied patient population. Second, the number of patients was small, and the factors not found to be significantly associated with the outcome here could be important in a larger population. Additionally, the sample was a heterogeneous group of patients, and the treatments were performed with palliative intent. Heterogeneity in terms of histology, stages and initial treatment, and the definition of SABR, which is both “radioresistant” and “radiosensitive” histologies, were included here and some of the regimens used comprised a relatively low dose compared with some other SABR reirradiation treatments. Also, 6 (40%) patients had concurrent metastasis and local recurrence. In terms of toxicity and survival, to prevent bias, reirradiation of the same lesion in the lung in all cases has been tried to be given here. As a result, our results may be even better than they seem because the patients had a worse stage, which may mean that we cannot give a reliable conclusion. However, it could be considered very successful in terms of local control because poor KPS shortens the time of hospital visits in such patients compared with other limited SABR studies. These parameters can affect tumor control and toxicity. Moreover, if the doses are kept as moderate, as in our study, considering the location of the tumor and previous OARs, rates of 20% for toxicity of grade 3 and above, seem quite reasonable.10,11 One patient who developed grade 3 toxicity was given BED10 85.5 Gy, and another was given 151.2 Gy. The patient who developed grade 4 toxicity received BED10 180 Gy. Grade 1 radiation pneumonia developed in only one patient among those who received doses of BED10 60 Gy and below. It was thought that the reason why our pneumonitis rates were low was due to the high-dose radiotherapy they received in the previous treatment (median 66 Gy) because these areas were already nonfunctional. As such, this may be a better value than in conventional radiotherapies.
Conclusions
In our multidisciplinary tumor boards, palliative radiotherapy is often recommended in our daily practice when local control is required and the patient displays progression under chemotherapy or chemotherapy is not enough due to poor KPS. In residual and recurrent tumors, the application of additional doses such as BED10 50 to 60 Gy may benefit local control. SABR is an important and relatively safe treatment option in small local recurrences.
Footnotes
Data Availability Statement
Data are available on request from the authors. Data are available on request due to privacy or other restrictions. Data are subject to third-party restrictions. Authors can confirm that all relevant data are included in the article and/or its supplementary information files. Data sharing is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethics committee approval was received for this study from the Ethics Committee of İstanbul Prof Dr Cemil Taşçıoğlu Şehir Hastanesi (approval number: 48670771-514.99 decision number 381, Date: 08.11.2021). This article complies with the Ethical Principles for Medical Research Involving Human Subjects of the World Medical Association Declaration of Helsinki. All included patients provided written informed consent for the treatment of their medical details. The patients were not required to give informed consent for this study because the study utilized the anonymous retrospective data obtained after each patient accepted the treatment by written consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
