Abstract
Keywords
Introduction
Portal vein embolization (PVE), first described by Makuuchi et al. 1 is a procedure that causes hypertrophy of the future liver remnant (FLR) in patients anticipated for extensive hepatectomy with FLR that is too small to result in normal liver function. PVE is performed to expand the indications for liver resection and lower the risk of postoperative liver insufficiency. 2 Additionally, patients with massive hepatocellular carcinoma (HCC) not appropriate for surgery or regional ablative therapy can be effectively treated with PVE combined with transarterial chemoembolization (TACE) with lower morbidity and a promising survival benefit. 3
The main PVE approaches include trans-hepatic (including contralateral and ipsilateral), trans-ileocolic, 4 trans-splenic, and transjugular approaches. 5
However, patients undergoing PVE are more likely to have a potential hepatic disease, such as thrombocytopenia or coagulopathies, than the normal population. Therefore, in trans-hepatic PVE procedures, bleeding is the most common postoperative complication, occurring in approximately 2% to 4% of patients after PVE.6,7
The risk of hemorrhage is reduced when materials such as embolization fibrin glue,2,8 n-butyl cyanoacrylate (NBCA),9,10 gelatin sponge particles,,11,12 vascular plugs, 13 and coils14-17 are used to embolize the tract. These various embolic materials not only increase procedure expenditure but also increase operation difficulty and duration.
Based on our experience with hepatic puncture, we devised a delayed catheter removal technique to control bleeding. This study aimed to assess the feasibility and safety of the delayed catheter removal technique for trans-hepatic PVE.
Materials and Methods
From November 2014 to December 2019, in a total of 278 consecutive patients (median age, 53 years; range, 18-76 years), the access tract of the percutaneous trans-hepatic portal vein was treated using the delayed catheter removal technique after trans-hepatic PVE. The reasons for PVE included HCC in 159 patients, cholangiocellular carcinoma in 83, metastases of gastrointestinal origin in 9, other primary malignancies in 10, and benign disease in 17. This retrospective study was conducted in accordance with the principles of the Declaration of Helsinki, and the study protocol was approved by the ethics committee of our hospital. Owing to the retrospective nature of the study, patient consent for inclusion was waived. All patient details were de-identified.
All PVE procedures were performed under local anesthesia. When embolic materials included NBCA, additional pethidine was used before PVE. A suitable portal venous branch was punctured with an EV needle (Hakko, Nagano, Japan) under ultrasound (US) guidance. A 0.035-inch guidewire was inserted into the portal vein without a vascular sheath, and a standard polyethylene 5-Fr angiographic catheter (Hanaco Medical, Tianjin, China) was placed at the splenomesenteric confluence to perform portography. An embolic agent, such as NBCA/lipiodol mixture, polyvinyl alcohol particles, gelatin sponge, and/or coils, was used to embolize the portal vein.
The Delayed Catheter Removal Technique
As is shown in Figure 1, Figure 1A shows that a standard polyethylene 5-Fr angiographic catheter is placed at the splenomesenteric confluence to perform portography. Figure 1B shows that after embolizing the targeted portal vein, the tip of the 5-Fr angiographic catheter was retraced to the liver parenchyma near the portal vein puncture site under fluoroscopy. Figure 1C shows that the end of the 5-Fr catheter was cut short and then knotted to create a marker near the skin to prevent bleeding and stop the catheter from sliding around. First, the knot was a marker to monitor the displacement of the catheter. Second, the knot prevented the catheter from moving into the patient. Third, it was a good anchor point for fixation by medical tape to prevent the catheter from moving out and falling out. Perihepatic hematoma formation was evaluated using US shortly, which was on standby. The next day, the catheter was gradually removed. At the first and the second withdrawal, the catheter was pulled out approximately 1 to 2 cm, and at the third withdrawal, it was pulled out completely. The intervals between 2 withdrawals of the catheters were approximately 2 to 3 h. The operators, who were involved in the procedures, included Y. F. Yang, N. Ge, C. J. He, Y.J. Zhang, and J. Huang with 20, 14, 10, 9, and 9 years of experience in interventional procedures, respectively. Huang with 20, 14, 10, 9, and 9 years of experience in interventional procedures, respectively.
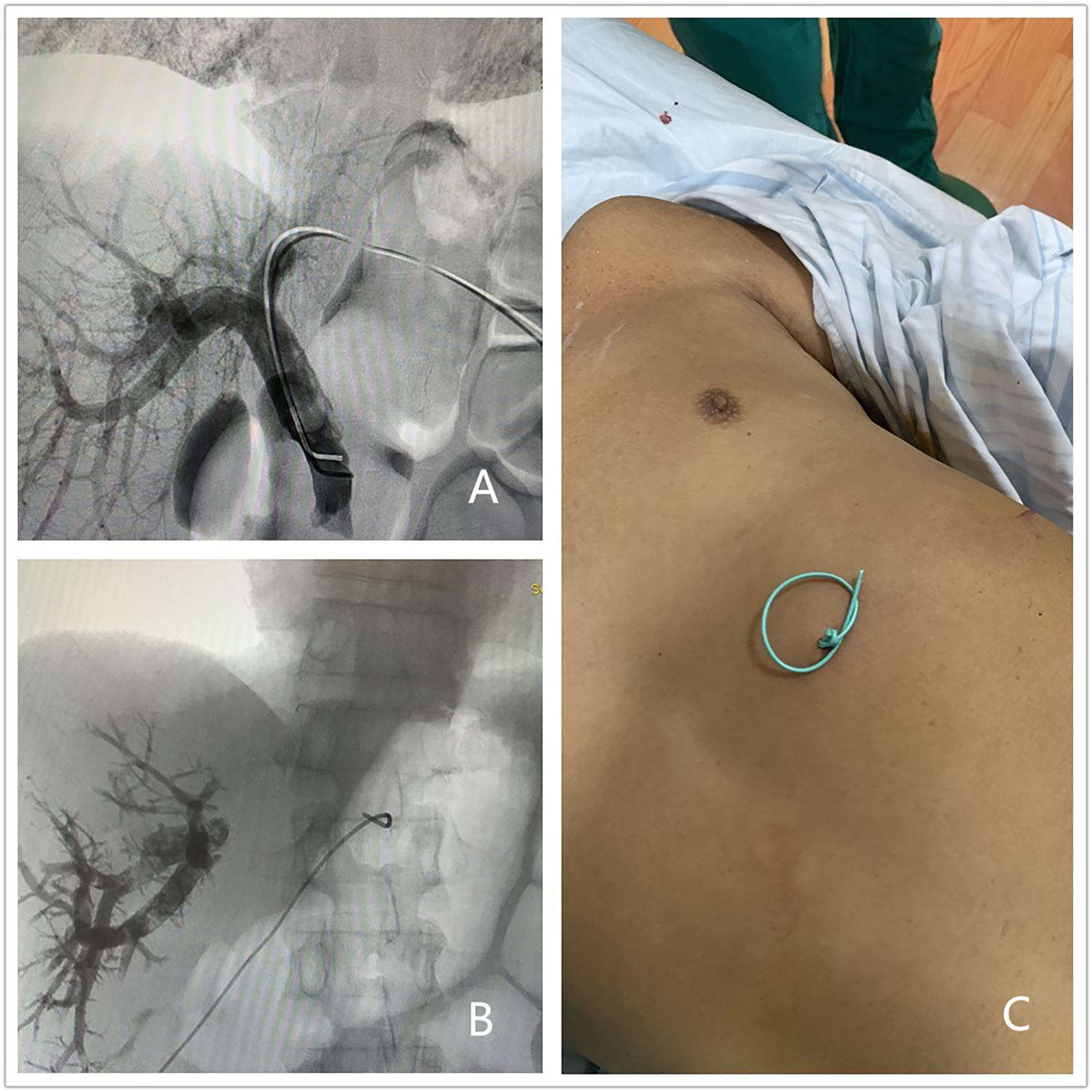
The delayed catheter removal technique. (A) A standard polyethylene 5-Fr angiographic catheter is placed at the splenomesenteric confluence to perform portography. (B) After trans-hepatic PVE, the tip of the 5-Fr catheter is retraced to liver parenchyma near the portal vein puncture site. (C) The end of the 5-Fr catheter is cut short and then knotted to create a marker near the skin to prevent bleeding and stop the catheter from sliding around.
Follow-up
Follow-up information was gathered to the point of the patient's surgery or up to the first follow-up after PVE. Before PVE, blood platelet count, prothrombin time (PT), activated partial thromboplastin time (aPTT), and international normalized ratio of each patient was pre-estimated to evaluate the possible existence of any potential bleeding disorder.
In patients who underwent follow-up examinations such as magnetic resonance imaging (MRI), computed tomography (CT), and/or US, the data were retrospectively reviewed to assess the existence of any complications. The results of baseline hemoglobin and hematocrit were examined before PVE, and follow-up results were examined 1 to 4 days after PVE, and then the comparison between the 2 results was performed to explore the existence of possible bleeding.
Reduction in hemoglobin and hematocrit levels by < 2 g/dL and 4%, respectively, was regarded as a slight decrease.
No hematoma on the penetrated liver surface on the instant US was regarded as a technical success. Major postoperative complications were defined as prolonged hospital stay due to procedure-related complications, massive hemorrhage, embolization of non-target portal vein, serious hepatic damage, perpetual adverse consequences, and demise. Slight ache or pyrexia, which could be treated or controlled with conservative treatment, was regarded as a minor complication. To evaluate these complications, medical records, laboratory findings, and imageological examinations such as CT, MRI, and/or US examination were reviewed.
Results
The trans-hepatic PVE procedure was successful in 278 patients, including 275 contralateral approach procedures and 3 ipsilateral approach procedures. A delayed catheter removal technique was successfully performed, and instant hemostasis was achieved in all cases. Instant US revealed no perihepatic hematoma in all patients, and the technical success rate was 100%.
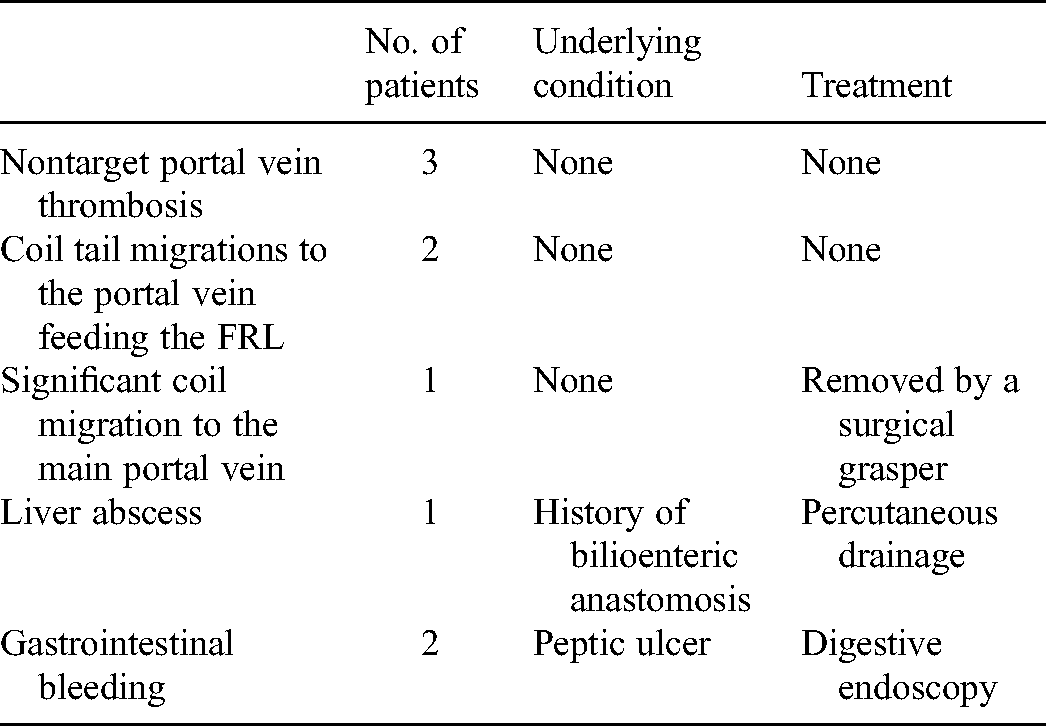
Major PVE-associated complications were found in 9 (3.24%) patients (Table 1). These included 2 coil tail migrations to the portal vein feeding the FLR, which did not hinder subsequent radical surgeries, 1 significant coil migration to the main portal vein that was successfully removed by a surgical grasper, 3 delayed nontarget portal vein thromboses that were found by follow-up CT, 2 gastrointestinal bleeding, and 1 liver abscess. The 2 gastrointestinal bleeding were due to peptic ulcer, which was verified by subsequent digestive endoscopy. The abscess was located in the right liver and was not located near the access site in the left liver. The patient had not received TACE therapy; however, 5 months previously, she had undergone choledochojejunostomy. So we believe the abscess was related to cholangitis. Seventy-eight patients complained of mild epigastric pain and/or fever after the procedure.
Major complications in 9 patients.
Sixty-eight patients had underlying bleeding conditions, showing a low blood platelet count and prolonged PT or aPTT. Follow-up blood examination, which was performed 1 to 4 days after the procedure, revealed that 9 patients had a decrease in hemoglobin and hematocrit levels of >2 g/dL and 4%, respectively. And among these 9 patients, 1 patient had a gastrointestinal hemorrhage. However, there was no patient with bleeding around the percutaneous puncture tract or hemoperitoneum owing to hemorrhage from the percutaneous trans-hepatic puncture tract of the portal vein on CT, MRI, and/or US examination.
PVE was performed to increase the volume of an initially insufficient FLR in 248 patients. Of these 248 patients, 190 (76.61%) underwent a successful surgery, while 58 (23.39%) patients did not undergo radical surgery, for disease progression, poor FLR hyperplasia, undetected metastatic tumor, or other reasons. In another 30 patients, PVE was performed for tumor treatment purposes.
Discussion
The main PVE approaches include trans-hepatic (including contralateral and ipsilateral), trans-ileocolic, trans-splenic, and transjugular approaches.
The first case of percutaneous splenoportography through the trans-splenic puncture tract was reported in 1951.18,19 Sarwar et al. 20 described the details of the PVE procedure through trans-splenic portal vein access. Trans-ileocolic PVE is invasive compared to trans-hepatic approach as the procedure requires general anesthesia. Additionally, a previously reported transjugular route is rarely used and can be proposed if it is impossible to perform trans-hepatic PVE because the access tract is obstructed by the tumor or impaired hemostasis. 21
According to the existing literature, most PVE procedures were performed using the trans-hepatic approach. However, patients undergoing PVE are more likely to have a potential hepatic disease, such as thrombocytopenia or coagulopathies, than the normal population. In the contralateral approach, the elevation of portal vein pressure after PVE increases the risk of bleeding from the portal vein. When performing percutaneous trans-hepatic procedures, hemorrhage is the most frequent complication in approximately 2% to 4% of patients.6,7 Occurring immediately or in a delayed manner, hemorrhage after percutaneous trans-hepatic procedures may present as subcapsular hematoma or hemoperitoneum. 22 It comes from several hemorrhage sources, including the intercostal and hepatic arteries and the portal and hepatic veins. The hemorrhage site is mostly located along the puncture tract.
Embolization of the percutaneous trans-hepatic access tracts of the portal vein to prevent hemorrhage has previously been reported. Various materials are utilized to embolize the access tracts after PVE, including fibrin glue,2,8 NBCA,9,10 gelatin sponge particles,11,12 vascular plugs, 13 and coils.14-17 The use of these various embolic materials will increase procedural expenditure, operation difficulty, and duration.
Based on our experience with hepatic puncture, we devised a delayed catheter removal technique to control bleeding. First, this technique is easy to perform. The tip of the 5-Fr catheter was retraced to the liver parenchyma near the portal vein puncture site, and then the end of the 5-Fr catheter (the procedure without the catheter sheath) was closed and fixed firmly, and was pulled out the next day. Second, compared with other embolic materials, this does not increase the cost. Third, if the hemorrhage is detected before pulling out the 5-Fr catheter, the percutaneous trans-hepatic portal venous access tract can be embolized by various embolic materials.
No perihepatic hematoma or hemoperitoneum, which has been frequently reported by other authors,6,7 was found in all patients. Therefore, it is anticipated that the delayed catheter removal technique can be safely and effectively utilized as a successful hemostasis as it is simple to use and free.
This study had some limitations. First, the delayed catheter removal technique was the sole method for embolizing the percutaneous trans-hepatic access tract of the portal vein; hence, there was no control group in this study to prove the advantage of the delayed catheter removal technique over the other embolization methods. Nevertheless, to the best of our knowledge, this is the first study to investigate the efficacy of the delayed catheter removal technique for use in trans-hepatic PVE access tracts. Second, this was a single-center, retrospective study. Third, in this study, we did not use a sheath. The sheath, which is larger than the catheter, could make a bigger access tract in FLR, which will make it challenging to deal with the access tract after PVE and increase damage to FLRs. However, many other centers performed PVE through a sheath rather than a 5-Fr catheter; therefore, this technique might not apply to cases with a larger diameter access site.
Conclusion
In conclusion, the delayed catheter removal technique is a free and relatively simple method that can be used as an alternate hemostasis technique in trans-hepatic PVE. This technique should be further evaluated and compared with other methods.
Footnotes
Abbreviations
Acknowledgments
No other than your co-authors is a significant contributor to the conduct of the study or preparation of the manuscript.
Declaration of Conflicting Interests
We declare that we have no financial and personal relationships with people or organizations that can inappropriately influence our work; there is no professional or other personal interest of any nature or kind in any product, service, and/or company that could be construed as influencing the position presented in, or the review of, the manuscript entitled.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This study is a retrospective study, which was conducted in accordance with the principles of the Declaration of Helsinki, and the study protocol was approved by the ethics committee of our hospital. Due to the retrospective nature of the study, informed consent was waived.
Data availability
The data that support the findings of this study are available on request from the corresponding author [YY]. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Animal research (Ethics)
Not applicable.
Consent to participate (Ethics)
Due to the retrospective nature of the study, informed consent was waived.
Consent to publish (Ethics)
Not applicable.
Plant reproducibility
Not applicable.
Clinical trials registration
Not applicable.
