Abstract
Introduction
Neuroblastoma is a common malignant tumor among infants and children, and the metastasis could possibly occur during childhood. 1 The dura mater metastasis of neuroblastoma is rare, but if it occurred, the tumor probably metastasizes to the entire dura mater around the brain.2–4 In some cases, the dura mater metastasis could be extensive, with diffuse involvement of the entire calvarium and skull base. This situation could be a great challenge in radiotherapy (RT) in delivering a homogeneous dose conformally to such a watermelon-peel-shaped target volume while sparing the whole brain and other organs at risk (OARs).
Given that neuroblastoma is a radiosensitive tumor, if the tumor metastasis only involved a limited area of the skull, the target could be easily irradiated with modern radiotherapy techniques. Many different radiation modalities and techniques have been performed for dosimetric discussion for the tumor involving the scalp or calvarium.5–11 IMRT could provide the best target-dose homogeneity and coverage among 3DCRT and HDR techniques in extensive scalp lesion irradiation. 5 RapidArc (RA) was selected as the optimal solution, with marginally better conformality, shorter delivery time, and lower monitor units than static IMRT for total dura irradiation. 12 For total scalp irradiation, the helical tomotherapy (HT) plan showed the best target coverage and conformity and low dose to the brain and the highest beam-on time and monitor units (MUs) compared with VMAT plan and lateral photon–electron plan. 9 In recent years, several studies on the clinical application of HT suggested that it has substantial dosimetric advantages in complex targets.13–15
However, not like the limited area of skull irradiation, the difficult challenge presented in RT for some very rare cases is that the tumor metastasis involves the entire calvarium and skull base. For these rare cases, the target is a completely enclosed volume that surrounds the entire brain, and total dura mater irradiation (TDMI) is needed. Whole-brain irradiation could cause significant problems on neurocognitive development among young children. 16 Memorial Sloan–Kettering Cancer Center employed a combined photon and electron technique for TDMI. 17 This technique could give very low dose to the brain and hippocampus. However, the inevitable inappropriate matching between photon fields and electron fields could produce an insufficient and inhomogeneous dose to the target. Considering that the patients who received TDMI are young children, improving the local control rate of tumor and reducing the radiation damage of OARs, especially for brain and hippocampus, are important. Thus, any attempt to deliver a high, conformal, and homogeneous dose to the targets or reduce the dose to OARs has great value and clinical significance.
In this study, nine young children's case datasets were collected, a dosimetric study was performed to compare the HT and RA plans, and the absolute doses achievable for these two techniques were reported. Several dosimetric parameters were selected and compared, and this study focused on the dose conformal indices and homogonous indices of the target, brain, brainstem, and hippocampus sparing, which are all relevant to neurocognitive function.
Material and Methods
Patient Selection
A total of nine cases that received HT treatment (Tomotherapy Hi-Art system) at Sun Yat-sen University Cancer Center were selected for dosimetric comparison from our database and fully anonymized for the purpose of this study. All these patients were diagnosed with dura mater metastasis of neuroblastoma with extensive diffusion involvement of the entire calvarium and skull base. Three are boys and six are girls. Their mean age was 4.8 years (range of 1-8 years). This retrospective study was approved by the Sun Yat-sen University Cancer Center Institutional Review Board (Approval No: YB2018-40), and all patient details were de-identified.
Computed Tomography (CT) Simulation
All patients were immobilized using a thermoplastic mask and a small vacuum bag for head and neck. The CT images were acquired with the head first and supine on position under a Philips Brilliance CT Big Bore (Philips Medical System, 96 Highland Heights). The slice thickness of the reconstructed CT image is 0.3 cm. All CT images were transmitted to Monaco treatment planning system (TPS; Monaco V5.0.2, Elekta AB) to delineate the clinical target volumes (CTVs) and OARs.
Delineation of Anatomical Structures
All organ structures were delineated by a trained physician. CTV comprised almost the entire calvarium and skull base. Planning target volumes (PTVs) were generated by expanded CTV with isotropic 3 mm margin and cropped to allow 0.3 cm of skin sparing to account for dose buildup in the absence of bolus. The expanded PTV is shown in Figure 1. Other OARs, including the lens, eyes, optical nerves, optical chiasm, brainstem, pituitary, brain, and hippocampus, were contoured following anatomical definitions.

Representative of planning computed tomography (CT) with PTV rendered in blue.
Planning Techniques
In this study, two techniques (tomotherapy and RA) were chosen for treatment planning and dosimetric comparison. The plans were optimized with different parameter combinations to achieve the best dose coverage of the target volume and spare OARs. Finally, the best combination of parameters was selected for the subsequent plan optimization. The details for each technique are as follows.
HT Plan
The HT plans were generated with tomotherapy planning system (Tomotherapy Hi@Art 5.1.1 system) with the energy of 6 MV photon beam. The major machine parameters involved in optimization were set as the field jaw width of 1.0 cm, the pitch value of 0.287, and the modulation factor of 3.8. The plan dose was calculated using convolution superposition method at fine mode and with a grid spacing of 0.273 cm × 0.273 cm × 0.3 cm.
RA Plan
RA plans were generated with Eclipse planning system (version 15.6, Varian Medical Systems, Inc.) with the energy of 6 MV photo beam of VitalBeam linac. The plans with four coplanar arcs of 360° with half field (RA4) were optimized to achieve the desired level of modulation required, as illustrated in Figure 2. Arcs 1 and 3 were full arcs that rotate clockwise with upper half field, while arcs 2 and 4 were full arcs that rotate counter-clockwise with another half field. The plan dose was calculated using AAA algorithm and with a grid spacing of 0.25 cm × 0.25 cm × 0.3 cm.

Illustration of RapidArc plan with 4 arcs and half field. (a) Transverse plane of PTV with half field. (b)Sagittal plane of PTV with diagram of MLC position of half field. (c) Arc 1&3 with clockwise rotation with upper half field. (d) Arc 2&4 with counter-clockwise rotation with another half field.
Planning Objectives
The prescription dose was set to 30.6 Gy at 1.8 Gy/fraction to the PTV, once per day and five fractions per week. The optimization constraints for PTVs were set as 100% of prescription dose must cover 95% of PTV volume at least. The PTV coverage by prescription dose for all plans were normalized to 95% to perform a reasonable comparison. The auxiliary optimization structure named DoseRing was used to constrain the high dose in the peripheral region of PTV and produce the high dose gradient to spare the normal tissue. DoseRing was defined by the region from 0.3 cm to 1.5 cm outside PTV. Another auxiliary optimization structure named OAR10 mm was created by shrinking 1 cm from the inner boundary of PTV to constrain the dose to the whole brain as low as possible. The OARs involved in optimization included bilateral lens, bilateral eyes, bilateral optical nerves, optical chiasm, bilateral hippocampus, pituitary, and brainstem. The bilateral lens was constrained by the max dose of 10 Gy, and the doses to the other organs were constrained as low as possible. The details of acceptable compliance criteria for the target and normal tissue planning doses are shown in Table 1.
The Dose Constrains for Treatment Planning.
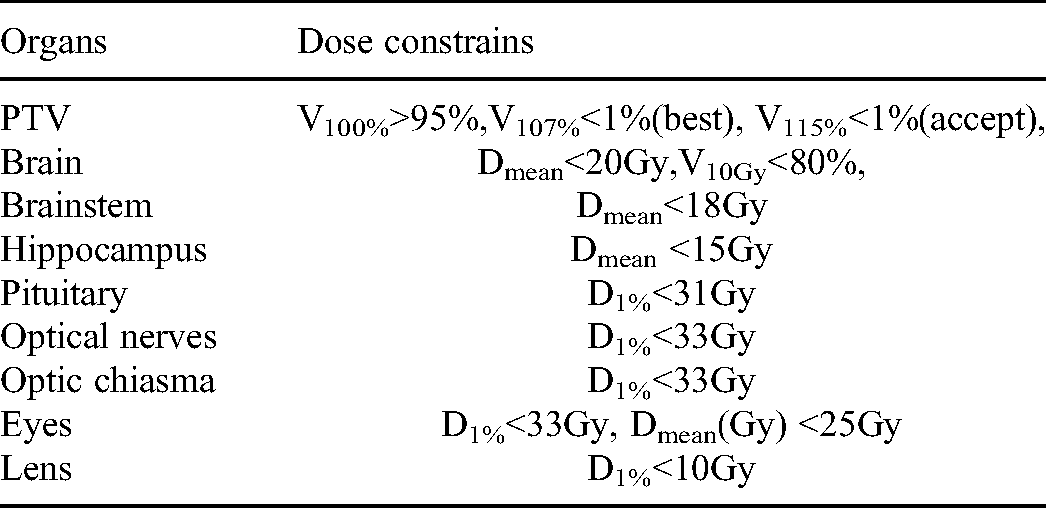
Plan Evaluation
For comparison purposes, the percentage volume covered by the prescription dose for PTV were rescaled to 95% for all plans from different TPSs. For PTV, the dosimetric analysis parameters were D2%, D98%, V107%, V95%, homogeneity index (HI), and conformity index (CI). The algorithm for CI
18
and HI
19
were described as follows.

D5% and D95% represent the doses that covered 5% and 95% of the target volume, respectively. PTV95% is the PTV that received 95% of the prescribed dose, and V95% is the volume that received 95% of the prescribed dose.
For OARs, the parameters analyzed included the following: (1) brain: the mean brain dose (Dmean) and the percentage volume of whole brain receiving dose at least 5 Gy (V5Gy), 10 Gy (V10Gy), 20 Gy (V20Gy), and 30 Gy (V30Gy); (2) hippocampus: the mean hippocampus dose (Dmean) and the percentage volume of hippocampus receiving dose at least 5 Gy (V5Gy); (3) lens: the maximum dose covering 1% volume of the organ (D1%); (3) optical chiasm, optical nerves, and eyes: the maximum dose covering 1% volume of the organ (D1%) and the mean dose (Dmean); (4) pituitary: the maximum dose covering 1% volume of the pituitary and the mean pituitary dose (Dmean); (5) brainstem: the percentage volume of brainstem receiving dose at least 5 Gy (V5Gy), the mean brainstem dose (Dmean), and the maximum dose covering 1% volume of the brainstem (D1%).
Delivery Efficiency Comparison
Machine MUs and plan delivery times were used for comparing the plan delivery efficiency.
Statistical Analysis
SPSS 19.0 software (IBM) was used for statistical data management and analysis. Two-tailed paired-sample t-test was performed to determine statistical significance, with P values < .05 considered as statistically significant. Data are presented as the mean over all patients with standard deviations (SD).
Results
Comparison of Dose Distribution in PTVs
The plans were evaluated and compared by dose-volume histogram and dose distribution on planning CT images. The representative dose distribution of these two techniques for a patient with PTV is shown in Figure 3. The spread of low dose to the brain was more restricted in HT plans than in RA4 plans, and dose conformity was comparable for both plans. The results of quantitative analysis of dosimetric metrics in PTV are listed in Table 2. The D98% and V95% of PTV are similar between the two techniques. However, the D2% of PTV was lower in HT plans than in RA4 plans, and the V107% of PTV was significantly lower in HT plans than in RA4 plans. The CI was equivalent for these two techniques. The HI was lower in HT plans than in RA4 plans. In summary, both techniques could achieve acceptable PTV coverage. The homogeneity of PTV in HT plans was inferior to that in RA plans. The DVH comparison of PTV between the two techniques for an exemplary case is shown in Figure 4.

Comparison of dose distribution in section planes and sagittal plane. The figures (a), (c), (e) represent dose distribution for RA4 (RapidArc plan with 4 coplanar full arcs with half-field technique, RA4) plans and the figures (b), (d), (f) represent dose distribution for HT plans. The dose was rescaled to 30.6 Gy as 100% (the prescription dose for PTV).

Dose-volume histogram comparison of PTV and critical structures between HT plans and RA4 plans on one exemplary case.
Target and OAR Dose Data in Addition to Monitor Units Requirements and Delivery Time Comparisons.

Abbreviations: Tomo, Tomotherapy plans; RA4, RapidArc plans with 4 arcs.
Note. P indicates significance on the two-tailed paired-sample t-test.
Comparison of Dose Distribution of Sparing OARs and Healthy Tissue
The results of quantitative analysis of OAR are shown in Table 2. All OARs met the dose constraints with acceptable range. The DVH comparison of the main OAR between the two techniques for a patient is shown in Figure 4. The D1% and Dmean of optic chiasm, bilateral optic nerves, bilateral eyes, and bilateral lens were similar between the two techniques, and only the D1% of bilateral lens, the Dmean of bilateral eyes, and the optic chiasm were slightly lower in HT plans than in RA4 plans.
The Dmean and V5Gy of the brain, brainstem and bilateral hippocampus were significantly lower in HT plans than in RA4 plans. HT reduced the Dmean of the brain, brainstem, pituitary, and bilateral hippocampus by 25%, 42%, 6%, and 48% compared with RA, respectively. HT also reduced the V5Gy of the brain, brainstem, and bilateral hippocampus from 93.75% to 66.07%, 90.32% to 46.55%, and 88.01% to 32.33% compared with RA4, respectively.
Comparison of Delivery Time and MUs
However, reduction in MUs and shorter treatment times were produced by RA4 plans compared with HT plans. In this study, the RA4 plans could save approximately 93.7% MUs compared with the HT plans of 15,044 MUs and save 66.7% treatment times compared with the HT plans of 1080 s.
Discussion
This study showed that HT and RA4 plans met the basic dosimetric compliance criteria. The HI of PTV was significantly better in HT plans than in RA4 plans (mean HI: 1.046 vs 1.088), and HT significantly reduced the Dmean and V5Gy of the brain, brainstem, and bilateral hippocampus compared with RA4.
For PTV, the biggest difference is mainly reflected in dose uniformity. The mean HI5−95% of PTV in HT plans was 1.046, significantly better than in RA4 plans, with mean HI5−95% of 1.088. These data indicated that HT plans present a more homogenous dose distribution within the target volume. The V107% for PTV was only approximately 3% (average) in the case of HT, whereas for RA4, it was approximately 30%. In addition, another hotspot metric D2% of PTV in HT was 32.58 Gy, which was slightly lower than the 33.45 Gy in RA4 plans. Such heterogeneity could be accepted due to the technical complexity in TDMI. All of these hotspots were within PTV, and they may be of advantage. The mean CI95% was comparable for both plans when all plans were normalized to such that 95% of the PTV received at least the prescribed dose of 30.6 Gy. However, the DVH curve of PTV in HT had sharper dose gradients than that in RA4 in Figure 4. As could be imagined, CIs may change in favor of HT when plans are normalized to the other dose (ie, D50% = 30.6 Gy). Thus, the plan was renormalized to D90%, D80%, D70%, D60%, and D50%, and then the corresponding CIs were calculated. As shown in Figure 5, the CIs in HT plans were better than in those in RA4 when the plan was normalized to these doses. Thus, CI was confirmed to be dependent on the plan normalization method. HT could provide higher dose homogeneity, consistent with other literature reports.8–10

The dependency of the mean conformity index on the plan normalization method.
For pediatric patients during cranial irradiation, the hippocampus should be avoided from irradiation as much as possible. The hippocampal injury in cranial irradiation could result in potential cognitive decline, especially in terms of short-term memory formation and recall. Literature reported that the deficits in learning, memory, and spatial processing observed in patients who underwent brain tumor radiotherapy are thought to be related to hippocampal injury.20–24 Yinai Gondi 20 evaluated the association between hippocampal dose and long-term neurocognitive function impairment and concluded that 7.3 Gy of equivalent dose to 2 Gy per fraction (EQD2) to the 40% of hippocampus could be used for the dosimetric threshold. Thus, many researchers attempted to practice with different irradiation techniques (IMRT, RA, coVMAT, VMDWAT, and tomotherapy) to reduce the dose to the bilateral hippocampus.5–7,25 In the present study, the dose to the hippocampus with HT plans was significantly reduced compared that with RA4 plans, and the mean dose to the bilateral hippocampus in HT plans was 6.56 Gy and the V5Gy was 32.33%, versus 12.01 Gy of mean dose and 88.73% of V5Gy in RA4, respectively. Therefore, the risk of causing cognitive decline for pediatric patients could be reduced with HT plans.
Radiotherapy-induced brain injury is also a side effect of brain tumor radiotherapy, especially in children. The acute side effects of RT to the brain include nausea, vomiting, and headache. 26 These symptoms are usually transient and could be cured with medicine. However, the principal latent side effects of RT to the brain, including radiation necrosis and cognitive deterioration, are permanent and irreversible. Hill reported that 24 Gy radiation to the whole brain could cause a median 13 points of intelligence quotient reduction at 5 years after RT. 27 Others reported that if 14 to 18 Gy was used, the toxicities could be lower. 28 However, the dose-volume data about brain subregions are very challenging, and the corresponding dose limit is unclear. Current evidence showed that the RT that induces irreversible cognitive decline could be reduced by decreasing the dose irradiated to the brain. 29 The results of the present study showed that the mean dose to the whole brain in HT plans was 13.99 Gy (45.7% of the prescribed dose) lower than 18.67 Gy (61% of the prescribed dose) in RA4 plans. In addition, the mean dose to the brain in HT was lower than that in photon/electron technique (45.7% vs 52%, rescaled to the prescribed dose). 17 The dose volume metric V5Gy of the whole brain in HT plan was 66.07%, lower than the 93.75% in RA4 plans. Therefore, compared with RA4 plans, HT plans could significantly reduce the dose to the whole brain and benefit patients. In addition, HT plans could provide more continuous and homogenous dose distribution than photon/electron technique, which may have dose uniformity problem at abutting field junctions.
Brainstem radiation toxicity and pituitary radiation toxicity are also clinically important. However, research about toxicity to quantitative dose and dose-volume showed that the key injury was mainly due to high radiation dose. 30 In the present study, the prescription dose was 30.6 Gy. Thus, the incident of main normal tissue injury is very rare. However, HT plans could significantly reduce the brainstem dose and pituitary dose relative to that of RA4 plans in terms of dose metric Dmean.
For optic pathway structures, only the mean dose of bilateral eyes and optic chiasm in HT plans was slightly lower than that in RA4 plans. However, the maximum dose (D1%) to bilateral lens, eyes, and optic nerves were comparable for both radiation techniques, but these doses were all within clinic dose tolerance. One of main possible reasons is that the target was irradiated by the tangential beam.
However, HT plan has more delivery times and more MUs than RA4 plan. This finding is consistent with the other reports about the delivery efficiency evaluation for HT plan versus other modalities. 31
This study has some limitations. First, for fair comparison of plans, common dose constraint goals were set for targets and OARs. Other optimization parameters (ie, weights and modulation factors) affect the plan quality, and these planning parameter settings are dependent on the dosimetrist's preferences. Second, mechanical parameter selection could also affect the results of plan quality. In this study, the jaw size in HT was 1.0 cm, not 2.5 cm. The aim was to produce higher plan quality. The estimation on an exemplary case showed that the plan with a jaw size of 1.0 cm has higher quality than that with a jaw size of 2.5 cm. For example, the CI95% (the closer the value is to 1, the better) of PTV could increase from 0.547 to 0.616, and the HI5−95% (the closer the value is to 1, the better) of PTV could decrease from 1.085 to 1.046. The Dmean and V5Gy of the brain could decrease from 15.36 Gy to 13.75 Gy and from 70% to 62.4%, respectively. The only drawback is that it prolongs the treatment time. For RA plans, four arcs with half-fields were chosen because the required dose could be easily produced with half field on the one hand. On the other hand, more arcs did not significantly increase the plan quality. Third, limited sample size may reduce the reliability of data and weaken the reliability of research results. In summary, dosimetric comparisons between RT techniques are not easy to perform in an objective manner due to innate unavoidable biases. In future work, the effort to reduce the innate unavoidable biases and improve plan quality could be developed, and this could go further to benefit patients.
Conclusions
HT and RA4 with half-field technique are all clinically feasible options for TDMI. HT may produce more homogenous dose to the target than RA4. However, any reduction in unnecessary dose to the brain and hippocampal structures could improve long-term cognitive decline in these young pediatric patients. Therefore, HT offers a priority option relative to RA4 because it provides significantly lower dose to the brain and hippocampal structures for this kind of a completely enclosed target volume, which surrounds the entire brain. The RA4 plan may be completed in a shorter time, but the dose to the brain or hippocampus should be evaluated if this plan is chosen for delivery.
Footnotes
Acknowledgments
We are very grateful to Shi-wang Guan physicist of the People's Hospital of JieYang and Zhu Liang physicist of the People's Hospital of GaoZhou for their help in the plan studies.
Availability of Supporting Data
Authors' Contributions
Design of the study and literature research: WZS, JZ and LXL, DXW. Treatment planning and acquisition of data: YXW, MNC, LC and JLW. Analysis and interpretation of the data: WZS,JZ, YXW and MNC. Critical review of data analyses: WZS and LC. Writing and revision of the manuscript: All authors. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China (grant number 12075329) and the Science and technology project of Guangdong Esophageal Cancer Institute (grant numberQ202008).
