Abstract
Introduction
Small cell lung cancer (SCLC) is a highly malignant and aggressive tumor with short doubling cycle and early distant metastasis. It has a high incidence in China, accounting for 13% of the total incidence of lung cancer. 1 With sensitivity to radiotherapy and chemotherapy, SCLC has a high treatment remission rate. But it is easy to relapse and has a high resistance rate of second-line chemotherapy. Without a standard plan for third-line and subsequent-line treatment, SCLC has a weak overall curative effect and a 5-year survival rate estimated to only 7%. 2
Paclitaxel is an antitumor drug with a definite effect on a variety of malignant tumors which reduces microtubule generation. It is a component of the standard regimen of non-SCLC. 3 Current studies have shown that paclitaxel has a specific effect on recurrent SCLC.2,4–6 Nanoparticle albumin-bound paclitaxel (Nab-P) is the first targeted chemotherapeutic agent based on a nanotechnology platform without solvent. It has the characteristics of high efficiency and low toxicity which can combine the insoluble paclitaxel with albumin through unique nanotechnology without using toxic solvents. This study retrospectively analyzed the clinical efficacy and adverse reactions of Nab-P as a single-agent third-line treatment of SCLC.
Materials and Methods
Patients
A total of 37 patients with recurrent SCLC admitted to Nanjing Chest Hospital from December 2016 to December 2019 were selected. Inclusion criteria for the current study were: (1) diagnosis of SCLC and (2) failed second-line treatment. Patients were subjected to the limited period group and extensive period group according to the following working definitions for 2 disease stages. The limited stage of SCLC was defined as the lesion that confined to 1 hemithorax and including the bilateral mediastinal and supraclavicular nodes. The extensive stage of SCLC was defined as the lesion that extends beyond these aforementioned boundaries.
The study was approved by the Ethics Committee of Nanjing Chest Hospital. All patients enrolled were provided with written informed consent.
Chemotherapy
For chemotherapy, 1 treatment circle took 21 days. Patients received 2 times of Nab-P single-agent intravenous infusion (130 mg/m2) at day 1 and day 8, and they rested for 1 week from day 15 to day 21. All patients in the current study received at least 2 circles of Nab-P chemotherapy. Adverse reactions during chemotherapy were observed and recorded.
Evaluation of Therapy Responses
First, chest computed tomography (CT) was performed for baseline imaging evaluation before treatment. Then CT was performed again at the end each chemotherapy cycle for efficacy evaluation. According to the Response Evaluation Criteria in a Solid Tumor standard, 7 patients were divided into complete response (CR), partial response (PR), stable disease (SD) and progression disease (PD). Objective response rate (ORR) was defined as the combined percentage of CR and PR in all patients. Disease control rate (DCR) was defined as the combined percentage of CR, PR and SD in all patients.
Adverse Reactions Evaluation
Hematological and biological tests were routinely done once a week during chemotherapy. The results were classified according to the fourth edition of the Common Toxicity Criteria of the National Cancer Institute.
Follow-up
Patients were followed up by telephone and outpatient clinic, which ended in December 2019. The 34 patients were followed up successfully. Progression free survival (PFS) is defined by the period from the day of receiving albumin paclitaxel treatment to disease progression or death caused by any other reasons. Overall survival (OS) is defined by the period from the day of receiving albumin paclitaxel treatment to death caused by any reason. If there is no disease progression when patient dies, the last time of follow-up will be recorded as the cutoff time.
Statistical Analysis
The data were statistically analyzed using SPSS 22.0 software. The categorical data were expressed in terms of the number of cases and the percentage (%). The Fisher Exact Test was used to compare OS and PFS between patient groups dichotomized by sex, disease stage, or eastern cooperative oncology group (ECOG) score. OS and PFS were calculated by the Kaplan–Meier method. P-values <.05 were considered significant.
Results
Efficacy
A total of 131 cycles of chemotherapy were completed in 37 patients. They consisted of 13 men and 24 women aged from 51 to 76 years old. The median age is 62 years old. There were 15 patients (40.54%) in limited stage, 22 patients (59.46%) in extensive stage, 29 patients (78.38%) with ECOG score of 0 to 1, and 8 patients (21.62%) with ECOG score greater than or equal to 2. There were 24 cases (64.86%) with smoking history and 13 cases (35.14%) without smoking history. Etoposide platinum-containing double-agent chemotherapy was used in the first-line regimen in 37 patients, topotecan alone in 14 patients, irinotecan in 15 patients, anlotinib in 1 patient, and original regimen in 7 patients (Table 1).
Baseline Characteristics of 37 SCLC Patients.

Abbreviatons: SCLC, small cell lung cancer; EN, etoposide + nedaplatin; EC, etoposide + carboplatin.
The number of chemotherapy cycles for each patient ranged from 2 to 4. Four cycles of chemotherapy were completed in 24 patients (64.86%), 3 cycles were completed in 9 patient (24.32%), and 2 cycles were completed in 4 patient (10.81%). There were 2 CR patients (5.41%), 7 PR patients (18.92%), 21 SD patients (56.76%), and 7 PD patients (18.92%) with the ORR of 24.32% and the DCR of 81.08% (Figure 1).
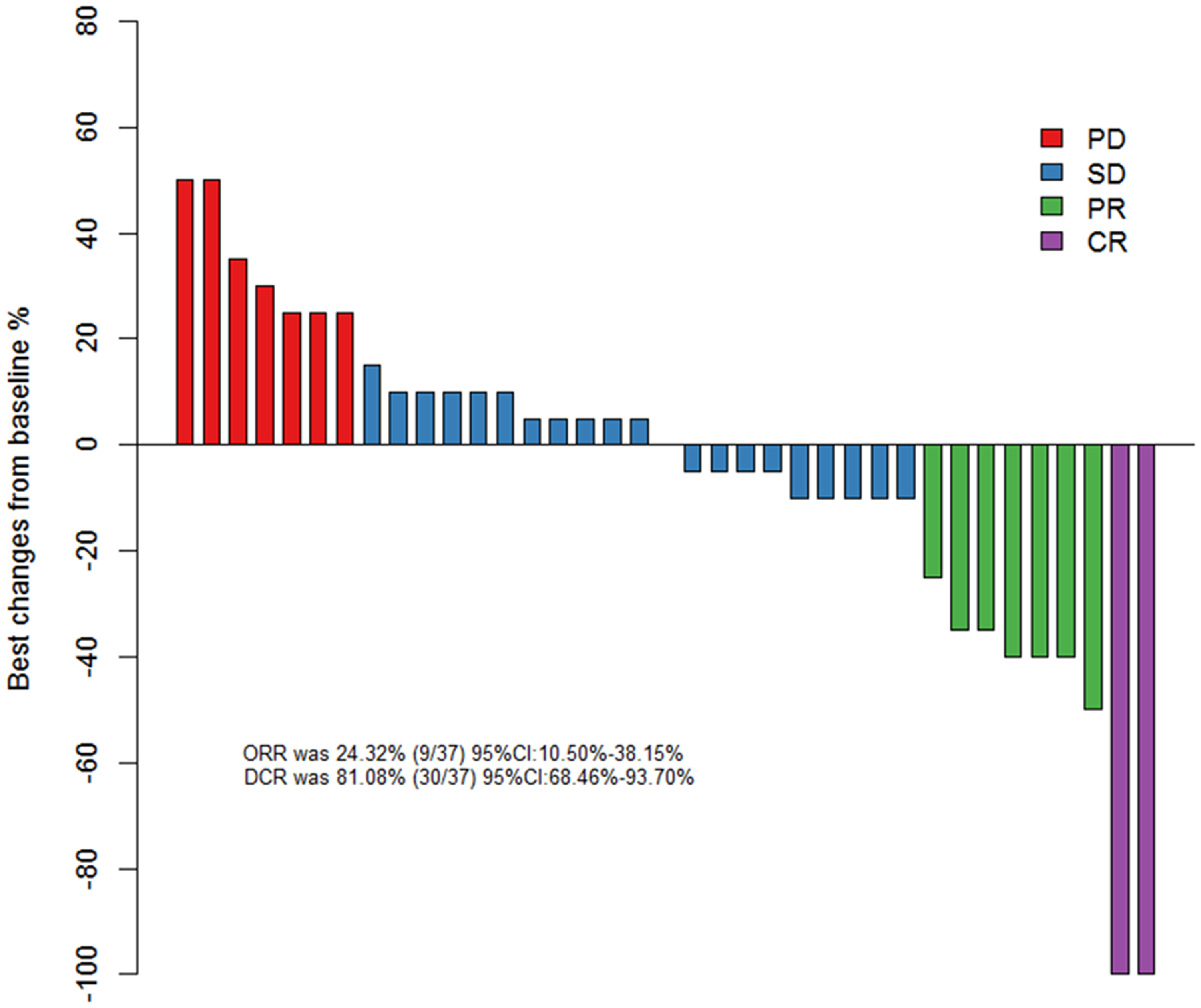
Waterfall plot of best change from baseline in 37 small cell lung cancer patients.
Follow-up was carried out from the start of treatment to December 2019. The median follow-up time was 7 months (4 to 10 months). For all patients, the median PFS (mPFS) was 5.0 months (95% confidence interval [CI] 4.0-6.0) and the median OS (mOS) was 9.0 months (95% CI 7.0-9.0) (Figure 2).

Survival curve of 37 patients.
Subgroup analysis showed that there was no significant difference (P > .05) of PFS and OS between extensive period group and limited period group, sex and ECOG score. But we can see that the mPFS and mOS of limited period group is greater than extensive period group, the median mPFS and mOS of patients with PS 0 to 1 points is greater than 2 to 3 (Figure 3).

Survival curve analysis of each subgroup.
Adverse Reactions
Bone marrow suppression was the primary adverse reaction. There were 36 patients (97.30%) with leukopenia and neutropenia, 32 cases (86.49%) with thrombocytopenia, and 31 cases (83.78%) with anemia. As for other adverse reactions, there were 29 cases (78.38%) with abnormal liver function, 32 cases (86.49%) with gastrointestinal reactions, 32 cases (86.49%) with joint and muscle pain, 33 cases (89.19%) with fatigue, and 27 cases (72.97%) with peripheral nervous system toxicity (Table 2). Except for bone marrow suppression, other adverse reactions were all classified as grade 1 to 2. Adverse reactions ≥ level 3 included leukopenia& neutropenia (24.32%), thrombocytopenia (5.41%) and anemia (2.70%). The patients with bone marrow suppression returned to normal after being treated with colony cell-stimulating factor hence the next cycle of medication was not disturbed. All patients tolerated the adverse reactions and completed at least 2 cycles of chemotherapy throughout the research period.
Adverse Reactions of Albumin-Bound Paclitaxel in Third-Line Treatment of Small Cell Lung Cancer.

Typical Case
A 58-year-old female patient had the CT scan of the right middle lung nodule with an encapsulated effusion on the right side before seeing a doctor (Figure 4). The result of bronchoscopy pathology confirmed small cell carcinoma. The blood tests for cancer biomarkers showed that neuron specific enolase (NSE) was 78.23 ng/mL and Pro-gastrin releasing peptide (GRP) was 168.23 pg/mL. She completed 6 cycles of etoposide and carboplatin chemotherapy. The best efficacy on this patient was PR and her PFS was 4 months. Then she received 2 cycles of topotecan chemotherapy, and the treatment efficacy was assessed as PD. Then, the blood tests for tumor biomarkers showed that NSE was 156.00 ng/mL, and Pro-GRP was 700.23 pg/mL. Nab-P chemotherapy was chosen as the third-line regimen for 4 cycles, and the efficacy was evaluated to be PR. The blood tests for tumor biomarkers showed that NSE was 32.89 ng/mL and Pro-GRP was 66.50 pg/mL.

(A) Chest CT before diagnosis; (B) Chest CT after 6 cycles of EC regimen chemotherapy; (C) Chest CT after 2 cycles of topotecan chemotherapy; (D) Chest CT after 4 cycles of Nab-P chemotherapy.
Discussion
SCLC is a neuroendocrine tumor from epithelial with high malignancy and poor prognosis. Approximately 70% of SCLC patients were diagnosed in the extensive-stage small cell lung cancer (ES-SCLC), which were inoperable, and only 30% of the patients were diagnosed in the limited stage small cell lung cancer (LS-SCLC). The median OS is estimated to be 15 to 20 months for LS-SCLC and 8 to 13 months for ES-SCLC, respectively. 8 SCLC patients were very sensitive to initial treatment, and chemotherapy was still the first choice for SCLC. 9 The total effective rate of traditional etoposide combined with platinum dual-drug chemotherapy could reach 60%-80%. 9 This chemotherapy method originated in a clinical study conducted in 1982 by the Southeastern Cancer Research Group in the United States. In the past few decades, standard chemotherapy has not changed significantly. Although the remission rate after the first chemotherapy treatment could reach more than 80%, most SCLC patients would relapse and developed drug resistance soon after the initial therapy with poor median OS. According to National Comprehensive Cancer Network (NCCN) guidelines, topotecan, irinotecan, gemcitabine, or paclitaxel were recommended for patients who relapsed within 3 months. Patients who recurred at 6 months could choose the initial treatment plan. Topotecan was the only chemotherapeutic drug approved by the Food and Drug Administration (FDA) and the European Union for the second-line treatment of SCLC. A meta-analysis on topotecan showed that the total effective rate of topotecan was 16.4% for sensitive and refractory relapsed patients. 10 The above research demonstrated that although topotecan was a recognized second-line treatment for SCLC, its efficacy was not satisfactory. There was no standard treatment plan for SCLC above third-line therapy, and the treatment effect was not satisfactory.
Paclitaxel was a commonly used chemotherapy drug for non-SCLC with the action mechanism of inhibiting the depolymerization of tubulin and blocking the cell cycle in M phase. It was also used in SCLC. A clinical study of a second-line method of irinotecan combined with paclitaxel for the treatment of recurrent SCLC showed a total effective rate of 21% in 51 patients, which was higher than the effective rate of topotecan for second-line treatment. 11
Compared with paclitaxel, Nab-P had a higher efficiency which was related to its unique pharmacological properties. Nab-P utilizes the unique natural transport mechanism of albumin to make paclitaxel act more on tumor tissues and consequently achieve a higher intratumor cell concentration without the use of toxic solvents, which can achieve low-efficiency attenuation effects. A Japanese study suggested that the ORR of Nab-P monotherapy was 33% for SCLC patients resistant to second-line therapy. 12 Another Japanese study reported lower ORR in 17 relapsed SCLC patients treated with Nab-P, which is 29.4%, and 17.6% patients suffered ≥3 level leukopenia. 13 A retrospective study conducted showed that patients could still approach DCR of 70% treated with the Nab-P-based regiment for second-line therapy and above. 14 A small conservational study focusing on Nab-P for relapsed SCLC reported a ORR of 36% and a DCR of 64%, but with 64% participants had anorexia and some even had severe neutropenia. 15
In this study, the ORR was 24.32% and the DCR was 81.08%, which was significantly higher than previous research programs. In the third-line treatment, there were still 2 patients who reached the efficacy as PR, indicating that they were still sensitive to Nab-P after being resistant to standard chemotherapy. A clinical study of anlotinib in the treatment of SCLC showed that the PFS and OS of anlotinib in SCLC patients were 4.1 months and 7.3 months. Our study shows the mPFS was 5.0 months and the mOS was 9.0 months of the patients who received Nab-P as the third-line chemotherapy. Both PFS and OS benefit. In terms of adverse reactions, bone marrow suppression of III-IV degree was the main reaction in hematological toxicity, and I-II degree adverse reactions were the left reactions in patients who could complete subsequent treatment after the colony cell-stimulating factor treatment. That the patient was well tolerated indicated the advantages of low toxicity and high efficiency of Nab-P in SCLC patients treated with multi-line therapy. At present, immunotherapy and antiangiogenesis therapy are also research hotspots in the third-line treatment of SCLC, which have achieved good clinical effects.14,15 As Nab-P has the characteristics of low toxicity and high efficiency, it can be considered in combination with antiangiogenesis or immunotherapy for better efficacy.
Our research has some limitations. Firstly, the current study was a single-center trial and all our patients were retrospectively recruited, which means that we should definitely conduct a multicenter, large-sample prospective study as the next step, to validate our results and further expand the sample size for more rigorous analyses. Secondly, the current study did set any forms of control groups, which might to some degree weaken the strength of our rights. Prospective controlled studies are needed.
Supplemental Material
sj-docx-2-tct-10.1177_15330338211050775 - Supplemental material for Nanoparticle Albumin Bound Paclitaxel in the Third-Line Treatment of Recurrent Small Cell Lung Cancer in Real-World Practice: A Single Center Experience
Supplemental material, sj-docx-2-tct-10.1177_15330338211050775 for Nanoparticle Albumin Bound Paclitaxel in the Third-Line Treatment of Recurrent Small Cell Lung Cancer in Real-World Practice: A Single Center Experience by Yuchao Wang, Li Li and Chunhua Xu in Technology in Cancer Research & Treatment
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nanjing Medical Science and Technique Development Foundation (grant number ZKX17044).
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
