Abstract
Keywords
Introduction
Despite the high incidence of gastric cancer in South Korea, the prognostic outcomes of gastric patients have improved 1 due to detection of early-stage gastric cancer with a generalized endoscopic screening program, 2 globally accepted D2 dissection as surgical standard, and efficient adjuvant chemotherapy. 3 According to the guidelines from different countries, however, there are inconsistencies in the recommendations of regimens in chemotherapy for gastric cancer patients. 4
Recently, 2 regimens of adjuvant chemotherapy after radical gastrectomy with D2 lymph node dissection in patients with resectable gastric cancer demonstrated significant oncologic advantages in randomized clinical trials conducted in Japan 5 and in Korea. 6 S-1 (TS-1), which is an oral agent comprised of tegafur, gimeracil, and oteracil, 7 was introduced in the ACTS-GC trial. The XELOX regimen, which consists of 2 drugs (capecitabine [XELODA] is an oral agent and oxaliplatin is an intravenous agent), was introduced in the CLASSIC trial. There seemed to be a chronological shift in regimens of adjuvant chemotherapy after those trials, and the 2 regimens have become the mainstream treatment in a large-volume gastric cancer center in Korea, since 2013. 8
However, there is not enough scientific evidence or high-quality evidence regarding the long-term benefits and indications for choosing between these 2 regimens for patients with various underlying diseases or comorbidities. Randomized phase 3 trials are essential for such conclusions; however, randomized phase 3 trials involve high cost and time requirements.
The aim of this study is to retrospectively review the characteristics of patients who received adjuvant chemotherapy (TS-1 or XELOX). We compared the long-term survival rates of patients who received TS-1 or XELOX during adjuvant chemotherapy. In addition, we investigated clinical indications that could guide clinicians in selecting regimens.
Materials and Methods
Patients
The prescriptions of adjuvant chemotherapeutic agent (XELOX or TS-1) were evaluated for 2021 patients who were diagnosed with gastric cancer and underwent curative resection at Samsung Medical Center between January 2010 and June 2017. The patients underwent R0 resection by radical gastrectomy with D2 lymph node dissection according to guidelines.9,10 We excluded patients who used other chemotherapeutic agents (not XELOX regimen or TS-1 monotherapy) during adjuvant treatment (n = 152), those who were diagnosed with remnant gastric cancer (n = 50) or double primary cancer (n = 39), and those who had history of treatment of other malignancy (n = 109). Those who received preoperative chemotherapy or radiotherapy (n = 72) and those who were diagnosed with stage IV cancer after operation (n = 138) also were excluded. The median follow-up period after surgery (n = 1461) was 56 months (range: 1-125 months).
Study Design and Data Collection
The clinicopathologic characteristics of age, sex, body mass index, American Society of Anesthesiologists (ASA) score, comorbidities, extent of gastric resection (subtotal or total), and tumor pathologic stage (including depth of invasion and lymph node metastasis) were reviewed in patient medical records. Tumor histology was dichotomized as differentiated, which included papillary adenocarcinoma and well or moderately differentiated adenocarcinoma, or undifferentiated, which included poorly or undifferentiated adenocarcinoma, signet ring cell carcinoma, mucinous carcinoma, and other types. Pathologic stage was classified according to the eighth edition of the American Joint Committee on Cancer classification. 11 The primary endpoint was overall survival (OS) after curative gastrectomy. Disease-free survival (DFS) was defined as the time elapsed from the operation date to the recurrence date or to the time of the last follow-up. We obtained the survival data of the patients included in the study from updated medical records and the National Statistical Office in South Korea. This study was approved by the Institutional Review Board of Samsung Medical Center, Seoul, Korea (SMC 2020-12-025), which waived the requirement for written informed consent from the patients owing to the retrospective study design.
Regimens of Adjuvant Chemotherapy
Adjuvant chemotherapy was initiated within 1 month (median and range were 1 month and 0 to 4 months for TS-1 and 1 month and 0-8 months for XELOX, respectively) after curative resection. The chemotherapeutic regimens were administered as described by the ACTS-GC trial 5 and the CLASSIC trial. 6 Patients treated with TS-1 (tegafur/gimeracil/oteracil) received 2 oral doses of 40 to 60 mg/m2 of body surface area (BSA) per day for 4 weeks, followed by 2 weeks of no chemotherapy. During the treatment weeks, the total amount of TS-1 changed according to patient BSA; those with BSA < 1.25 m2 received 80 mg daily; those with 1.25 m2 ≤ BSA < 1.50 m2 received 100 mg daily; and those with BSA ≥ 1.50 m2 received 120 mg daily. This 6-week cycle (8 cycles total) was repeated for the first year after surgery. Patients who received XELOX received eight 3-week cycles of oral capecitabine (XELODA, 1000 mg/m2 twice daily on days 1-14 of each cycle) plus intravenous oxaliplatin (130 mg/m2 on day 1 of each cycle). Reductions of doses, modifications of schedules, or interruptions of adjuvant chemotherapy were conducted based on the clinicians’ decisions considering the events that occurred during treatment.
Follow-Up
The patients were recommended to undergo follow-up examinations every 3 or 6 months for the first year and then every 6 months until the fifth year after surgery. The follow-up evaluations included physical examination, laboratory tests (including gastrointestinal tumor markers), chest X-ray, and abdominal and pelvic enhanced computed tomography scan (AP-CT) scan. The upper gastrointestinal endoscopy was performed annually. After approximately 5 years following surgery, assessments were performed annually or once every 2 years. To confirm recurrence, a tissue biopsy was performed if possible. Further work-up included magnetic resonance imaging, chest CT scans, bone scans, and positron emission tomography.
Statistical Analysis
We used the χ2 test or Fisher's exact test for categorical variables and Student's t-test for continuous clinicopathologic variables. Five-year overall survival and recurrence-free survival were calculated by the Kaplan-Meier method. The log-rank test was utilized for univariate analysis. Variables with P < .05 on univariate analysis were included for multivariate analysis using the Cox proportional hazards model with backward logistic regression to identify independent prognostic factors. The P-values less than .05 were considered significant in statistical assessment. Statistical analysis was carried out using SPSS version 25.0 for Windows (SPSS).
Results
Patient Baseline Characteristics
Of 1461 patients with stage IB-III gastric cancer, 825 (56.5%) received TS-1 and 636 (43.5%) received XELOX as adjuvant chemotherapy after radical surgery. Table 1 shows the clinicopathologic characteristics of the 2 groups (TS-1 vs XELOX). The patients in the TS-1 group were significantly older (60.9 vs 54.9 years, P < .001) and comprised a larger proportion of ASA scores of 2 or greater (64.0% vs 57.9%, P = .011) than did patients in the XELOX group. The patients in the XELOX group underwent more total gastrectomies (42.8% vs 35.6%, P = .006) and were diagnosed with more advanced T and N stages, resulting in a larger proportion of advanced stage patients than in the TS-1 group (stage III and stage II, 56.6% and 43.1% in XELOX, 35.3% and 57.0% in TS-1, P < .001). Regarding comorbidities of the studied patients, hypertension was more frequent in the TS-1 group than in the XELOX group (36.1% vs 28.8%, P = .003).
Clinicopathologic Characteristics of 1461 Gastric Cancer Patients Who Underwent Adjuvant Chemotherapy (TS-1 Versus XELOX).

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; LN, lymph node; TS-1, tegafur/gimeracil/oteracil; XELOX, capecitabine (XELODA) with oxaliplatin.
Data are expressed as mean ± standard deviation or number (percentage).
*Chi-square test, †Fisher's exact test, ‡Student's t-test.
Univariate and Multivariate Analyses of OS and DFS
Although 5-year OS and DFS were significantly different between the TS-1 and XELOX groups (TS-1 vs XELOX: OS, 81.9% vs 75.4%, P = 0.013; DFS, 79.3% vs 69.3%, P < .001), type of regimen was not an independent prognostic factor in multivariate analysis (Table 2). BMI (P = .003), depth of invasion (T stage, P < .001), lymph node metastasis (N stage, P < .001), and cerebrovascular disease (P = .010) were significant factors in multivariate analysis of OS. Extent of resection (total or subtotal, P = .028), T stage (P = .024), and N stage (P < .001) were independent prognostic factors of DFS (Table 2).
Univariate and Multivariate Analyses of Overall Survival and Disease-Free Survival of 1461 Gastric Cancer Patients Who Underwent Adjuvant Chemotherapy (TS-1 or XELOX).

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; HR, hazard ratio; LN, lymph node; 95% CI, 95% confidence interval; TS-1, tegafur/gimeracil/oteracil; XELOX, capecitabine (XELODA) with oxaliplatin.
Comparison of OS and DFS Between TS-1 and XELOX Groups at Each Pathologic Stage
There were no significant differences in DFS for pathologic stage IIA (P = .196), IIB (P = .161), IIIA (P = .226), IIIB (P = .568), or IIIC (P = .289), as shown in Figure 1. No differences in OS rate for pathologic stages IIIA (P = .796), IIIB (P = .701), and IIIC (P = .397) were observed between the TS-1 and XELOX groups (Figure 2). However, there was a significant difference in OS rate between the 2 study groups diagnosed with stage IIA or IIB (5-year OS in TS-1 vs XELOX: stage IIA, 93.4% vs 99.1%, P = .024; stage IIB, 80.3% vs 91.5%, P = .015).

Disease-free survival of patients according to adjuvant chemotherapy regimen (5-year disease-free survival, TS-1 vs XELOX): (A) stage IIA (93.0% vs 97.3%, P = .196), (B) stage IIB (79.7% vs 87.9%, P = .161), (C) stage IIIA (70.2% vs 63.5%, P = 0.226), (D) stage IIIB (59.0% vs 55.4%, P = .568), and (E) stage IIIC (28.4% vs 24.7%, P = .289).
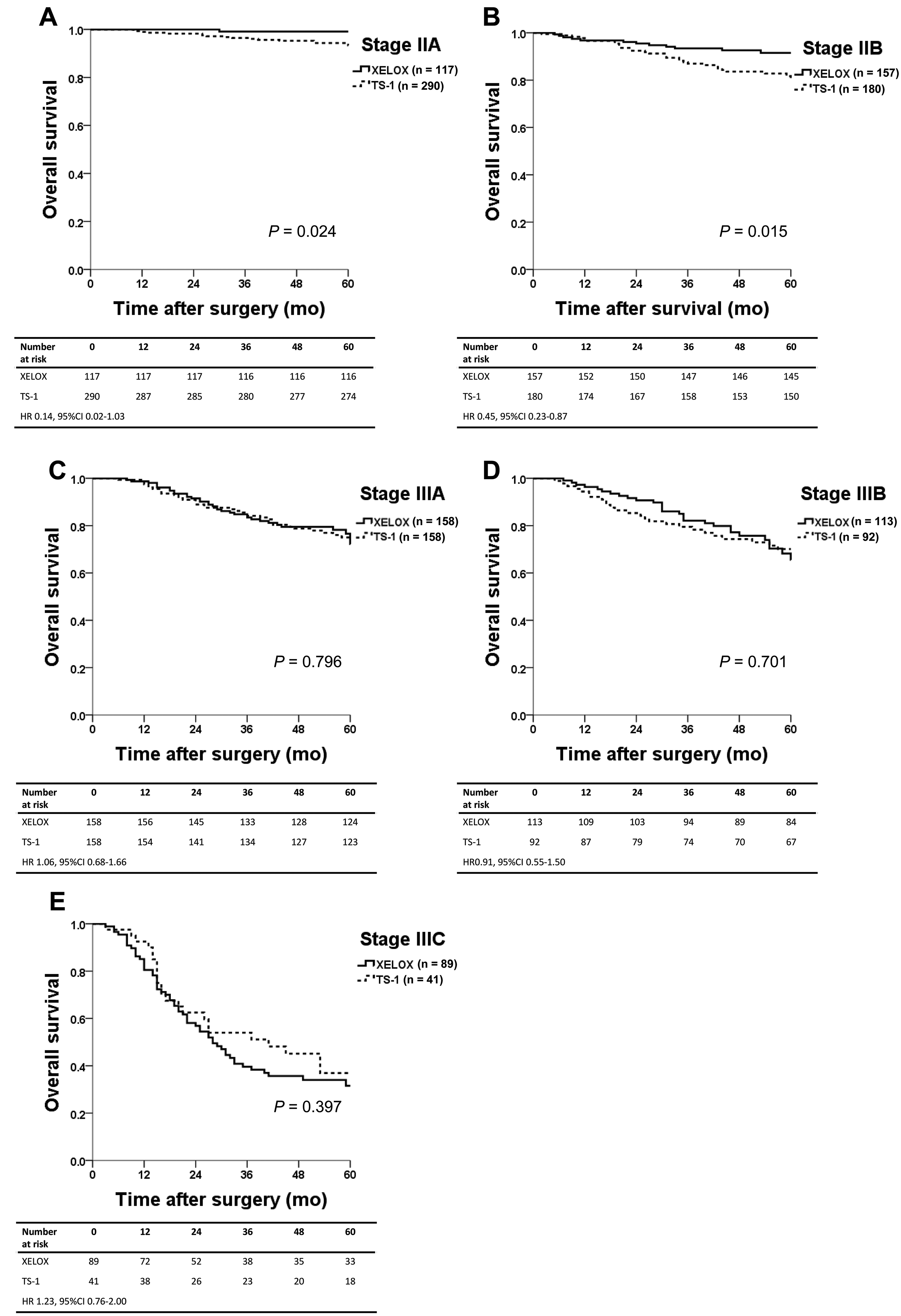
Overall survival of patients according to adjuvant chemotherapy regimen (5-year overall survival, TS-1 vs XELOX): (A) stage IIA (93.4% vs 99.1%, P = .024), (B) stage IIB (80.3% vs 91.5%, P = .015), (C) stage IIIA (75.0% vs 76.6%, P = .796), (D) stage IIIB (70.1% vs 65.6%, P = .701), and (E) stage IIIC (36.9% vs 31.6%, P = .397).
Subgroup Analysis of Patients Diagnosed With Stage II
As there was a significant difference in OS in stage II patients (n = 744), we performed further subgroup analysis of those patients (Table 3). Findings similar to those of the entire cohort were shown in the TS-1 group with stage II. Stage II patients who received TS-1 were significantly older (P < .001) and had a higher prevalence of ASA scores of 2 or greater (P = .004) than those who received XELOX. Cardiac disease was more prevalent in the TS-1 group than in the XELOX group (P = .006). Patients with stage IIA were more prevalent in the TS-1 group, and patients with stage IIB were more prevalent in the XELOX group (P < .001).
Clinicopathologic Characteristics of Stage II Gastric Cancer Patients (n = 744) Who Underwent Adjuvant Chemotherapy (TS-1 Versus XELOX).

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; TS-1, tegafur/gimeracil/oteracil; XELOX, capecitabine (XELODA) with oxaliplatin.
Data are expressed as mean ± standard deviation or number (percentage).
*Chi-square test, †Fisher's exact test, ‡Student's t-test.
In multivariate analysis of OS, type of chemotherapeutic regimen was a significant independent factor (hazard ratio: 0.47, 95% confidence interval: 0.25-0.89, P = .021, Table 4). Among variables and comorbidities, age (P = .003) and pathologic stage (P < .001) were significant in multivariate analysis.
Univariate and Multivariate Analyses of Overall Survival of Stage II Gastric Cancer Patients (n = 744) Who Underwent Adjuvant Chemotherapy (TS-1 or XELOX).

Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; CI, confidence interval; HR, hazard ratio; LN, lymph node; N/A, not applicable; TS-1, tegafur/gimeracil/oteracil; XELOX, capecitabine (XELODA) with oxaliplatin.
The causes of death in stage II patients (n = 63) are listed in Supplemental Table 1. Overall, 64.0% of the patients in the TS-1 group and 61.6% in the XELOX group died due to disease progression after recurrence. In the TS-1 group, 3 patients died due to sepsis, 2 patients died due to liver failure, and 2 patients died due to aggravation of chronic lung disease.
Discussion
In this study, we compared the long-term efficacy of the TS-1 and XELOX regimens as adjuvant chemotherapy in patients after curative resection of gastric cancer. The patients who received TS-1 were mostly older and had higher ASA scores than the patients who received XELOX. Furthermore, more patients diagnosed with higher pathologic stage received XELOX than TS-1. However, we did not identify significant differences in DFS or OS between the 2 regimens except in the OS rates of stage IIA and stage IIB patients. Type of regimen during adjuvant chemotherapy (TS-1 vs XELOX) was not an independent prognostic factor of OS or DFS in the entire cohort. On the other hand, type of chemotherapeutic regimen was an independent prognostic factor of OS, along with pathologic stage, in stage II gastric cancer patients.
Recently, there have been studies introducing various adjuvant chemotherapy regimens. In Japan, studies used several options (such as S-1 plus docetaxel, 12 S-1 plus oxaliplatin, 13 and XELOX 14 ) for chemotherapy in stage III gastric cancer based on toxicity profile, pattern of recurrence, and efficacy by histological type. 15 The RESCUE-GC study of China will compare the oncologic efficacies between S-1 monotherapy and SOX. 16
There are a few retrospective studies that showed significant efficacy difference between TS-1 and XELOX. One retrospective multicenter study showed that the 3-year DFS of stage IIIB or IIIC patients who received XELOX was significantly higher than that of those who received TS-1. 17 In another study, XELOX tended to be superior to TS-1 in terms of 3-year DFS of stage IIIC gastric cancer after D2 gastrectomy, although the difference was not statistically significant. Pathologic N stage and cycles of chemotherapy completion were independent prognostic factors of OS. 18 Similar findings were shown in another retrospective study demonstrating that XELOX showed significant benefit for DFS and OS in the pathologic N3 group and stage IIIC patients. 19 In general, the results of previous studies have shown that the 2 regimens have similar efficacies, while monotherapies tended to be less capable of inhibiting recurrence than dual regimens.
On the other hand, a recent study demonstrated that TS-1 and XELOX regimens did not show significant difference in longer than-5-yr DFS after curative gastrectomy in patients with stage II or III gastric cancer. The researchers found that pathologic T and N stages and completion of planned chemotherapy were prognostic factors for recurrence. 20 Although there was a significant difference in OS between XELOX and TS-1 groups of stage II patients in our cohort, there was no significant difference in stages IIIA-IIIC, and no significant difference in DFS was found between the 2 groups at any stage.
These survival discrepancies among previous studies might be due to different AJCC stages (seventh or eighth), treatment compliance of patients, slight differences in planned adjuvant chemotherapy completion rates, dose reduction, and duration of chemotherapy. Currently, 8 courses of S-1 monotherapy is the standard treatment for stage II disease in Japan, 15 based on the results of the JCOG1104 study, which compared 4 versus 8 courses of S-1 monotherapy but did not demonstrate the noninferiority of 4 courses of treatment for stage II patients. 21
This study has limitations. It is a single-center retrospective study; however, it analyzed the largest number of patients in any study to date. There might be selection and information bias due to the study design. Although we preformed multivariate analysis of variables that might affect the prognosis of the analyzed patients, several baseline characteristics such as age, ASA score, extent of gastrectomy, and pathologic stage were not well balanced between 2 study groups, which might affect the rationality and reliability of this study results. Therefore, additional large-scale, multicenter, prospective studies or well-designed clinical trials are needed to confirm these differences in survival. Lastly, the information on adjuvant chemotherapy regimens used in this study is limited. When preparing additional evidence-based studies, clinicians should investigate side effects, compliance rate, duration of chemotherapy, and completeness of planned chemotherapy.
In conclusion, we compared long-term OS and DFS between monotherapy (TS-1) and a combined regimen (XELOX) as adjuvant chemotherapy after D2 gastrectomy. Similar efficacies were for these 2 regimens in each substage, except for OS in stages IIA and IIB. Because type of chemotherapeutic regimen was an independent prognostic factor of OS in stage II patients, the XELOX regimen can be considered as adjuvant chemotherapy for stage II patients.
Supplemental Material
sj-docx-2-tct-10.1177_15330338211039679 - Supplemental material for Comparison of Long-Term Efficacy in S-1 and Capecitabine With Oxaliplatin as Adjuvant Chemotherapy for Patients With Gastric Cancer After Curative Surgery: A Retrospective, Single-Center Observational Study
Supplemental material, sj-docx-2-tct-10.1177_15330338211039679 for Comparison of Long-Term Efficacy in S-1 and Capecitabine With Oxaliplatin as Adjuvant Chemotherapy for Patients With Gastric Cancer After Curative Surgery: A Retrospective, Single-Center Observational Study by Sung E. Oh, Ji Y. An, Min-Gew Choi, Jun H. Lee, Tae S. Sohn and Jae M. Bae in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Author Contributions
Ji Yeong An contributed to conception and critical revision of this study. Sung Eun Oh collected and analyzed the data and drafted the work. Jun Ho Lee, Min-Gew Choi, Tae Sung Sohn, and Jae Moon Bae investigated and resolved questions related to the accuracy or integrity of all parts of the work. All authors gave final approval for this version of the manuscript to be published.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable
Ethics Approval
This study was approved by the Institutional Review Board of Samsung Medical Center, Seoul, Korea (SMC 2020-12-025-001).
Consent to Participate
This investigation waived the requirement for written informed consent from the patients owing to the retrospective study design.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
