Abstract
Background:
Monoclonal antibodies targeting cluster of differentiation (CD) proteins have been incorporated into standard treatments for multiple types of hematologic malignancies, including acute lymphoblastic leukemia (ALL). This systematic review and meta-analysis investigated the efficacy of using CD-targeted antibodies for ALL.
Materials and Methods:
The EMBASE and MEDLINE databases were searched for research papers using immunotherapy- and ALL-related terms from inception to July 2021. Eligible studies were randomized, controlled trials (RCTs) or cohort studies in which ALL patients received CD-targeted immunotherapy or conventional chemotherapy as the induction or salvage therapy. The reports had to report our primary outcomes of interest: overall survival (OS), relapse-free survival (RFS), or complete remission (CR), with the patient number for each outcome. The effect estimates with 95% confidence interval (CI) from each study were combined to calculate the pooled-effect estimate, using the Hantel-Maenszel method.
Results:
Five RCTs and 9 retrospective cohort studies were eligible for the meta-analysis. ALL patients given CD-targeted immunotherapy in the induction or salvage therapy had significantly higher OS and RFS rates than those administered conventional chemotherapy only, with pooled odds ratios (OR) of 2.11 (95% CI, 1.76-2.53; I2, 0%) and 2.25 (95% CI, 1.62-3.14; I2, 61%), respectively. The rates of achieving CR and minimal residual disease negativity were also higher for the immunotherapy group, with pooled ORs of 1.70 (95% CI, 1.07-2.69; I2, 79%) and 2.98 (95% CI, 1.17-7.58; I2, 90%), while developing less risk for febrile neutropenia (pooled OR, 0.22; 95% CI, 0.08-0.58; I2, 84%). Subgroup analyses revealed that all antibody types yielded dramatically better OS rates than those for patients administered chemotherapy alone.
Conclusions:
The ALL patients receiving CD-targeted immunotherapy as induction or salvage therapy had significantly higher response rates and survival outcomes, as well as lower odds of acquiring febrile neutropenia, than the patients given conventional chemotherapy.
Introduction
Acute lymphoblastic leukemia (ALL) is a malignant hematologic neoplasm of the lymphoid progenitor cells. It is the most common form of leukemia in children and the second most common form of acute leukemia in adults, with an annual incidence rate of 1.7 per 100,000 person-years in the USA. 1 It follows a bimodal distribution, with the first peak in childhood and the second around 50-60 years of age. 2
Due to a lack of specific morphology and cytochemistry, immunophenotypic analysis is essential for the diagnosis and classification of ALL patients. For B-lymphoid lineage ALL, the common immunotypic markers are CD19, cytoplasmic CD22, cytoplasmic CD79a, and CD20 with surface immunoglobulin for mature B cells. 3,4 However, none of these immunophenotypes are specific to the disease. Co-expression of the immunophenotypes or strong intensity of markers help improving the accuracy of the diagnosis. The majority of cases show that the lymphoblasts express CD10, surface CD22, CD24, PAX5, and TdT. 5 Cytoplasmic CD3, which is considered lineage-specific, and CD7 are common immunotypic markers for T-lymphoid lineage ALL. Both CD4 and CD8 are often positive on the T cell lymphoblast, but not exclusively specific for T-ALL. 3,4,6 The positivity of markers depends on the level of differentiation of the leukemic cells, such as pro B-ALL with CD19, cCD79a, cCD22, and nuclear TdT, common B-ALL with CD10, and pre B-ALL with cytoplasmic μ chain. 7
Despite the widely-known efficacies of the current chemotherapeutic regimens for pediatric ALL, with 5-year survival rates exceeding 90%, the adult survival rate is much lower (35%-45%). 8 -11 As a well-established cornerstone of the treatment of ALL, several conventional chemotherapeutic regimens generally have fair outcomes, but with poorer outcomes for high-risk genetic profile cases, such as Philadelphia chromosome (Ph)-like ALL with 5-year event-free survival of 20% and 5-year overall survival of 23%. 1,12 -15
Immunotherapy has contributed to major advances in the treatment of various hematologic malignancies as a result of several surface antigens being expressed on malignant cells, including ALL. Multiple agents which target cell surface proteins, especially cluster of differentiation (CD) proteins such as CD19, CD20, and CD22, have been proved to generate favorable outcomes in ALL patients. This has led to a gradual increase in 5-year survival rates, and the agents have consequently recently been incorporated in the standard of care guidelines for ALL patients. 2,16 -19 The mechanisms of monoclonal antibody-based drugs are antibody-dependent cytotoxicity, complement activation, and induction of apoptosis. 5 With the development of techniques, a monoclonal antibody-based drug can now be combined with a radioisotope such as ibritumomab tiuxetan, which is linked to yttrium-90 20 or a cytotoxic agent such as inotuzumab ozogamicin (InO) which is combined with calicheamicins, 19 or be bound to 2 different types of antigen at the same time to form what is termed a bispecific monoclonal antibody like blinatumomab. 21 -23 The summary of immunotherapy drugs in ALL is provided in Supplementary Table S1.
However, there has been no systematic review and meta-analysis comparing the efficacies and toxicities of chemotherapy alone with those of immunotherapy with/without conventional chemotherapy. The present systematic review and meta-analysis was therefore conducted to summarize the available data and compare the efficacies and toxicities of these 2 therapeutic strategies during the induction or salvage therapy.
Methods
Data Sources and Searches
Two investigators (B.P., P.V.) individually searched all published studies indexed in the EMBASE, MEDLINE, PubMed, and ScienceDirect databases from inception to June 2021. The search terms drew upon terms associated with immunotherapy and ALL. Supplementary Data 1 details the exhaustive search strategy lists that were utilized. The study was performed in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analysis) statement 24 and the study protocol was registered with the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY) (registration number INPLASY202170011).
Selection Criteria
To qualify for the meta-analysis, studies had to be either randomized, controlled studies (RCTs) or cohort studies (prospective or retrospective), and have 2 groups of ALL patients: 1 group which received CD-targeted immunotherapy for the induction or salvage therapy, and another group which did not receive any CD-targeted immunotherapy for the induction or salvage therapy. For both groups, the studies needed to have reported our primary outcomes of interest—overall survival (OS), relapse-free survival (RFS), or complete remission (CR)—by reporting the number of patients in each group for each outcome. The secondary outcomes of interest were minimal residual disease (MRD) negativity and grade 3-5 febrile neutropenia, details of which were gathered if they were available. Study eligibility was individually examined by 3 investigators (B.P., P.V., W.O.); disagreements were resolved by consensus.
Data Extraction
A standardized data collection form was used to extract the following data: last name of the first author; year of publication; number of participants in each group; number of male and female participants in each group; average participant age for each group; disease statuses of the included participants; chemotherapeutic regimens employed; type and dose of immunotherapy used during the induction; countries where the studies were conducted; study period (years); and number of participants with outcomes of interest in each group.
Definitions of Outcomes
CR was defined as a patient who had <5% of bone marrow lymphoblasts, the absence of circulating blasts or extramedullary disease, an absolute neutrophil count >1.0 × 109/L, and a platelet count >100 × 109/L. 16 OS rate was defined as the percentage of patients who were still alive after diagnosis at a particular time of interest. RFS rate was defined as the proportion of patients who had CR without relapse or death at a particular time of interest. 13,14,20 -22 For the OS and RFS rates, the time of interest was the longest of 6 months, 1 year, 2 years, 3 years, or 4 years with available results in each study. 17 -19,25 -33 The MRD negativity rate was defined as either <0.01% bone marrow lymphoblasts, confirmed by cytometry or immunoglobulin, or T-cell receptor gene rearrangements in bone marrow samples. 17,19,25,26,30,32 Finally, febrile neutropenia was defined as an absolute neutrophil count <1.0 × 109/L, and either a single temperature >38.3° C or a sustained temperature of at least 38.0° C for more than 1 hour, according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events (version 4.0). 34
Quality Assessment
Two investigators (P.V., W.O.) evaluated the quality of each study, using the Jadad scale for RCTs and the Newcastle-Ottawa scale for cohort studies. 35,36
Statistical Analysis
Review Manager 5.3 software (The Cochrane Collaboration, United Kingdom) was used for all statistical analyses. The pooled odds ratio (OR) and the associated 95% confidence interval (CI) was calculated by combining the effect estimates and 95% CI from each study using the Mantel-Haenszel method. 37 As it is likely that there could be interstudy heterogeneity, a random-effect model was used instead of a fixed-effect model. Statistical heterogeneity was determined using Cochran’s Q test, accompanied by the heterogeneity (I 2 ) statistic. The I 2 statistical value quantifies the proportion of the total variation across studies which is explained by study heterogeneity more than by random chance, with I 2 values of 0%-25% representing insignificant heterogeneity, 26%-50% low heterogeneity, 51%-75% moderate heterogeneity, and >75% high heterogeneity. 38 The presence of publication bias was to be visualized by a funnel plot if there was a sufficient number of eligible studies for the meta-analysis. A subgroup analysis based on the target of immunotherapy, disease status, and age groups were also to be performed if there were enough studies. However, funnel plots and Egger’s test were eventually not used to evaluate the publication bias due to the limited number of included studies (less than 10) for each outcome.
Results
Search Results
The systematic search in the EMBASE, MEDLINE, PubMed, and ScienceDirect databases identified 25,977 articles, from which 10,558 duplicates were removed. This resulted in 15,419 articles for the title and abstract review. Subsequently, 15,265 articles were excluded as the article type and study design did not fulfill the inclusion criteria, leaving 154 articles for a full-length article review. 140 of those were discarded as they lacked the outcomes of interest. Finally, 14 eligible studies (5 RCTs 17 -19,25,26 and 9 retrospective cohort studies 27 -33,39,40 ) fulfilled the eligibility criteria for our meta-analysis. Of those, 7 compared blinatumomab (a CD3/CD19 bispecific T-cell engager) to chemotherapy 18,25,27 -29,39,40 ; 4 compared rituximab (an anti-CD20 monoclonal antibody) to chemotherapy, 17,26,30,31 1 compared epratuzumab (an anti-CD22 monoclonal antibody) to chemotherapy 32 ; and 2 compared (InO; an anti-CD22 antibody-drug conjugate) to chemotherapy 19,33 ; Figure 1 illustrates the full literature review and selection process.

Study identification and literature review process.
Baseline Patient Characteristics
All 14 included studies were composed of 1,596 patients who received any of the CD-targeted immunotherapies as induction or salvage therapy, and another 2,764 patients who did not receive any CD-targeted immunotherapy for the induction or salvage therapy. The age of the participants varied greatly (immunotherapy group: 1 to 81 years, and chemotherapy group: 1 to 83 years). This was because some studies recruited only pediatric and adolescent cases whereas others recruited only elderly patients. Thirteen of the eligible studies included only B-cell precursor ALL patients, 17 -19,25 -32,39,40 while 7 recruited only Ph-negative ALL patients. 17,18,27,29,30,33,40 Moreover, 4 of the eligible studies enrolled only de novo patients, while 6 included relapsed, refractory patients. 18,19,27,28,39,40 The study characteristics and quality assessment results are summarized in Tables 1 to 3.
Characteristics and Participants of Studies That Compare Anti-CD19 Therapy to Conventional Therapy.

Abbreviations: ALL, Acute lymphoblastic leukemia; AYAs, Adolescents and young adults; BCP, B-cell precursor; CD, Cluster of differentiation; CMT, Chemotherapy; CR, Complete response; CRh, Complete response with partial hematologic recovery; CVT, Conventional therapy; FLAG, Fludarabine cytarabine and filgrastim; GMALL, German multicenter study group for adult ALL; GRAALL, Group for Research on Adult Acute Lymphoblastic Leukemia; HD-MTX, High-dose methotrexate; HiDAC, High-dose cytarabine; MRD, Minimal residual disease; M/F, Male/Female; NA, Not available; NILG, Northern Italy Leukemia Group; OS, overall survival; Ph, Philadelphia chromosome; RCT, Randomized controlled trial; R/R, Relapsed/refractory; TKI, Tyrosine kinase inhibitor; UKALLR, United Kingdom Acute Lymphoblastic Leukemia Regimen; USA, United States of America.
Characteristics and Participants of Studies That Compare Anti-CD20 Therapy to Conventional Therapy.
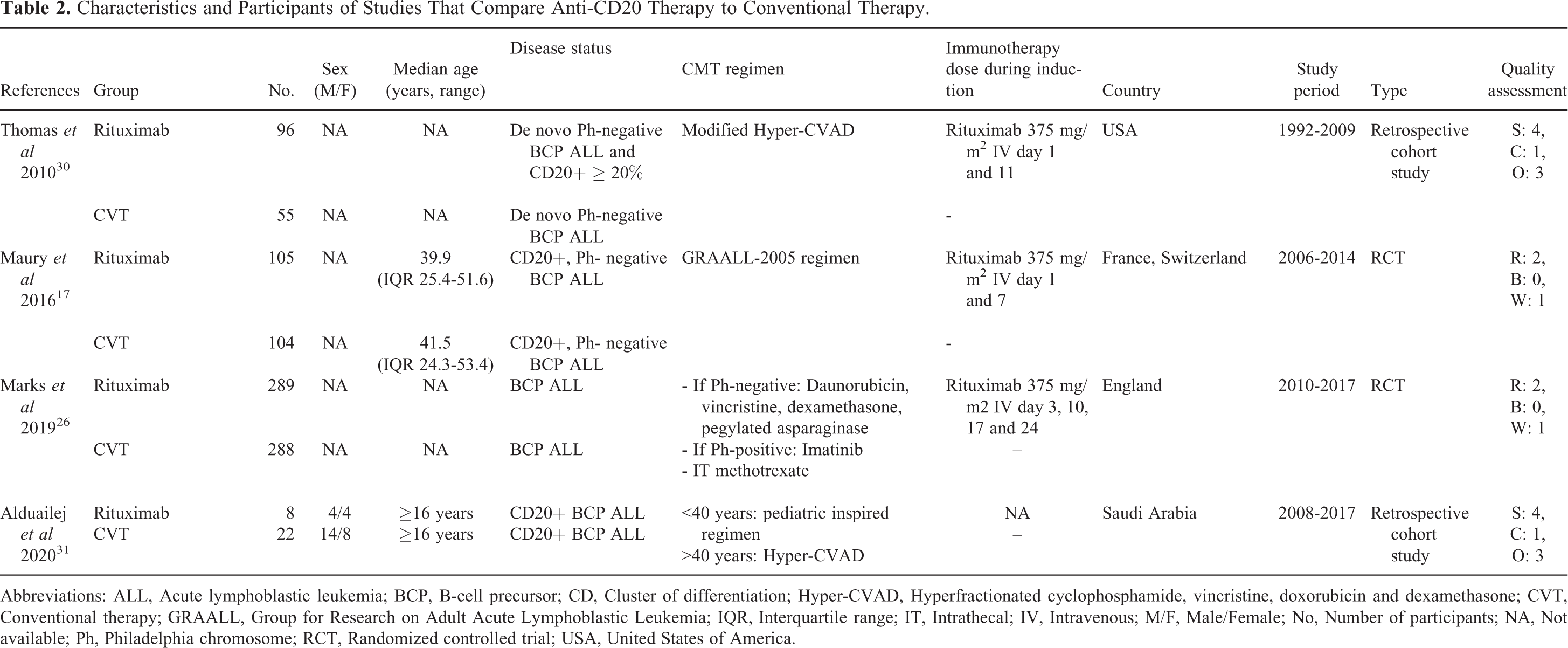
Abbreviations: ALL, Acute lymphoblastic leukemia; BCP, B-cell precursor; CD, Cluster of differentiation; Hyper-CVAD, Hyperfractionated cyclophosphamide, vincristine, doxorubicin and dexamethasone; CVT, Conventional therapy; GRAALL, Group for Research on Adult Acute Lymphoblastic Leukemia; IQR, Interquartile range; IT, Intrathecal; IV, Intravenous; M/F, Male/Female; No, Number of participants; NA, Not available; Ph, Philadelphia chromosome; RCT, Randomized controlled trial; USA, United States of America.
Characteristics and Participants of Studies That Compare Anti-CD22 Therapy to Conventional Therapy.

Abbreviations: ALL, Acute lymphoblastic leukemia; AraC, Cytarabine; BCP, B-cell precursor; CD, Cluster of differentiation; COG, Children’s Oncology Group; CVT, Conventional therapy; FLAG, Fludarabine, cytarabine and filgrastim; Hyper-CVAD, Hyperfractionated cyclophosphamide, vincristine, doxorubicin and dexamethasone; HiDAC, High-dose cytarabine; MXN, mitoxantrone; NA, Not available; Ph, Philadelphia chromosome; RCT, Randomized controlled trial; R/R, Relapsed/refractory; USA, United States of America.
Drug Regimens Used During Induction or Salvage Therapy
Various chemotherapeutic regimens were used for the induction or salvage therapy, with the most common being hyper-CVAD (hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone); FLAG (fludarabine, cytarabine, and granulocyte-stimulating factor), with or without anthracycline; high-dose cytarabine; high-dose methotrexate; and pediatric-inspired regimens. A group of Ph-positive ALL patients in an included study received 600 mg/day of imatinib continuously. 26
The immunotherapy regimens differed for each drug. For rituximab, all studies administered 375 mg/m 2 per dose. As to blinatumomab, most studies administered 9 μg/day for the first 7 days, and 28 μg/day via continuous intravenous infusion over the following 4 weeks. Epratuzumab was administered at 360 mg/m 2 per dose, while InO was administered either at 1.3 or 1.8 mg/m 2 (as described by Jabbour et al), 33 or 0.8 mg/m 2 on Day 1 followed by 0.5 mg/m 2 on Days 8 and 15 (as described by Kantarjian et al). 19
Outcomes of CD-Targeted Immunotherapy as Induction or Salvage Therapy on ALL Patients
The OS rates were reported as a 6-month rate by 1 study, 18 a 1-year rate by 2 studies, 27,28 a 2-year rate by 3 studies, 19,25,29 a 3-year rate by 3 studies, 30,31,33 and a 4-year rate by 1 study. 17 RFS rates were reported as a 6-month rate by 1 study, 18 a 2-year rate by 2 studies, 25,29 a 3-year rate by 3 studies, 26,30,33 and a 4-year rate by 1 study. 17 The pooled meta-analysis found that OS and RFS were significantly better in patients who received immunotherapy, with pooled ORs of 2.11 (95% CI, 1.76-2.53; I 2 , 0%) and 2.25 (95% CI, 1.62-3.14; I 2 , 61%), respectively. Patients who received any CD-targeted immunotherapy in the induction or salvage therapy also had 1.70-fold odds of obtaining CR compared to those who did not receive any immunotherapy (95% CI, 1.07-2.69; I 2 , 79%). 17 -19,26 -28,31 -33,39,40 In addition, the rate of achieving MRD negativity was significantly higher for the patients who received any immunotherapy (pooled OR, 2.98; 95% CI, 1.17-7.58; I 2 , 90% 17,19,25,26,30,32 ; Figure 2A-D).

Forest plot of the meta-analysis of incorporating immunotherapy into the induction or salvage therapy versus chemotherapy only. (A) OS rate; (B) RFS rate; (C) CR rate; and (D) rate of MRD negativity.
Interestingly, the immunotherapy treatment carried fewer odds of acquiring febrile neutropenia and thrombocytopenia (pooled OR, 0.22; 95% CI, 0.08-0.58; I 2 , 84% 19,25,32 and 0.38; 95% CI, 0.25-0.57; I 2 , 0%, 18,19 respectively). The infection and discontinuation rates favored immunotherapy treatment but without statistical significance (pooled OR, 0.62; 95% CI, 0.24-1.64; I 2 , 92% 17 -19,25,32 and 0.71; 95% CI, 0.15-3.31; I 2 , 88%, 18,19 respectively; Figure 3A-D). Only 1 study reported the incidence of anemia in both arms in which the rate was decreased in the immunotherapy arm. 19

Forest plot of the meta-analysis of incorporating immunotherapy into the induction or salvage therapy versus chemotherapy only. (A) infection rate; (B) febrile neutropenia rate; (C) thrombocytopenia rate; and (D) discontinuation rate.
Subgroup Analysis Based on Type of Immunotherapy Used in the Induction or Salvage Therapy
Subgroup analyses were performed based on the immunotherapy type (anti-CD19, anti-CD20, or anti-CD22). Greater survival outcomes were achieved in patients who received blinatumomab in the induction or salvage therapy, including OS (pooled OR, 2.02; 95% CI, 1.63-2.52; I 2 , 0%) 18,25,27 -29 and RFS (pooled OR, 2.77; 95% CI, 2.00-3.84; I 2 , 0%). 18,25,29 The immunotherapy patients had higher odds of achieving CR with near-significance (pooled OR, 1.61; 95% CI, 0.99-2.63; I 2 , 70% 18,27,28,39,40 ; Figure 4A-C).

Forest plot of the meta-analysis of incorporating blinatumomab into the induction or salvage therapy versus chemotherapy only. (A) OS rate; (B) RFS rate; and (C) CR rate.
Patients who received rituximab in the induction or salvage therapy had significantly greater rates of OS (pooled OR, 2.27; 95% CI, 1.24-4.16; I
2
, 36%)
17,30,31
and RFS (pooled OR, 1.87; 95% CI, 1.07-3.25; I
2
, 72%).
17,26,30
However, although the odds of achieving CR (pooled OR, 1.40; 95% CI, 0.81-2.43; I
2
, 0%)
17,26,31
and MRD negativity (pooled OR, 1.48; 95% CI, 0.72-3.03; I
2
, 72%)
17,26,30
were also better, they did not reach statistical significance

Forest plot of the meta-analysis of incorporating rituximab into the induction or salvage therapy versus chemotherapy only. (A) OS rate; (B) RFS rate; (C) CR rate; and (D) rate of MRD negativity.
In the case of anti-CD22 immunotherapy, a significantly greater rate of OS (pooled OR, 2.53; 95% CI, 1.58-4.06; I 2 , 0%) 19,33 with a lower rate of febrile neutropenia (pooled OR, 0.36; 95% CI, 0.24-0.52; I 2 , 0%) 19 , 32 was observed with the immunotherapy group. Moreover, the odds of achieving CR (pooled OR, 1.98; 95% CI, 0.40-9.76; I 2 , 93%) 19,32,33 and MRD negativity (pooled OR, 3.88; 95% CI, 0.70-21.48; I 2 , 85%) 19,32 were also elevated, but they were not statistically significant (Figure 6A-D).

Forest plot of the meta-analysis of incorporating anti-CD22 antibody into the induction or salvage therapy versus chemotherapy only. (A) OS rate; (B) CR rate; (C) rate of MRD negativity; and (D) febrile neutropenia rate.
Subgroup Analyses
Two subgroup analyses were performed for relapsed/refractory ALL patients and adult ALL patients. For relapsed/refractory ALL patients, better OS, RFS and CR rates were achieved in those who received blinatumomab compared to conventional chemotherapy (Supplementary Data 2), 18,25,27,28 while those who received anti-CD22 treatment attained CR rate similar to the full analysis (Supplementary Data 2). 19,32 However, subgroup analysis could not be performed for rituximab since all of the patients were newly-diagnosed.
For adult patients, similar OS, RFS, and CR rates compared to the main analysis were observed. Infection and febrile neutropenia rates also appeared to be similar. However, the statistical significance of MRD negativity rate was lost (pooled OR of 2.13; 95% CI, 0.59-7.72) due to the reduced number of studies (Supplementary Data 3). 17 -19,26 -29,33
Discussion
This meta-analysis is the first to review all available RCTs and cohort studies to compare the outcomes of patients who received CD-targeted immunotherapy with those of patients given conventional chemotherapy. We found that all of the immunotherapy data available in the studies was significantly associated with better OS and RFS rates. Through the pooled analysis, we found that the use of immunotherapy as a part of the induction or salvage therapy also increased the odds of obtaining CR and MRD negativity while reducing the odds of acquiring febrile neutropenia. According to the previously published meta-analyses, blinatumomab 41 and InO 42 are effective for acute lymphoblastic leukemia with limited toxicity. However, these studies considered both patients with acute lymphoblastic leukemia and B-cell non-Hodgkin lymphoma and did not compare the outcomes with conventional chemotherapy therapy. 41,42 Moreover, other types of CD-targeted immunotherapy which were not previously conducted were also taken into consideration in our study.
There are several reasons why these novel agents contribute to better outcomes. CD19 and CD22 were shown to be vastly expressed in all subtypes of B-cell ALL. 43 -45 There is a considerable amount of evidence that blinatumomab can bind to T-cells and tumor cells, forming a complex. This induces the T-cells to start the apoptosis cascade, even without T-cell receptor specificity or without major histocompatibility complex class I molecules that have exceptionally high potency. 46 Moreover, cells without CD19 were also found not to be affected by the drug. 47 -49 Blinatumomab is also known to increase inflammatory cytokine production, such as IL-2, which plays a major role in T and NK cell cytolytic activity. 50,51 A hypothesis that may explain the subgroup analysis of blinatumomab showing better results for OS, RFS, and CR with statistical significance is that blinatumomab has multiple action pathways compared to other CD-targeted monoclonal antibodies. 46 For instance, sequential blinatumomab and hyper-CVAD induced CR rate for 100% with 2-year remission and OS rates at 79% and 86%, respectively, in newly diagnosed Ph-negative B-ALL from a phase 2 study. 52 As to antibodies targeting CD22, InO, being an antibody-drug conjugate, induced apoptosis mainly by the toxic effector molecule being bound to the antibody. 53,54 In contrast, epratuzumab has been shown to be a B-cell activation and signaling modulator, and it can partially stimulate antibody-dependent cellular cytotoxicity. 55
While CD20 was only fairly expressed in B-cell ALL, it is widely known that rituximab can induce apoptosis by triggering protein tyrosine kinases and proapoptotic proteins such as caspases. 43,44,56 -58 CD20 is also known to induce complement-mediated cytotoxicity and antibody-dependent cellularity which, in turn, eliminates malignant B-cells. 58 -61
Regarding the relative efficacies of the drugs, mixed results were found from indirect comparisons between blinatumomab and InO from TOWER and INO-VATE-ALL trials in terms of their CR and OS rates. 62,63 Therefore, additional, direct, head-to-head studies need to be performed as there is limited data to make comparisons of the immunotherapies.
Apart from the immunotherapy treatments included in this meta-analysis, a chimeric antigen receptor (CAR) T-cells therapy is an appealing therapeutic option for ALL. Tisagenlecleucel is an anti-CD19 CAR T-cell therapy which was approved for relapsed/refractory B-ALL patients, with a 3-month overall remission rate of 81%. The rates of 12-month event-free survival and OS were 50% and 76%, respectively. 64 Additionally, in a preclinical study, anti-CD19 CARs can successfully transduce hematopoietic stem cells from human umbilical cord blood and lead to CAR expression on myeloid cells, T cells, and NK cells in an animal model. 65 This advanced cellular therapy will improve the outcome of the treatment in the near future.
Regarding the methods for combining the dichotomous outcomes for the meta-analysis, the Mantel-Haenszel method is generally preferred due to its strength of the ability to combine several types of outcomes, such as odds ratios, risk ratios, or risk differences, while maintaining its statistical properties when there are few events. Moreover, it can analyze the pooled outcomes even when no events were observed in one or both arms, in contrast to the inverse-variance methods. On the other hand, it is susceptible to zero-correction bias toward no difference and over-estimating variances of study estimates when there exist studies with no events in one or both groups. Note that although the Peto method does not have zero-correction bias, it is beneficial only when the 3 criteria, which are, small intervention effects, rare events (<1%), and similar numbers in each group of the study, are met. Therefore, the Peto method is not recommended as a default approach and is generally used only when such criteria are satisfied. 37,66
Our results should be cautiously interpreted as there are some study limitations. First, approximately half of the eligible studies are retrospective cohort studies that used a historical cohort as a group without immunotherapy treatment; this might create bias. Second, there was high statistical heterogeneity, conceivably due to the differences in the design, population characteristics, and chemotherapeutic regimens of the eligible studies. Moreover, 3 of the studies included pediatric or adolescent cases which tend to have better outcomes compared to adult cases. However, due to the scarce data available for pediatric cases, more studies are warranted to investigate the outcomes solely in this patient population. Finally, due to the limited number of studies, some adverse events and subgroup analyses might be underpowered and be the cause of non-significance for several results. Therefore, more studies on the usage of immunotherapy as a part of the induction or salvage therapy and the outcomes of ALL patients are certainly demanded.
Conclusions
The current systematic review and meta-analysis illustrated that patients with ALL who received CD-targeted immunotherapy in the induction or salvage therapy had significantly higher OS and RFS rates plus reduced odds of acquiring febrile neutropenia, relative to those given conventional chemotherapy. The use of immunotherapy as a part of the induction or salvage therapy also increased the odds of obtaining CR and MRD negativity. While these findings suggest that antibody-based immunotherapy is an attractive choice for the treatment of ALL, more studies are required to determine the significance of the outcomes and toxicities of this type of treatment.
Supplemental Material
Supplemental Material, sj-docx-1-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-docx-1-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-2-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-2-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-3-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-3-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-4-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-4-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, sj-pdf-5-tct-10.1177_15330338211037434 - Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-5-tct-10.1177_15330338211037434 for Efficacy of Targeted Immunotherapy as Induction or Salvage Therapy in Acute Lymphoblastic Leukemia: A Systematic Review and Meta-Analysis by Ben Ponvilawan, Pongthep Vittayawacharin, Pattaraporn Tunsing and Weerapat Owattanapanich in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
All authors designed the study. Ben Ponvilawan, Pongthep Vittayawacharin, and Weerapat Owattanapanich collected the data. Weerapat Owattanapanich performed the statistical analyses. Ben Ponvilawan drafted the manuscript and prepared the final version. Weerapat Owattanapanich and Pongthep Vittayawacharin made critical revisions. All authors read and approved the final manuscript. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. Consent for publication: Not applicable because this study does not directly involve human subjects. The need for ethics approval by an institutional board review was waived as this study does not directly involve human subjects. The study protocol was registered with the International Platform of Registered Systematic Review and Meta-Analysis Protocols (INPLASY) (registration number INPLASY202170011).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
