Abstract
Objective:
To investigate the cellular and molecular epidemiology of acute leukemias in vulnerable populations of children and adolescents in Oaxaca de Juarez, Mexico.
Material and Methods:
Descriptive, cross-sectional and retrospective study, conducted from 2014 to 2018 in which profiles of molecular and immunophenotypic aberrations were investigated in children and adolescents diagnosed with acute leukemia, by evaluating 28 molecular abnormalities by HemaVision-Q28 multiplex RT-PCR kit and standardized EuroFlow Immunophenotyping of bone marrow cells.
Results:
We included 218 patients, with 82.5% younger than 14 years and 17.5% adolescents. The median age was 9 years and a main peak of incidence was recorded at age of 4 to 5 years. B-cell acute lymphoblastic leukemia was diagnosed in 70.64% of all cases, acute myeloid leukemia was in 22.48%, T-cell acute lymphoblastic leukemia in 6.42%, and mixed lineage acute leukemia in 0.46% of cases. Overall, chromosomal translocations were positive in 29.82% of cases. While 65.31% of patients with acute myeloid leukemia reported aberrancies, only in 18.83% of B-cell acute lymphoblastic leukemia cases genetic abnormalities were obvious. Surprisingly, most prevalent translocations in B-cell acute lymphoblastic leukemia were t(9;22) in 20.7%, followed by t(4;11) in 17.2% and t(6;11) in 13.8%, whereas patients with acute myeloid leukemia showed t(15;17) in 40.6% and t(8;21) in 21.9%. In contrast, an homogeneous expression of t(3;21) and t(6;11) was recorded for T-cell acute lymphoblastic leukemia and mixed lineage acute leukemia cases, respectively. Except for t(1;19), expressed only by pre-B cells, there was no association of any of the studied translocations with differentiation stages of the B-leukemic developmental pathway.
Conclusion:
Our findings identify near 50% of patients with acute lymphoblastic leukemia at debut with high-risk translocations and poor prognosis in B-cell acute lymphoblastic leukemia as well as an unexpected increase of acute myeloid leukemia cases in young children, suggesting a molecular shift that support a higher incidence of poor prognosis cases in Oaxaca.
Keywords
Introduction
Acute leukemias are a group of very heterogenous hematological disorders, exhibiting differences in morphology, immunophenotype, cytogenetic and chromosomal aberrancies, biological and clinical behavior, treatment response, and prognosis. 1,2 Diagnosis must be addressed by integral analysis of lab data and clinical criteria, considering the lower limit for the percentage of malignant blasts should be higher than 20%. 3
Historically, important discoveries in cancer research has been established in leukemia, as the description of first chromosomal aberration, 4 first selective cancer therapy by using tyrosine-kinase inhibitors, 5 discovery of cancer stem cells, 6 role of tumor microenvironment during malignancies 7,8 and more recently, immunotherapy approaches to eliminate leukemic cells. 9
Globally, survival rates and leukemia incidences have increased due the opportune diagnosis, better risk stratification, enhanced clinical management, and monitoring of minimal/measurable residual disease (MRD). 10 Cases of pediatric and adolescent acute leukemias have increased during the last years and deaths still occurring even upon a good therapy management in patients from Mexico and Latin America. 11 Novel Health metrics as disability-adjusted life year (DALY) which is a measure of burden diseases per population appointed childhood cancer with the highest proportions in Central Latin America; as 90% of children live in low- and middle-income countries as is the case of Mexico, DALYs proportion is even higher. 12 Interestingly, in developed countries, overall survival rate is around 80% where Hispanics have highest rates and leukemias with worse prognosis, whereas the genetic vulnerability, environmental, and immune surveillance factors behind of them are still under research. 13
Fundamental classification identifies myeloid, lymphoid, and ambiguous lineage leukemias (previously termed as mixed phenotype leukemias) where the most frequent entity is B-cell acute lymphoblastic leukemia (B-ALL).
14
More recent, multiparametric approaches have allowed the characterization of hidden entities as early-T progenitor (ETP) leukemia.
15
Important criteria for patient stratification is age, number of peripheral blood leukocytes, and cytogenetics. While genetic abnormalities can be detected in more than 20% of patients with B-ALL mostly by implementing conventional karyotype and polymerase chain reaction (PCR) and sequencing technologies, the rest still remaining.
16
Based on the molecular profiling, several subtypes can be identified and confer a risk factor.
17,18
Most frequent B-ALL abnormalities include ETV6-RUNX1, BCRL-ABL, and E2A-PBX1 and MLL rearrangements as well as hyper- and hypodiploidy.
1,19
Habitually, translocation t(12;21)(p13;q22)/ETV6-RUNX1(TEL-AML1) is the most common rearrangement reported in B-ALL with favorable prognosis while t(9;22) (q34;q11.2)/BCR-ABL1 occurs in young adult patients (20%-30%) with a very low frequency in children (1%-3%) and provide poor prognosis.
19,20
BRC-ABL fusion protein has reveal its oncogenic ability to initiate leukemia, it has different breakpoints: p190BCR/ABL e1a2/e3a2 and p210BCR/ABL b2a2/b3a2, where childhood leukemias exhibit p190 more often.
21
There are patients with similar transcriptomic profile to those to have Philadelphia (Ph) chromosome-positive but lacks oncoprotein and are called as Ph-like.
22
Remarkably, Ph-like patients have shown abnormalities in IKZF1, TCF3, EBF1, PAX5, and VPREB1 with resistance to
Nevertheless, the major clinical challenge in leukemia is relapse, which occurs in around 30% in pediatric patients with acute lymphoblastic leukemia (ALL). 24 -26 According to clinical criteria, more than 50% of ALL Mexican patients are classified as high risk of relapse. Interestingly, relapse frequency in Mexican standard-risk patients is 55%, suggesting a substratification of these patients. 16
In this study, we aim to unravel the cellular and molecular profiles of acute leukemias in Oaxaca, focused on biomolecular risk factors associated with poor prognosis that may contribute the increasing incidence of relapse and early death cases in vulnerable populations of the region.
Materials and Methods
Biological Specimens
Bone marrow aspirates were obtained from 218 pediatric patients with acute leukemia suspect as a part of the routine protocol for diagnosis from the Hospital de la Niñez Oaxqueña and Hospital Regional Presidente Juárez ISSSTE under parents informed consent. This study was approved by the ethics committee at the Hospital Infantil de México Federico Gómez (HIM72012/035 SSA.1060). All procedures were conducted according to the World Medical Association Declaration of Helsinki. Age, gender, and patient characteristics are shown in Supplementary Table 1.
Immunophenotype of Acute Leukemias
Flow cytometry staining was performed with the standardized EuroFlow ALOT (acute leukemia orientation tube) antibody panel (CD45-OC515, CD3-Pacific Blue, CyMPO-FITC, CyCD79a-PE, CD34-PerCP/Cy5.5, CD19-PE/Cy7, CD7-APC, SmCD3, APC/C750) and further tubes 1-3, 27 samples were acquired by using a FACSCanto II (Becton Dickinson), and data were analyzed by using Infinicyt 2.0 (Cytognos).
RNA Extraction
Total RNA was extracted from whole bone marrow samples by using the InviTrap Spin Cell RNA Mini Kit according to the manufacture recommendations.
Molecular Translocations
Screen for 28 common molecular abnormalities were determined by multiplex real-time polymerase chain reaction (RT-PCR) by using HemaVision-Q28 (DNA Diagnostic) according to the manufacture recommendations. This platform include detection of del1(p32)(STIL-TAL1), t(1;11)(p32;q23)(MLL-EPS15), t(1;11)(q21;q23)(MLL-MLLT11), t(1;19)(q23;p13)(TCF3-PBX1), t(3;5)(q25;q34)(NPM1-MLF1), t(3;21)(q26;q22)(RUNX1-MECOM), t(4;11)(q21;q23)(MLL-AFF1), t(5;12)(q33;p13)(ETV6-PDGFRB), t(5;17)(q35;q21)(NPM1-RARA), t(6;9)(p23;q34)(DEK-NUP214), t(6;11)(q27;q23)(MLL-MLLT4), t(8;21)(q22;q22)(RUNX1-RUNX1T1), t(9;9)(q34;q34)(SET-NUP214), t(9;11)(p22;q23)(MLL-MLLT3), t(9;12)(q34;p13)(ETV6-ABL1), t(9,22)(q34;q11)(BCR-ABL1), t(10;11)(p12;q23)(MLL-MLLT10), t(11;17)(q23;q21)(MLL-MLLT6), t(11;17)(q23;q21)(ZBTB16-RARA), t(11;19)(q23;p13.1)(MLL-ELL), t(11;19)(q23;p13.3)(MLL-MLLT1), t(12;21)(p13;q22)(ETV6-RUNX1), t(12;22)(p13;q11)(ETV6-MN1), t(15;17)(q24;q21)(PML-RARA), inv(16)(p13;q22)(CBFB-MYH11), t(16;21)(p11;q22)(FUS-ERG), t(17;19)(q22;p13)(TCF3-HLF), and t(X;11)(q13;q23)(MLL-FOXO4).
Data Analysis
Data analysis were performed using Prism 6.0 statistic software.
Results
Immunophenotype and Characteristics of Local Pediatric Leukemias
A total of 218 pediatric and adolescent leukemia cases presented during 2014 to 2018 for diagnosis and follow-up were included in this study. The median age of general pediatric population was 9 years. Interestingly, 82.5% were younger than 14 years and 17.5% were adolescents. Of all cases, most frequent leukemia entity is B-ALL (70.64%, n = 154), followed by myeloid leukemias (22.48%, n = 49), T-cell acute lymphoblastic leukemia (T-ALL; 6.42%, n = 14), and mixed-linage leukemia (0.46%, n = 1; Figure 1A). Neither children nor adolescent patients presented differences in leukemia subtypes or gender, however, we noted that 80% of the childhood T-ALL were male (Supplementary Figure 1A and B and Table 1). Incidence in pediatric male and female was practically the same (46.94% and 53.06%, respectively). Interestingly, we identified a peak for leukemia incidence at age of 4 to 5 years with more than 30 cases, this age was the same for acute myeloid leukemia (AML), however, we noted a slight incidence of T-ALL in older patients (between 8 and 9 years of age). B-cell acute lymphoblastic leukemia exhibited 2 main peaks of incidence, one at 6 to 7 years and other at age of 10 to 11 years (Figure 1B) and still being the more frequent childhood leukemia, however, myeloid entities have become more recurrent during puberty.
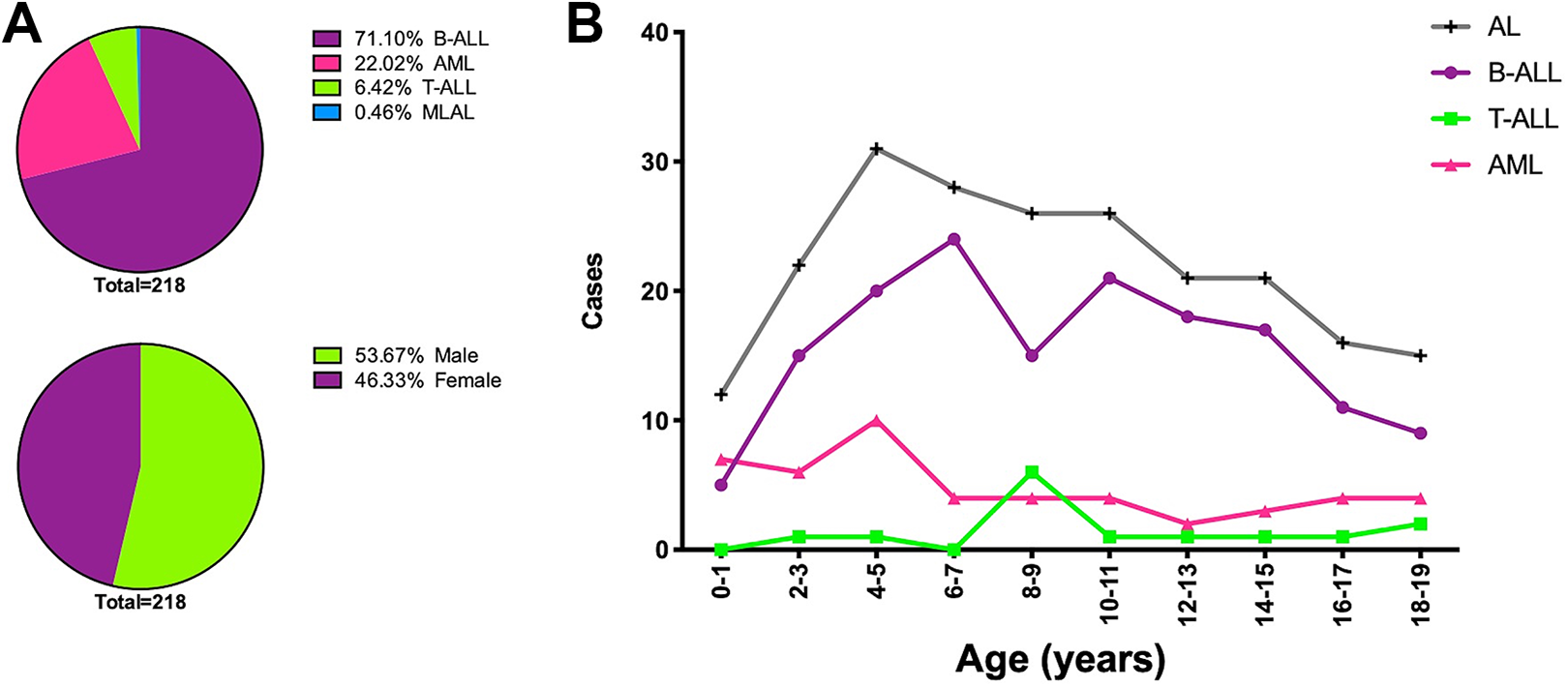
Distribution of acute leukemias (AL) by their immunophenotype. a) Case frequencies and gender distribution of B-cell and T-cell acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML) and mixed-lineage acute leukemia (MLAL) in pediatrics from Oaxaca. b) Childhood B-cell and T-cell acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML) and mixed-lineage acute leukemia (MLAL) by age are shown.
Childhood and Adolescent Leukemia Frequencies.

Abbreviations: AML, acute myeloid leukemia; B-ALL, B-cell acute lymphoblastic leukemia; MLAL, mixed-linage acute leukemia; T-ALL, T-cell acute lymphoblastic leukemia.
High Frequency of BCR-ABL Rearrangements in Pediatric B-ALL
In all, 28 major molecular aberrancies reported for acute leukemias were investigated by using a multiplex RT-PCR approach. Of all explored chromosomal translocations, we detected molecular aberrancies only in 29.82% of cases (Figure 2A). Interestingly, we found that 65.31% of cases with AML resulted positive for chromosomal rearrangements, specially t(15;17) (PML-RARα) followed for t(8;21) (AML1-ETO) and t(9;22) (BCR-ABL). In contrast, only 18.83% and 21.43% were found in B-ALL and T-ALL, respectively (Figure 2B). Surprisingly, the most common translocation in B-ALL was t(9;22) in 20.7%, followed by t(4;11) in 17.2% (Figure 2C and Table 2). Moreover, a number of translocations commonly associated with myeloid malignancies were found in patients with B-ALL: t(3;21), t(6;11), t(6;9), t(8;21), t(9;11), and t(16;21). Although their immunophenotypes do not suggest mixed linage leukemias, the aberrant expression of myeloid markers such as CD13, MPO, CD33, and CD66c can occur more often among these cases (Table 2 and Supplementary Figure 3). In general, the most frequent molecular alterations were t(15;17), t(9;22), and t(8;21), Figure 2C. Unexpectedly, 2 different rearrangements were detected in 7 patients (Supplementary Table 1 and Supplementary Figure 3). When we compared translocations incidence and age, we noted that molecular aberrancies were more often detected in children (75%) compared with adolescents (25%; Figure 3A). However there is a peak for AML associated at 4 years old while B-ALL-associated translocations exhibit 2 main ages, 9 and 14 years (Supplementary Figure 2A). Apparently, one-third of patients with leukemia carry any of the 28 common translocations and surprisingly 1 molecular group strongly associated with poor prognosis (Philadelphia positive [Ph+]) is the more frequent in pediatric B-ALL from the southeast of Mexico.

Distribution of major molecular translocations in childhood leukemias. a) Chromosomal translocations frequencies per leukemia subtype. b) Percentage of cases positive for translocations. c) Number of cases per translocations among patients.
Molecular Aberrancies in Childhood Leukemias.

Abbreviations: AML, acute myeloid leukemia; B-ALL, B-cell acute lymphoblastic leukemia; MLAL, mixed-linage acute leukemia; T-ALL, T-cell acute lymphoblastic leukemia.

Leukemia-associated translocations per age at diagnosis. Frequencies of positive cases for molecular aberrancies per leukemia subtype and age at diagnosis in childhood and adolescents. B-ALL, of B-cell acute lymphoblastic leukemia; AML, acute myeloid leukemia; T-ALL, T-cell acute lymphoblastic leukemia; MLAL, mixed-lineage acute leukemia.
BCR-ABL B-ALL Shows Heterogeneous Immunophenotype
More than 50% of the bad prognosis are attributed to t(9;22), t(4;11), and t(6;11) rearrangements. Immunophenotype analysis shows that blast with BCR-ABL rearrangements were classified as Pro-B (CD34+CD19+) and/or Pre-B (CD34−CD19+) and leukemias with coexistence of both leukemic precursors ProB-/Pre-B (Table 3). In other words, noncommon lymphoid differentiation stage was associated for the BCR-ABL positive pediatric B-ALL. Aberrant myeloid markers such as CD13, MPO, CD66c, and CD58 were also explored, interestingly, CD58 appeared to be consistently positive while CD13 showed heterogeneity among this particular molecular profile, however, CD66c appeared to be more heterogeneous among the malignant Ph+ population. Nevertheless, CD58 was also a positive marker for B-cell leukemias which carried the t(4;11) rearrangement (Figure 4). Same heterogeneity was observed in those leukemias which carried the t(6;11) translocation which occurs in proB, preB, and proB/PreB stage while CD58 was consistently expressed.
Immunophenotypic Characteristics of Molecular Subgroups of B-ALL.

Abbreviation: B-ALL, B-cell acute lymphoblastic leukemia.
a ProB (CD34+CD10+/−CD19/CD79a+); PreB (CD34-CD10+/−CD19/CD79a+).
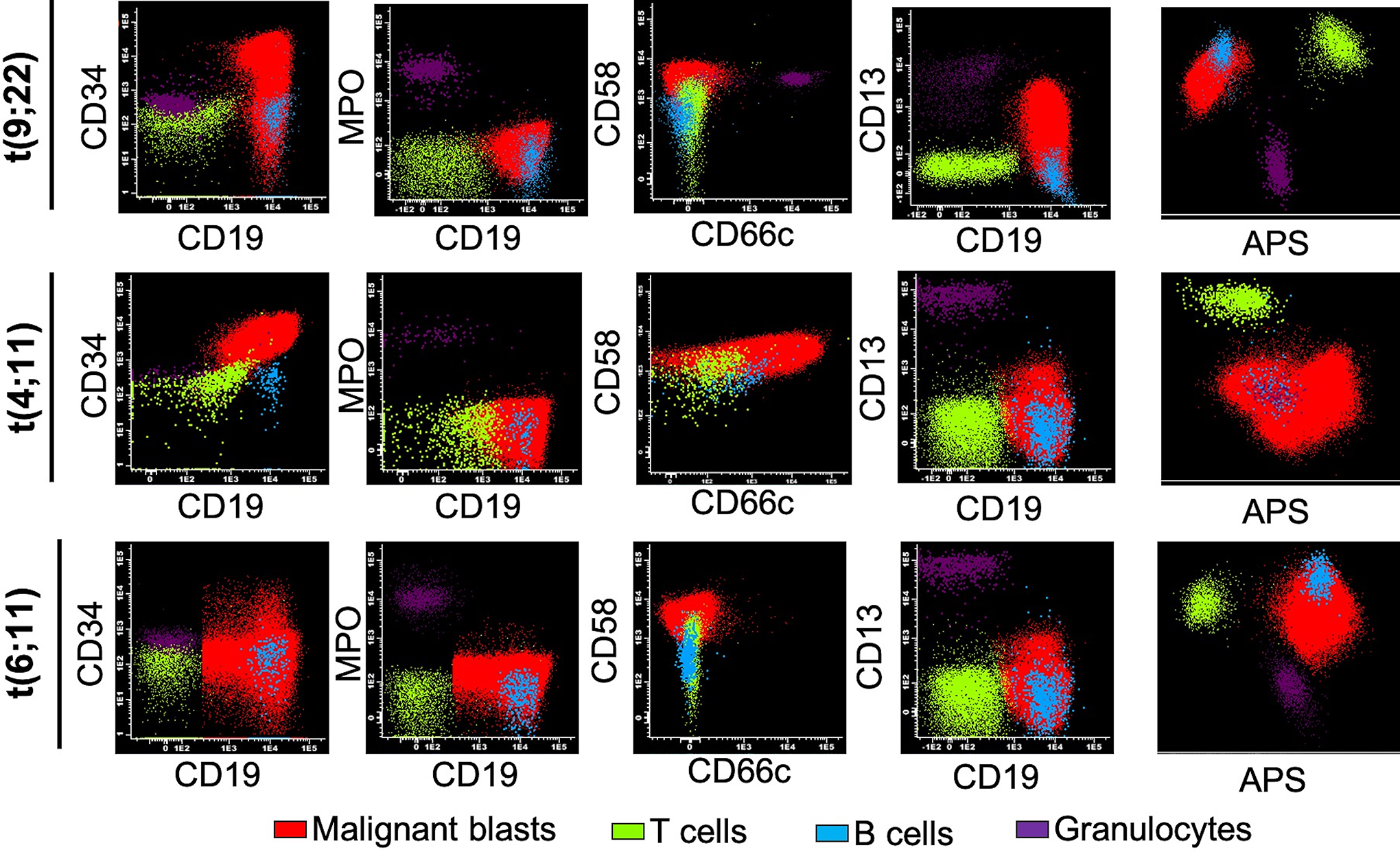
Heterogeneity in immunophenotypic characteristics among B-ALL molecular subgroups. Representative plots of positive leukemias for chromosomal translocations t(9;22), t(4;11) and t(6;11) are shown. Expression of CD34, CD19, CD10, MPO, CD13, CD58 and CD66 were analyzed by FACS. APS, automatic population separator.
In conclusion, immunophenotype characteristics do not correlate with a specific molecular aberrancy or differentiation stage, at least, not by using the standardized Euroflow antibody panel.
Discussion
Acute leukemias are the major cause of death in children from Latin America and the rest of the world. Hispanic children show high incidence and bad prognosis, 28 and Mexico has reported the highest mortality rates. 11 Novel metrics have recently pointed to Latin America as one of the regions whit high DALYs rate due to childhood leukemia. 12 Major reports about incidence and mortality are from Mexico City and the North of the Country, but little is known about the leukemia characteristics, molecular, and cellular epidemiology at the southeast of Mexico, where socioeconomic conditions are low-to-very low. 26,29,30 Although Popular Medical Insurance provided by the Mexican government covers the treatment of children with leukemia since 2005, deaths continue to be recorded and remain as a national health priority. 31,32
Here, we analyzed the immunophenotype, molecular characteristics and age in 218 pediatric patients recruited from 2 hospitals in Oaxaca, Mexico, during 2014 to 2018. As expected, B-cell ALL was the most frequent leukemia subtype representing more than 70% of all cases. Remarkably, a substantially higher frequency of myeloid leukemias (22.48%) compared with previous studies conducted in Mexico City and Nuevo León (10%-13%). 2,30,33 Perez-Saldivar reported a major peak at age of 2 for lymphoid leukemias, while for myeloid leukemias at 10. 30 Interestingly, we recorded a major peak at age of 6 to 7 for B-cell leukemias, similar to what was observed in Nuevo León, 33 and a slight peak for myeloid entities around ages 4 to 5. Adolescents were analyzed separately as unfavorable features worse outcomes have been associated with such age group. 34 Pui et al reported around 10% of adolescent patients with ALL at diagnosis, whereas T-ALL were greatly represented (23%). 34 We noted a higher occurrence of leukemia during puberty (17.5%) but T-ALL was not the major leukemia subtype in this group, as represented around 10% of cases. Intriguingly, 80% of the childhood (0-14 years old) T-ALL were males. Distinct reports from Mexico coincide with a lower frequency of T-ALL in adolescents (3%-7%), 35 -37 contrary to what occurs in high-income countries (>20%). 15,34,38
For childhood leukemogenesis should exist certain susceptibility and a period for vulnerability after exposition to induction factors. Considering that during the first 3 years of life, immune system is maturing, some clones of lymphoid progenitors should suffer damage and occasioning any mutation so, the estimated time for B-ALL is between 2 and 4 years. The delay in the beginning of the disease in our population might be explained by the time of susceptibility to infections. 39 The “unified theory” of leukemia affirms that cancer in developed countries can be preventable if chronic inflammation is avoided just if a correct prime of immune system occurs. 40 However, in a low-to-middle-income countries, additional inflammatory sources may trigger secondary hits, as important contact with several microorganisms, environmental contaminants, diet, and microbiota is often occurring, allowing them to function as drivers. Of note, two-thirds of cancer cases are caused by random mutations due to errors during DNA replication, 41 yet unfavorable environmental conditions increase the probability of this event, as inflammation could enhance the proliferation in normal CD34+ progenitors cells. 42 Malnutrition has been suggested as a predictor factor for early mortality but further studies should be addressed as it remains controversial. 43
Here we shown for the first time the cellular and molecular epidemiology of leukemias from the southeast from Mexico by using an in vitro diagnostic test validated for the screening of 28 translocations in parallel with the standardized flow cytometry EuroFlow diagnostic algorithm. Chromosomal aberrancies are normally found in about 20% of patients with B-ALL 16,37 and 50% to 60% in AML. 44 In our study, we were able to detect same proportions in both leukemia entities, however, we noted that almost 70% of pediatric AML were positive for any translocation. In general, t(15;17) was the most frequent leukemia-associated translocation which result in PML-RARα fusion products characteristic from acute promyelocytic leukemia. Frequency of this subtype of AML is variant (15%-60%), and studies have reported a high incidence in Latinos, European, and African populations; importantly, survival is relatively high (up to 75%). 45 The second more frequent translocation was t(8;21) which produce the fusion protein AML1-ETO usually associated with FAB M2 morphology. Clinical outcome has resulted variable and represents a very heterogeneous disease. According to the reports from St. Jude’s, t(8;21) represents the 13% of the childhood AML subtypes. 46 Nevertheless, we noted that 21% of pediatrics AML carry this rearrangement. A further analysis should be addressed to verify if clinical behavior is similar with previous reports as both are consider with favorable prognosis, of note, incorporation of novel technologies as next generation sequencing (NGS) will unravel the rest of molecular abnormalities in myeloid leukemias.
Although there is not available information about molecular epidemiology and prevalence of gene rearrangements in our country, independent studies have reported the frequency of t(12;21), t(1;19), t(4;11), and t(9;22) as the most explored translocations for pediatric B-ALL because of their prognosis value. 37,47 -49 Internationally, t(4;11) is the most frequent translocation found in B-ALL (2%-20%) followed by t(21;21) in 15% to 12%. 19 In the report from Bekker-Mendez et al, they found a notable prevalence of t(12,21) and t(1;19) among 282 cases, 37 very surprisingly, we noted that most prevalent rearrangement was t(9;22) in 20.7% among B-ALL cases with any rearrangement, while the reported prevalence is 1.8% to 3.2% of the patients. 19,37,50 The translocation t(9;22) produces a BCR-ABL fusion protein, which confers bad prognosis and occurs in the 3% to 5% of the cases of childhood B-ALL, 19 of note, prevalence of BCR-ABL increases with age and is more common for chronic myeloid leukemia (CML). 20 Treatment with tyrosine kinase inhibitors (TKIs) or hematopoietic stem cell and progenitor transplantation usually increase overall survival in these patients. There are 2 major breakpoints that could happen in the BCR gene, for CML rupture often occur between exons 12 and 16 and producing a bigger fusion protein (p210); in contrast, for ALL a smaller product is formed (p190), to date other products have been described as p230. 51 More than 90% of patients with B-ALL carry the p190, which creates a more aggressive leukemia when bone marrow cells are transformed. 52 A very recent report from Arana-Trejo et al published that prevalence of BCR-ABL was 13.2% in 394 cases from different hospitals in Mexico. 53 Is clear that Mexican patients exhibit high prevalence of t(9;22) rearrangements, however, this proportion is higher in Oaxaca (Figure 5). We need to analyze different breakpoints that occur in our patients and perform further clinical correlations. In addition, as several myeloid-associated rearrangements were detected in patients with B-ALL immunophenotype, further sequencing and single-cell analysis may help to confirm these results and to investigate whether the cases showing 2 different molecular rearrangements result from the development of two simultaneous leukemias.

Concluding model. a) B-ALL: representative distribution of most common translocations from international studies (including other regions in Mexico) and the b) shift in Oaxaca.
In order to determine if an unique signature could reveal certain phenotypic characteristics in this group of patients, we analyzed the expression of CD34, CD10, and CD19 to distinguish pre-B from pro-B stages as adult B-ALL Ph+ are commonly classified as proB (CD34+CD10+CD19+) blasts with aberrant expression of CD13. 54 However, we noted that BCR-ABL could be present in both pre-B and pro-B blasts during childhood. The expression of CD13 and CD66c correlates with adult B-ALL Ph+ 55 , however, we didn’t see same pattern in our pediatric samples. Interestingly, CD58 was consistently positive among BCR-ABL patients, however CD58 was also expressed in other molecular groups. Of note, more patients should be incorporated to ensure this expression pattern and explore other markers. Despite it has passed 10 years since the description of Ph-like by gene expression analysis, 22,23 transcriptomics is available in a very few institutions, so little is known about real frequency of this novel leukemia subtype. Interestingly, Ph-like is supposed to be 3 times more common than Ph+ and it is associated with patients of Hispanic descent with CRLF2 rearrangements and GATA3. 56,57 In the next years, we hope to integrate novel approaches to detect BCR-ABL1-like signatures and then identify its immunophenotype. On the other hand, we found that CD13, CD66c, or CD58 were not consistently expressed among patients with t(6;11), MLL rearrangements have been associated to lineage infidelity and mixed lineage leukemias.
Taking all together, integral diagnosis should be mandatory to accurate treatment election and follow-up during MRD. Molecular testing has important therapeutic implications by providing prognosis factors, however, a deeper study should be done to know if this rearrangements in our local leukemias have the same clinical outcomes as the internationally reported. To date, more than 250 different molecular aberrancies have been found to be associated with leukemogenesis and a comprehensive knowledge about their molecular epidemiology is still missing. Incorporation of “old-fashion” techniques as conventional karyotyping or aneuploidy which lack in our study and the avant-garde NGS, 58 next generation flow cytometry (NGF) 59 and why not to start to think in combined technologies as PrimeFlow 60 to detect RNA targets in a particular population of cells by using flow cytometry, will help to enhance survival and will allow to find a more precise patient stratification and appropriate follow-up.
Regarding the report from Curado et al, Mexico has the highest mortality rates in Latin America 11 , this could be explained because the majority of hospitals do not have strategies for MRD detection during follow-up and chemotherapy schemes do not include molecular profiling. Treatment failure and very early relapse are often observed in 26.2% of Mexican pediatric patients, while global mortality is 36.1%. 24 The reasons of this poor survival could be variable, so novel parameters should be included for a better patient stratification, management, and follow-up. In the high-income countries, overall survival of childhood leukemias is around 80%, despite the big advances in diagnosis and treatments. Mexico have a major challenge in pediatric oncology as cure is only achieved in less than 65% of patients.
Additionally, our research group is working now on a novel approach to predict response to therapeutic drugs in the context of the bone marrow microenvironment by using organoids as leukemic cells can create “sanctuaries” where they can hide from chemotherapy or immunosurveillance, with this low-cost tool, we can help to optimize chemotherapy regimen to avoid toxicity and detect drug-resistance phenotypes in a more real biological context. For example, we can include a TKIs-sensivity testing in those Ph-negative to investigate if they are possible Ph-like patients which may be sensible to kinase inhibitors. So, novel laboratory test should be prepared to impact in the clinical management of the patients.
The future of Laboratory Medicine will use cellular, genetic, and molecular classification on behind of modern tools as NSG, NSF, and the drug response prediction to provide integrative information to ensure precision medicine and accelerate the discovery of novel leukemic entities in those which reminds with “normal” karyotype and then unravel the regional and national epidemiology of ETP leukemias or the Ph-like subtype.
Conclusions
A shift is occurring in the biomolecular paradigm of childhood leukemias in Oaxaca, emphasizing the higher incidence of factors associated with bad prognosis that could be linked with early relapse and lower survival.
Supplemental Material
Supplemental Material, Revised_Supplementary_Files - Poor Prognosis Biomolecular Factors Are Highly Frequent in Childhood Acute Leukemias From Oaxaca, Mexico
Supplemental Material, Revised_Supplementary_Files for Poor Prognosis Biomolecular Factors Are Highly Frequent in Childhood Acute Leukemias From Oaxaca, Mexico by Gerardo Juárez-Avendaño, Nuria Citlalli Luna-Silva, Euler Chargoy-Vivaldo, Laura Alicia Juárez-Martínez, Mayra Noemí Martínez-Rangel, Noemí Zárate-Ortiz, Edith Martínez-Valencia, Briceida López-Martínez, Rosana Pelayo and Juan Carlos Balandrán in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
GJA and NCSL are cofirst authors. GJA, NCLS, LAJM, BLM, RP, and JCB performed the analysis and wrote the manuscript. GJA, MNMR, NZO, and EMV performed samples processing. NCSL and ECV provided patient samples and clinical discussion. GJA, LAJM, and EMV provided reagents and critical discussion. This study was approved by the Ethics Committee at the Hospital Infantil de México Federico Gómez (approval no. HIM72012/035 SSA.1060). Written informed consent was obtained from all patient’s parents prior to enrollment in the study. Both Gerardo Juárez-Avendaño and Nuria Citlalli Luna-Silva equally contributed to the work.
Acknowledgments
The authors acknowledge to the professional staff from Laboratorio Juárez. JCB acknowledges the scholarship provided by CONCYTEP and Laboratorio Juárez.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was conducted with funding from the National Council of Science and Technology Grant CONACYT PRONAII 302941.
Supplemental Material
Supplemental material for this article is available online.
