Abstract
Background:
Plastic scintillator detector (PSD) Exradin W1 has shown promising performance in small field dosimetry due to its water equivalence and small sensitive volume. However, few studies reported its capability in measuring fields of conventional sizes. Therefore, the purpose of this study is to assess the performance of W1 in measuring point dose of both conventional IMRT plans and VMAT SRS plans.
Methods:
Forty-seven clinical plans (including 29 IMRT plans and 18 VMAT SRS plans with PTV volume less than 8 cm3) from our hospital were included in this study. W1 and Farmer-Type ionization chamber Exradin A19 were used in measuring IMRT plans, and W1 and microchamber Exradin A16 were used in measuring SRS plans. The agreement between the results of different types of detectors and TPS was evaluated.
Results:
For IMRT plans, the average differences between measurements and TPS in high-dose regions were 0.27% ± 1.66% and 0.90% ± 1.78% (P = 0.056), and were −0.76% ± 1.47% and 0.37% ± 1.34% in low-dose regions (P = 0.000), for W1 and A19, respectively. For VMAT SRS plans, the average differences between measurements and TPS were −0.19% ± 0.96% and −0.59% ± 1.49% for W1 and A16 with no statistical difference (P = 0.231).
Conclusion:
W1 showed comparable performance with application-dedicated detectors in point dose measurements for both conventional IMRT and VMAT SRS techniques. It is a potential one-stop solution for general radiotherapy platforms that deliver both IMRT and SRS plans.
Introduction
Quality assurance (QA) is critical to the implementation of sophisticated techniques like intensity-modulated radiation therapy (IMRT) and stereotactic radiation therapy (SBRT/SRS). Errors among various stages of radiotherapy may cause serious injuries to patients. 1 -3 Some errors can be detected by pre-treatment dose verification termed as patient-specific quality assurance (PSQA). Currently, a variety of methods are utilized for PSQA, including 2D or 3D dose reconstruction methods, point dose verification, and transmission dose verification. 4 Among them, point dose verification is the most widely used method because of its relatively high sensitivity to errors, ease of use, and reliability. 5
Commonly used dosimeters (ionization chambers, diodes, and radiochromic films) have different scopes of applications. Some types of detectors are not suitable for extremely small fields due to large sensitive volumes. 6 Radiochromic film has been considered as a reference dosimeter when it comes to spatial resolution, but it is subject to errors induced during calibration and reading. 7 While diode detector is suitable for the measurement of small fields, it usually over-response in large fields due to high Z material. 8 Therefore, performing PSQA for general radiation platforms, SRS/SBRT capable Linacs for example often requires switching detectors during the procedure. The consequent extra labor provides an incentive to search for a relatively universal instrument.
The plastic scintillator detector (PSD) is characterized by its small sensitive volume and near water equivalence, making it a suitable device for small field dosimetry. The current commercial PSD, Exradin W1 has proven its good dosimetric characteristics. 9,10 Its performance in PSQA for small field applications, including VMAT SRS, gamma knife, cyberknife, and small field proton radiotherapy, has been investigated thoroughly. However, very few studies focused on W1’s potential in non-small field conditions. This study aimed to investigate the feasibility of using W1 in both scenarios.
Material and Methods
The Exradin W1 PSD used in this study was designed and manufactured by Standard Imaging (USA), with a sensitive volume of 0.0024 cm3 and a nearly water equivalent scintillating fiber material. The Cerenkov light, which was the biggest challenge when using scintillation detectors, has now been corrected by the spectral method. The impact of Cerenkov light can be limited to 0.7% by dividing the radiation signal into blue light (Cerenkov light) and green light signal (radiation signal light) 11 and therefore the Cerenkov correction factor (or Cerenkov light radio, CLR) were determined by the following formula (1). 12 This correction function has been integrated into the Super Max electrometer (Standard Imaging, USA) to display the corrected reading directly.
For comparison, 2 ionization chambers, Exradin A19 (Standard Imaging, USA) and Exradin A16 (Standard Imaging, USA), were used in this study. The characteristics of all detectors are listed in Table 1.
Detector Information.

Treatment Planning
A total of 47 clinical plans from our center were selected for this study, including 18 VMAT SRS plans, and 29 conventional IMRT plans. The CT images for treatment planning were acquired on a 32-detector row big bore CT scanner (Philips, Netherlands) with 1 mm slice thickness. The PTV volumes in VMAT SRS plans ranged from 1.11 to 7.07cm3 [equivalent diameter range (12.8 mm, 23.4 mm)] and the fractional doses were 8-12Gy; PTV volumes in IMRT plans ranged from 30.4 to 835cm3 and the fractional doses were 1.8-3.2Gy. Both coplanar and non-coplanar 6MV flattening filter-free arc fields at 1400MU/min dose rate were used for VMAT SRS plans, and regular IMRT plans were designed with 7 to 10 6MV coplanar beams at 600MU/min dose rate. The dynamic jaw positions during jaw tracking were extracted from TPS and MU-weighted average field size was defined. The field sizes of conventional IMRT plans ranged from 4.04 × 5.28 cm2 to 10.76 × 10.26 cm2, with an average field size of 6.06 × 9.55 cm2, and jaws were fixed as 3 × 3 cm2 for VMAT SRS plans to minimize the uncertainty induced by inaccurate modeling of collimator scattering factor Sc .
All VMAT SRS plans targeted brain metastases. Among the IMRT plan targets, there were 8 pulmonary lesions, 6 esophageal lesions, 3 pancreatic lesions, 3 pelvic lesions, 3 osseous lesions, 2 gastric lesions, 2 hepatic lesions, 1 cerebral lesion, and 1 soft tissue lesion. All radiotherapy plans were designed with Varian Eclipse v.13.5 system. In all plans, 100% of the prescription dose covered at least 95% of the PTV. All measurements were performed on an EDGE (Varian Medical Systems, USA) medical linear accelerator at our center.
Calibration of W1
Cerenkov light correction and absolute dose calibration for W1 were performed according to the user manual provided by the manufacturer. As previous studies described, 12,13 the Cerenkov light ratio (CLR) was determined by the formula (1) as follows:

where SC1max40 and SC1min40 refer to the signals for longest and shortest optical fiber placements under 40 × 40 cm2 field irradiation collected from channel 1(blue light), similarly, SC2max40 and SC2min40 were from channel 2 (green light) with 40 × 40 cm2 field.
For absolute dose calibration, the Gain value is calculated by the following formula (2),
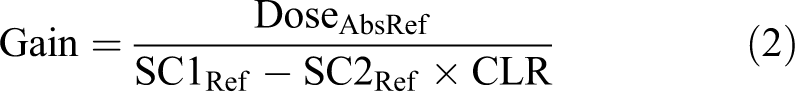
where DoseAbsRef refers to a known absolute dose under reference condition, and SC1Ref and SC2Ref were corresponding signals from the 2 channels.
Finally, the Dose can be given by formula (3):

where the SC1 and SC2 signals from the 2 channels were directly read from the Super Max electrometer.
For determining the known dose, a Farmer-Type chamber Exradin A19 was used. According to the literature, 14 W1 was calibrated using a 6MV FFF beam and a 4 × 4 cm2 field for VMAT SRS plans whereas a 6MV beam and a 10 × 10 cm2 field were used for IMRT plans. Since PSD has a slight temperature dependency, all experiments were carried out at a constant temperature of 22°C with the variation being less than 1°C, so that the temperature dependency was irrelevant. After the calibration, the CLR value and Gain value were determined for subsequent measurements.
Patient Plan Verification
W1 and A19 were used to measure IMRT plans, and W1 and microchamber Exradin A16 were used in VMAT SRS plans. A stereotactic dose verification phantom (SDVP, 20 × 20 × 10 cm3, Standard Imaging, USA), which is made of water equivalent material, was used in the study. For a more comprehensive evaluation, point doses were measured in both high and low dose regions for IMRT plans. High-dose measurements were performed in a region where the doses were high and uniform, any points that received less than 95% of the prescribed fractional dose were excluded. A low-dose point was located in an OAR structure and was several centimeters away from the border of PTV. Since VMAT SRS plans have a very steep dose gradient outside the high-dose area which could greatly induce measurement uncertainty, only points in the high-dose region were measured for VMAT SRS plans. For VMAT SRS, the dose distributions calculated with TPS were verified with Monte Carlo (MC) simulation and film measurement as previously described. 15,16 The difference between the measurements and TPS calculations was analyzed to evaluate the accuracy of W1.
The dose verifications were also performed using conventional QA tools in this study. VMAT SRS dose distributions were measured with EBT3 film (Ashland, USA) in the coronal plane. Films were calibrated in FilmQA Pro software (Ashland, USA) with 9 films irradiated with 0 cGy to 1600 cGy. Films for VMAT SRS plans were cut to dimensions of 63.5 × 63.5 mm2 and inserted into the center slabs of SDVP, which were comprised of 2 slabs of 5 mm thickness plus 5 slabs of 2 mm thickness and each of them have a recessed pocket to hold the film. Irradiated films were scanned using an Epson model V750 PhotoPerfection document scanner (Epson America, USA). The difference in dose distribution was quantified using gamma analysis with 2%/2 mm criteria, and only points received doses higher than 10% of maximum dose were included; IMRT dose distributions were measured with Delta 4 phantom (Scandidos, Sweden), which consists of 3 crossing planes filled with diodes inside a cylindrical polymethylmethacrylate (PMMA) phantom. The comparison between the TPS calculations and the measurements was performed with the Delta 4 software, following manufacturer’s instruction. IMRT dose distributions were analyzed with Delta 4 software using gamma analysis with 3%/3 mm criteria and only points received doses higher than 10% of maximum dose were included. 17
Statistical Analysis
The differences between the measured values (relative to the TPS calculations) of different detectors, were evaluated using Wilcoxon signed-rank test. Data analysis was performed using SPSS 17.0 software package (SPSS Inc. USA). A P-value of less than 0.05 was considered statistically significant.
Results
The measurement results of the IMRT plans are shown in Table 2. The average gamma passing rate of the IMRT plans was 98.99% ± 1.34%. In high-dose measurements, the differences between all W1 measurements and TPS calculations were within the departmental tolerance of 3%, whereas 2 out of 29 A19 measurements exceeded the tolerance. The average differences between measured and calculated doses were 0.27% ± 1.66% for W1 and 0.90% ± 1.78% for A19. The Exradin W1 had a smaller difference than A19, but no statistical difference was observed (P = 0.056). In low-dose measurements, the average differences between measured and calculated doses were −0.76% ± 1.47% for W1 and 0.37% ± 1.34% for A19. In low-dose measurements, A19 achieved a better agreement with TPS than W1 did (P = 0.000).
IMRT Plans High-Dose Point and Low-Dose Measurements.
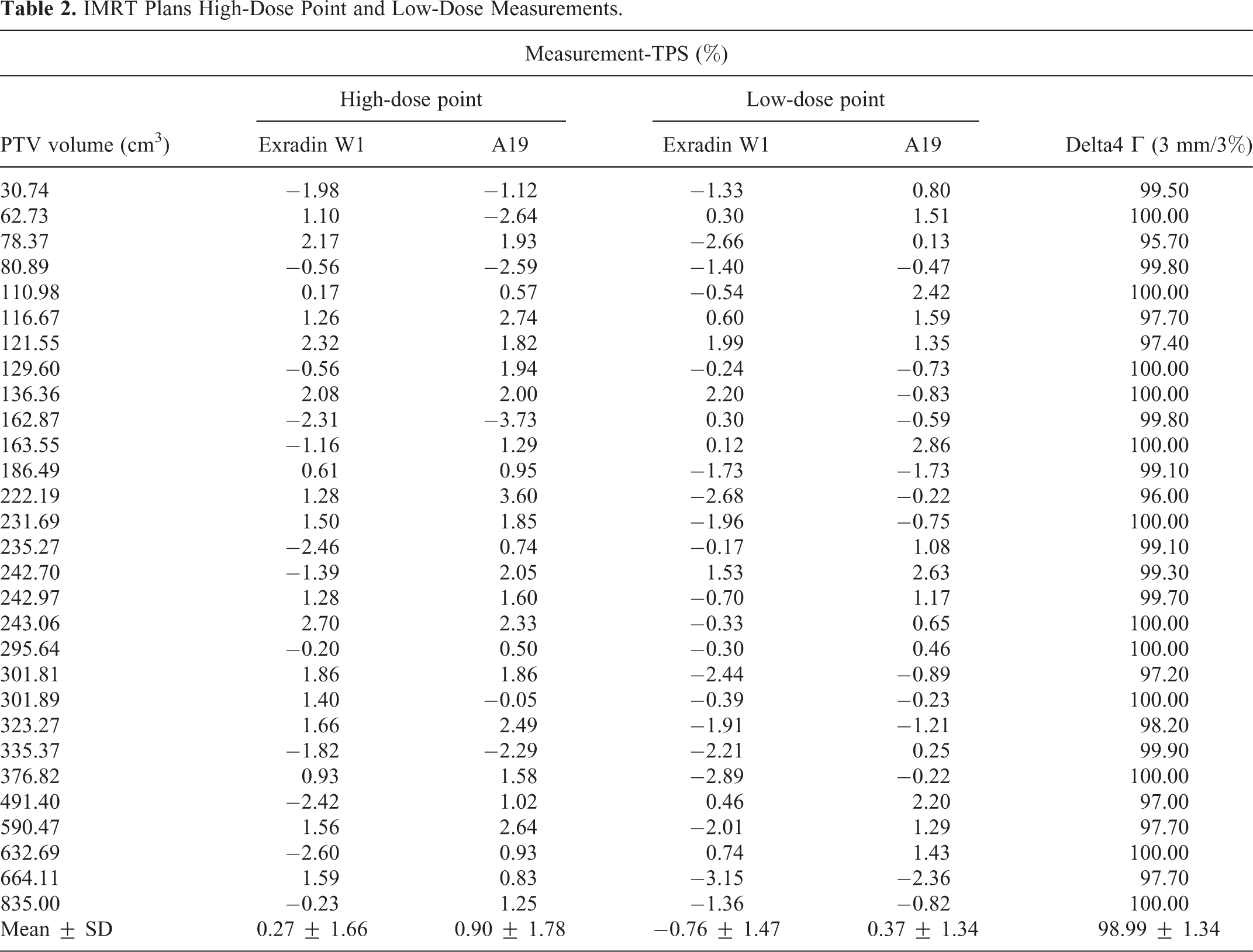
The measurements of VMAT SRS plans are shown in Table 3. The average 2%/2 mm gamma passing rate was 98.84% ± 1.34%. The point doses measured with W1 and A16 showed a good agreement with TPS, and none of the measurements exceeded the 3% tolerance. The average difference between W1 and TPS was −0.19% ± 0.96%, and that value for A16 was −0.59% ± 1.49%. No significant difference was observed between W1 and A16 (P = 0.231).
VMAT SRS Plans Point Dose Measurements.

All QA measurements for the IMRT plans are plotted in Figure 1 and those for the VMAT SRS plans are plotted in Figure 2. The W1 has a lower response than the A19 in measuring IMRT plans in both high-dose and low-dose measurements, but the deviation between the 2 detectors was acceptable. When measuring SRS plans, the W1 resulted in good agreement with the TPS calculation and the A16.

Relevant difference between point dose measurements and TPS calculations for conventional IMRT plans (*P < 0.05).
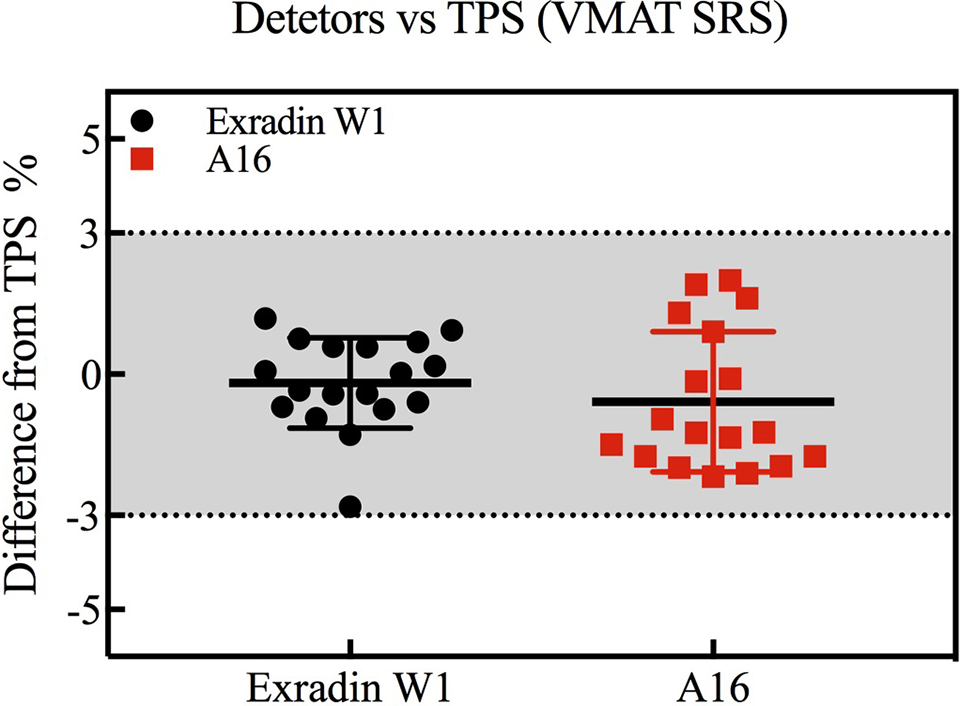
Relevant difference between point dose measurements and TPS calculations for VMAT SRS plans.
Discussion
A plastic scintillator detector needs to be calibrated before absolute dose measurement due to the Cerenkov effect, which is field-size dependent. According to the research of Snyder et al, 14 the authors found a low sensitivity to the Cerenkov factor under different sizes of calibration fields, but the residual deviations suggested that the calibration should be done under a condition similar to which the measurements will be made in. Therefore, a 10 × 10 cm2 field was used for IMRT plan verification, and a 4 × 4 cm2 field was used for VMAT SRS plan verification in this study.
Exradin W1, the only commercially available plastic scintillator detector, has shown better performance than other detectors such as pinpoint chambers, diodes, and microchambers, in measuring small fields. 18 -20 However, studies on using W1 for conventional IMRT plans are sparse. In this study, the W1 showed good consistency with the TPS calculations and had an acceptable deviation from the A19. The possible cause of this deviation might be the leakage current of the dosimeter which is more prominent in low-dose measurements. The experimental results for VMAT SRS plans showed that the W1 has similar accuracy to the A16, and additionally the relative differences between the TPS and the W1 were more centrally distributed around the average value, which is in consistence with the results of Qin et al. 19
The Exradin W1 plastic scintillator detector is made of non-magnetic materials, and the small sensitive volume and water equivalence make it a promising candidate for MR-Linac. The angle-independency of PSD in magnetic fields was proved in the previous studies. 21,22 Meanwhile, Therriault-Proulx et al 21 found that magnetic fields can cause an inevitable influence on the Cerenkov effect for PSD, which can be corrected effectively in the calibration. While after the correction of the Cerenkov effect, there was still a response error of about 2.4%, which was better than the results of ionization chambers. Regarding the field size dependence in magnetic fields, the study by Yoon et al 22 considered W1 unsuitable for fields larger than 10.5 × 10.5 cm2, because the output factor measured by W1 with the magnetic field was 3% different from the that without the magnetic field. One of the reasons may be the influence of the magnetic field on the dose deposition itself. 23 On the other hand, Koniarová and Konček 10 found that the optoelectronic components of PSD are sensitive to radiation. The noise generated by optoelectronic components depends on the distance between the beam center and the photodiode and the size of the field. In that study, a 2.5% deviation was observed under the 10 × 10 cm2 field at 1 m away from the beam center. In our experiments, we placed the photodiode as far as possible from the beam center and added extra custom-made shielding, thus the noise was minimized.
The measurements in this study covered a wide range of field sizes. In the current practice of PSQA, two or more different types of radiation detectors are usually required to cover such a wide range. Exradin W1 PSD can potentially replace other detectors to avoid switching devices when verifying both IMRT and SRS/SBRT plans.
Compared with the Exradin A16 microchamber, the Exradin W1 has a smaller sensitive volume and better water equivalence. However, the data in this study did not indicate that Exradin W1 was superior to the Exradin A16 in the verification of the SRS plan. The volume of the smallest PTV in this study was 1.11cm3, with an equivalent diameter of 12.8 mm. This relatively large size did not push those detectors to their limits. According to Therriault-Proulx et al, 24 the relative differences between the output factors measured by the W1 and the A16 were not significant until a 4 mm diameter collimator was used. On the other hand, the maximal field size tested was 10.76 × 10.26 cm2. Further study is needed if W1 will be used to measure fields beyond this size.
Conclusion
The Exradin W1 plastic scintillator detector sustains accuracy in different field sizes, making it suitable for verifying both conventional IMRT and SRS/SBRT plans. It could potentially be a one-stop solution for general radiotherapy platforms that deliver both IMRT and SRS plans.
Footnotes
Authors’ Note
Zike Huang and Jian Qiao are co-first authors, who contributed equally to this study. All analyses were based on retrospective data, thus no ethical approval are required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial support from Cancer Precision Radiotherapy of China International Medical Foundation (grant number: HDRS2020030202).
