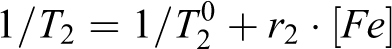Abstract
Bifunctional magnetic/fluorescent core-shell silica nanospheres (MNPs) encapsulated with the magnetic Fe3O4 core and a derivate of 8-amimoquinoline (N-(quinolin-8-yl)-2-(3-(triethoxysilyl) propylamino) acetamide) (QTEPA) into the shell were synthesized. These functional MNPs were prepared with a modified stöber method and the formed Fe3O4@SiO2-QTEPA core-shell nanocomposites are biocompatible, water-dispersible, and stable. These prepared nanoparticles were characterized by X-ray power diffraction (XRD), transmission electron microscopy (TEM), thermoelectric plasma Quad II inductively coupled plasma mass spectrometry (ICP-MS), superconducting quantum interference device (SQUID), TG/DTA thermal analyzer (TGA) and Fourier transform infrared spectroscopy (FTIR). Further application of the nanoparticles in detecting Zn2+ was confirmed by the fluorescence experiment: the nanosensor shows high selectivity and sensitivity to Zn2+ with a 22-fold fluorescence emission enhancement in the presence of 10 μM Zn2+. Moreover, the transverse relaxivity measurements show that the core-shell MNPs have T2 relaxivity (r2) of 155.05 mM−1 S−1 based on Fe concentration on the 3.0 T scanner, suggesting that the compound can be used as a negative contrast agent for MRI. Further in vivo experiments showed that these MNPs could be used as MRI contrast agent. Therefore, the new nanosensor provides the dual modality of magnetic resonance imaging and optical imaging.
Introduction
Compared with the dual-function molecular probes, the introduction of the nanoparticles makes more advantages of dual-function probes. In addition, as a new type of nanoparticles, magnetic fluorescent composite nanoparticles have attracted extensive attention in recent years. These composite nanoparticles take the advantages of molecular fluorescence, magnetic properties and nano-scaled materials, and can provide a potential platform for the construction of novel drugs for biological separation, targeting, biological imaging, tumor cell localization and even tumor therapy. Many interesting works have been reported based on these nanocomposites. Dual functional and multifunctional magnetic core shell Fe3O4@SiO2 nanocomposites as a special carrier have attracted more and more attention. 1 -6 The silica shell outside the iron oxide core not only increases the and biocompatibility and stability of nanoparticles in water environment, but also provides convenient sites for modifying ligands in biomedical applications, such as biological separation, 7 -9 drug targeting, 10 -14 cell separation, 8 enzyme immobilization 15 -18 and protein purification. 19 In addition, Fe3O4@SiO2 MNPs are more and more studied because they are easy to prepare and friendly to aqueous solution, 20 and can be designed into various shapes and sizes. On the other hand, some of which have been approved by FDA for clinical use. Fe3O4@SiO2 NPs which have superparamagetism have gradually become good drug agents, and FDA has approved the use of some of them in clinical practice. 21 As a solid-state chemical sensor, sensor modified Fe3O4@SiO2 nanoparticles have many attractive advantages in heterogeneous solid-liquid system. Such nanoparticles are easily obtained by hydrolysis reaction, meanwhile, different functional groups are introduced in the preparation process. For example, magnetic resonance contrast agents and fluorescent organic dyes can be constructed together into such nanoparticles, which produces a good platform for many potential applications. 20
A variety of transition metal ions play a wide range of roles in the environment and biological physiological processes. 22 -24 Among them, Zinc ions is the second most abundant transition metal ion in human body. 25,26 In human body, most zinc are closely bound to enzymes and transcription factors, 27 -30 but many chelatable Zn(II) ions are still dissociated in certain cells and play important roles in biological systems. 31 -38 In the human body, the brain is one of the organs with the highest zinc content. Studies showed that the concentration of free zinc ions in the brain is about 0.1-0.5 mM, and in the serum is about 12 μM. Zinc ions are components of many enzymes in human body. It is widely involved in cell proliferation and differentiation, nucleic acid and protein synthesis and many other important physiological activities. 39 -41 Zinc ion is closely related to the central nervous system, for example, the decrease of zinc concentration in serum and plasma will increase the risk of Alzheimer’s disease and Parkinson’s disease. 42,43 Therefore, intense research efforts have been devoted to the development of chemosensors for Zn2+ ion detection. Due to the unique role of Zinc ions in physiological function, it is of great significance to detect and image Zinc ions in biological samples. In order to probe Zinc ions in vivo and in vitro, a large number of scientific researches have been carried out. 44 Among those reported Zn2+ sensors, quinoline derivatives are commonly chosen, such as 6-methoxy-(8-ptolunesulphonamido) quinoline (TSQ) 45 and its derivative (Zinquin series). 46 -48 These derivatives containing quinoline ring have been used for the fluorescence detection of zinc ions. It is obvious that the quinoline ring of supramolecular system, especially 8-aminoquinoline, has a very good selectivity for zinc ions. 49 However, some studies showed that zinc ion fluorescent probes containing quinoline ring have poor solubility in aqueous solution and are also interfered by other metal ions. In addition, many of the reported probes are difficult to work in the cellular environment. Therefore, there is still a great demand for the development of zinc ion fluorescent probes with wider versatility and higher performance.
With those concepts in mind, our strategy is to synthesize uniform dual-functionalized nanoparticles which permit dual-modality detections by incorporating organic fluorescent dye a chloroacetyl derivate of 8-amimoquinoline, N-(quinolin-8-yl)-2-(3-triethoxysilyl) propylamino) acetamide (QTEPA) into silica-coated magnetite core-shell (Figure 1). We utilized a common stöber method to synthesize the Fe3O4@SiO2 silica-coated MNPs. However, Rastogi et al, 50 have reported the use of silica functional with the same fluorophore, QTEPA (Chemical structure shown in Figure S1), for the detection of Zn2+. In our work, the difference is superparamagnetic Fe3O4 was added as the core. These QTEPA modified MNPs, as far as we known, are the first to be reported for not only exhibiting good selectivity and sensitivity for Zn2+, but also of potential possibility to work as a T2 contrast agent.

Schematic illustration of the formation of Fe3O4@SiO2-QTEPA.
Experimental
The experimental details were provided in the supporting information.
Results
Characterization of the Modification MNPs
The TEM image of magnetic Fe3O4 obtained in this study is shown in Figure 2 (left). The result showed that the shapes of Fe3O4 nanoparticles are nearly spherical, and the average particle size is about 7 nm. 51 After the Fe3O4 NPs were modified with II, TEM revealed that the Fe3O4 were successfully encapsulated in silica shell, and the average particle size was about 20-30 nm as shown in Figure 2 (right). The TGA results indicated that the grafted ratio of onto Fe3O4@SiO2 is about 10% (Figure S2).

TEM images of Fe3O4 (left) and Fe3O4 @SiO2-QTEPA (right) (bar = 100 nm).
The suspension prepared with Fe3O4 has good stability and can be kept for several months without obvious change. Dynamic light scattering (DLS) experiments showed that the hydrodynamic diameter of Fe3O4 NPs had almost no change in the first 2 months, and there was no obvious aggregation in 5 months. However, the stability Fe3O4@SiO2-QTEPA is much less than pure Fe3O4, and the suspensions only maintain about 3-4 days (Figure S3).
Zeta potential (Ζ) reflects the degree of repulsion between adjacent, similarly charged particles in the dispersion. Particles with high Ζ (negative or positive) are electrically more stable than those with low Ζ, that is, the solution or dispersion of the former is more likely to resist aggregation. The zeta potential of Fe3O4 in aqueous solution is −18.8 mV, while Fe3O4@SiO2-QTEPA aqueous solution has a net-negative charge at the value of −1.25 mV. Therefore, Fe3O4 NPs show higher water dispersibility than Fe3O4@SiO2-QTEPA do. This is also a proof that the Fe3O4 was successfully grafted by SiO2-QTEPA. This suggests that mutual repulsion between the macromolecules and subsequent lack of interactions. 52
The crystallographys of Fe3O4 and Fe3O4@SiO2-QTEPA were verified by powder XRD (Figure 3). As shown in Figure 3, the diffraction patterns of both particles exhibit 6 peaks at 2θ of 30.1º, 35.5º, 43.1º, 53.6º, 57.2º, and 62.7º respectively, corresponding to standard inverse spinel phase of Fe3O4 (220) (311) (400) (422) (511) and (440) surface of the diffraction peak. The diffraction patterns and the relative intensities of all diffraction peaks of the 2 samples indicated that there were phases in both Fe3O4 and Fe3O4@SiO2-QTEPA nanoparticles which are consistent with the crystal anti-spinel structure of magnetite (Fe3O4) and maghemite (γFe2O3). The results also showed that the crystal Fe3O4 were encapsulated in the core-shell structure. Compared with the Fe3O4, all the diffraction peaks of Fe3O4@SiO2-QTEPA are weaken, which due to the surface of silica and organic matter. And the broad peak between 20º and 25 º (2θ) is assigned to the mesoporous silica oxide shell.
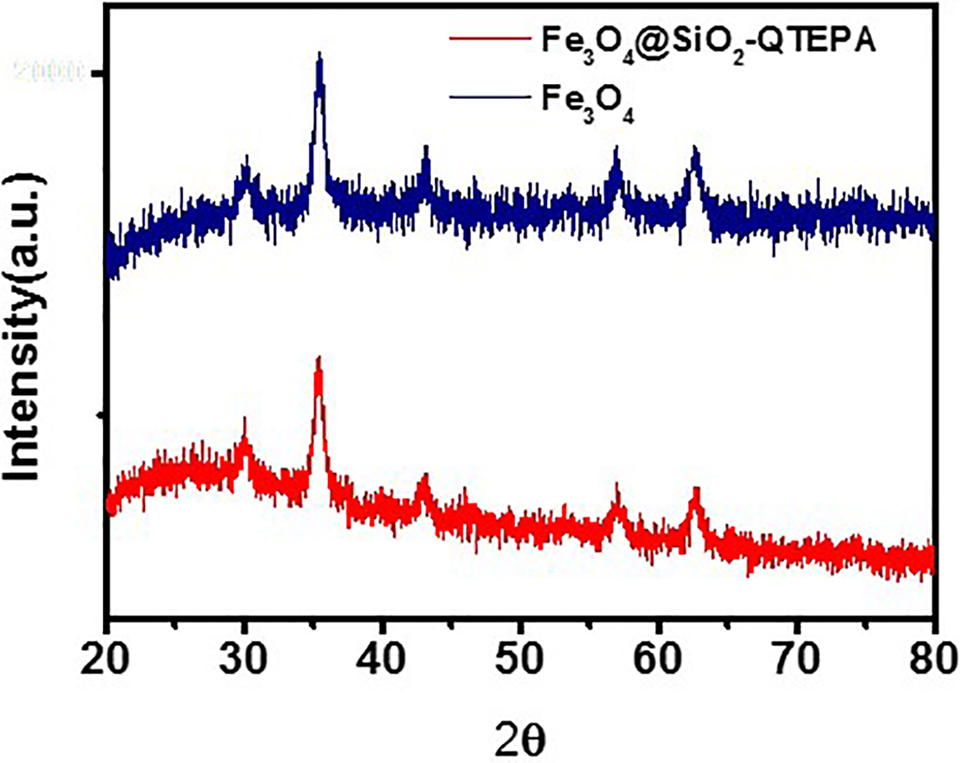
X-ray diffraction pattern of Fe3O4 and Fe3O4@SiO2-QTEPA.
FTIR spectroscopy is used to confirm that QTEPA silicon is successfully immobilized on the Fe3O4@SiO2 nanoparticles. The FT-IR spectra of Fe3O4@SiO2 and Fe3O4@SiO2-QTEPA are shown in Figure 4. In Figure 4 (red line), the broad band centered at 3410.4 cm−1 represents silane and any form of water adsorbed on the surface of Fe3O4@SiO2 nanoparticles. The band at 1643.3 cm−1 represents the bending mode of O-H vibrations. The strong band centered at 1103.04 cm−1 represents the structural Si-O-Si vibration, while the Si-OH vibration band appears in the 802-950 cm−1 region. Compared with the infrared spectrum of Fe3O4@SiO2, the N-H stretching vibration band of Fe3O4@SiO2-QTEPA appears at 3402 cm−1 (black line) with the broad band of the silanol and moisture on the surface. The band at 1531.4 cm−1 is C = C aromatic stretching band, while the carbonyl band at 1662.3 cm−1 belongs to the aliphatic C-H stretching band. The FT-IR spectra clearly showed that II was successfully modified on the surface of MNPs.

FTIR spectra of Fe3O4@SiO2 (red line) and Fe3O4@SiO2-QTEPA (black line).
Magnetic Properties Fe3O4@SiO2-QTEPA
The magnetization curve of Fe3O4@SiO2-QTEPA is shown in Figure 5. The room temperature magnetization (m) curve of the samples with applied magnetic field (H) shows that the saturation magnetization (MS) is 15.2 emu/g. Due to the existence of core-shell structure, the room temperature magnetization Fe3O4@SiO2-QTEPA is lower than that of naked Fe3O4 nanoparticles (69.7 emu/g). Remanence and coercivity are almost invisible in the amplification curve (inset in Figure 5), which indicated that both nanoparticles have superparamagnetism at room temperature. So it can be considered that Fe3O4@SiO2-QTEPA NPs have enough magnetic attraction to effectively perform magnetic separation and can be used in nuclear magnetic resonance imaging.

Room temperature magnetization curves of pure Fe3O4 powder and Fe3O4@SiO2-QTEPA.
The superparamagnetism of magnetic NPs has a strong contrast effect on the surrounding tissues, which enables T2 -weighted MRI to display tumor tissues in any plane of the body. 53,54 Therefore, we measured the transverse relaxation time (T 2) to evaluate the negative-contrast effect of Fe3O4@SiO2-QTEPA. In order to evaluate the performance and effect of Fe3O4@SiO2-QTEPA as T2 contrast agent, we measured the transverse relaxation time (T 2) under 3 T magnetic field, and calculated the transverse relaxivity (r 2) according to the following equation 55 :
where T 2 0 is the standard relaxation time of pure water, [Fe] is the concentration of Fe3+ calculated of Fe3O4@SiO2-QTEPA NPs (treated with nitric acid digestion). Each point in the Figure 6 is the transverse relaxation rate (1/T2) of the solution at different concentrations, and the straight line is the linear relationship between the reciprocal of relaxation time and Fe3+ concentration. The linear fitting transverse relaxation rate is 155.05 mM−1 s−1 (Figure 6), suggesting that the Fe3O4@SiO2-QTEPA NPs could be used as a T 2-shortening agent to effectively relax the spin of water proton. These results showed that although Fe3O4 is coated by silica shell, it can still contact with water molecules, and the existence of shell structure only slightly affects the relaxation of Fe3O4.

T2 relaxivity plot of aqueous of Fe3O4@SiO2-QTEPA.
The T 2-weighted magnetic resonance images of Fe3O4@SiO2-QTEPA in terms of different Fe concentrations are shown in Figure 7. The Fe3O4@SiO2-QTEPA NPs have good dispersion in water, which enabled us to detect the relaxivity of the particles in the solution at 3.0 T MR system. It is obvious from the Figure 7 that the Fe3O4@SiO2-QTEPA NPs displayed a signal enhancement in the T2 weighted MRI image with the decrease of iron concentration, which means that MNPs may be used as the negative contrast agent of MRI.

T2-weighted MRI images of Fe3O4@SiO2-QTEPA in water.
In Vitro and In Vivo MRI
The diagnostic potential of Fe3O4@SiO2-QTEPA was first explored in vitro by testing the negative-contrast effect in MCF-7 cells. To evaluate the possibility of using Fe3O4@SiO2-QTEPA nanoparticles as contrast agent to detect cancer cells, MCF-7 cells incubated with Fe3O4@SiO2-QTEPA nanoparticles suspension with different concentrations ([Fe] = 0, 0.0025, 0.0050, 0.0075 0.0100, 0.0125, 0.0150, 0.0175, 1.020, 0.0225, 0.025 mM) were detected by nuclear magnetic resonance (NMR). It can be clearly seen from the Figure 8 that the MR signal of MCF-7 cells treated by this method gradually decreased with the increase of Fe concentration. This suggested that Fe3O4@SiO2-QTEPA NPs have a good spatial resolution for T2 MRI, which is consistent with the data reported in the literature based on iron oxide NPs.

T2-weighted MRI images of Fe3O4@SiO2-QTEPA in MCF-7 cells for 3 h.
Further, we carried out in vivo experiments. Fe3O4@SiO2-QTEPA suspension was injected into the mice via the tail vein and the changes of MR signal decline in the target organ liver were measured before and after injection (Figure 9A). The results showed that Fe3O4@SiO2-QTEPA accumulated in the normal liver, while no signal appeared in other organs, which confirmed that the MNPs eventually targeted and gathered in the liver. Figure 9A is an in vivo MRI image of mouse liver. Figure 9A shows the images of mice liver in vivo MRI. The images were acquired 1, 5, 7 and 12 h respectively after tail vein injection of the Fe3O4@SiO2-QTEPA suspension at 1 mg /mL Fe3O4@SiO2-QTEPA concentration, equivalent to a dose of 5 mg Fe3O4@SiO2-QTEPA/kg of the mice body weight, the color becomes darker after injection, and the signal cavity appears in the liver substance. The liver of mice was normal gray signal, while the signal becomes darker and daker after injection. After 1 h injection, the signal intensity in the liver decreased obviously. Compared with 5 and 7 hours after injection, 1 hour was the time that the MNPs accumulation in the normal liver arrived maximum value, and the MNPs gradually metabolized in the normal liver after 24 hours (Figure 9B).
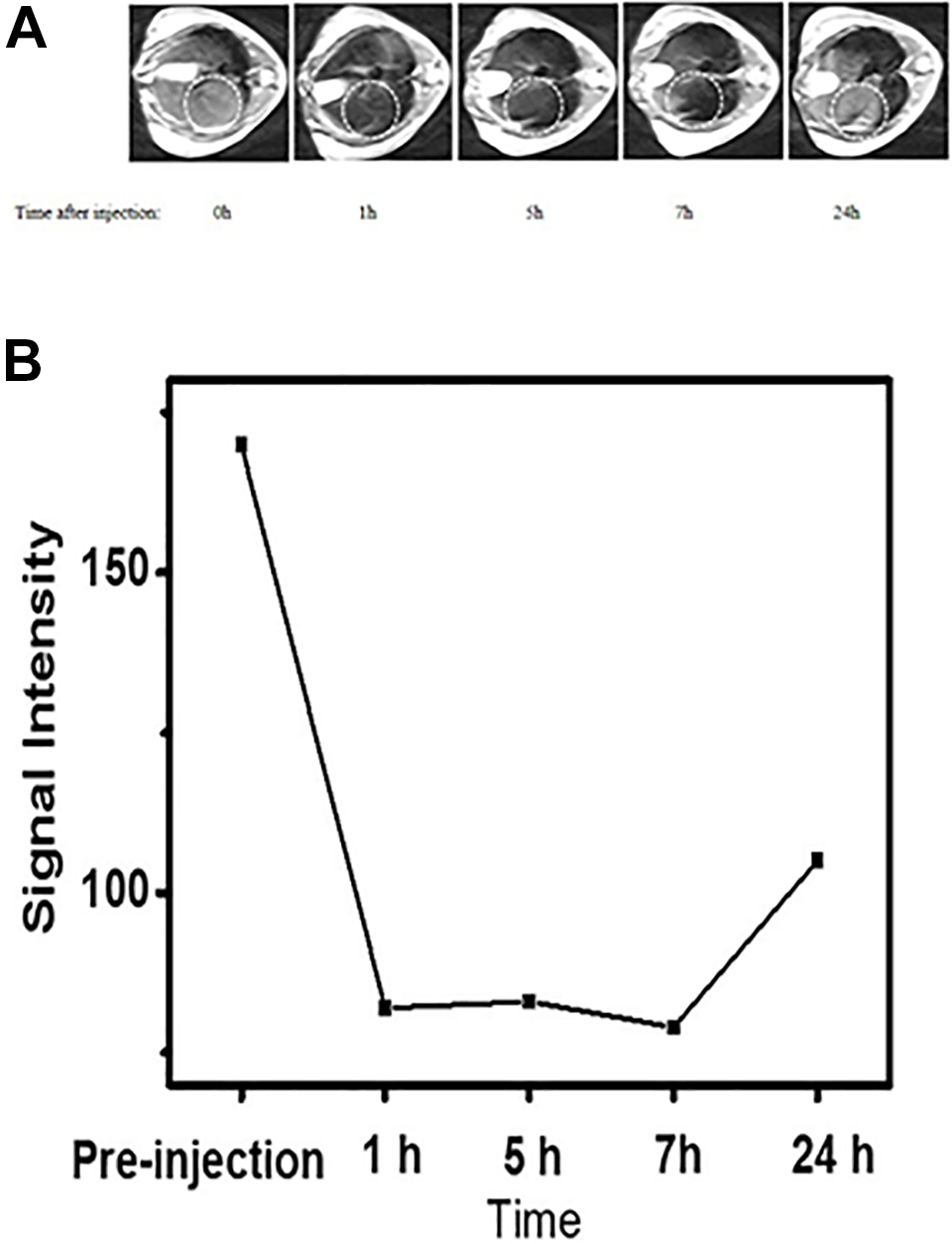
A, MR imaging of mouse liver regions after systemic administration of 150 µL of Fe3O4@SiO2-QTEPA (1 mg /mL). B, The time-dependent drop in MR T2 signal intensities measured in the liver after the administration of Fe3O4@SiO2-QTEPA.
Zn2+ Fluorescence Sensing
The fluorescence experiments of all metal ions were carried out in MNPs buffer solution (100 µg of Fe3O4@SiO2-QTEPA in 3 mL HEPES buffer). Fluorescence titration of Zn2+ was conducted using this suspension. Fe3O4@SiO2-QTEPA showed a rather weak fluorescence signal in the range between 475 nm and 510 nm without Zn2+ ions. However, after adding Zn2+ to the concentration of 10 μM, the fluorescence intensity of the solution increased by a factor of about 22 times at 490 nm, and the maximum emission shifted from 420 nm to 490 nm (Figure 10A). We also determined the fluorescence spectra the Fe3O4@SiO2-QTEPA suspension in presence of Mn2+, Fe3+, Ag+, Cr3+, Co2+, Cu2+, Cd2+, Fe2+, Hg2+, Pb2+and Ni2+. After adding these above metal ions to 20 μM separately, the changes of fluorescence intensity at 490 nm are shown in Figure 10B. After adding different metal ions, only the addition of Hg2+ increased the fluorescence intensity slightly. Adding other metal ions to the Fe3O4@SiO2-QTEPA suspension did not cause obvious change of fluorescence spectra. In addition, the enhancement of fluorescence intensity due to the addition of Zn2 + ions was not affected by the subsequent addition of other metal ions with concentrations of 20 μM except K+, Ca2+, Na+, Mg2+ (200 μM). (Figure S7 in the Supporting Information.)
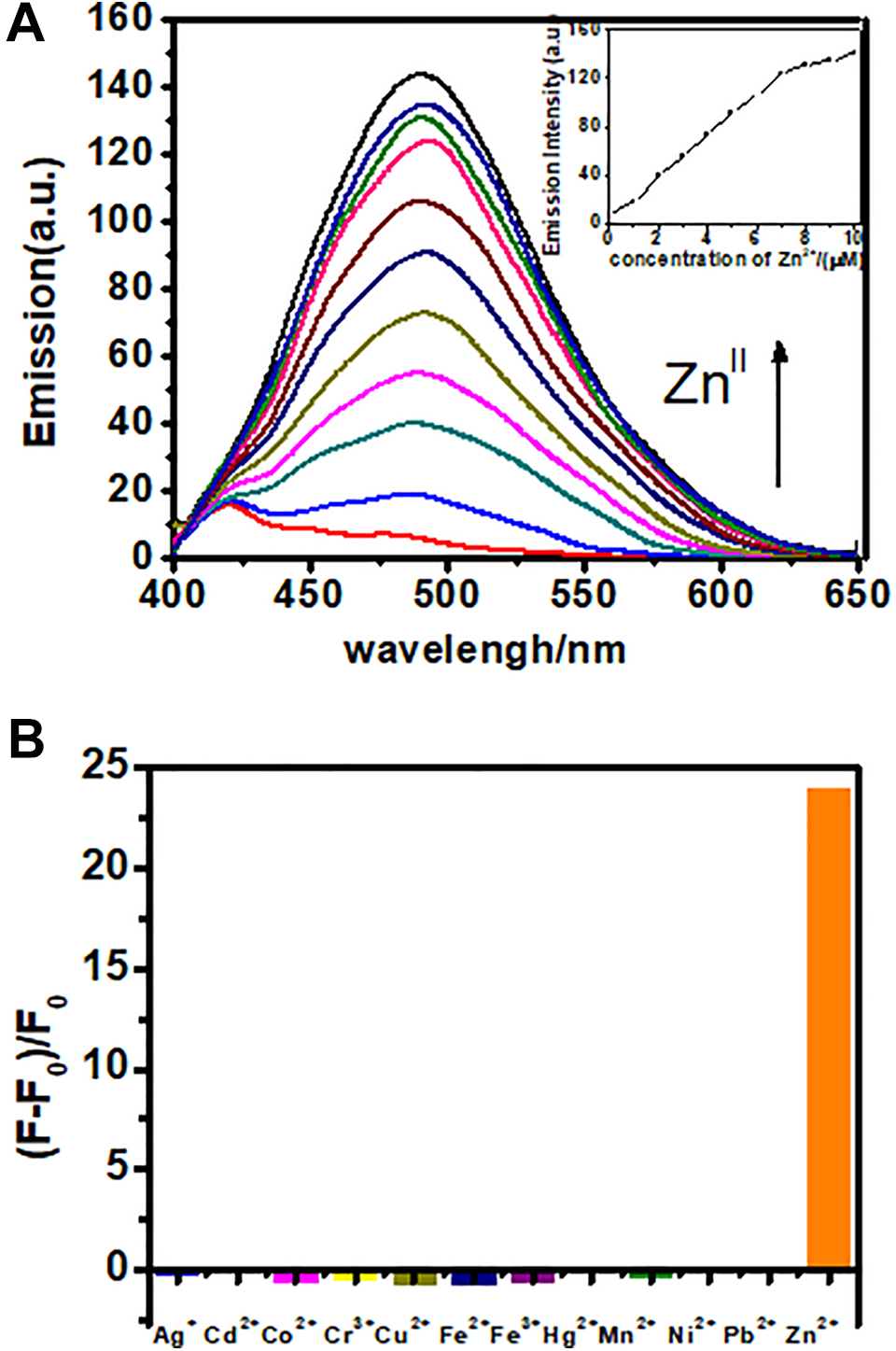
A, Fluorescence emission spectra of Fe3O4@SiO2-QTEPA (100 μg/3 mL) in the presence of Zn2+ from 1 μM to 10 μM in HEPES buffer. B, Bar graph of fluorescence emission intensity for 12 different metals showing metal selectivity profile of Fe3O4@SiO2-QTEPA in HEPES (5 mM, pH = 7.4) with the concentration of 20 μM for each metal.
We applied Fe3O4@SiO2-QTEPA NPs for fluorescent imaging of Zn2+ in living cells to demonstrate the Fe3O4@SiO2-QTEPA NPs with practical application potential in the buffer system. Hela cells were incubated with Fe3O4@SiO2-QTEPA NPs at 37ºC for 4 h and washed thoroughly by PBS buffer to remove the extra Fe3O4@SiO2-QTEPA NPs. Then with the addition of Zn2+, the living cells were incubated for another 20 minutes. The results showed that Fe3O4@SiO2-QTEPA NPs could penetrate the cell membrane for imaging Zn2+ in living cells, free Fe3O4@SiO2-QTEPA NPs in living cells showed very weak fluorescence (Figure 11B), a bright fluorescence was observed in the cells (Figure 11C) after adding Zn2+ in the above cells. Finally, TPEN (a good masking agent for Zn2+) solution was added to the above cells for incubating another 20 minutes and fluorescence decreased (Figure 11D). These results demonstrated that Fe3O4@SiO2-QTEPA NPs can be applied for dual-mode in vitro imaging of Zn2+ ions in living cells and potentially sensitive in vivo MRI imaging as well. The comparison of Fe3O4-SiO2-QTEPA with another similar materials in the literatures were summarized in Figure S4.

Bright image (A) and fluorescence images of Hela cells incubated with Fe3O4@SiO2-QTEPA NPs in the absence (B) and presence (C) of Zn2+, and TPEN was added to eliminate Zn2+ (D). Scale bar is 10 μM.
Conclusions
In summary, we demonstrate bifunctional nanoparticles based on silica-coated Fe3O4 MNPs which can be used as biocompatible magnetic T2 MRI contrast agents and for imaging Zn2+ in living cells. To the best of our knowledge, these QTEPA functionalized MNPs are the first to be reported for not only exhibiting good selectivity and sensitivity for Zn2+, but also of potential possibility to work as a T2 contrast agent. This kind of core-shell magnetic nanoparticles has the comprehensive ability of fluorescence and MR imaging, and has the potential application for drug loading, which provides a platform for the research of targeting and biological imaging materials in biological systems. These results provide considerable foundation and reference for the construction of bifunctional magnetic nanoparticles. The magnetic nanoparticles modified by appropriate fluorescent probe molecules have good biocompatibility and can be used in fluorescence imaging and nuclear magnetic resonance imaging.
Supplemental Material
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211036539 - Silica-Coated Fe3O4 Nanoparticles as a Bifunctional Agent for Magnetic Resonance Imaging and ZnII Fluorescent Sensing
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211036539 for Silica-Coated Fe3O4 Nanoparticles as a Bifunctional Agent for Magnetic Resonance Imaging and ZnII Fluorescent Sensing by Lin Qiu, Shuwen Zhou, Ying Li, Wen Rui, Pengfei Cui, Changli Zhang, Yongsheng Yu, Cheng Wang, Xiang Wang, Jianhao Wang and Pengju Jiang in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
The use of the animal in current study was approved by the Ethics Board of Tongji University School of Medicine. Approval number KS18183.
Acknowledgments
We thank the Jiangsu Key Research and Development Plan (Society Development, No. BE2018639), Science & Technology Support Program of Changzhou (Application Basic Research CJ20190033), Natural Science Foundation of Jiangsu (grant number BK20190566) and the International Scientific Cooperation Project of Changzhou Scientific Bureau (grant numbers CZ20190004, CZ20190009). This work was also supported by The Qing Lan Project of Jiangsu Province.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Jiangsu Key Research and Development Plan (Society Development, No. BE2018639), Science & Technology Support Program of Changzhou (Application Basic Research CJ20190033), Natural Science Foundation of Jiangsu (grant number BK20190566) and the International Scientific Cooperation Project of Changzhou Scientific Bureau (grant numbers CZ20190004, CZ20190009).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.