Abstract
Hypoxia is an important feature of the tumor microenvironment, and is closely associated with cell proliferation, angiogenesis, metabolism and the tumor immune response. All these factors can further promote tumor progression, increase tumor aggressiveness, enhance tumor metastatic potential and lead to poor prognosis. In this review, these effects of hypoxia on tumor biology will be discussed, along with their significance for tumor detection and treatment.
Introduction
Hypoxia is a common and important feature of solid malignancies. Tissue is generally classed as hypoxic if the oxygen tension falls below 10mmHg, compared to 40-60mmHg in most normal tissue. 1 In tumors, hypoxia arises due to a mismatch between oxygen delivery and consumption. Hypoxia is closely associated with tumor progression, increased aggressiveness, enhanced metastatic potential, resistance to radio- or chemo-therapy and lower overall survival of various tumor types. 2 -4 Hypoxia also has the ability to facilitate the selection of apoptosis-resistant cell clones and induce tumor metastasis. 5
Hypoxia may be either acute or chronic. Acute hypoxia is perfusion-related and occurs through transient opening and closing of tumor vessels. Chronic hypoxia is diffusion-related, as the oxygen demand associated with tumor cell proliferation creates a maximum distance beyond which oxygen cannot diffuse. 6 -8 Chronic hypoxia is long-term or possibly irreversible, and generally occurs when the tumor cells are distant from functional vessels.
Tumor cells response to hypoxia depends in part on the duration of exposure. Hypoxic tumor cells may undergo necrosis or reoxygenation, and during these hypoxia-reoxygenation cycles, they may acquire a more aggressive phenotype. 9
Tumor Microenvironment (TME)
Solid malignancies consist of transformed cells embedded in a mass of connective tissue or mesenchyme, which forms the tumor microenvironment, and includes vascular endothelial cells (ECs), cancer-associated fibroblasts (CAFs), immune cells and extracellular matrix (ECM). 10 Both parenchyma and the mesenchyme are morphologically, phenotypically, and functionally heterogeneous at multiple levels: between patients, between primary tumors and their metastases, and even within a single tumor. 11,12 As tumor cells proliferate, they recruit nearby non-tumor cells into collaborative processes to foster a specific tumor microenvironment conducive for local growth and metastasis to distant organs. This collection of tumor cells interacting with host cells forms an abnormal organ-like structure. 13 It is now evident that tumor microenvironment supports tumor growth, causes heterogeneity and contributes to the resistance to systemic therapies. 14
Hypoxia and Proliferation
Oxygen is required for efficient AT production, and hence hypoxia is unsurprisingly linked to reduced cell proliferation. In the classical Pasteur effect, 15 while yeast growth is reduced under anaerobic conditions, their rate of sugar metabolism is increased. In tumors, as demonstrated by Waki et al, well oxygenated cells, which are closely adjacent to functional blood vessels, have a higher proliferation rate, while hypoxic cancer cells, generally located 100-200 µm away from functional blood vessels and/or close to necrotic areas, have a relatively low proliferation rate. 16 Several other studies have also revealed the mutually exclusive pattern between cell proliferation and hypoxia in both microscopic (avascular) and macroscopic (vascularized) tumors. 17 -20 Further, tumors that are hypovascular may appear dormant, with cells either dividing very slowly or resting in G0, 21 -24 though it has also been found that dormancy can result from high proliferation being offset by apoptosis, preventing growth beyond a threshold size. 25,26
The picture of an anti-correlation between hypoxia and proliferation has also been confirmed with radiolabeled tracers. Using autoradiography, Fatema et al showed that 18 F-misonidazole ( 18 F-FMISO, a hypoxia probe) was distributed mostly in the tumor center, whereas 3 H-Fluorothymidine ( 3 H-FLT, a proliferation probe) was found in the periphery. 27 Others also observed a mismatch in the intratumoral distribution of 18 F-FLT and 18 F-FMISO in lung cancers 28 -31 (Figure 1).
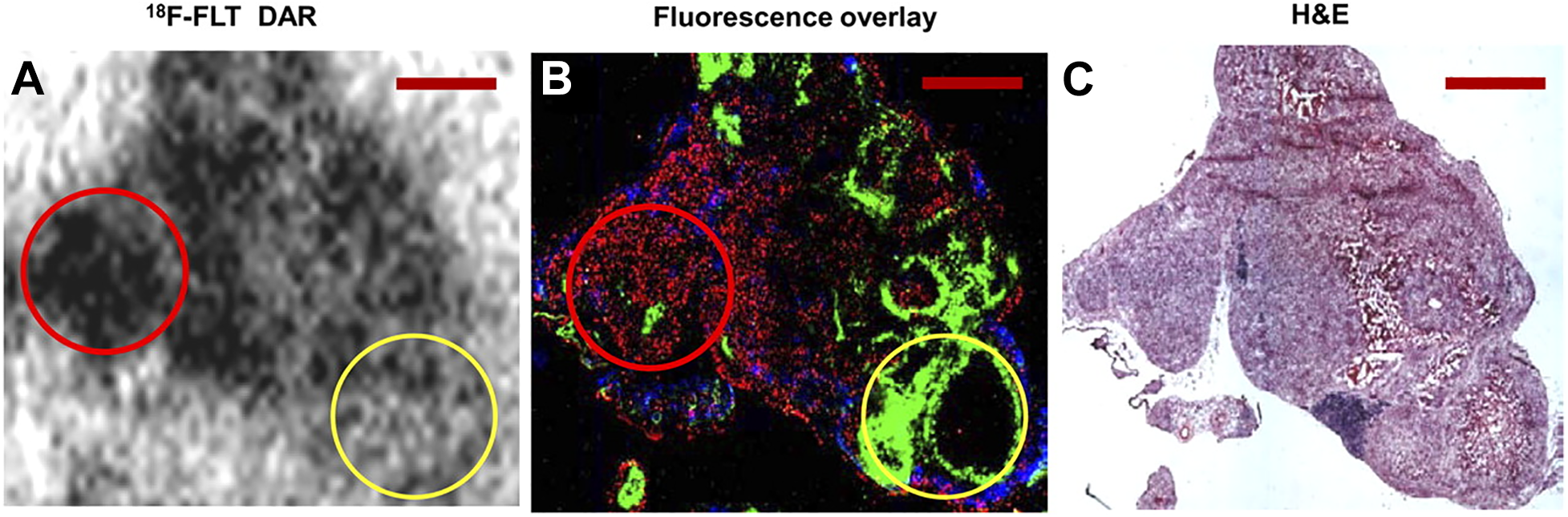
Relationship between hypoxia and cell proliferation. Images derived from a xenograft of HTB177, human lung cancer cell line: (A) 18 F-FLT autoradiography images; (B) A fluorescence overlay showing the hypoxic marker pimonidazole (green); proliferation marker bromodeoxyuridine (red); and blood perfusion marker Hoechst 33342 (blue); (C): H&E staining. Red circle: Well oxygenated (pimonidazole-negative) area is co-localized with high cell proliferation (by high levels of 18 F-FLT uptake and bromodeoxyuridine binding); Yellow circle: Hypoxic (pimonidazole-positive) area is co-localized with low cell proliferation (by lower 18 F-FLT uptake and bromodeoxyuridine binding). This research was originally published in JNM. 28 Huang T, Civelek AC, Li J, et al. Tumor microenvironment-dependent 18 F-FDG, 18 F-fluorothymidine, and 18 F-misonidazole uptake: a pilot study in mouse models of human non-small cell lung cancer. J Nucl Med. 53, 1262-8 (2012) © SNMMI.
Hypoxia and Angiogenesis
Angiogenesis is the process through which new vessels are formed, mainly from existing vessels by means of sprouting. 32 ECs lining the luminal surface of blood vessels are normally quiescent but during development, wound healing, and disease, can be activated to enter the angiogenic process. 33
Tumors initially form in a pre-vascular phase: when the tumor diameter is less than 1 mm, there is no vessel formation, and cells mainly depend on permeation to acquire nutrients. As the tumor grows, vessels develop and cells acquire nutrients through angiogenesis. If angiogenesis fails, the absence of oxygen or other nutrients, restricts tumor size to 2-3mm. 34 -36 Studies by Li et al have shown that the hypoxic status is closely associated with blood perfusion (Figure 2). In a peritoneal metastatic colorectal cancer animal model, they found that microscopic tumors (< 1 mm in diameter) were avascular and extremely hypoxic, while in those of greater size (1-4 mm in diameter), vessels had developed, and hypoxia was minimal 19,20 (Figure 3). As tumor volume increased, the rapid expansion of tumor cells exceeded the local blood supply, and the inefficient tumor vascular system could not provide enough oxygen and other nutrients, resulting in hypoxia in macroscopic tumors.

Hypoxia correlates with low blood perfusion. Images derived from a xenograft of HT29, a human colorectal carcinoma cell line. A, A fluorescence overlay, showing the hypoxic markers pimonidazole (green) and CA9 (red); and blood perfusion marker Hoechst 33342 (blue). Necrotic tissue is marked “N.” B, Relationship between hypoxia and blood perfusion. The scattergram represents the decile-binned mean and 95% CIs (confidence interval) of paired fluorescence intensity values acquired on an 8-Am square pixel-by-pixel basis. •: Hoechst 33342 versus pimonidazole; ˆ: CA9 versus pimonidazole. Reproduced with permission from Cancer Res. 19 Li XF, Carlin S, Urano M, Russell J, Ling CC, O’Donoghue JA. Visualization of hypoxia in microscopic tumors by immunofluorescent microscopy. Cancer Res. 67, 7646-7653 (2007).

Relationship between hypoxia and angiogenesis. Images derived from a xenograft of HT29, a human colorectal carcinoma cell line. A, A fluorescence overlay, showing the hypoxic markers pimonidazole (green) and CA9 (red); and blood perfusion marker Hoechst 33342 (blue). B, Immunohistochemistry staining for CD31 (blood vessel marker). Hypoxic (high pimonidazole and CA9 binding) areas are avascular with little to no blood perfusion (red arrow), while well oxygenated (little to no pimonidazole or CA9 binding) areas have vasculature with extensive blood perfusion (green arrow). Reproduced with permission from Cancer Res. 19 Li XF, Carlin S, Urano M, Russell J, Ling CC, O’Donoghue JA. Visualization of hypoxia in microscopic tumors by immunofluorescent microscopy. Cancer Res. 67, 7646-7653 (2007).
Angiogenesis is activated in response to hypoxia via the HIF1 pathway, and the consequent production of vascular endothelial growth factor (VEGF) both tumor and stromal cells. Tumor-activated ECs have distinctive gene expression profiles and luminal cell-surface receptors, resulting in abnormalities in tumor vasculature. 37 This abnormal vascular network is characterized by leaky and compressed blood and lymphatic vessels leads to further regional hypoxia, creating a vicious cycle. As a result, tumor cells move through periods of hypoxia-reoxygenation that produce a more aggressive phenotype. 38
Hypoxia and Metabolism
Low extracellular pH is another feature of solid tumors and hypoxia leads to acidosis in the tumor microenvironment. 39,40 Most cancers are characterized by a high rate of glycolysis, as low oxygen levels force tumor cells to shift from oxidative phosphorylation to glycolysis, with a resultant production of metabolic acids. 41 -43 Like increased angiogenesis, these changes are mediated by hypoxia-inducible factor 1α (HIF-1α) driven transcription, which leads to upregulation of both glucose transporters and genes involved in glycolysis. 44 Li et al have observed a strong association between hypoxia, glucose transporter-1 (GLUT-1) and 18 F-fluorodeoxyglucose ( 18 F-FDG) uptake 43,45 (Figure 4).

Relationship between glucose metabolism, hypoxia and cell proliferation. Circle: Hypoxic (high pimonidazole binding) area is co-localized with high 18 F-FDG uptake and high GLUT-1 expression. Square: Relatively non-hypoxic (lower pimonidazole binding) area is co-localized with lower 18 F-FDG uptake and lower GLUT-1 expression. This research was originally published in JNM. 43 Li XF, Ma Y, Sun X, Humm JL, Ling CC, O’Donoghue JA. High 18 F-FDG uptake in microscopic peritoneal tumors requires physiologic hypoxia. J Nucl Med. 51, 632-38 (2010). © SNMMI.
The mechanistic/mammalian target of rapamycin (mTOR) pathway is a central signaling pathway that controls metabolic processes. Multiple points along the glycolytic pathway are influenced by mTOR via regulation of critical transcription factors such as HIF-1α. 46 The expression of HIF-1α is dependent on mTORC1 and mTORC2. 47 Both mTORC1 and mTORC2 play important roles in glucose uptake and glycolytic metabolism of tumor cells. 48,49 Thus, application of mTOR inhibitors to control tumor metabolism and tumor microenvironment will become innovative anti-tumor strategy. 50
Even in the absence of hypoxia, oncogene activation may also promote glycolysis through upregulation of enzymes and cell surface nutrient transporters. 39 Therefore, tumor acidity depends on both physiological and genetic factors. Sengupta et al revealed a more complex tumor metabolism which has significant heterogeneity. However, they found little correlation between FDG uptake and lactate release. 51 This suggests that tumor cells have metabolic plasticity: They can use a variety of anabolic and catabolic pathways to adapt to energetic needs and availability of nutrients. 52 Pathways available for tumor cells to produce adenosine triphosphate (ATP) include oxidative phosphorylation, glutaminolysis, and fatty acid metabolism. Investigating and monitoring tumor metabolism, therefore, can be an essential step leading to a deeper understanding, better diagnostics, and more effective therapeutic strategies for tumor treatment.
Hypoxia and the Immune System
Cancer-associated inflammation is involved in all stages of tumorigenesis and development, which may cause genomic instability, cell proliferation, angiogenesis, enhancement of anti-apoptotic pathways and cancer dissemination. 53- 56 Hypoxia can enhance the resistance of tumor cells to immune attack and evade immunosurveillance. 57 Hypoxia-inducible factors (HIFs), the key mediator of hypoxia signaling, regulate a series of genes that are responsible for tumor immunity under hypoxic condition. 58
Studies indicate that intratumoral hypoxic regions are infiltrated by high levels of myeloid-derived suppressor cells (MDSCs), tumor-associated macrophages (TAMs), and T-regulatory (Treg) cells. 57,59 Under long-term exposure to hypoxia, activated HIF-1α may reduce and inactivate cytotoxic lymphocytes, including NK cells and CD8+ T cells, and result in the inhibition of their anti-tumor efficacy. 60- 62 Furthermore, increased production of lactate in TME can inactivate cytotoxic lymphocytes, such as NK cells and CD8+ T cells, thus resulting in the reduction of their anti-tumor effect. 63,64 Mahiddine et al 65 found that relief of tumor hypoxia can unleash the tumoricidal potential of neutrophils. Furthermore, there is robust evidence that hypoxia has a strong impact on the induction of HIF-1α-dependent PD-L1 expression in tumor cells, macrophages, and dendritic cells, which is a crucial mechanism to block antitumor immunity by cancer cells. 66- 69
Hypoxia increases glycolytic rate of B cells, which reduces proliferation, increases apoptosis, and impairs class-switching. HIF will be activated at key points in the life of B cells, which may shape and control B cell fate and function. Constitutive HIF stabilization via deletion of Vhl reduces GC B cells and impairs affinity maturation, class-switching to the pro-inflammatory IgG2c isotype, recall response and early memory B cell formation. Thus, hypoxia induced over activation of HIF is detrimental to B cell function. 70,71
The effect of hypoxia on Dendritic cells (DCs) in the tumor microenvironment has not been well described. Current reports suggest that hypoxia may be beneficial to the ability of DC to induce immune response. 72,73 Given that the effect of hypoxia was mediated by HIF1α expression, HIF1α modulates survival, migration, antigen presentation, interferon synthesis and differentiation of DCs. 74 HIF1α has also been found to positively regulate the migratory capability of human monocyte-derived DCs. 75
Detecting Tumor Hypoxia
Several methods can be used for hypoxia detection: Polarographic oxygen electrodes: the gold standard for hypoxia detection, this is an invasive method which directly measures tissue oxygen pressure. This method is only used for scientific research, and can not be applied to clinical. Positron emission tomography (PET): a widely used imaging technology in clinic, which is a non-invasive, three-dimensional, direct assessment of intratumoral oxygen levels. By employing different radiotracer probes to directly report cell oxygen levels, rather than through hypoxia mediated phenotype changes,
76
PET has high sensitivity and specificity toward regional hypoxia and also can monitor the whole tumors. However, its spatial resolution is relatively low.
77-
79
At present, widely used radiotracer probes for hypoxia imaging include 18F-FDG, 18F-FMISO, 18F-Fluoroazomycin-arabinofuranoside (18F-FAZA), 18F-2-(2-nitro-1H-imidazol-1-yl)-N-(2,2,3,3,3-pentaflfluoropropyl)-acetamide (18F-EF5), 131I-labeled iodo-azomycin galactopyranoside (131I-IAZGP). Immunofluorescence and immunohistochemical methods: an indirect manner to measure hypoxia in tissue sections by assessing hypoxia-related protein expression, such as exogenous hypoxic markers (pimonidazole, EF-5) and endogenous cell markers (HIF-1, VEGF, CA9, GLUT-1/3). This method is only used to evaluate the hypoxic state of tissue in vitro.
Potential Treatment Exploiting Tumor Hypoxia
Several therapeutic strategies targeting hypoxia have been developed. It may be possible to directly deliver O2 into the tumor through different methods such as the catalytic decomposition of endogenous hydrogen peroxide (H2O2) and light-triggered water splitting. 80
There are also many ongoing hypoxia-targeted therapy trials involving several kinds of drugs with different mechanisms. HIF inhibitors are an important type of hypoxia targeted therapy drugs and a large number of studies have shown that HIF inhibitors (such as PX-478, LW6), either as a single agent or in combination with other therapeutic agents, exhibited promising anti-tumor effects. 81- 84
Hypoxia-activated prodrugs (HAPs) are bioreductive drugs that are selectively activated under hypoxic conditions and that can accurately target the hypoxic regions of solid tumors. Researches on HAPs is currently underway. Representative drugs include tirapazamine, evofosfamide. A large number of pre-clinical and clinical studies have reported exciting anti-tumor effects, regardless of single-agent application or combination therapy. 85- 87
Other strategies targeting hypoxia are also under study. As mentioned above, mTOR pathway plays a key role in tumor metabolism, mTOR targeted drugs are essential for regulating the tumor microenvironment under hypoxia. 88 Jain et al. 13,89- 91 have proposed 2 strategies to alleviate hypoxia: vascular normalization and vessel decompression. They conducted a lot of research to improve the efficacy of anti-vascular treatment (anti VEGF/VEGFR drugs, such as bevacizumab, sunitinib), and suggested that strategies aimed at alleviating tumor hypoxia while improving perfusion may enhance the outcome of radiotherapy, chemotherapy, and immunotherapy. Since hypoxia has a strong impact on the induction of HIF-1α-dependent PD-L1 expression in tumor cells, blockade of PD-L1 may thus represent a novel approach for cancer immunotherapy. 69,92
These kinds of studies are still in their early phase and need to be further investigated to overcome challenges for successful clinical translation. Further studies of hypoxia in tumors are of utmost importance in both biological and therapeutic implications.
Conclusion
Hypoxia is an important property of solid malignancies, and is widely involved in all aspects of tumorigenesis and development. Lack of vascularity is the main cause of hypoxia, which in turn leads to low cell proliferation, increased glucose metabolism, immunosuppression and resistance to immune attack. At present, a variety of methods are developed to detect tumor hypoxia, among which, PET imaging technology using radiotracer probes has great advantages due to its non-invasive, three-dimensional assessment and high sensitivity and specificity and has been widely applied in clinic. For therapeutic purposes, hypoxia has been used as a target to develop a variety of therapeutic strategies. Hypoxia-activated prodrugs (HAPs) have been developed and validated since 1960s. Many types of HAPs are already in phase 3 clinical trials. The application of hypoxia targeted therapy will open up a new way for tumor treatment.
Footnotes
Abbreviations
Authors’ Note
Yue Li performed the literature search and wrote the manuscript; Long Zhao performed the literature search and figure editing; Xiao-Feng Li contributed to write and revise the manuscript; all authors had approved the final manuscript to be submitted. This is a review article with no original animal and human studies.
Acknowledgments
We thank Dr. James Russell PhD from Memorial Sloan Kettering Cancer Center (New York, NY, USA) for critical reading the article. The authors’ research is supported in part by a grant from Shenzhen People’s Hospital for “Climbing” Program (X-FL) and a Shenzhen Science and Technology Project grant (JCYJ20190806151003583)(X-FL).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
