Abstract
Objective:
The purpose of this meta-analysis was to study the prognostic effects of androgen receptor splicing variant 7 (AR-V7) on metastatic castration-resistant prostate cancer (mCRPC) under different treatment options (chemotherapy, hormone therapy).
Methods:
We conducted a systematic search of PubMed, EMBASE and Cochrane databases for clinical studies up to June 4, 2021, and used prostate-specific antigen (PSA) progression free-survival (PSA-PFS), radiologic PFS (r-PFS), overall survival (OS) and PSA response rate (PSA RR) as the main endpoints. Subgroup analyses were conducted based on the source of the specimens. STATA v.15 software was used for data analysis.
Results:
Twenty-one studies were included in this meta-analysis, with a total of 1578 samples. In the abiraterone (AA)/enzalutamide (E) treatment group, AR-V7 positive patients had worse PSA-PFS (hazard ratio [HR] = 3.40; 95% confidence interval [95%CI] 2.56-4.51; P < 0.05) and worse r-PFS (HR = 2.69; 95%CI 1.70-4.24; P < 0.05) and OS (HR = 3.02; 95%CI 1.73-5.30; P < 0.05). Multivariate Cox regression results showed that AR-V7 positive status was an independent risk factor for OS in the AA/E treatment group. In the taxane treatment group, AR-V7-positive and negative patients had similar PSA-PFS (HR = 0.87; 95%CI 0.46-1.63; P = 0.657), r-PFS (HR = 1.01; 95%CI 0.53-1.96; P = 0.965) and OS (HR = 1.50; 95%CI 0.89-2.52; P = 0.127). For AR-V7-positive patients, the difference in OS between taxane and AA/E treatment was not statistically significant (HR = 1.03; 95%CI 0.52-2.06; P = 0.930). However, multivariate Cox regression results suggested that for AR-V7-positive patients, taxane therapy was a protective factor for OS (HR = 0.35; 95%CI 0.20-0.60; P < 0.05).
Conclusion:
The expression of AR-V7 indicates a poor prognosis and is an independent risk factor for OS in AA/E-treated mCRPC patients. However, AR-V7 positive status does not play the same role in taxane-treated patients. In addition, compared to AA/E, taxane treatment is a protective factor for OS in AR-V7-positive patients. AR-V7 may thus be an effective biomarker for treatment prognosis in patients with mCRPC.
Introduction
Androgen receptor (AR) is commonly found in normal prostate and prostate cancer cells and plays an important role in prostate cancer. Castration therapy with the androgen synthesis inhibitor abiraterone (AA) and the AR signaling inhibitor enzalutamide (E) confers survival advantages in such diseases. 1 -4 However, 10%-20% of patients with metastatic castration-resistant prostate cancer (mCRPC) show primary resistance to castration therapy, and all prostate cancers eventually develop secondary resistance. 5 During this process, AR changes, such as AR gene amplification, key point mutations and expression of androgen receptor splicing variants (ARVs), are multiple mechanisms that lead to castration resistance. Currently, more than 20 different ARVs have been identified. 6 The most widely studied ARV is androgen receptor splicing variation 7 (AR-V7). AR-V7 retains the N-terminal domain and DNA binding domain, but lacks the C-terminal ligand binding domain, so it is hormone-independent and can be activated without ligand. 7 -9 AR-V7 is expressed and transcribed at a high level in mCRPC cells and can lead to continuous activation of the AR signaling pathway, which is an important mechanism underlying resistance to androgen deprivation therapy (ADT) in prostate cancer.
A previous study indicated that AR-V7 positive status was associated with worse disease progression and shorter survival time. 10 It is worth noting that the expression of AR-V7 does not affect the clinical outcome of patients receiving taxane chemotherapy. 11,12 At present, clinical applications targeting AR-V7 are not yet common. Therefore, this study analyzes the influence of AR-V7 expression on the prognosis of mCRPC patients under different treatment options (AA/E, taxane) in order to provide evidence-based medical evidence to further guide the choice of treatment options.
Methods
Search Strategy
Two independent researchers systematically searched studies included in PubMed, EMBASE and Cochrane databases up to June 4, 2021. The search keywords used were “metastatic castrate resistant prostate cancer (mCRPC),” “Androgen Receptor Splicing Variant 7 (AR-V7),” and “prognosis.” According to the different requirements of each database, the search strategies were changed accordingly, and potentially relevant articles were also sought in the references of relevant studies.
Inclusion and Exclusion Criteria
Studies were included if they conformed to the principles of PICO(S): participants, interventions, comparisons, outcomes (study design). 13 Inclusion criteria were as follows: (1) Participants: patients who were diagnosed with mCRPC; (2) Interventions: treated with AA/E or taxane; (3) comparisons: AR-V7-positive and AR-V7-negative patients; (4) outcomes: prostate-specific antigen-progression-free survival (PSA-PFS), radiologic PFS (r-PFS), overall survival (OS) and PSA response rate (PSA RR); (5) study design: clinical research based on the prognosis of AR-V7 expression in patients with mCRPC. Exclusion criteria: (1) use of a non-AR-V7 marker; (2) non-mCRPC patients; (3) lack of data, or (4) original data impossible to obtain from the author; (5) case reports, letters, conference abstracts, reviews, animal experiments, expert comments.
Data Extraction and Quality Assessment
Two researchers independently extracted data from the studies. For each included study, the following information was extracted: first author, year of publication, research type, total number of specimens, number of AR-V7-positive specimens, number of AR-V7-negative specimens, sample source, treatment strategy, information on outcomes (PSA-PFS, r-PFS, OS, PSA RR). The Newcastle-Ottawa Scale (NOS) scoring system was used to evaluate the quality of the included studies. 14
Statistical Analysis
STATA v.15 software was used for statistical analysis. The odds ratio (OR) was used to express dichotomous variables. Hazard ratios (HRs) for PSA-PFS, r-PFS and OS were extracted from survival curves and calculated using the method presented by Tierney et al. 15
The confidence interval (CI) was set at 95%, and P < 0.05 was defined as statistically significant. The Chi-square test or Cochrane Q test was used to evaluate heterogeneity. I2 < 50%, P > 0.10 was defined as no significant heterogeneity, and non-heterogeneous data was evaluated using the fixed-effects model; otherwise, the random effects model was used. 16,17 Subgroup analysis was performed based on the source of the specimen, while sensitivity analysis was conducted on the results of more than 6 cases included in the literature to assess the stability of the outcome. In addition, Funnel plot and Begg’s tests were used to test the publication bias of the included studies. 18
Results
Study Selection and Characteristics
A total of 21 studies were included in this meta-analysis (18 prospective studies 10 -12,19 -33 and 3 retrospective studies 34 -36 ). A total of 1578 samples were analyzed; AR-V7-positive group, n = 557; AR-V7-negative group, n = 1021. The basic characteristics of the included studies are summarized in Table 1. A flow diagram of study selection is shown in Figure 1. All included studies had NOS scores ≥ 6. The prognostic outcomes of receiving AA/E treatment are summarized in Table 2. The prognostic outcomes of receiving taxane chemotherapy are summarized in Table 3. PSA RR according to the different treatments are summarized in Table 4. For AR-V7-positive patients, the comparison of overall survival after taxane and AA/E treatment is summarized in Table 5. Forest plots of all outcomes are shown in Figures 2 –5. The sensitivity analysis of these results and the published bias are shown in Figures 6–7.
Characteristics of Included Studies.
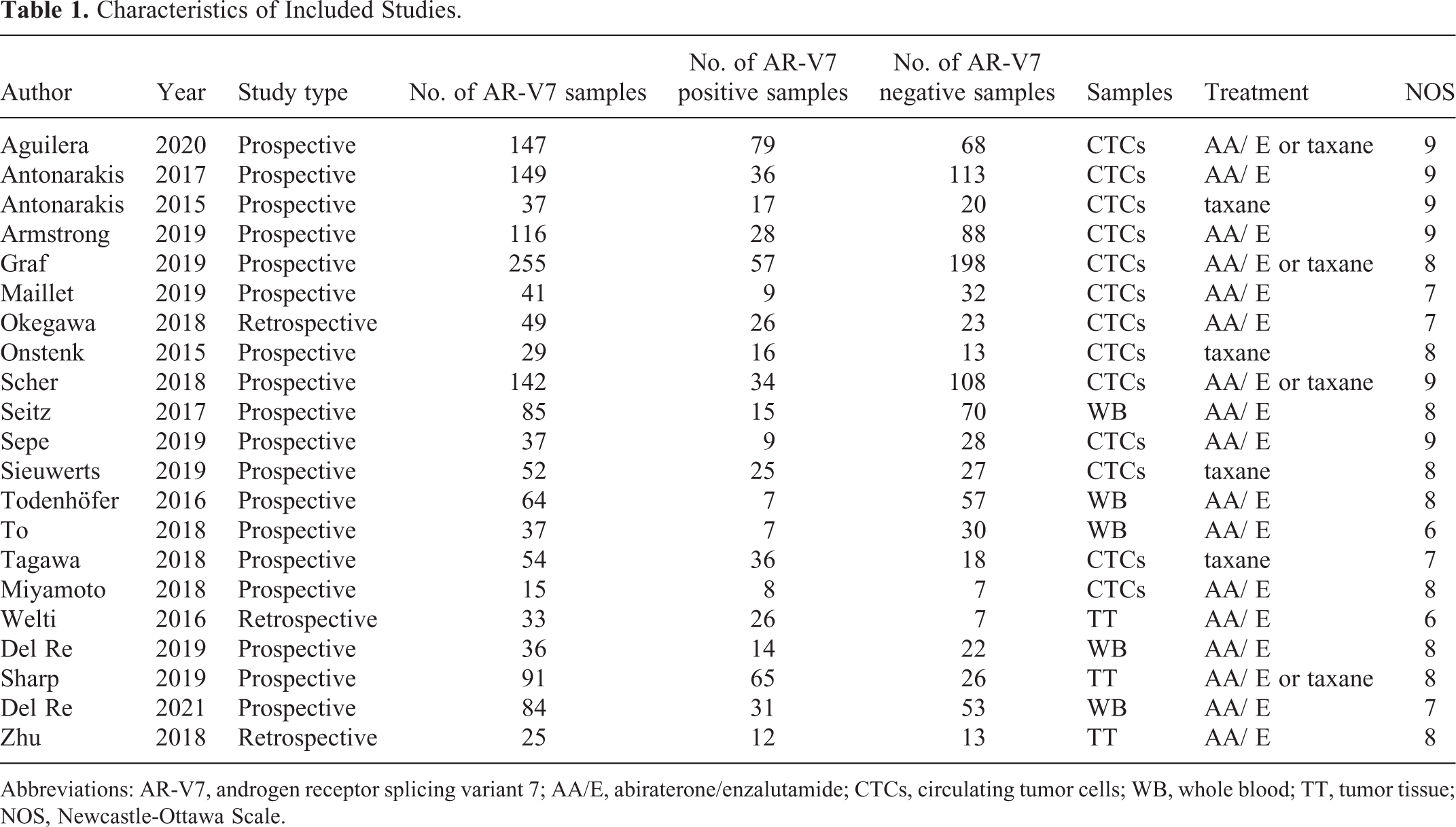
Abbreviations: AR-V7, androgen receptor splicing variant 7; AA/E, abiraterone/enzalutamide; CTCs, circulating tumor cells; WB, whole blood; TT, tumor tissue; NOS, Newcastle-Ottawa Scale.

Flow diagram of study selection.
Prognostic Outcomes of AR-V7 in the AA/E-Treated Cohort.

Abbreviations: AR-V7, androgen receptor splicing variant 7; AA/E, abiraterone/enzalutamide; CTCs, circulating tumor cells; WB, whole blood; TT, tumor tissue; PSA-PFS, PSA progression-free survival; r-PFS, radiologic progression free survival; OS overall survival.
a Multivariate Cox model.
Prognostic Outcomes of AR-V7 in the Taxane-Treated Cohort.

Abbreviations: AR-V7, androgen receptor splicing variant 7; AA/E, abiraterone/enzalutamide; CTCs, circulating tumor cells; TT, tumor tissue; PSA-PFS, PSA progression free survival; r-PFS, radiologic progression free survival; OS, overall survival.
a Multivariate Cox model.
PSA RR (AR-V7 Positive vs. Negative).
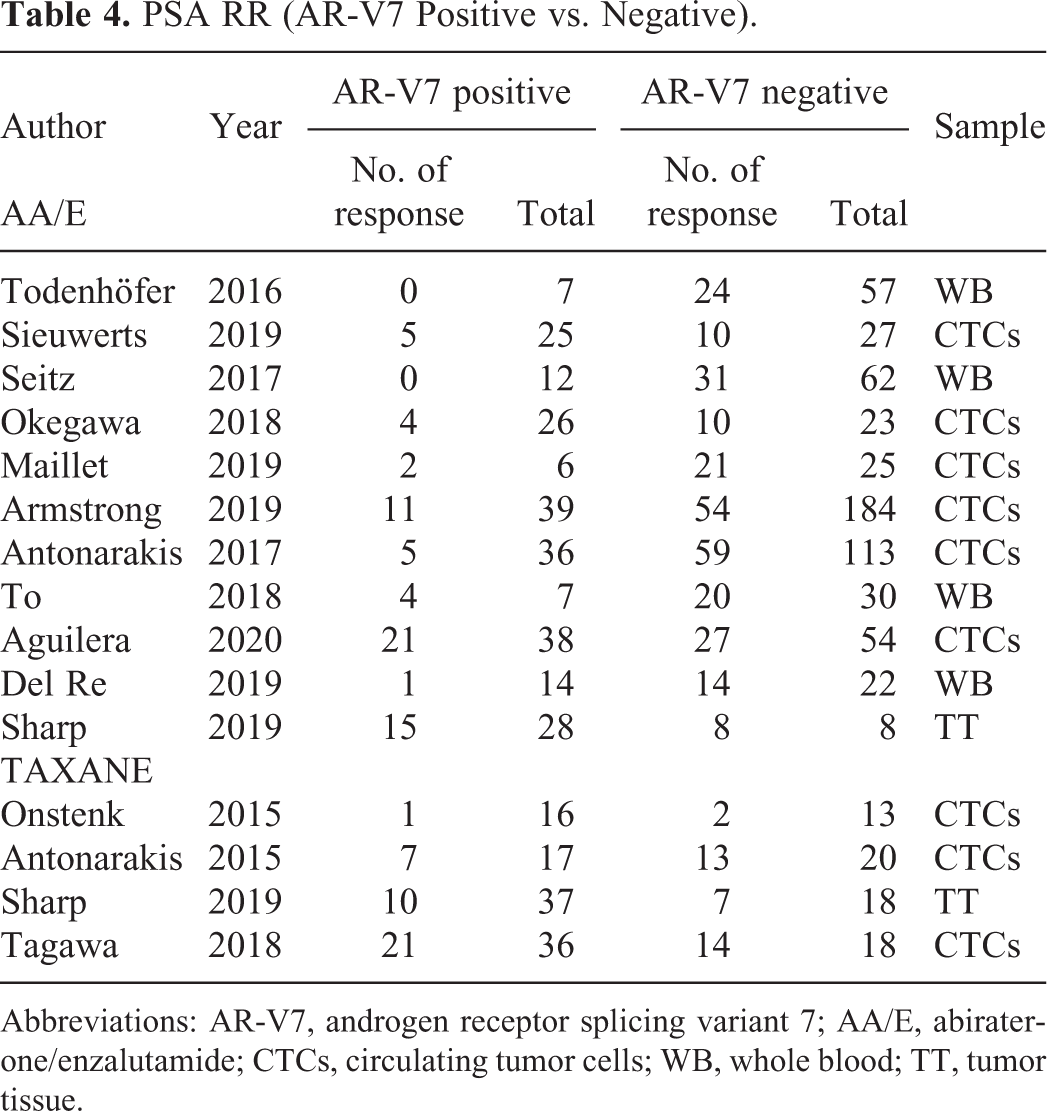
Abbreviations: AR-V7, androgen receptor splicing variant 7; AA/E, abiraterone/enzalutamide; CTCs, circulating tumor cells; WB, whole blood; TT, tumor tissue.
Taxane vs AA/E-Treated AR-V7-Positive Patients.

Abbreviations: AR-V7, androgen receptor splicing variant 7; AA/E, abiraterone/enzalutamide; HR, hazard ratio.
a Multivariate Cox model.

Forest plots of prognostic outcomes in the AA/E-treated cohort (AR-V7 positive vs. negative). (A) PSA-PFS, (B) multivariate Cox model for PSA-PFS, (C) r-PFS, (D) multivariate Cox model for r-PFS, (E) OS, (F) Multivariate Cox model for OS.

Forest plots of prognostic outcomes in the taxane-treated cohort (AR-V7 positive vs. negative). (A) PSA-PFS, (B) multivariate Cox model for PSA-PFS, (C) r-PFS, (D) OS, (E) Multivariate Cox model for OS.

Forest plots of PSA RR (AR-V7 positive vs. negative). (A) AA/E-treated cohort, (B) Taxane-treated cohort.

Forest plots of AR-V7-positive patients (taxane vs AA/E). (A) OS, (B) Multivariate Cox model for OS.

Sensitivity analysis of clinical outcomes. (A) PSA-PFS in the AA/E-treated cohort, (B) r-PFS in the AA/E-treated cohort, (C) OS in the AA/E-treated cohort, (D) OS in the taxane-treated cohort, (E) PSA RR in the AA/E-treated cohort (AR-V7 positive vs. negative).
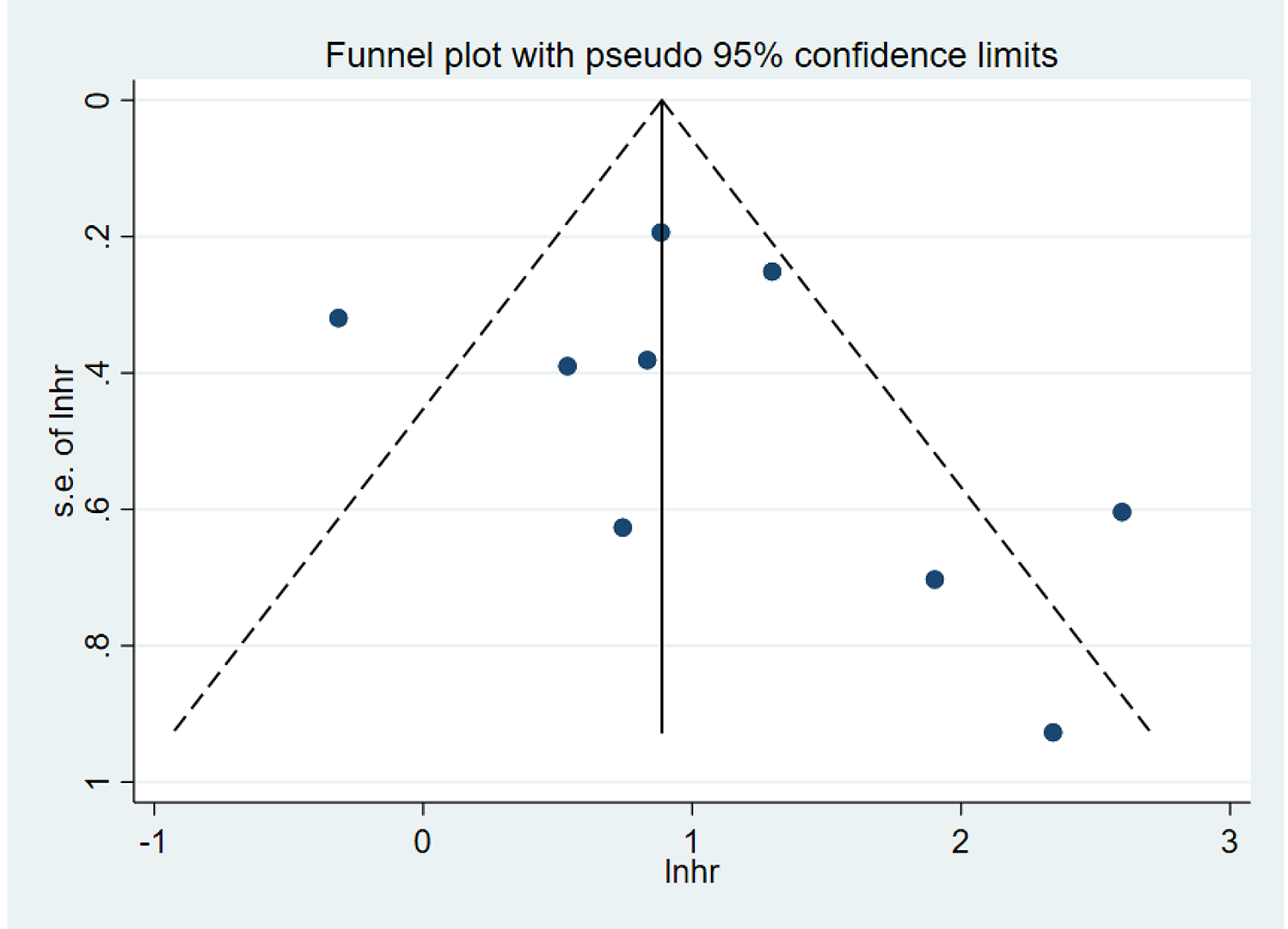
Funnel plot of publication bias based on r-PFS of the AA/E-treatment group (Begg’s test, P = 0.532).
Prognostic Outcomes of AA/E Treatment Groups
Eight studies reported PSA-PFS. 19,23,24,26,27,32,34,36 The results showed that the AR-V7-positive group had worse PSA-PFS than the AR-V7-negative group, and the difference was statistically significant (HR = 3.40; 95%CI 2.56-4.51; P < 0.05). No significant heterogeneity was observed (I2 = 30.6%); therefore, a fixed effects model was used (Figure 2A). This result indicated that in ARV7 positive mCRPC patients, AA/E treatment may not bring more benefit in PSA-PFS. Moreover, the results of multivariate Cox regression analysis showed that AR-V7 was not an independent risk factor for PSA-PFS in the AA/E treatment group (HR = 2.45; 95%CI 0.37-16.29; P = 0.354; Figure 2B). 24,31,34 Studies included in the multivariate Cox regression analysis is insufficient and with high heterogeneity (I2 = 88.2%). In the future, high-level evidence- based medicine evidence is needed to confirm the result.
Ten studies reported r-PFS. 19 -21,23 -27,32,34 The results showed that the AR-V7-positive group had worse r-PFS than the AR-V7-negative group, and the difference was statistically significant (HR = 2.69; 95%CI 1.70-4.24; P < 0.05). Heterogeneity was observed (I2 = 70.4%); therefore, the random effects model was used (Figure 2C). Subgroup analysis based on samples circulating tumor cells [CTCs], whole blood [WB] and tumor tissue (TT) were consistent with the primary outcome which suggested that sample sources may not affect the accuracy of the test. The results of multivariate Cox regression analysis indicated that AR-V7 was an independent risk factor for r-PFS in the AA/E treatment group (HR = 3.67; 95%CI 1.17-11.50; P < 0.05). The forest plot is shown in Figure 2D. 20,34 Limited by the number of included literature, this result needs further confirmation.
There were 12 studies reporting OS. 10,19 -22,24 -27,32,34,35 The results showed that the AR-V7-positive group had worse OS than the AR-V7-negative group, and the difference was statistically significant (HR = 3.02; 95%CI 1.73-5.30; P < 0.05). Heterogeneity was observed (I2 = 91.9%); therefore, the random effects model was used (Figure 2E). Subgroup analysis was also conducted based on sample source (CTCs, WB, TT) and was consistent with the primary outcome. Moreover, the results of multivariate Cox regression analysis suggested that AR-V7 is an independent risk factor for poor OS in the AA/E treatment group (HR = 4.46; 95%CI 3.13-6.34; P < 0.05). 20,31,33,34 The forest plot is shown in Figure 2F. This result indicated that AR-V7 may be an effective biomaker in predicting the survival of mCRPC patients with AA/E treatment.
Prognostic Outcomes of Taxane Treatment Groups
Four studies reported PSA-PFS for patients treated with taxane. 11,12,24,32 The results showed that the PSA-PFS in the AR-V7-positive and negative groups was not significantly different (HR = 0.87; 95%CI 0.46-1.63; P = 0.657). Heterogeneity was observed (I2 = 74.6%); therefore, the random effects model was used (Figure 3A). The results of multivariate Cox regression analysis indicated that AR-V7 was not an independent risk factor for PSA-PFS in the taxane treatment group (HR = 0.84; 95%CI 0.25-2.83; P = 0.779). 11,24 The forest plot is shown in Figure 3B.
There were 4 studies reporting r-PFS 11,24,29,32 for taxane-treated patients. These results showed that r-PFS of the AR-V7-positive and negative groups was not significantly different (HR = 1.01; 95%CI 0.53-1.96; P = 0.965). Heterogeneity was observed (I2 = 69.7%); therefore, the random effects model was used (Figure 3C).
Six articles reported OS. 10 -12,22,24,32 The results showed that the AR-V7-positive and negative groups had similar OS (HR = 1.50; 95%CI 0.89-2.52; P = 0.127). Heterogeneity was observed (I2 = 72.4%); therefore, the random effects model was used (Figure 3D). Multivariate Cox regression indicated that AR-V7 was not an independent risk factor for OS in the taxane treatment group (HR = 1.61; 95%CI 0.86-3.01; P = 0.135). 11,28 The forest plot is shown in Figure 3E.
The above results did not suggest a prognostic role for AR-V7 in taxane treatment. However, limited by the number of included literature and the existence of heterogeneity, it should be more conservative to draw a conclusion.
PSA RR in Different Treatments
There were 15 studies reporting PSA RR, including 11 with AA/E treatment groups 19 -21,23,24,26,28,30 -32,34 and 4 with taxane treatment groups. 11,12,29,32 The results showed that the PSA RR of AR-V7-positive patients in the AA/E treatment groups was lower than the AR-V7-negative group (OR = 1.43; 95%CI 1.10-1.85; P < 0.05). Heterogeneity was not observed (I2 = 0%); therefore, the fixed effects model was used. Subgroup analysis based on sample source (CTCs, WB, TT) was consistent with the primary result. The PSA RR of AR-V7-positive patients in the taxane treatment group was similar with AR-V7-negative patients (OR = 1.53; 95%CI 0.88-2.64; P = 0.125). No heterogeneity was observed (I2 = 0%); therefore, a fixed effects model was used (Figure 4A and B). In mCRPC patients treated with taxane, AR-V7 may not play the same role in predicting PSA RR as in AA/E treatment group. In addition, results above were consistent with the outcome of PSA-PFS in AA/E and taxane treatment.
Comparison of Prognosis for AR-V7-Positive Patients in Different Treatment Groups
Three studies reported the OS for AR-V7 positive-patients in the taxane and AA/E treatment groups. 10,11,22 The results showed that OS of the 2 treatment groups was similar (HR = 1.03; 95%CI 0.52-2.06; P = 0.930). Heterogeneity was observed (I2 = 62.0%); therefore, the random effects model was used. Interestingly, the results of multivariate Cox regression suggested that in AR-V7-positive patients, taxane chemotherapy was a protective factor for OS (HR = 0.35; 95%CI 0.20-0.60; P < 0.05). No heterogeneity was observed (I2 =0%); therefore, a fixed effects model was used (Figure 5A and B). For AR-V7-positive mCRPC patients, taxane may bring more survival benefit than AA/E, which bring a light on how to choose treatment options in these patients.
Sensitivity Analysis and Publication Bias
Sensitivity analysis was performed on the results of more than six included studies, and we found the results were relatively stable (Figure 6A-E). The results for r-PSA after AA/E treatment were selected for evaluation of publication bias, but no obvious publication bias was observed. (Begg’s test, P = 0.532). This result is shown in Figure 7.
Discussion
According to the 2020 version of the European Association of Urology (EAU) consensus, diagnosis of CRPC requires the following—(i) serum testosterone below 50 ng/dl or 1.7 nmol/L; (ii) 3 consecutive rises in PSA 1 week apart, with two 50% increases over the nadir, and PSA >2 ng/ml; or 2 or more bone lesions or one soft tissue lesion on imaging. 37 Treatment decisions in CRPC are not one-way, but are based on multifactorial considerations, such as previous treatment regimens; genetic alterations; and drug resistance. 38 Previous studies have shown that the median time to bone metastases in nonmetastatic CRPC (nm-CRPC) patients is 25-30 months, and 33%-46% of nm-CRPC patients progress to metastatic CRPC within 2 years of diagnosis. 39 -41 Managements of patients with nm-CRPC are as follows: (i) prostate-specific androgen doubling time (PSA-DT) ≤ 10 mo, survival benefit will achieve by adding modern anti-androgens to ADT (SPARTAN, 42 PROSPER, 43 ARAMIS 44 ); (ii) PSA-DT > 10 mo, regular monitoring is better than early intervention. 37 For mCRPC, first-line treatments include abiraterone (COU-AA-302 trial), 45 enzalutamide (PREVAIL trial), 46 docetaxel chemotherapy 47,48 and Sipuleucel-T, 40 which showed a significant survival benefit. In the 2020 EAU guidelines, the recommended strength of these treatments is strong in mCRPC. 37 Although treated with standard first lines of therapy, patients with mCRPC will eventually progress. How should mCRPC patients choose second-line or other sequential therapy after disease progression? The TROPIC trial showed that patients treated with a maximum of 10 cycles of cabazitaxel had significantly better OS, compared to mitroxantrone treatment after docetaxel-based therapy. 49 Similarly, abiraterone, 50 enzalutamide (AFFIRM trial), 3 radium-223 (ALSYMPCA study) 51 also achieved survival benefits for patients after docetaxel. For mCRPC that progresses within 1 year after treatment with AA/E, the CARD study demonstrated that the choice of cabazitaxel as a sequential treatment is clear 52 ; for patients who response to AA/E for over 1 year, radium-223 or cabazitaxel are both reasonable options. 37 Other sequential treatment options for patients who already have docetaxel chemotherapy or first-line novel hormonal therapy options are: olaparib (a poly ADP ribose polymerase inhibitors) for patients with mutation in DNA-repair genes, 53,54 and PSMA therapy. 55,56 Both treatment options showed promising results for the selected patients. In this present study, we focused on the relationship between the expression of AR-V7 and prognosis under different treatment options, wherein we conducted a meta-analysis to determine whether AR-V7 markers can effectively guide treatment decisions for mCRPC patients.
AR-V7 can be activated without a ligand, which is an important mechanism underlying androgen resistance and ADT resistance in prostate cancer, and ultimately leads to a poor prognosis. This research study examined the PSA RR of AR-V7-positive patients in different treatment groups. The results suggested that the PSA RR of AR-V7-positive patients in the AA/E treatment group was significantly lower than that of AR-V7-negative patients. However, in the taxane treatment group, the difference between the 2 patient groups was not statistically significant. Maillet et al further studied the dynamic of PSA response as a function of time according to AR-V7 status and showed that almost all AR-V7-positive patients with an initial PSA response had a shorter PSA response time compared with AR-V7-negative patients. 23 PSA response is closely related to PSA-PFS. A study by Armstrong et al indicated that even if there were AR-V7-positive patients whose PSA decreased by more than 50% from baseline, the PSA-PFS of such patients was still very short, with only 9% of patients having a PFS greater than 6 months. 20 During the treatment, the proportion of tumor patients expressing AR-V7 gradually increases. Onstenk et al detected AR-V7 in the CTCs of all patients treated with AA, while this proportion was only 35% in patients who had not received AA treatment. 12 Previous studies had speculated that in the process of transforming hormone-sensitive prostate cancer (HSPC) to CRPC, the status of AR-V7 in patients could be used to predict prognosis and treatment response. 35 The present meta-analysis showed that the PSA-PFS, r-PFS and OS of AR-V7-positive patients after treatment with AA/E were significantly worse than those of AR-V7-negative patients. The results using the multivariate Cox model also suggested that AR-V7-positive status is an independent risk factor for poor r-PFS and OS. Therefore, the results of this study support the promotion of AR-V7 expression in the progression of mCRPC, which ultimately leads to a poor prognosis in the AA/E treatment group.
Our research also studied the PSA RR in the taxane treatment group. The results suggested that the PSA RR in AR-V7-positive patients was similar with negative patients. The question is whether AR-V7-positive patients are more resistant to taxane treatment. A study by Marín-Aguilera et al pointed out that high expression of AR-V7 during taxane treatment was related to poor r-PFS and was consistent with the conclusion drawn in the TAXYNERGY trial. 24,29 It was hypothesized that taxane may exert its anti-tumor effect by blocking transport of AR in the microtubule network from the cytoplasm to the nucleus. Therefore, to some extent, taxane treatment may lead to cross-resistance of AR target therapy and taxane chemotherapy in CRPC patients. 57 -61 In contrast, a study by Antonarakis et al proved that AR-V7 status has nothing to do with the initial resistance to taxane, and the clinical outcome of AR-V7-positive patients was not significantly different from that of negative patients. These authors also proposed that AR-V7-positive patients treated with taxane had better outcomes than patients treated with AR-targeted therapy; however, this outcome was not significantly different among negative patients. 11 Our study analyzed the prognosis of the taxane treatment group, and the results showed that PSA-PFS, r-PFS and OS of AR-V7-positive patients was not significantly different from negative patients. Therefore, our study does not support the conclusion that promotion of AR-V7 expression in the progression of mCRPC during taxane treatment ultimately leads to a poor prognosis.
In the present study, 238 AR-V7-positive patients were enrolled in the AA/E treatment group, with a PSA RR of 28.57%, while 106 people were enrolled in the taxane treatment group, with a PSA RR of 36.79%; the difference was statistically significant (P = 0.005). It can be seen that in the taxane treatment group, the PSA RR was much higher than in the AA/E treatment group. Will the difference in PSA RR bring survival benefits? Graf et al found that AR-V7 positive patients had better OS after taxane treatment than after AR target therapy. 22 Antonarakis et al pointed out that taxane was more effective in AR-V7-positive patients, but this advantage was not observed in AR-V7-negative patients. 11 Our study further analyzed the OS of AR-V7-positive patients in the 2 treatment regimens, with results suggesting that the OS of AR-V7-positive patients under taxane treatment was similar to that of patients in the AA/E treatment group. However, the results of multivariate Cox regression analysis excluding interfering factors (full-length AR expression level, history of ADT preparations, doctors’ preference) suggested that taxane is a protective factor for OS in AR-V7-positive patients. 10,11,22
CTCs can spread in blood, so that tumor-derived substances can be obtained without invading tumor tissues. Changes in CTC counts during treatment are also predictive biomarkers for the prognosis of cancer patients. 62 Therefore, most previous studies were based on CTCs to detect AR-V7 mRNA. 10 -12,19,20,22 -25,27 -29,34 However, the count of CTCs is closely related to tumor burden. Selecting CTCs to detect AR-V7 may cause a certain degree of selection bias. 1 Using CTCs to detect AR-V7, the extraction and separation process is complex and expensive. Therefore, choosing a highly sensitive, specific and easy-to-operate method will be more conducive to clinical applications. In addition to CTCs, AR-V7 transcription can also be detected in tumor tissues, exosomes and plasma. 21,35,63 Recent studies have reported extraction of AR-V7 directly from WB. 26,30,31 One advantage of this method is that it can also reflect the overall expression of AR-V7 in the blood. In addition, the detection of AR-V7 in WB does not rely on detectable CTCs and their enrichment and separation, and it is convenient to obtain materials. 26 In the present meta-analysis, 5 studies reported that samples were collected from WB. In order to verify the effect of these methods (CTCs, WB and TT) on the results, subgroup analysis was carried examining PSA-PFS, r-PFS, OS and PSA RR after AA/E treatment, based on the source of the specimen. The subgroup analysis results were consistent with the primary results. Therefore, this study supports the consistency and effectiveness of both methods (CTCs and WB).
Due to resistance mechanisms mentioned above, the treatment of mCRPC is now no longer a single therapy, and multiple lines of therapy have been increasingly reported. Enzalutamide plus abiraterone could sensitizes enzalutamide-resistant PCa, and was tested successfully in the PLATO trial (NCT01995513). 64 ADT has also been used in combination with taxane chemotherapy for a long time to treat patients with high tumor burden. 65 Moreover, a multi-armed clinical (STAMPEDE, NCT00268476) attempt to compare the efficacy of multiple combination of drugs included ADT with radiotherapy, docetaxel and abiraterone and ADT with enzalutamide, abiraterone and prednisolone. 66,67
However, before making clinical decisions on mCRPC patients, knowing the status of AR-V7 is still helpful to choose appropriate treatments and can further improve the patient’s prognosis. In the future, additional research is needed to discover new biomarkers to predict the resistance of AR-V7-negative patients to ADT and to provide support for such patients’ medication decisions.
Wang et al had conducted a meta-analysis on prognostic value of AR-V7 in 2020. 68 They concluded that in AR-V7 positive patients treated with novel hormone therapy had worse PSA RR, OS and PFS which is consistent with ours. In this present study, we focused on mCRPC patients receiving AA/E or taxane-based chemotherapy. In addition to explore the prognostic value of AR-V7, the novelty of this study is that we compare the OS of AR-V7 positive patients treated with AA/E and taxane and conclude that AR-V7 positive patients treated with taxane chemotherapy can achieve a better OS, which may be suggestive for clinical decision making in the future. Moreover, the results of sensitive analysis and subgroup analysis also support our conclusion and further provide robust evidence on the reliability of our results.
Conclusions
AR-V7 can be used as a predictive biomarker for ADT resistance, wherein expression of AR-V7 is related to the poor prognosis of this treatment. Clinically, medication decisions can be modified according to the expression of AR-V7 in order to improve the prognosis of mCRPC patients. In the future, more research is needed to demonstrate the superiority and efficiency of extracting AR-V7 from whole blood in order to promote the application of AR-V7 detection in clinical practice.
Footnotes
Authors’ Note
RJL and QH contributed equally to this manuscript. Study design, data extraction and analysis: RJL and QH. Manuscript writing: RJL, QH, SYL, WPM and BX. Manuscript reviewed and revised: RJL, QH and MC. All authors approved the final version and its submission. There is no need for ethical permission in meta-analysis. Our study is registered with INPLASY (registration number: INPLASY202160021). These data can also be found at: doi.10.37766/inplasy2021.6.0021.
Acknowledgments
We thank all the study authors for the support in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The National Natural Science Foundation of China (No. 81872089, 81370849, 81672551, 81300472, 81070592, 81202268, 81202034); 6 talent peaks project in Jiangsu Province, Jiangsu Provincial Medical Innovation Team (CXTDA2017025); Natural Science Foundation of Jiangsu Province (BK20161434, BL2013032, BK20150642 and BK2012336).
