Abstract
Purpose:
Human umbilical endothelial cells (HUVECs) have been proved to be an effective whole-cell vaccine inhibiting tumor angiogenesis. In this study, we fused HUVECs with human lung adenocarcinoma cells A549 s, aiming at preparing lung cancer vaccine to achieve dual effects of anti-tumor angiogenesis and specific immunity to tumor cells.
Methods:
A549 cells were induced by ethyl methane sulfonate (EMS) and 8-azaguanine (8-AG) to get hypoxanthine guanine phosphoribosyl transferase (HGPRT) auxotrophic A549 cells. Then Fused HGPRT auxotrophic A549 cells with primary HUVEC cells by combining electrofusion with polyethylene glycol (PEG). Afterward the fusion cells were screened by HAT and HT selective medium and sorted by flow cell sorter to obtain high-purity HUVEC-A549 cells. Finally, HUVEC-A549 cells were identified by karyotype analysis and western blotting.
Results:
The fusion efficiency of HUVEC-A549 cells prepared by combining electrofusion with polyethylene glycol (PEG) was significantly higher than that of electrofusion and PEG (43.0% vs 17.60% vs 2.71%, P < 0.05). After screened by HAT and HT selective medium and sorted by flow cell sorter, the proportion of HUVEC-A549 cells can count for 71.2% ± 3.2%. The mode of chromosomes in HUVEC-A549 cells was 68, and the chromosome was triploid. VE-cadherin and platelet endothelial cell adhesion molecule-1 (CD31) were highly expressed in HUVECs and HUVEC-A549 cells, but not in A549 cells.
Conclusions:
These results indicate that HUVEC-A549 cells retain the biological characteristics of human umbilical vein endothelial cells and A549 cells. It can be used in the experimental study of lung cancer cell vaccine.
Keywords
Introduction
Lung cancer is the leading cause of cancer death in both men and women worldwide, accounting for 1.8 million new cases and 1.6 million deaths annually. 1 Non-small cell lung cancer (NSCLC) accounts for about 85% of all lung cancers. 2 With the emergence of various targeted therapies 3 -5 and the effective application of immunotherapy 6 in some patients with advanced NSCLC, great progress in the treatment of NSCLC have been achieved in the past 2 decades. However, the overall cure and survival rates for NSCLC remain low, particularly in metastatic disease. 7 Hence, there is an urgent need for an effective treatment.
Tumor blood vessels are a key target for cancer therapeutic management. 8 Angiogenesis is known as one of the hallmarks in cancer which could play a key role in providing oxygen and nutrients for tumor cells. 9 It has been shown that tumor cannot grow without sufficient development of new blood vessels. However, because tumor vascular endothelial cells have been in tumor microenvironment for a long time, their phenotypic and functional characteristics have significantly changed, including some changes in immunological characteristics, such as decreased expression of endothelial cell adhesion molecules and poor antigen presentation function. 10,11 Tumor vascular endothelial cells are the first barrier for immune cells and immunotherapeutic drugs to enter tumor tissue. Therefore, the immunological characteristics of tumor vascular endothelial cells may be related to tumor cells escaping immune surveillance and resisting immune killing. 12 Accordingly, targeting angiogenesis, especially endothelial cells, could be considered as a common therapeutic target in tumors. Tumor vaccine is one of the most important methods of tumor immunotherapy. In previous studies, 13 -15 the vaccine prepared by endothelial cells can not only induce humoral immunity to produce specific antibodies, but also induce specific cellular immunity to activate cytotoxic lymphocyte (CTL) to produce anti-angiogenesis effect, so as to achieve the purpose of anti-tumor.
Most of the studies on human vascular endothelial cells are based on human umbilical vein endothelial cells (HUVECs). However, primary cultured HUVEC cells has some limitations. First of all, the growth of primary HUVEC cells in vitro is slow and the life span is limited. 16 After several months of culture, it is difficult to avoid aging, which greatly restricts the progress and operability of the experiment. Secondly, it is difficult to maintain a stable state in the process of cell culture. 17 It was found that the expression of intercellular adhesion molecule 1(ICAM-1) decreased further with the further culture and passage in vitro. 18 Third, the primary umbilical vein endothelial cells represent the characteristics of neovascularization, which cannot completely reflect the nature of adult endothelial cells.
If we can obtain a cell line that can maintain the enough characteristics of endothelial cells and also not be limited by aging problems, the above limitations will be well solved. Based on this idea, we consider prepare immortalized human umbilical vein endothelial cells. A549 cell is one of non-small cell lung cancer cell line, and its morphological characteristics are epithelioid, polygonal, adherent growth. It belongs to subculture cell line and can be stably subcultured. So, we want to prepare the fusion cells of human umbilical vein endothelial cells (HUVEC) and human lung adenocarcinoma cells (A549). The fusion cell will have the characteristics of tumor cell and endothelial cell, and can be stably subcultured. What’s more, the fusion cells can be used to prepare lung cancer vaccine, achieving dual effects of anti-tumor angiogenesis and specific immunity to tumor cells.
Materials and Methods
Cell Lines and Cell Culture
Human lung adenocarcinoma cells line A549, human umbilical vein endothelial cell fusion cell line Eahy926, were purchased from ATCC (American Type Culture Collection, VA, USA). These cells were cultured in DMEM medium supplemented with 10% fetal calf serum plus 1% ampicillin in a humidified atmosphere with 5% CO2 at 37 °C. Primary human umbilical vein endothelial cell (HUVEC) isolated sterile from umbilical cord of newborn after delivery, was cultured in endothelial cell growth medium-2 (EGM-2, purchased from LONZA, art no: cc-3162) including 0.1% hEGF, 0.04% hydrocortisone, 0.1% CA-1000, 2% FBS, 0.4% hFGF-B, 0.1% VEGF, 0.1% R3-IGF-1, 0.1% heparin and 0.1% ascorbic acid (Lonza, Basel, Switzerland).
A549 Cells Mutagenesis by Ethyl Methane Sulfonate (EMS)
A549 cells in logarithmic growth phase were digested and inoculated in 6 well plates, then EMS was added when the cell confluence reached 60%-80%. According to the references, the final concentration of EMS was set at 200, 250, 300 μg/ml, then each concentration was treated for 24, 36 and 48 hours respectively. The dose of EMS at 70% cell survival rate were used as mutagenic dose. The survival rate of A549 cells was calculated by trypan blue staining. After repeated experiments, EMS with 250 ug/ml of final concentration acting for 48 hours was used as mutagenic dose.
HGPRT Auxotrophic A549 Cells Induced by 8-AG
The mutant A549 cells were treated by DMEM medium containing 8-azaguanine (8-AG). 8-AG with final concentration of 1.25, 2.5, 5.0, 10.0, 20.0, 40.0 and 60 μg/ml was added in turn every 2 days, until the cells could grow stably in the 60.0 μg/ml 8-AG. Finally, stable hypoxanthine guanine phosphoribosyl transferase (HGPRT) auxotrophic A549 cells were obtained. Sensitivity detection of HGPRT auxotrophic A549 cells to HAT: the survival rate of A549 cells was detected from the first day to eleventh day after cultured with HAT selective medium (Gibco, 21060-017).
Polyethylene Glycol (PEG) Induced Cell Fusion
HUVEC cells and HGPRT auxotrophic A549 cells were mixed in the centrifuge tube at a ratio of 1:2 with the final cell density of 3*105/100 μl. The mixed cells were washed by PBS, then 2 ml 50% PEG4000 solution (Sigma, P7181) preheated at 37°C was slowly added within 45-60 s, and shaken while adding. The centrifuge tube was heated in a 37°C water bath for 2 min, then 30 ml DMEM medium was added into the centrifuge tube to stop the reaction within 90 s to dilute and terminate the effect of PEG. The cell suspension was stationary for 5 min, and then centrifuged for 3 min at 1200 rpm/min. The cell precipitation was resuspended with 10 ml DMEM medium and cultured in the incubator.
Exploration of Electrofusion Conditions
Cell electrofusion instrument (American BTX company, ECM-2001) was used to prepare the fusion cells of HUVEC and A549. Selection of alternating current (AC) voltage: The mixed cells were added into sterile 3.2 mm microslide. Set the instrument to AC mode, and then set different gradients of AC voltage to observe whether the cells were arranged in a string of beads under the inverted microscope. When the cells were arranged in 2 or 3 beads, the AC voltage was recorded. Selection of direct current (DC) voltage: After the determination of the AC voltage of electrofusion, several DC voltage gradients were set, and other conditions are consistent. Finally, the efficiency of electroporation was detected by flow cytometry, and cell survival rate was calculated by trypan blue staining, so as to select the best DC voltage.
Electrofusion to Prepare the Fusion Cells
HUVEC cells and HGPRT auxotrophic A549 cells were mixed at a ratio of 1:2 in cell fusion buffer (0.3 M D-glucose,0.1 mM CaCl2,0.1 mM MgCl2,1 mM Hepes, and PH 7.0-7.4), with the final cell density was 3*105/100 μl. The cell suspension was centrifuged for 3 min at 1200rpm/min, and the supernatant was discarded, then 500 µl fusion buffer was added to mix evenly. Set the parameters of the electric fusion instrument: AC voltage was 50 V, and the time was 25 s; DC voltage was 800 V, and the time was 30 μs, with 3 times of repeat time. After electrofusion, the cells were stationary in microsides for about 3 min, and then centrifuged for 3 min at 1200 rpm/min. The cell precipitation was resuspended with 10 ml DMEM medium and cultured in the incubator.
The Combination Electrofusion and PEG to Prepare the Fusion Cells
Set the parameters of the electric fusion instrument: AC voltage was 50 V, and the time was 25 s; DC voltage was 800 V, and the time was 30 μs, with 1 time of repeat time. Electrofusion was performed according to the above parameters. Then 50% PEG4000 solution preheated at 37°C was slowly added into electrofusion cells within 45-60 s. The cell suspension was heated in a 37°C water bath for 1 min, then 30 ml DMEM medium was added into the centrifuge tube to stop the reaction within 90 s to dilute and terminate the effect of PEG. The cell suspension was stationary for 5 min, and then centrifuged for 3 min at 1200 rpm/min. The cell precipitation was resuspended with 10 ml DMEM medium and cultured in the incubator.
Screening of Fusion Cells
After the fused cells adhered to the wall, the culture medium was removed, and then cultured in 1x HAT selective medium. After that, half of HAT selective medium was changed every 3-4 days, and HT selective medium was used 15 days later. After cultured in HT selective medium for 1 week, the fusion cells were cultured with DMEM medium. At this time, most of cells were fusion cells of HUVEC and A549 (HUVEC-A549 cells).
Detection of Fusion Rate of HUVEC-A549 Cells by Flow Cytometry
The fusion cells were harvested and washed 3 times by PBS, then 50µl PBS was added into each flow tube to prepare single cell suspension. CD31-FITC antibody was added into each flow tube and incubated in dark for 30 minutes at 4°C. The cells were washed 3 times by PBS, then samples were detected by flow cytometry (American Becton Dickinson company).
Cell Sorting of HUVEC-A549 Cells
Clean flow cell sorter (American BD FACS Aria II) 1 day before sorting to maintain a sterile sorting environment. HUVEC-A549 cells were stained with CD31-FITC, meanwhile negative control group (A549) and positive control group (HUVEC) were set up. Pre-experiment should be done before sorting to optimize the determination conditions of flow cytometry and establish sorting gate. The positive rate of HUVEC-A549 cells was recorded. The HUVEC-A549 cells after sorting were centrifuged at 1200 rpm/min for 5 min, then maintained in DMEM supplemented with 10% Fetal Bovine Serum (FBS) in a humidified atmosphere with 5% CO2 at 37 °C.
Karyotype Analysis
Eahy926 cells was established by fusing primary human umbilical vein cells with a thioguanine-resistant clone of A549 by exposure to polyethylene glycol (PEG). Eahy926 cells served as a positive control to analyze the number of chromosomes of HUVEC-A549 cells we prepared. HUVEC-A549 and Eahy926 cells were treated with 0.4 μg/ml colchicine, trypsinized and collected. After hypotonic treatment with 0.075 mol/L potassium chloride (KCl) for 10 min at 37 °C, cells were fixed with 3:1 mixture of methanol: acetic acid for 3 times. Then, cells were dropped on precooled glass slides to obtain metaphase chromosome spreads. Chromosomes were stained with Giemsa and imaged by a microscope with a 100X objective lens. The chromosomes of each cell sample should be taken at least 20 visual field.
Western Blotting
Briefly, A549, HUVEC-A549, and HUVEC cells were collected, then washed in PBS and lysed in RIPA buffer containing 1 mM PMSF. Total protein concentration was quantified with the Enhanced BCA Protein Assay Kit. The cell lysates were separated by SDS-PAGE and transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked in Tris-buffered saline containing 0.1% Tween-20 (TBS-T) and 5% nonfat milk for 2 h at room temperature, and incubated with primary antibodies against VE-cadherin (Catalog number. 66804-1-Ig, Proteintech Group) at dilution of 1:1000, CD31 (Catalog number. 11265-1-AP, Proteintech Group) at dilution of 1:1000 and GAPDH (Catalog number. 60004-1-Ig, Proteintech Group) at dilution of 1:20000 overnight at 4 °C, followed by incubation with horseradish peroxidase-conjugated secondary antibodies at 37 °C for 1 h. The blots were detected using the enhanced chemiluminescence system (Millipore). VE-cadherin and CD31 were the marker protein for HUVEC cells, which were high expressed in the HUVEC cells. GAPDH served as an internal reference protein.
Technical Roadmap for Preparation of HUVEC-A549 Fusion Cell
Technical roadmap for preparation of HUVEC-A549 fusion cell was shown in Figure 1. First, mutation of A549 cells were induced by EMS, then induced by 8-AG, so as to get HGPRT auxotrophic A549 cells. After that, the fusion cells of HGPRT auxotrophic A549 cells and HUVEC cells were prepared by the method of combination electrofusion and PEG. Finally, high purity fusion cells were obtained by screening of HAT and HT selective medium and cell sorting of flow cell sorter.
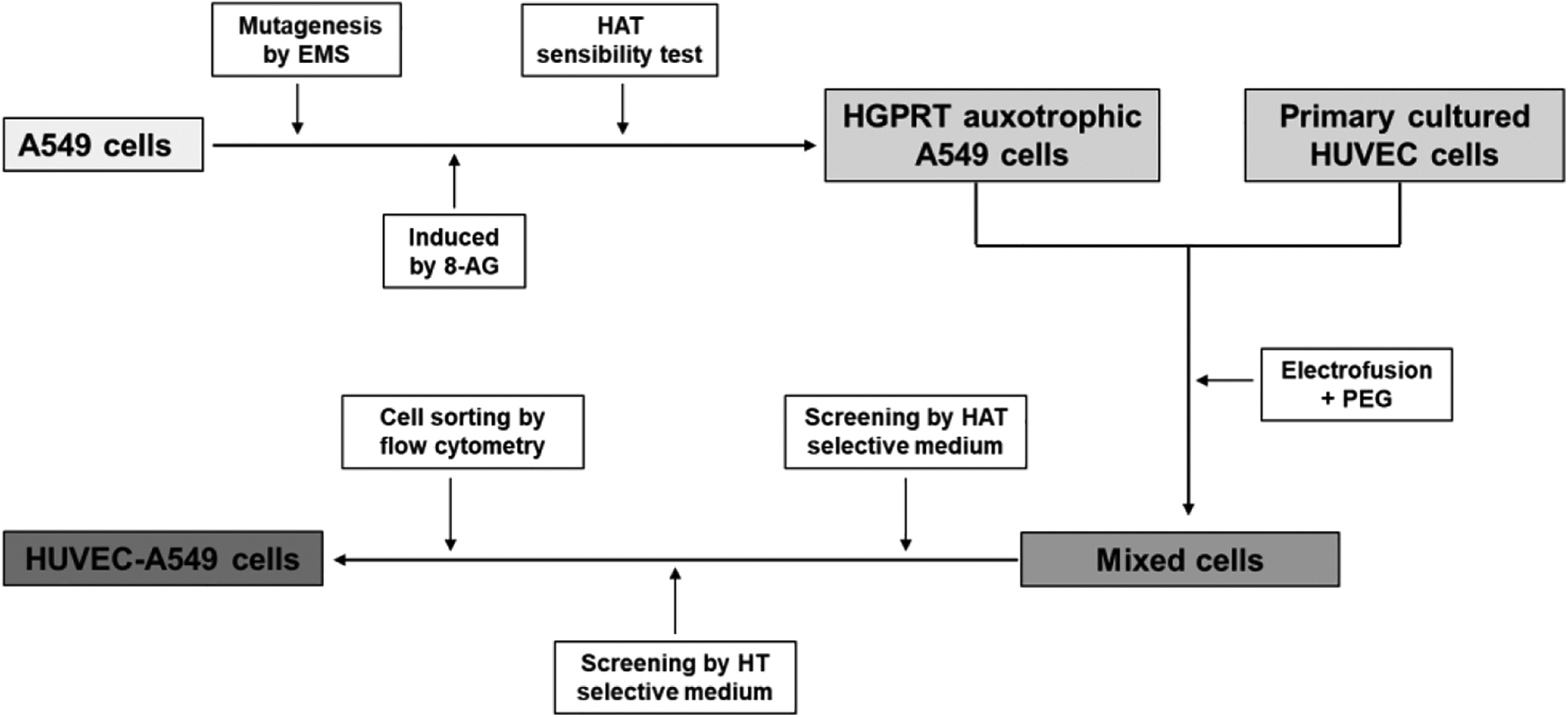
Technical roadmap for preparation of HUVEC-A549 fusion cells.
A549 cells were induced by EMS and 8-AG to get HGPRT auxotrophic A549 cells. Then Fused HGPRT auxotrophic A549 cells with primary HUVEC cells by a method of electrofusion combined with PEG. Afterward the mixed cells were screened by HAT and HT selective medium and sorted by flow cell sorter to obtain high-purity HUVEC-A549 cells.
Results
Constructing of HGPRT Auxotrophic A549 Cells
HGPRT auxotrophic A549 cells, which were obtained from A549 cells induced by 8-AG, couldn’t survive in HAT selective medium, while non-mutated A549 cells can grow stably (Figure 2), suggesting that we succeeded in establishing HGPRT auxotrophic A549 cells.

Sensitivity of HGPRT auxotrophic A549 cells to HAT.
The cell survival rate of HGPRT auxotrophic A549 cells and non-mutated A549 cells cultured in HAT selective medium for 11 days. HGPRT auxotrophic A549 cells couldn’t survive in HAT selective medium, while non-mutated A549 cells can grow stably.(The red line was HGPRT auxotrophic A549 cells and the blue line was non-mutated A549 cells).
The Electrofusion Method and Results
When the cells were arranged in a string of 2 or 3, which was beneficial to improve the efficiency of cell fusion, the AC voltage was 50 V and the time was 25 s. When the DC voltage was 700 V (field strength: 2187 V/cm), 800 V (field strength: 2500 V/cm), 900 V (field strength: 2812 V/cm), and the efficiency of cell fusion were 14.6%, 18.8%, 20.7% respectively. The efficiency of cell fusion under different DC voltage were shown in the Figure 3. However, when the DC voltage was 900 V, cell death increased significantly. Hence, we chose the 800 V DC voltage as electroporation conditions. Finally, electrofusion conditions were as follow: the AC voltage was 50 V and the time was 25 s; the DC voltage was 900 V (field strength: 2500V/cm), and the time was 30 μs, with 3 times of repeat time.

The efficiency of cell fusion under different DC voltage. A-C, The CD31 positive rates of the fused cells were detected by flow cytometry at direct current (DC) voltage of 700 V, 800 V, and 900 V. The CD31 positive rates of fusion cells represented the efficiency of electroporation (the red line was the negative control cell, and the blue line was the fused cell). D, One-way ANOVA analysis showed that the efficiency of cell fusion were significantly different under DC voltage of 700 V, 800 V, and 900 V.
The Fusion Efficiency of the 3 Methods
The fusion efficiency of PEG, electrofusion, electrofusion combined with PEG were 2.71%, 17.60% and 43.0% respectively (Figure 4A-C). One-way ANOVA analysis showed that the efficiency of cell fusion among 3 methods were significantly different (Figure 4D).

The efficiency of cell fusion under different methods. A-C, The CD31 positive rates of the fused cells prepared by PEG, electrofusion, and electrofusion combined with PEG, were detected by flow cytometry (the red line was the negative control cell, and the blue line was the fused cell). D, One-way ANOVA analysis showed that the efficiency of cell fusion among 3 methods were significantly different.
The HUVEC-A549 Cells After Cell Sorting
Before cell sorting, the proportion of fusion cells was 43.0% ± 2.0%, however after cell sorting, the proportion of fusion cells could reach 71.2% ± 3.2%, as shown in Figure 5. The purity of HUVEC-A549 fusion cells was significantly improved by flow cytometry sorting. The morphology of HUVEC-A549 cells was slender and spindle shaped, as shown in Figure 6.

The proportion of HUVEC-A549 fusion cells before and after cell sorting. A-B, Before cell sorting, the proportion of fusion cells was 43.0% ± 2.0%, while after cell sorting, the proportion of fusion cells could reach 71.2% ± 3.2% (the red line was the negative control cell, and the blue line was the fused cell, and the yellow line was positive control cell). C, There were significant differences between the proportion of HUVEC-A549 fusion cells before cell sorting and after cell sorting.
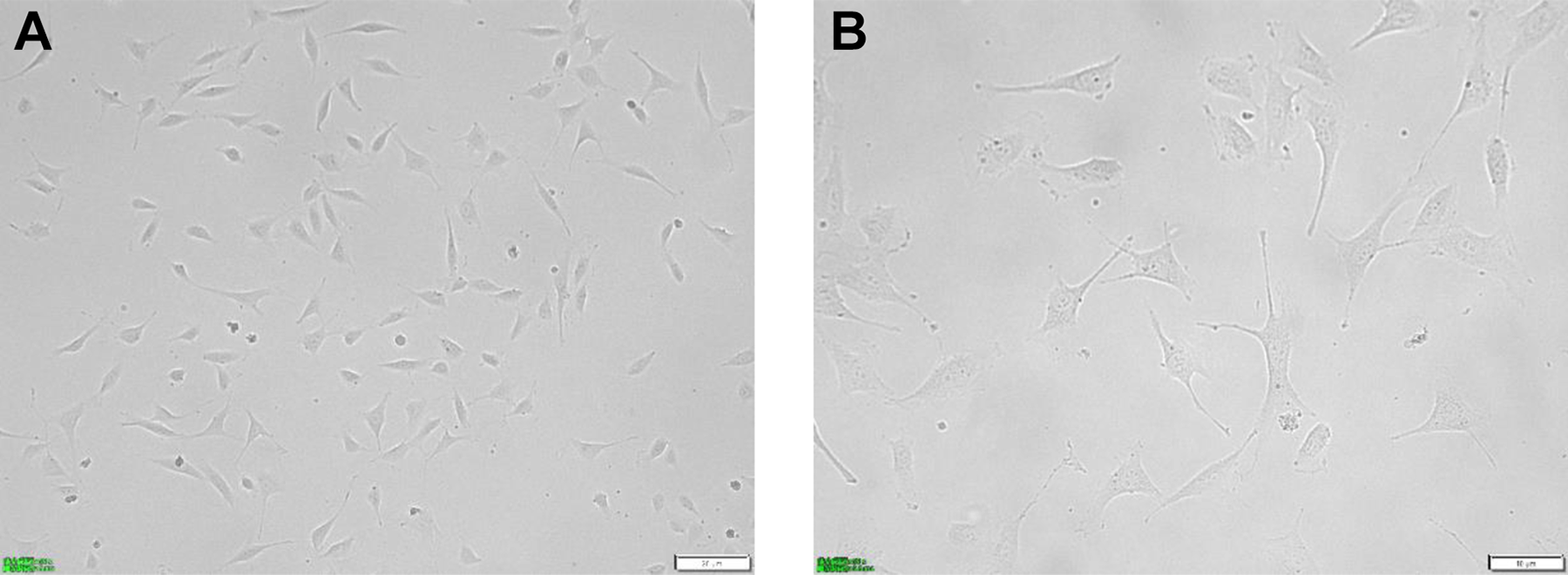
The morphology of HUVEC-A549 cells after cell sorting. A, The morphology of HUVEC-A549 cells under 200 times inverted microscope. B, The morphology of HUVEC-A549 cells under 400 times inverted microscope.
Identification of HUVEC-A549 Cells
Karyotype analysis of HUVEC-A549 cells and Eahy926 were shown in the Figure 7. The average number of chromosomes in HVEC-A549 cells were 68.20 ± 1.135, and the average number of chromosomes in Eahy926 cells were 69.00 ± 0.667. Then they were compared by student T test, and there was no statistical difference between them (t = 1.922, P = 0.071). According to the number of chromosomes in 30 visual fields, the mode of chromosomes in fusion cells is 68. After cell fusion, the number of chromosomes is non-tetraploid, and the chromosome is usually triploid. VE-cadherin and CD31 were highly expressed in human umbilical vein endothelial cells, but not in A549 cells. VE-cadherin and CD31were highly expressed in the fusion cells, as shown in the Figure 8. These results indicate that the fusion cells retain the biological characteristics of human umbilical vein endothelial cells.

Karyotype analysis of HUVEC-A549 cells and Eahy926. A, The number of chromosomes in HVEC-A549 were observed under 100 times oil microscope by karyotype analysis experiment. B, The number of chromosomes in Eahy926 (positive control) were observed under 100 times oil microscope by karyotype analysis experiment. C, There were no statistical differences between the number of chromosomes of HVEC-A549 and Eahy926.

To detect the marker protein of HUVEC-A549 by western blot. A, VE-cadherin and CD31 in HUVEC-A549 (HU-A) cells were detected by western blot. VE-cadherin and CD31 were carried out in the same batch of experiment (western blot), so we used same control GAPDH. B and C, VE-cadherin and CD31 were both higher expressed in Hu-A than those of HUVEC.
Discussion
HUVECs have been proved as an effective whole-cell vaccine inhibiting tumor angiogenesis. 13,19 However, Primary cultured HUVECs divide a very limited number of passages before entering replicative senescence, and the cost of cell culture is high, which limit its application for clinical situation.
The biological phenomenon of cell fusion plays a crucial role in several physiological processes, including wound healing, tissue regeneration and tumor metastasis. 20 In recent years, 21 studies have shown that cell fusion can be spontaneous in the development of tumor. The fusion between tumor cells, as well as the fusion between tumor cells and host cells (such as macrophages, lymphocytes, endothelial cells, etc.), the fused cells can obtain new biological characteristics or new genes. Fusion cells are beneficial to tumor metastasis in certain degrees, so they directly or indirectly promote the metastasis of tumor cells to new organs or tissues. It is suggested that the fusion cells formed by endothelial cells and tumor cells may play an important role in tumorigenesis and development. In this study, the preparation of fusion cells of HUVECs and A549 s is an important step. Methods of artificial induced cell fusion to prepare fusion cells commonly used are PEG chemical induction method, 22,23 electrofusion method, 24 virus induced cell fusion. 25
In our experiment, the fusion efficiency and cell survival rate of PEG chemical induction method and electrofusion method were compared. It was found that although PEG chemical method was easy to operate, the efficiency of cell fusion was relatively lower than that of electrofusion method (2.71% vs 17.6%). Electrofusion method has more advantages in the preparation of HUVECs and A549 s. On the one hand, the fusion efficiency of cells has been greatly improved, which can reach 17.6%. On the other hand, the damage to cells is lower and the cell activity is good in the process of cell fusion. What’s more, we found that if the cells after electrofusion were induced by PEG, the fusion efficiency can increase to 43.0%. The combination of electrofusion and PEG significantly improved the fusion efficiency (P < 0.05).
Because the fusion cells have the antigen expression of the 2 kinds of cells, the fusion cells of HUVECS and A549 s have the expression of CD31. Therefore, flow cytometry can be used to detect CD31 molecules in fusion cells to determine the fusion efficiency. The results of western blot showed that the fusion cells expressed CD31 and VE-cadherin, indicating that the fusion cells maintained the characteristics of endothelial cells. The karyotype analysis showed that the number of chromosomes in fusion cells was 86, but it was not tetraploid, which indicated that the chromosomes were integrated or lost, which was generally triploid. Through the protein detection of fusion cells and karyotype analysis of chromosomes, it shows that the fusion cells have been integrated, not simply superimposed. The fusion cells have the characteristics of human umbilical vein endothelial cells, such as the expression of CD31 and VE-cadherin, and the proliferation of A549 cells. Therefore, the fusion cells of HUVECs and A549s (HUVEC-A549) can be used in subsequent experiments. In the next study, we will prepare the fusion cells as lung cancer vaccine, achieving dual effects of anti-tumor angiogenesis and specific immunity to tumor cells.
In conclusion, we used an innovative approach to prepare the fusion cells of HUVECs and A549 s, which had better cell fusion efficiency than the previous methods. Moreover, the fusion cells not only retained the characteristics of HUVEC cells, but also can be stably subcultured. In addition, the fusion cells can be used to prepare lung cancer vaccine, achieving dual effects of anti-tumor angiogenesis and specific immunity to tumor cells.
Therefore, the fusion cells of HUVECs and A549 s had a good application prospect.
Footnotes
Authors’ Note
Our study did not require an ethical board approval because it did not contain human and animal trials.
Acknowledgments
Thanks to the State Key Laboratory of Biotherapy of Sichuan University for the strong support of this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
