Abstract
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors. Investigating the underlying molecular mechanism is essential for the treatment and prognosis of HCC. Emerging evidence suggests that microRNAs (miRNAs) play pivotal roles in cancer progression. Down-regulation of miR-936 has been found in several cancers, which serves as a tumor suppressor to inhibit the development of cancers. However, the clinical significance and functional roles of miR-936 in HCC have not been determined. To explore this, the expression of miR-936 in HCC tissues and cells was detected by RT-qPCR. Cell Counting Kit-8 (CCK-8) assay, cell migration and cell cycle analysis were performed to evaluate the effects of miR-936 on the growth of HCC cells. The targets of miR-936 were predicted using the miRDB database and confirmed by luciferase reporter experiments. The protein expression of targets was determined by western blot. The results showed that miR-936 was significantly decreased in HCC tissues and cell lines. Low expression of miR-936 was associated with the advance progression and poor survival of HCC patients (P = 0.0036). Functional study revealed that overexpression of miR-936 inhibited the proliferation, migration (decreased to ∼0.26 fold) and induced cell cycle arrested in G1 phase (from 35.3% to 44.7%) of HCC cells. Additionally, miR-936 targeted the 3′-untranslated region (UTR) of jagged-1 (JAG1) and reduced the expression of JAG1 (decreased to ∼0.35 fold). JAG1 was found to be up-regulated in HCC tissues and was inversely correlated with the expression of miR-936 (Pearson r = −0.4633; P = 0.0007). The anti-cancer effects of miR-936 on the proliferation of HCC cells were partially reversed by the rescue of JAG1. Therefore, these results suggested that miR-936 might be a potential target for HCC treatment.
Introduction
Hepatocellular carcinoma (HCC) is a highly fatal malignancy with high prevalence and death rate. 1 -4 Cirrhosis, infection with hepatitis C and hepatitis B have been found to contribute to the development of HCC. 3 Even great research efforts have been devoted to the diagnosis and treatments of HCC, such as surgery and radio-chemotherapy, the clinical outcomes of HCC patients, especially for those diagnosed with advanced stages, still remain poor. Therefore, identification of novel effective therapeutic targets of HCC is of great importance.
As a class of single-stranded nucleotides, microRNAs (miRNAs) act as post-transcriptional negative regulators of gene expression and play important roles in both physiological and pathological processes. 5 In principle, miRNAs bind the 3′-untranslated region (UTR) of targeted mRNAs, leading to mRNA degradation or translation inhibition. 6,7 Increasing evidence has suggested that alterations of miRNAs expression result into uncontrolled cell proliferation, differentiation and ultimately, contributed to the initiation of cancers. 8 -12 Recently, the critical function of miRNAs in HCC has become a hot topic in cancer research field. 13 miRNAs act as tumor suppressors or oncogenes in HCC tumorigenesis via targeting key pathways that involved in cancer development. 14 -18 Aberrant expression of miRNAs was correlated with the progression and clinical outcome of HCC patients. Therefore, exploring cancer-related miRNAs might provide novel insights into the mechanism underlying HCC and benefit the treatment of HCC patients.
miR-936 is located at chromosome 10q25.1 with the length of 22 nucleotides. Interestingly, miR-936 is a newly identified cancer-related miRNA and its dysfunction has been confirmed in several cancers. 19 -22 Overexpression of miR-936 suppressed the proliferation and invasion of non-small cell lung cancer by targeting E2F2. 19 miR-936 negatively regulated CKS1 and induced cell cycle arrest in glioma. 22 Furthermore, the tumor-suppressive role of miR-936 was also found in epithelial ovarian cancer and laryngeal squamous cell carcinoma by inhibiting the malignant phenotypes of cancer cells. 20,21 However, the roles and the underlying mechanism of miR-936 in HCC have not been fully understood.
In the present study, the expression of miR-936 was detected in paired HCC tissues and adjacent non-tumor tissues. The correlation between the level of miR-936 with the prognosis of HCC patients was also analyzed. Moreover, the effects of miR-936 on the growth of HCC cells were evaluated by in vitro gain-of-function. Mechanism study identified JAG1 as a target of miR-936 that mediated the function of miR-936 in HCC. Our findings provided clues to develop novel therapeutic targets for HCC.
Materials and Methods
Tissue Samples
This study was approved by the Ethics Committee of Luoyang Central Hospital Affiliated Zhengzhou University. A total of 50 paired HCC tissues and adjacent non-tumor tissues were obtained from the patient diagnosed with HCC who underwent surgery at Luoyang Central Hospital Affiliated Zhengzhou University between January 2012 and December 2013. None of these patients have been treated with chemotherapy or radiotherapy before surgical resection. All tissues were immediately frozen in liquid nitrogen and stored at −80ºC. This study was performed accordance with the Declaration of Helsinki. Written informed consent was obtained from all the participants regarding the use of the tissue samples. This study was approved by the Ethics Committee of Luoyang Central Hospital Affiliated Zhengzhou University (approval number LYCH20120314-A).
Cell Culture and Transfection
HCC cell lines including HepG2, 97-L, Hep3B, Huh-7 and a hepatocellular cell line THLE-3 were purchased from the American Type Culture Collection (Manassas, VA, USA). Cells were grown in Dulbecco’s Modified Eagle’s medium (DMEM) supplemented with 10% of fetal bovine serum (FBS, Gibco, Thermo Scientific, Shanghai, China), 100 U/mL penicillin and 100 μg/mL streptomycin (Gibco, Thermo Scientific, Shanghai, China). Cells were cultured at 37ºC with 5% CO2.
miR-936 mimics and mimic negative control (miR-NC) were obtained from GenePharma Co., Ltd. (Shanghai, China). The expression vector of pcDNA3.1-JAG1 was constructed by inserting the full length of JAG1 cDNA into the backbone of pcDNA 3.1 vector. All transfection procedures were performed using the Lipofectamine 2000 (Invitrogen, Grand Island, NY, USA) according to the manufacturer’s instruction. Briefly, miRNA (50 nM) or plasmid (100 μg) were diluted in 100 µl OPTI-MEM (Thermo Scientific, Shanghai, China), mixed with 10 µl of lipofectamine and incubated at room temperature (RT) for 15 min. The mixture was added into the cells and cultured for 48 h before further analysis.
Cell Counting Kit-8 (CCK-8) Assay
HCC cells transfected with miR-936 mimics or miR-NC were seeded into the 96-well plate with 1,000 cells per well. Cells were maintained at 37ºC in an atmosphere with 5% CO2. CCK-8 reagent was added into the medium at different time points (1-, 2-, 3-, 4- and 5 days) followed by incubating at 37ºC for additional 3 h. The absorbance of each well at the wavelength of 450 nm was detected with the spectrophotometer (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
Total RNA or miRNA was extracted from tissues or cells using TRIzol reagent (Thermo Fisher Scientific, Inc., Shanghai, China) or miRNeasy Mini Kit (Qiagen, Hilden, Germany), respectively. Total RNA was converted into cDNA using the RevertAid First Strand cDNA Synthesis kit (Thermo Fisher Scientific, Inc., Shanghai, China) according to the manufacturer’s instructions. qPCR was performed using the SYBR Green PCR Master Mix (Bio-Rad Laboratories, Inc., Hercules, CA, USA) on the 7900 Real-time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc., Shanghai, China). Small nuclear U6 RNA and GAPDH served as the controls for the normalization of miR-936 and JAG1, respectively. Relative gene expression was calculated with the 2−ΔΔCT method.
Dual-Luciferase Reporter Assay
The wild-type (WT) or mutant 3′-UTR fragment of JAG1 containing the predicted miR-936 binding sequences was chemically synthesized and cloned into the pmirGLO luciferase vector, respectively. HCC cells were seeded into the 24-well plate and co-transfected with miR-936 mimics or miR-NC and pmirGLO-WT-3′-UTR-JAG1 or pmirGLO-Mut-3′-UTR-JAG1. After transfection for 48 h, cells were harvested and the luciferase activity was determined using the Dual-Luciferase Reporter Assay Kit (Promega, USA) according to the manufacturer’s instructions. The activity of firefly luciferase was normalized to that of Renilla.
Targets Prediction
The targets of miR-936 were predicted using the miRDB online database (http://mirdb.org). Choosing the “Search by miRNA name” option and inputting the name of miRNA as “miR-936.” The possible targets of miR-936 including the target details were presented.
Western Blot Analysis
Total proteins were extracted from cells using the NP-40 lysis buffer (50 mM Tris-HCl (pH 7.5), 120 mM NaCl, and 1% (v/v) NP-40) containing protease inhibitor. The protein concentration was determined using the bicinchoinic acid protein assay kit (Beyotime, Shanghai, China). Equal amount of proteins (20 μg) were separated on 15% SDS-PAGE, and then transferred onto the nitrocellulose membranes. Following blocking with 5% non-fat milk at RT for 1 h, the membrane was incubated with primary antibody against JAG1 (1:1000 dilution; sc-390177; Santa Cruz Biotechnology, USA) or GAPDH (1:2000 dilution; sc-47724; Santa Cruz Biotechnology, USA) at 4ºC overnight. After washing twice with Tris-buffered saline with 0.1% of Tween 20 (TBST), membrane was incubated with goat anti-mouse HRP-conjugated secondary antibody at RT for 1 h. The protein signals were detected using the Enhanced Chemiluminescence Detection System (Pierce, Rockford, IL, USA). GAPDH was also checked as the loading control.
Cell Cycle Analysis
HCC cells transfected with miR-936 mimics or miR-NC were harvested, washed with PBS and fixed with 70% ice-cold ethanol at 4ºC overnight. After washing with PBS, cells were stained with 1 mg/ml RNase A and 50 mg/ml PI at RT for 30 min. The cell cycle progression was determined using the Cell Lab Quanta SC flow cytometry (Beckman Coulter, Inc.). The cell cycle prolife was analyzed using the ModFit software (LT, V3.3, Beckman Coulter, Inc.).
Cell Migration
HCC cells transfected with miR-936 mimics or miR-NC were seeded into the 6-well plate and cultured at 37ºC with 5% CO2. Scratches were generated using a 1,000 pipette tip. The cell debris was removed and cells were cultured overnight. Cell migration was captured with the light microscope (×40 magnification; Eclipse TS-100, Nikon Corporation).
Statistical Analysis
The data were presented as mean ± standard deviation and analyzed using the SPSS statistics software (Version 21.0; IBM Corp). Comparisons between 2 groups were determined by Student’s t test, and differences between multiple groups were performed by one-way analysis of variance (ANOVA) followed by Tukey post hoc test. The correlation between miR-936 and JAG1 was analyzed with the Spearman’s correlation test. P < 0.05 was considered as statistically significance.
Results
miR-936 Was Underexpressed in HCC Tissues and Cell Lines
To investigate the potential involvement of miR-936 in HCC, the expression of miR-936 in paired HCC tissues and adjacent non-tumor tissues was detected by RT-qPCR assay. As presented in Figure 1A, paired student’s t test showed that compared with the non-tumor tissues, the relative expression of miR-936 was significantly decreased in HCC tissues (Figure 1A). Furthermore, miR-936 was significantly lower in high-grade HCC tissues compared with that of lower-grade ones (Figure 1B). Additionally, the expression of miR-936 was also examined in normal THLE-3 and HCC cell lines HepG2, 97-L, Hep3B, and Huh-7. As indicated in Figure 1C, obviously down-regulated expression of miR-936 was observed in HCC cell lines compared with that of the THLE-3 cells (Figure 1C).

The expression of miR-936 was down-regulated in HCC tissues and cells. A, The relative expression of miR-936 in 50-paired HCC tissues and non-tumor tissues was detected by RT-qPCR. B, Patient with higher grade of HCC carried lower level of miR-936. C, RT-qPCR analysis was performed to detect the expression of miR-936 in HCC cell lines and normal cell THLE-3. Significance was determined by comparing the miR-936 expression level with that of the control THLE-3 cells. D, The Kaplan Meier survival curves of HCC patients based on miR-936 expression. ***P < 0.001.
The clinical value of miR-936 was also evaluated by analyzing the relationship of miR-936 expression with the prognosis of HCC patients. All patients were classified into low miR-936 expression group (n = 32) and high miR-936 expression group (n = 18) according to the median value of miR-936 expression. Kaplan-Meier survival analysis indicated that the 5-year overall survival of HCC patients harboring lower miR-936 expression was relative shorter than those patients expressing higher level of miR-936 (Figure 1D). Collectively, these results demonstrated that down-regulation of miR-936 might be correlated with the progression of HCC.
Overexpression of miR-936 Inhibited the Proliferation and Induced Cell Cycle Arrest of HCC Cells
To investigate the biological role of miR-936 in the malignancy of HCC, gain-of-function analysis was performed by transfecting miR-936 mimics into both HepG2 and Huh-7 cells considering the relative lower level of miR-936 in these cell lines. As indicated, miR-936 was significantly increased with the transfection of miR-936 mimics (Figure 2A). The effects of miR-936 on the proliferation of HCC cells were evaluated by the CCK-8 assay. As shown in Figure 2B and C, both HepG2 and Huh7 cells transfected with miR-936 mimics showed suppressed cell proliferation compared with cells expressing miR-NC. Moreover, to evaluate the influence of miR-936 on the migration of HCC cells, in vitro migration assay was performed with HCC cells expressing miR-936 mimics or miR-NC. The results showed that in comparison with the control cells, exogenous miR-936 expression significantly reduced the migration of both HepG2 and Huh7 cells (Figure 2D). Consistently, the invasion of HCC cells was also significantly inhibited with the overexpression of miR-936 compared with that of control group (Figure 2E). To evaluate whether the reduced growth of HCC cells with miR-936 overexpression was due to cell cycle arrest, flow cytometry analysis was performed and the data showed that cells transfected with miR-936 have more distribution in G1 but less in S phase (Figure 2F). Altogether, these findings suggested that miR-936 played a suppressive role in the malignant phenotypes of HCC cells.

Overexpression of miR-936 inhibited the growth of HCC cells. A, The transfection efficiency of miR-936 in HepG2 and Huh7 cells was validated by RT-qPCR. B and C, CCK-8 assay was performed to detect the proliferation of HCC cells after transfection of miR-936 mimic or miR-NC. D, The migration of HCC cells was reduced with the transfection of miR-936 compared with cells expressing miR-NC. E, The invasion of HCC cells transfected with miR-NC or miR-936 mimics was detected with the Boyden chambers. F, Overexpression of miR-936 induced G1 cell cycle arrest in both HepG2 and Huh7 cells. ***P < 0.001.
miR-936 Bound the 3′-UTR of JAG1 and Inhibited the Expression of JAG1
To elucidate the underlying molecular mechanisms by which miR-936 attenuated the malignancy of HCC, the potential targets of miR-936 were predicted via the online miRDB database. A total of 506 predicted targets for miR-936 were found. Among them, the 3′-UTR of JAG1 showed putative complementary binding sequence of miR-936 (Figure 3A), which ranked top among all the targets. JAG1 was identified as an important Notch ligand that activated the Notch signaling pathway and played an oncogenic role in the development of cancer. 23 Additionally, overexpression of JAG1 has been reported in a variety of cancers and associated with the worse prognosis of cancer patients. 23 Thus, down-regulating the expression of JAG1 might be a promising therapeutic strategy in cancer treatment. Therefore, JAG1 was selected to explore its relationship with miR-936 as well as its function in HCC. To valid this, RT-qPCR assay was performed to check the mRNA level of JAG1 in HCC cells with the overexpression of miR-936. The data demonstrated that transfection of miR-936 in both HepG2 and Huh7 cells significantly decreased the mRNA level of JAG1 compared with cells expressing miR-NC (Figure 3B). Consistently, the protein expression of JAG1 was also decreased following the transfection of miR-936 in HCC cells (Figure 3C). To further verify JAG1 as a target of miR-936, the 3′-UTR fragment of JAG1 that contains the wild-type or mutated binding site of miR-936 was cloned into the luciferase vector, respectively. Dual-Luciferase reporter assay showed that transfection of miR-936 markedly reduced the luciferase activity of vectors harboring WT but not mutated 3′-UTR of JAG1 (Figure 3D and E). This data suggested the specific binding between miR-936 and the 3′-UTR of JAG1.

JAG1 was a target of miR-936 in HCC. A, The 3′-UTR of JAG1 contained the complementary binding sequence of miR-936. B, The mRNA level of JAG1 in both HepG2 and Huh7 cells was decreased with the transfection of miR-936. C, Overexpression of miR-936 reduced the protein abundance of JAG1 in HCC cells. The protein abundance of JAG1 was qualified by normalizing to that of GAPDH. Data was obtained from 3 independent experiments. D and E, The luciferase activity of cells expressing WT-3′-UTR was significantly repressed with the overexpression of miR-936. ***P < 0.001.
miR-936 Inversely Correlated With the Level of JAG1 in HCC Tissues
Since JAG1 was identified as a target of miR-936 in HCC cells, the relationship between the expressions of miR-936 and JAG1 was determined. The mRNA levels of JAG1 in 50 paired HCC tissues and adjacent non-tumor tissues were detected by RT-qPCR. As shown in Figure 4A, JAG1 mRNA expression was notably higher in HCC tissue than that of the non-tumor tissues. To evaluate the correlation between miR-936 and JAG1, spearman test was performed. A significant inverse correlation was noted between the levels of JAG1 and miR-936 in HCC tissues (Figure 4B). Moreover, both the mRNA and protein levels of JAG1 were also higher in HCC cell lines than that of the normal cell (Figure 4C and D). These results indicated the up-regulation of JAG1 in HCC.

JAG1 was highly expressed in HCC. A, The mRNA levels of JAG1 in paired HCC tissues and adjacent non-tumor tissues were examined by RT-qPCR assay. B, Spearman test was performed to evaluate the correlation between the expression of miR-936 and JAG1. C and D, Both the mRNA and protein abundance of JAG1 was increased in HCC cells compared with that of the normal cell THLE-3. The protein abundance of JAG1 in different cell lines was qualified by normalizing to that of GAPDH (Right panel of Figure 4D). ***P < 0.001.
Overexpression of JAG1 Attenuated the Anti-Cancer Effects of miR-936 on HCC Cells
To verify whether the tumor-suppressive effects of miR-936 in HCC was mediated by repressing JAG1, both HepG2 and Huh7 cells were co-transfected with miR-936 mimics and either pcDNA 3.1-JAG1. Both RT-qPCR and western blot analysis were performed to confirm the recovery of JAG1 in HCC cells with the transfection of pcDNA3.1-JAG1 (Figure 5A and B). As shown in Figure 5C and D, reintroduction of JAG1 expression significantly attenuated the suppressive role of miR-936 in the proliferation of both HepG2 and Huh7 cells. Accordingly, these results indicated that the inhibitory effects of miR-936 on the malignant behaviors of HCC cells at least partially was mediated by repressing JAG1.
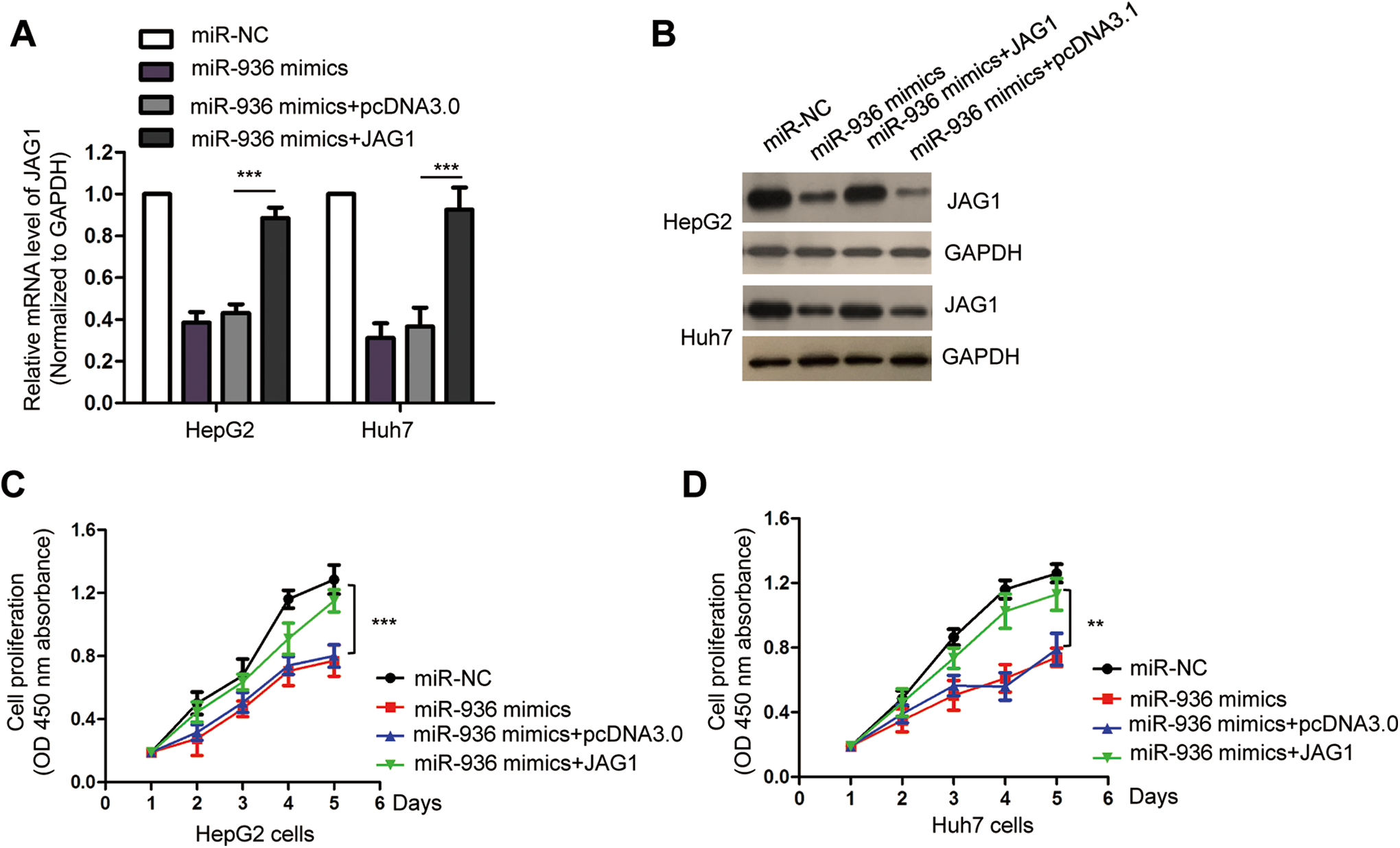
Reintroduction of JAG1 abolished the anti-cancer effects of miR-936 on the growth of HCC cells. A and B, The rescue of JAG1 expression was confirmed by RT-qPCR and western blot. C and D, Co-transfection of JAG1 attenuated the suppressive role of miR-936 in the proliferation of both HepG2 and Huh7 cells. ***P < 0.001.
Discussion
HCC is a highly invasive disease with up to 70% recurrent rate within 5 years. 24 Nowadays, great efforts have been made to develop effective therapeutic strategies to improve the outcomes of HCC patients. Given the critical function of miRNAs in regulating gene expression, miRNAs are considered as promising candidates for the diagnosis and prognosis of cancers. 25,26 Investigating the function and characterizing the underlying mechanisms by which miRNAs contribute to HCC progression may provide novel insights into the pathogenesis of HCC. Here, the expression and related functional mechanism of miR-936 in HCC were explored.
miR-936 has been reported as a tumor-suppressive miRNA that is dysregulated in several cancers. 19 -22 A recent study showed that miR-936 was down-regulated in NSCLC tissues and correlated with the advanced progression of NSCLC patients. 19 Overexpressed miR-936 induced cell cycle arrest, inhibited proliferation and invasion of NSCLC cells. 19 The anti-cancer function of miR-936 was also established in epithelial ovarian cancer (EOC), which inhibited the aggressiveness of EOC cells by targeting FGF-2-mediated suppression of PI3K/Akt pathway. 21 In the present study, miR-936 was underexpressed in HCC tissues and cell lines. Low expression of miR-936 was significantly correlated with the higher grade of HCC. Moreover, patients with lower level of miR-936 showed shorter 5-year overall survival rate compared with those harboring higher miR-936 expression. These findings indicated the potential application of miR-936 as a biomarker for the prognosis of HCC. Further study is encouraged to evaluate whether the level of miR-936 in blood, which is more easily detectable, can be used in the diagnosis of HCC. Additionally, since hepatitis B virus (HBV) infection plays an important role in the initiation and development of HCC, it would be interesting to explore the correlation between HBV and the aberrant expression of miR-936 in HCC. The mechanisms that are involved in the down-regulation of miR-936 in HCC also deserves further studies.
Consistent with the decreased expression of miR-936 in HCC, gain-of-function analysis showed that miR-936 overexpression inhibited the proliferation and migration of HCC cells, suggesting the tumor-suppressive role of miR-936 in HCC. In vivo mice experiments with stably expressed miR-936 are necessary to clarify the anti-cancer effects of miR-936. Identifying target genes of miR-936 is essential for understanding the function of miR-936 in the development of HCC. In this study, miR-936 was found to bind the 3′-UTR of JAG1 and negatively modulated the expression of JAG1 in HCC cells. JAG1 is a key factor involved in both physiological and pathological conditions. 27,28 Notably, overexpression of JAG1 has been found in a variety of cancers and regulates the malignant behaviors of cancer cells. 28 -30 Consistently, we found that JAG1 was overexpressed in HCC tissues and inversely correlated with that of miR-936 in HCC tissues. The anti-cancer effects of miR-936 in the proliferation of HCC cells was reversed by JAG1 overexpression, suggesting JAG1, at least partially, mediated the tumor-suppressive role of miR-936 in HCC.
In conclusion, our findings in this study revealed that miR-936 was down-regulated in HCC tissues and cell lines. Decreased miR-936 was associated with the poor clinical features of HCC patients. miR-936 exerted inhibitory effects on the growth of HCC cells by suppressing JAG1. These results uncovered the tumor-suppressive function of miR-936 in HCC via targeting JAG1. Since overexpression of miR-936 inhibited the malignant behaviors of HCC cells including proliferation. These results suggested that blocking the function of miR-936 might be a promising strategy for the treatment of HCC in the future. Additionally, highly expressed miR-936 reduced the migration and invasion of HCC cells, it would be interesting to determine the effects of miR-936 on the metastasis of HCC. Drug resistance has been a major challenge in the treatment of HCC, considering the tumor-suppressive function of miR-936 in HCC, the effects of miR-936 on the chemo-resistance of HCC is also interesting to be determined. Further investigation is necessary to clarify the role of miR-936 in inhibiting the progression of HCC, as well as the regulation of JAG1 by miR-936 by in vivo validation. Additionally, to evaluate the potential clinical significance of miR-936/JAG1 axis in the diagnosis and prognosis of HCC, larger clinical sample size is needed.
Footnotes
Authors’ Note
Our study was approved by the ethics committee of Luoyang Central Hospital Affiliated Zhengzhou University (Approval no. LCH20120452). All patients provided written informed consent prior to enrollment in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
