Abstract
Background:
Radiation therapy is a highly cost-effective treatment for cancer, but the existence of radio-resistant cells remains the most critical obstacle in radiotherapy. We have been established clinically relevant radioresistant (CRR) cell lines by exposure to a stepwise increase of fractionated X-rays. We are trying to overcome the radio-resistance by analyzing the properties of these cells. In this study, we tried to evaluate the effects of hydrogen peroxide (H2O2) on the CRR cells because this can evaluate the efficacy of Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas (KORTUC) that treats H2O2 before irradiation. We also established H2O2-resistant cells to compare the radiation and H2O2 resistant phenotype.
Materials and Methods:
We used human cancer cell lines derived from hepatoblastoma (HepG2), oral squamous cell carcinoma (SAS), and cervical cancer (HeLa). We established HepG2, SAS, and HeLa CRR cells and HepG2, SAS, and HeLa H2O2-resistant cells. To evaluate their sensitivity to radiation or H2O2, high-density survival assay, or WST assay was performed. CellROXTM was used to detect intracellular Reactive Oxygen Species (ROS).
Results:
CRR cells were resistant to H2O2-induced cell death but H2O2-resistant cells were not resistant to irradiation. This phenotype of CRR cells was irreversible. The intracellular ROS was increased in parental cells after H2O2 treatment for 3 h, but in CRR cells, no significant increase was observed.
Conclusion:
Fractionated X-ray exposure induces H2O2 resistance in CRR cells. Therefore, it is necessary to carry out cancer therapy such as KORTUC with the presence of these resistant cells in mind, and as the next stage, it would be necessary to investigate the appearance rate of these cells immediately and take countermeasures.
Introduction
Radiation therapy is a highly cost-effective single-modality treatment for cancer. Ionizing radiation (i.e. X-rays or γ-rays) is commonly used for cancer treatment because it can pass through tissue, break chemical bonds, and remove electrons from atoms to become ionized. Cancer treatment utilizing fractionated radiation therapy has distinct advantages over a single administration of radiation because the former can increase the anticancer therapeutic effect and decrease the occurrence of side effects in normal tissues. 1 Empirically, in conventional fractionated radiation therapy, approximately 1.8-2 Gy of radiation is administered daily, resulting in 9-10 Gy administered per week, which amounts to approximately 54-60 Gy for a total of 6 weeks. Although radiation therapy may be curative in various cancer types, the resistance of cancer cells to radiation remains a major therapeutic problem. 2,3 In order to destroy the radioresistant cells, chemotherapeutic agents are also used for cancer treatment (radio-chemotherapy).
One of the first events that occur after irradiation is the induction of DNA damage. Ionizing radiation can directly interact with critical cellular targets, such as DNA. This results in energy transfer, which induces atom ionization and subsequent biological changes (direct action). Additionally, ionizing radiation instantaneously induces the formation of water radiolysis products that contain reactive oxygen species (ROS) such as superoxide anions, hydrogen peroxide (H2O2), and hydroxyl radicals. These ROS exhibit high reactivity to a variety of cellular macromolecules, including DNA, lipids, and proteins. These free radicals can interact with macromolecules, such as DNA, and induce biological changes (indirect action). In the presence of O2, ionizing radiation converts hydroxyl radicals, superoxides, and organic radicals to H2O2 and organic hydroperoxides. 4 H2O2 is converted into hydroxyl radicals by a Fenton-type reaction. The hydroxyl radicals induce cellular lesions which are similar to lesions induced by ionizing radiation. 5 Hydroxyl radicals induce both chromosome breaks and the formation of numerous micronuclei in dividing cells.
In order to elucidate the mechanisms underlying tumor cell radioresistance, clinically relevant radioresistant (CRR) cell lines were developed by exposure to a stepwise increase of fractionated X-rays. 6 CRR cells proliferated for more than a month when exposed to 2 Gy/day of X-rays. Recently, it was shown that CRR cells are resistant to H2O2. 7 Conversely, it is unknown whether H2O2-resistant CRR cells are also resistant to X-rays. In this study, H2O2-resistant cancer cell lines were developed to evaluate their sensitivity to radiation.
Ogawa et al. developed a unique radiosensitization treatment using a H2O2 solution (Oxydol)-soaked gauze referred to as Kochi Oxydol-Radiation Therapy for Unresectable Carcinomas (KORTUC I). KORTUC I was applied to superficially exposed and unresectable neoplasms, such as malignant melanoma and malignant fibrous histiocytoma (MFH). The highly radioresistant human osteosarcoma cell line, HS-Os-1 was also subjected to KORTUC. 8 KORTUC I induces radiosensitization of radioresistant tumors by blocking antioxidative enzymes such as peroxidases, which results in local oxygen production. A new injectable radiosensitizer (KORTUC II) may convert radioresistant tumors into radiosensitive tumors. 9 In this study, the effects of H2O2 on the radioresistance of CRR cells was evaluated, which may be extrapolated to evaluate the efficacy of KORTUC for cancer treatment.
Materials and Methods
Cell Culture
Human cancer cell lines derived from hepatoblastoma (HepG2), oral squamous cell carcinoma (SAS), and cervical cancer (HeLa) tissues were obtained from the Cell Resource Center for Biomedical Research, Institute of Development, Aging and Cancer, Tohoku University. The CRR cells are referred to by their cell line name followed by an R to denote their radioresistance. The HepG2-8960-R and HepG2-R cells were independently established from HepG2 cells. The SAS-R and HeLa-R cells were derived from SAS and HeLa cells, respectively, by exposing the cells to gradually increasing X-ray doses. 10 The cells were maintained in Roswell Park Memorial Institute (RPMI) 1640 medium (Nacalai Tesque Inc., Kyoto, Japan) supplemented with 5% fetal bovine serum (Gibco Invitrogen Corp., Carlsbad, CA, USA) in a humidified atmosphere at 37°C with 5% CO2. Exponentially growing cells were used in all of the experiments. X-ray irradiation was conducted with a dose of 1 Gy/min in a 150-KVp X-ray generator (MBR-1520 R, Hitachi, Tokyo, Japan) with a total filtration of 0.5 mm aluminum and 0.1 mm copper. H2O2-resistant cell lines were obtained utilizing a conditioning method. Briefly, parental cells were cultured in medium containing increased levels of H2O2 in a stepwise fashion. CRR cell lines were developed by exposing cells to a stepwise increase in fractionated radiation from 0.5 Gy/day to 2 Gy/day. We hypothesized that the H2O2-resistant phenotype of CRR cells originated during the development of the CRR cell lines. In order to test this hypothesis, we obtained 2 types of cell lines. One cell line was exposed to 0.5 Gy/day × 10 (total dose 5 Gy) of X-rays and another cell line was exposed to 0.5 Gy/day × 10 + 1 Gy/day × 10 (total dose 15 Gy) of X-rays. Both of these cell lines were assayed for their sensitivity to H2O2 utilizing a water-soluble tetrazolium salt (WST) assay.
High-Density Survival Assay
The sensitivity of cells to X-rays was determined utilizing a modified high-density survival (MHDS) assay as previously reported. 11
WST Assay
The effect of H2O2 on cell proliferation was monitored with a WST assay, which was conducted using the Cell Count Reagent SF (Nacalai Tesque Inc., Kyoto, Japan) according to the manufacturer’s protocol. Briefly, 5 × 103 exponentially growing cells were plated in a 96 well plate 24 hours before the experiment. Next, the cells were incubated for 48 hours in RPMI 1640 medium containing H2O2. Subsequently, 10 µL of Cell Count Reagent SF was added to each well and the optical density of each well was determined with a Multiskan JX microplate reader (Thermo Fisher Scientific Inc., MA, USA). In order to determine the radiosensitizing effects of H2O2 in CRR cells, the cells were treated with H2O2 for 48 hours followed by exposure to 10 Gy of X-rays.
Intracellular ROS Assay
CRR cells were treated with or without 100 μM of H2O2 and the intracellular ROS levels were determined using the CellROX™ Green Reagent (Thermo Fisher Scientific, Inc., PA, USA) according to the manufacturer’s protocol. Fluorescent images of the cells were obtained using a BZ-8000 Fluorescence Microscope (KEYENCE, Osaka, Japan).
Statistics
The data from 3 independent experiments are expressed as the mean ± standard deviation (SD). Three independent experiments were performed with cells derived from 3 different cultures. The data for comparison may be the same in some figures. It was used as a representative example because it has been reproduced more than once. We also took this step because it is extremely difficult to conduct all the experiments on the same day. Comparison between the 2 groups was performed using the student’s t-test. One-way ANOVA with Dunnett’s test was performed for multiple comparisons. P < 0.05 was considered significant.
Results
All of the Tested CRR Cell Lines Were Resistant to H2O2-Induced Cell Death
In order to determine the sensitivity of CRR cells to H2O2, WST assays were conducted (Figure 1). The WST assay results showed that all of the CRR cell lines were significantly resistant to H2O2 compared to the corresponding parental cell lines. The H2O2 half maximal inhibitory concentration (IC50) values for the HepG2, HepG2-8960-R, and HepG2-R cells were approximately 13, 25, and 45 μM, respectively. The H2O2 IC50 values for the SAS and SAS-R cells were approximately 20 and 30 μM, respectively. Lastly, the H2O2 IC50 values for the HeLa and HeLa-R cells were approximately 5 and 15 μM, respectively.
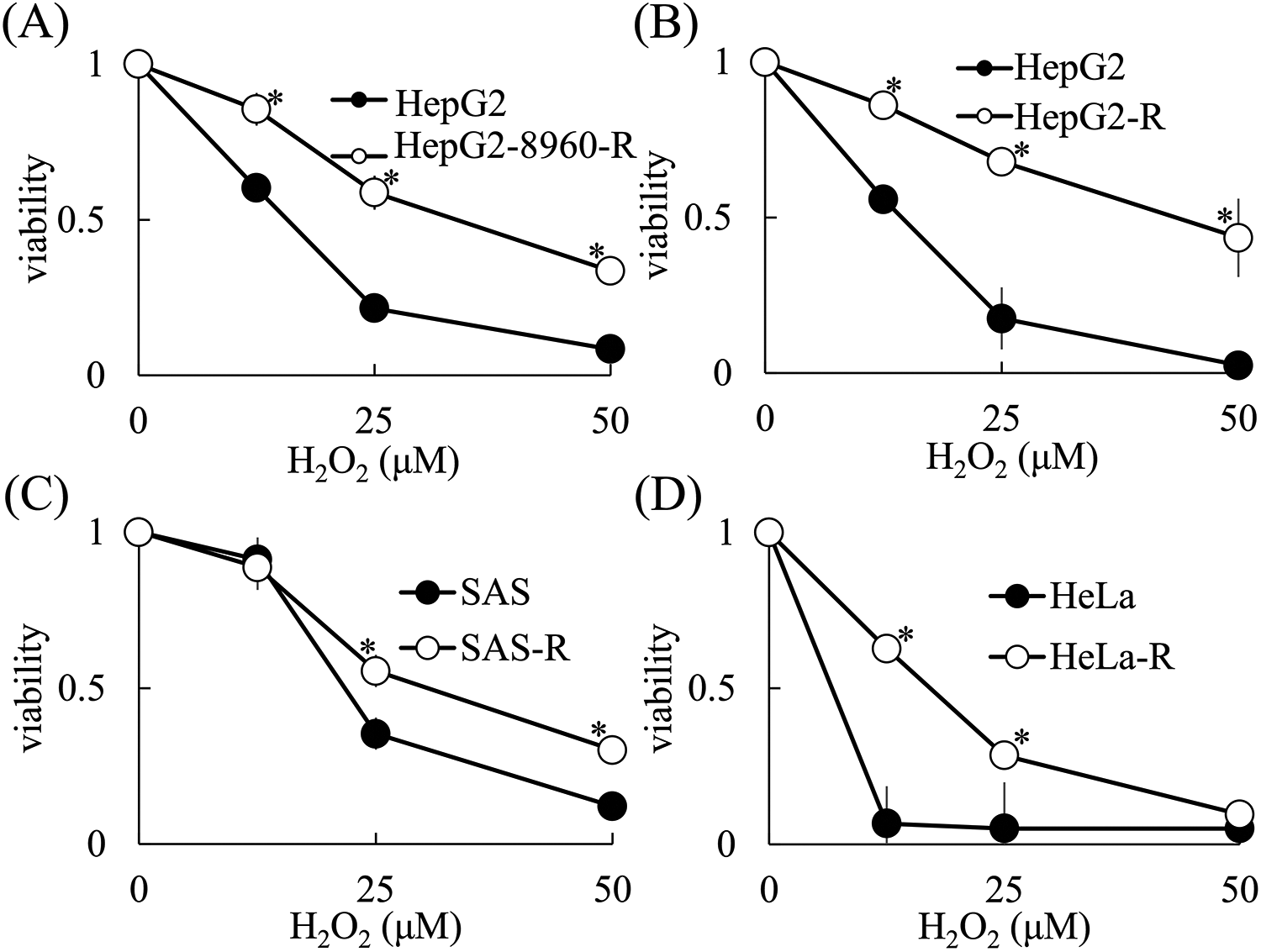
CRR cell lines are resistant to H2O2-induced cell death. Cells (5 × 103) were incubated with H2O2 for 48 hours and a WST assay was performed according to the manufacturer’s protocol. A, H2O2 sensitivity of parental HepG2 and CRR HepG2-8960-R cells. B, H2O2 sensitivity of parental HepG2 and CRR HepG2-R cells. C, H2O2 sensitivity of parental SAS and CRR SAS-R cells. D, H2O2 sensitivity of parental HeLa and CRR HeLa-R cells. The data represent the mean ± SD from 3 independent experiments and are presented relative to the control parental cell lines. *: p < 0.05 using Student’s t test (vs. Parental cells).
H2O2-Resistant Cell Lines Were Not Resistant to X-Ray-Induced Cell Death
Several distinct cell types, such as murine lens, monkey kidney, and hamster ovarian cells, have been found to acquire H2O2 resistance after conditioning with H2O2. 12 -15 In order to determine whether H2O2-resistant cells were also resistant to X-rays, we generated H2O2-resistant cells from HepG2, SAS, and HeLa cells. As shown in Figure 2, we successfully obtained H2O2-resistant cells, which are referred to by their cell line name followed by H2O2 to indicate their H2O2 resistance. HepG2-H2O2, SAS-H2O2, and HeLa-H2O2 cells continued to proliferate in medium containing up to 50 μM of H2O2, respectively. An MHDS assay revealed that the X-ray sensitivity of HepG2-H2O2 cells was nearly identical to that of parental HepG2 cells (Figure 3). Additionally, Figure 3 shows that SAS-H2O2 and HeLa-H2O2 cells exhibited similar X-ray sensitivity as their parental SAS and HeLa cells, respectively.

Generation of H2O2-resistant cell lines. The sensitivity of cells to H2O2-induced cell death was determined by WST assays. The H2O2-resistant cell lines are referred to as their cell line name followed by H2O2. The cells (5 × 103) were incubated with H2O2 for 48 hours and a WST assay was performed according to the manufacturer’s protocol. A, H2O2 sensitivity of parental HepG2 and HepG2-H2O2 cells. B, H2O2 sensitivity of parental SAS and SAS-H2O2 cells. C, H2O2 sensitivity of parental HeLa and HeLa-H2O2 cells. The data represent the mean ± SD of 3 independent experiments and are presented relative to the control parental cell lines. *: p < 0.05 using Student’s t test (vs. Parental cells).
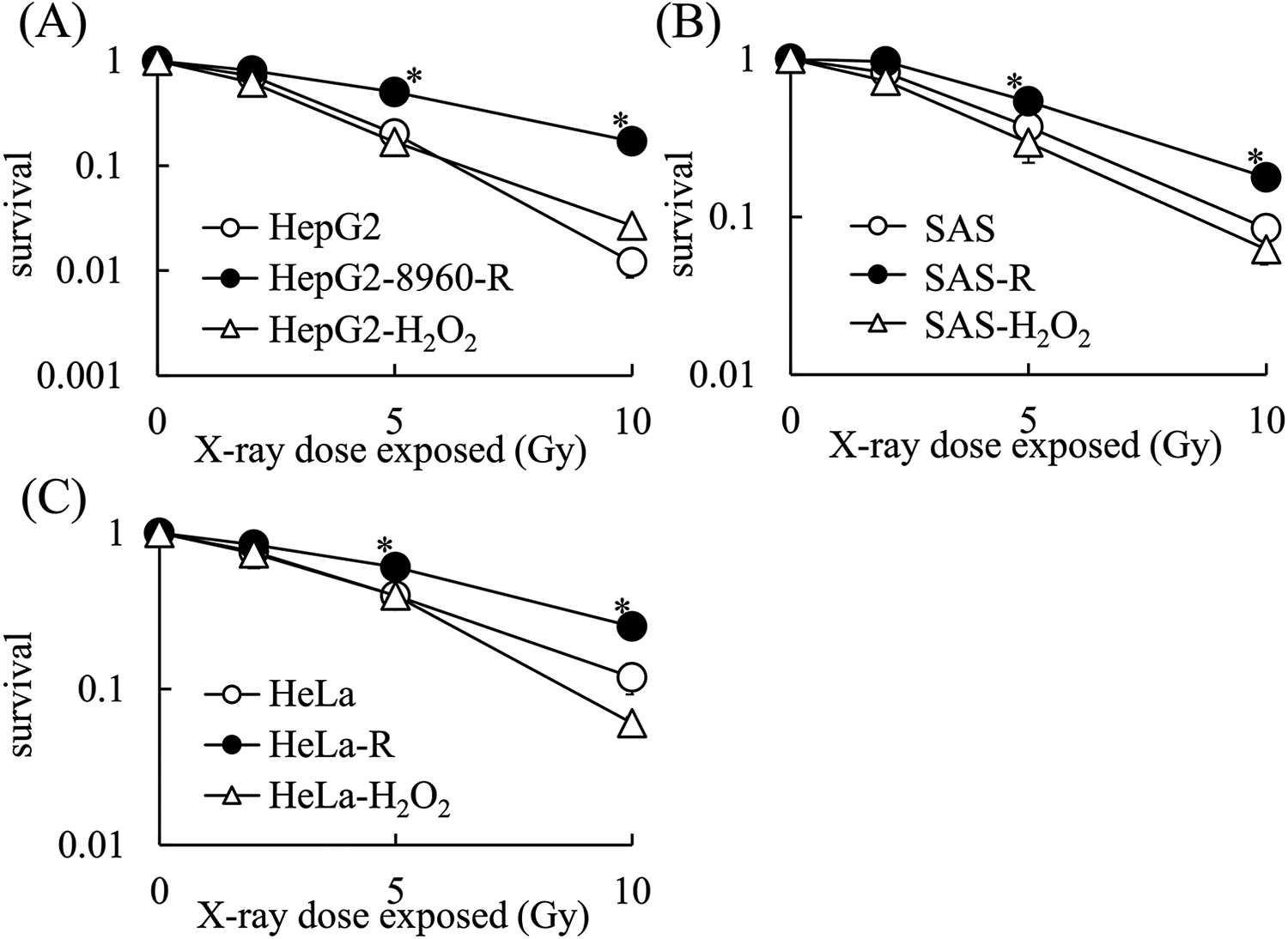
H2O2-resistant cell lines are not resistant to X-ray-induced cell death. A, X-ray sensitivity of HepG2, CRR HepG2-8960-R, and HepG2-H2O2 cells. B, X-ray sensitivity of SAS, CRR SAS-R, and SAS-H2O2 cells. C, X-ray sensitivity of HeLa, CRR HeLa-R, and HeLa-H2O2 cells. The data represent the mean ± SD of 3 independent experiments and are presented relative to the control parental cell lines. *: p < 0.05 using Dunnett’s test (vs. Parental cells).
Exposure to Fractionated X-Rays Induced H2O2 Resistance in Cancer Cells
HepG2 cells exposed to 0.5 Gy/day × 10 of fractionated X-rays (referred to as HepG2-0.5 cells) acquired H2O2-resistant phenotype (Figure 4). The sensitivity of HepG2-0.5 cells to H2O2-induced cell death was nearly identical to that of the HepG2-8960-R cells. Further exposure of HepG2-0.5 cells to 1 Gy/day of fractionated X-rays (referred to as HepG2-0.5-1 cells) did not increase the H2O2 resistance. On the other hand, SAS cells did not acquire H2O2 resistance upon exposure to 0.5 Gy/day × 10 of fractionated X-rays (referred to as SAS-0.5 cells). However, additional exposure of these cells to 1 Gy/day × 10 of fractionated X-rays (referred to as SAS-0.5-1 cells) induced H2O2 resistance. In addition, the exposure of HeLa cells to 0.5 Gy/day × 10 of fractionated X-rays (referred to as HeLa-0.5 cells) induced H2O2 resistance.
The H2O2 Resistance Phenotype of the CRR Cell Lines Was Irreversible
In order to maintain the X-ray resistance of CRR cells, they were exposed daily to 2 Gy of X-rays (maintenance irradiation, MI). In order to test whether H2O2 resistance in CRR cell lines is reversible, we cultured CRR cells without MI for 3 months, and then determined the H2O2 sensitivity of these cells by WST assays. The CRR cells cultured without MI for 3 months were still radioresistant (data not shown). WST assays indicated that the H2O2 sensitivity of HepG2-8960-R cells cultured without MI (referred to as HepG2-8960-R-NoIR cells) was nearly identical to that of HepG2-8960-R cells (Figure 5A). Furthermore, WST assays indicated that the H2O2 sensitivity of HepG2-R cells cultured without MI (referred to as HepG2-R-NoIR cells) was nearly identical to that of HepG2-R cells (Figure 5B). Moreover, WST assays indicated that the H2O2 sensitivity of SAS-R cells cultured without MI (referred to as SAS-R-NoIR cells) was nearly identical to that of SAS-R cells (Figure 5C). In contrast, WST assays showed that the H2O2 sensitivity of HeLa-R cells cultured without MI (referred to as HeLa-R-NoIR cells) was reduced compared to that of HeLa-R cells. Thus, HeLa-R-NoIR cells were more resistant to H2O2 compared with HeLa-R cells. These results suggested that H2O2 resistance in CRR cell lines was irreversible.

Exposure to fractionated X-rays induced H2O2 resistance in cancer cells. Cells (5 × 103) were incubated with H2O2 for 48 hours and a WST assay was performed according to the manufacturer’s protocol. A, H2O2 sensitivities of parental HepG2 cell, 0.5 Gy/day × 10 of fractionated X-rays pretreated HepG2 (HepG2-0.5) cell, 0.5 Gy/day × 10 plus 1 Gy/day × 10 of fractionated X-ray pretreated HepG2 (HepG2-0.5-1) cell and CRR HepG2-8960-R cell. B, H2O2 sensitivities of parental SAS, 0.5 Gy/day × 10 of fractionated X-rays pretreated SAS (SAS-0.5) cell, 0.5 Gy/day × 10 plus 1 Gy/day × 10 of fractionated X-ray pretreated SAS (SAS-0.5-1) cell and CRR SAS-R cells. C, H2O2 sensitivity of parental HeLa, 0.5 Gy/day × 10 of fractionated X-rays pretreated HeLa (HeLa-0.5) cell and CRR HeLa-R cell. The data represent the mean ± SD of 3 independent experiments and are presented relative to the control parental cell lines. *: p < 0.05 using Dunnett’s test (vs. Parental cells).
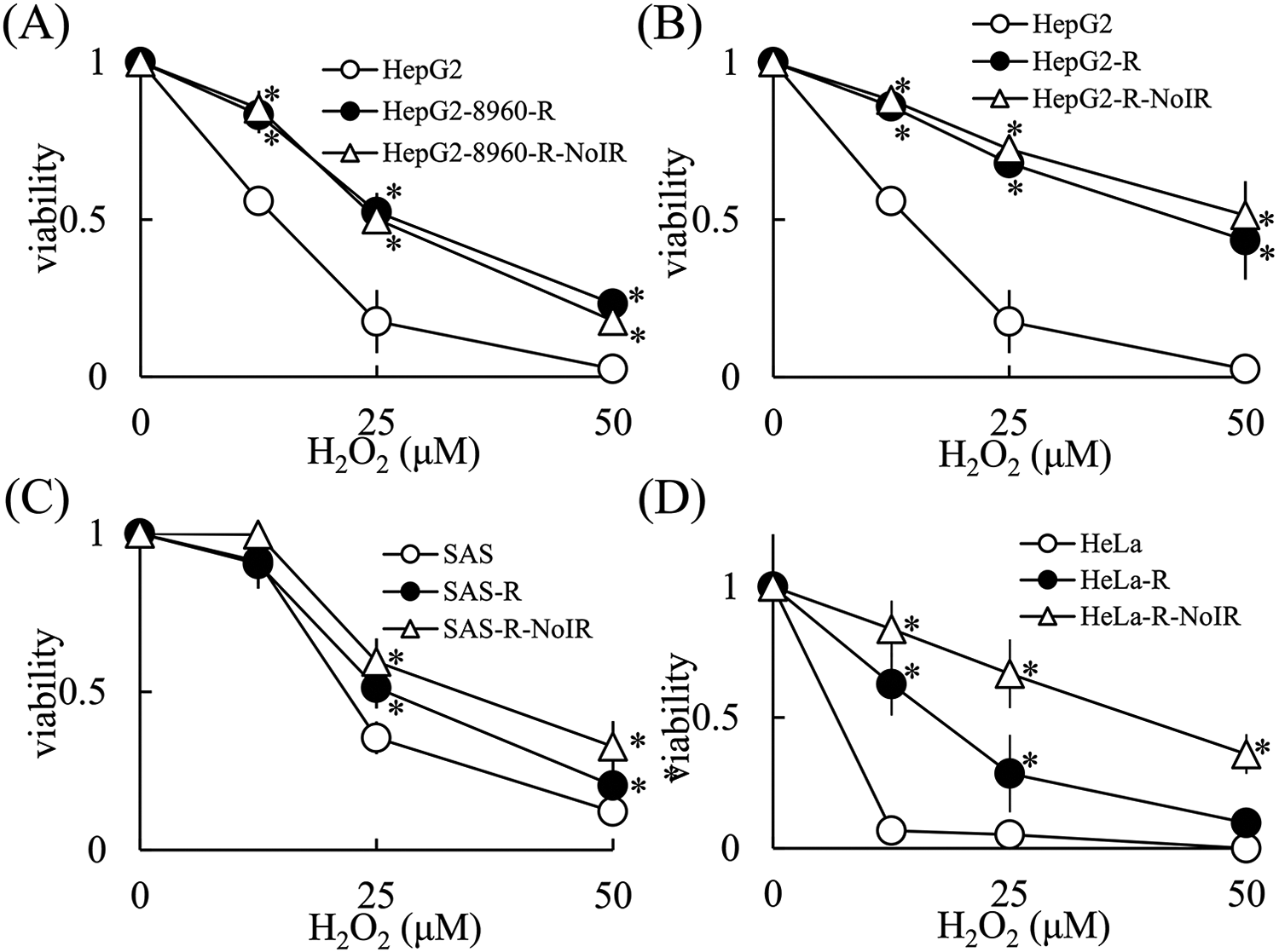
The H2O2 resistance phenotype of CRR cell lines was irreversible. Cells (5 × 103) were incubated with H2O2 for 48 hours and a WST assay was performed according to the manufacturer’s protocol. A, H2O2 sensitivity of parental HepG2, CRR HepG2-8960-R, and HepG2-8960-R-NoIR cells. B, H2O2 sensitivity of parental HepG2, CRR HepG2-R, and HepG2-R-NoIR cells. C, H2O2 sensitivity of parental SAS, CRR SAS-R, and SAS-R-NoIR cells. D, H2O2 sensitivity of parental HeLa, CRR HeLa-R, and HeLa-R-NoIR cells. The data represent the mean ± SD of 3 independent experiments and are presented relative to the control parental cell lines. *: p < 0.05 using Dunnett’s test (vs. Parental cells).
Exposure to X-Rays Induced H2O2 Resistance in CRR Cell Lines
The effects of H2O2 after X-ray exposure were evaluated utilizing WST assays. As expected, 10 Gy of X-rays sensitized parental HepG2, SAS, and HeLa cells to H2O2 (Figure 6). In contrast, X-rays induced further H2O2 resistance in all of the tested CRR cell lines including HepG2-8960-R, HepG2-R, SAS-R, and HeLa-R.

Exposure of CRR cells to X-rays enhanced their resistance to H2O2-mediated cell death. Following incubation with H2O2 for 1-h, cells (5 × 103) was irradiated with 10 Gy of X-rays and incubated extra 48 hours. WST assays were then performed according to the manufacturer’s protocol. A, H2O2 sensitivity of parental HepG2 and HepG2 irradiated cells. B, H2O2 sensitivity of CRR HepG2-8960-R and CRR HepG2-8960-R irradiated cells. C, H2O2 sensitivity of HepG2-R and HepG2-R irradiated cells. D, H2O2 sensitivity of Parental SAS and SAS irradiated cells. E, H2O2 sensitivity of CRR SAS-R and SAS-R irradiated cells. F, H2O2 sensitivity of parental HeLa and HeLa irradiated cells. G, H2O2 sensitivity of CRR HeLa-R and HeLa-R irradiated cells. The data represent the mean ± SD of 3 independent experiments and are presented relative to the control parental or CRR cell lines. *: p < 0.05 using Student’s t test (vs. no irradiated cells).
The Kinetics of Intracellular ROS Production in H2O2-Treated Parental and CRR Cells
The intracellular ROS levels were quantitated using the CellROXTM Green Reagent. In untreated cells, the amount of intracellular ROS was higher in HepG2-8960-R cells compared to HepG2 cells (Figure 7). After treatment with 100 μM of H2O2 for 3 hours, the intracellular ROS level was increased in HepG2 cells compared to untreated cells. Additionally, the intracellular ROS level decreased in HepG2 cells after H2O2 treatment for 24 hours. In contrast, in HepG2-8960-R cells, a significant increase in intracellular ROS was not observed after H2O2 treatment for 3 or 24 hours. These tendencies were also observed in SAS, SAS-R, HeLa and HeLa-R.

The kinetics of intracellular ROS production in H2O2-treated parental and CRR cells. Cells were treated with or without 100 μM of H2O2 for 3 or 24 hours and the intracellular ROS levels were determined using the CellROX™ Green Reagent according to the manufacturer’s protocol.
Discussion
One of the main obstacles of radiation therapy for cancer treatment is the existence of radioresistant cells. In order to understand the characteristics of radioresistant cells, CRR cell lines have been generated. Screening for anticancer drugs against CRR cell lines elucidated that all CRR cells were resistant to docetaxel (an inhibitor of microtubule depolymerization) compared with their parental cells. 16 Recently, Tomita et al. found that the CRR cell lines, SAS-R and HeLa-R, were resistant to H2O2 compared to their corresponding parental SAS and HeLa cell lines, respectively. 7 They suggested that the membrane status is a major factor conferring H2O2 resistance in CRR cells. In this study, we generated CRR cell lines by exposing parental cell lines to a stepwise increase in fractionated X-rays from 0.5 to 2 Gy/day. We then examined whether our established CRR cell lines were resistant to H2O2. We found that SAS-R, HeLa-R, HepG2-8960-R and HepG2-R cell lines were all resistant to H2O2. We also found that the H2O2-resistant phenotype is an irreversible characteristic of the CRR cell lines. Our results (Figure 4) indicated that CRR cells acquired H2O2 resistance during the development of radioresistance, suggesting that tumors subjected to radiotherapy might possess H2O2 resistance.
In this study, we developed H2O2-resistant cell lines and evaluated their sensitivity to radiation. Unexpectedly, the H2O2-resistant cells did not exhibit radioresistance compared to their parental cells (Figure 3). Exposure of cells to ionizing radiation leads to the formation of ROS (including H2O2), which is associated with radiation-induced cytotoxicity. 17,18 Therefore, ROS scavengers are one of the important factors that protect cells against ROS-induced injury during ionizing radiation exposure. Previous studies showed that H2O2-resistant cells had increased catalase expression. 19,20 Catalase is a potent antioxidant enzyme that converts H2O2 into hydrogen and water. Tomita et al. reported that catalase activity was lower in CRR cells compared to parental cells, despite higher H2O2 resistance. Previous reports showed that H2O2-induced DNA damage is distinct from ionizing radiation-induced DNA damage. 21 H2O2 produces hydroxyl radical damage in the form of DNA single-strand breaks. 22 DNA double-strand breaks (dsbs) induced by ionizing radiation are the most deleterious lesion that induces cell death. H2O2 does not produce DNA dsbs. It has been reported that there was an increase in γH2AX-positive cells in the parent cell line after irradiation, but not in CRR cells. 22 From these results we hypothesize that CRR cells can efficiently repair not only DNA dsbs but also DNA single-strand breaks. Therefore, CRR cells are resistant to H2O2.
Radiotherapy is one of the most common treatments for cancer. However, the existence or induction of radioresistant cells is one of the major problems to overcome in this therapy. Therefore, approaches utilizing a sensitizer in combination with radiation therapy have been proposed, and many drugs have been tested for this purpose. As we previously reported, Everolimus is one candidate. 23 H2O2 is another candidate and KORTUC therapy is currently being used to treat multiple cancer types. 24 -26 In biological systems, H2O2 is converted into a more reactive species (e.g. hydroxyl radical) via the Fenton reaction, which is a transition metal-catalyzed reaction. 27 These reactive species injure vital molecules including DNA, proteins, and membrane lipids, which leads to the destruction of cellular integrity. Several enzymes scavenge these toxic oxidants or transform them into nontoxic metabolites; e.g. catalase decomposes H2O2 to water and molecular oxygen. Since hydroxyl radicals are a direct product of H2O2 catalysis, H2O2 may generate greater amounts of hydroxyl radicals compared to radiation therapy. Ogawa theorized that when tumors are irradiated in the presence of H2O2, the activities of antioxidative enzymes such as peroxidase and catalase are blocked, and oxygen molecules are produced at the same time via the H2O2 effect. This results in oxidative damage to low-LET radioresistant tumor cells, thereby rendering them highly sensitive to irradiation. 8 It should be noted that the overall levels of intracellular ROS in CRR cells were abundant compared to the levels in parental cells (Figure 7). Treatment with H2O2 increased intracellular ROS in HepG2 cells but not in HepG2-8960-R cells (Figure 7). We hypothesize that antioxidative enzymes are blocked in parental cells, which renders them highly sensitive to radiation. Due to the overall high amount of intracellular ROS in CRR cells, treatment with H2O2 might have no effect on CRR cells. Tomita et al. reported that catalase activity was lower in CRR cells compared to parental cells. 28 High levels of intracellular ROS may inactivate catalase in CRR cells. We hypothesize that internal ROS scavenging activity, such as via catalase activity, may not be a major factor contributing to H2O2 resistance in CRR cells. In CRR cells, it has been reported that the amount of Fe2+ was lower than that in the parental cells. 29 Furthermore, regarding the amount of hydroxyl radicals, it has been reported that the amount of hydroxyl radicals in CRR cells is lower than that in the parental cells. 28,29 These results indicate that the Fenton reaction is one of the key factor in the resistant phenotype in CRR cells because it generates hydroxyl radicals and oxidize the plasma membrane finally leads to cell death.
In this study, we found that fractionated X-ray exposure induced H2O2 resistance in CRR cells. Therefore, it is necessary to carry out cancer therapy such as KORTUC with the presence of these resistant cells in mind, and as the next stage, it would be necessary to investigate the appearance rate of these cells immediately and take countermeasures.
Footnotes
Authors’ Note
This study does not include animal studies or human studies that require ethical review. The origin of the cell lines used in this study was stated in materials and methods section.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI (Grant-in Aid for Scientific Research C: 19K12326 and 16K00538 to Y.K.; No. 19K10318 to K.T.).
