Abstract
Objective:
To compare the short-term clinical effects between totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis, and laparoscopic-assisted radical gastrectomy with Roux-en-Y anastomosis; to explore the safety, feasibility and short-term effect of totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis.
Methods:
Data of 75 patients who underwent totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis, and 95 patients who underwent laparoscopic-assisted radical gastrectomy with Roux-en-Y anastomosis by the same surgical team were analyzed. During the modified Roux-en-Y anastomosis, the stomach separation and regional lymph node dissection were completed under a laparoscope; the specimen was placed in a bag; gastrojejunostomy was completed; the subumbilicus hole was enlarged to 3 cm; the specimen was taken out; then, the proximal and distal ends of the small intestine were moved outside of the abdominal wall to complete the small intestine-small intestine end-to-side anastomosis.
Results:
All 170 operations were successful. The differences in the time of anastomosis and the number of dissected lymph nodes between the 2 groups were not statistically significant (P > 0.05), but in the totally-MA group the amount of bleeding and the length of incision significantly decreased (P < 0.05). The recovery time as measured by breathing unassisted, drinking fluids and getting out of bed was significantly shorter than those in the laparoscopic-assisted group (P < 0.05), and the pain score 1 day after surgery was significantly lower than that of the laparoscopic-assisted group (P < 0.05). One case of duodenal stump leakage and 1 case of esophagojejunostomy leakage were found in the laparoscopic-assisted group. In the totally-MA group, there were no complications such as anastomotic leakage, anastomotic stenosis or anastomotic bleeding, but 2 patients with double primary carcinoma underwent joint radical resection.
Conclusion:
Compared with laparoscopic-assisted surgery, totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis has the advantages of being safer and less traumatic, with associated reductions in bleeding and pain.
Keywords
Introduction
The surgical methods of gastric cancer include open radical gastrectomy and laparoscopic radical gastrectomy. Results from multiple studies suggest that there is no significant difference in the short-term effect and long-term prognosis between laparoscopic gastrectomy and open gastrectomy. 1 -3 Because it has the advantages of being less traumatic and painful, and associated with quicker recovery times, coupled with the rapid development of laparoscopic technology, laparoscopic radical gastrectomy has been widely used in clinic. Its clinical feasibility and safety are recognized. 4,5 The reconstruction of the digestive tract after laparoscopic radical gastrectomy has also become a hot topic in the field of surgery. 6,7
At present, the methods of laparoscopic gastrectomy and gastrointestinal reconstruction include Billroth I, Billroth II and Roux-en-Y anastomosis. The choice of anastomotic method is related to the location of the tumor, the progress of the tumor, the constitution of the patient and the technical proficiency of the surgeon, 8 but there is no consensus regarding the preferred method. A meta-analysis reveals that there is no difference among the 3 methods in the occurrence of postoperative dumping syndrome. Roux-en-Y anastomosis is superior to Billroth I and Billroth II in preventing bile reflux and residual gastritis. This kind of reflux often causes basic reflux gastritis and esophagitis, and even increases the canceration of remnant stomach or the esophagus. Therefore, Roux-en-Y is considered a safer anastomotic method in clinical practice. 9
At present, there are 2 main procedures for laparoscopic gastrectomy and digestive tract reconstruction: totally laparoscopic gastrectomy and laparoscopic-assisted gastrectomy. 10,11 However, there are many anastomotic devices used in totally laparoscopic radical gastrectomy and digestive tract reconstruction that are expensive, difficult to use, and require higher technical competence from surgeons. To some extent, this limits its clinical application. Meanwhile, laparoscopic-assisted radical gastrectomy requires a 5–7 cm abdominal longitudinal incision. Therefore, laparoscopic-assisted radical gastrectomy is widely used in clinic, during which the stomach separation and regional lymph node dissection are completed under a laparoscope. Then the reconstruction of the digestive tract is completed by making a 5–7 cm auxiliary incision in the middle of the upper abdomen. However, in recent years, with the optimization of laparoscopic equipment and the maturity of laparoscopic operation technology of surgeons, the application of totally laparoscopic gastrectomy has gradually increased. 12 In this operation, lymph node dissection, stomach separation and digestive tract reconstruction are completed under a laparoscope, which has a clear field of vision and results in less blood loss. The excised specimens are bagged and removed via the enlarged umbilical incision, causing little trauma and offering an obvious cosmetic benefit.
Considering the disadvantages of the 2 methods, our department actively explores the totally laparoscopic radical gastrectomy and improves Roux-en-Y anastomosis. The modified procedure is as follows: the stomach separation and regional lymph node dissection are completed under a laparoscope; the specimen is placed in a bag; gastrojejunostomy (esophagojejunostomy) is completed; the subumbilicus hole is enlarged to 3 cm; the specimen is taken out; finally, the proximal and distal ends of the small intestine are taken outside the abdominal wall to complete the small intestine-small intestine end-to-side anastomosis. This procedure saves anastomotic apparatuses and reduces the difficulty of the operation.
From November 2017 to November 2019, 75 cases of totally laparoscopic radical gastrectomy and modified Roux-en-Y anastomosis were performed in our department. In this study, these cases were compared with 95 gastric cancer patients who underwent Roux-en-Y laparoscopic-assisted radical gastrectomy, to explore the safety, feasibility and short-term effects of totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis.
Methods
Patient Information
In this retrospective analysis study, from November 2017 to November 2019, we collected the data from 75 patients with primary gastric cancer who were treated with laparoscopic radical gastrectomy and modified Roux-en-Y anastomosis (referred to as the “totally-MA group”), and 95 primary gastric cancer patients who were diagnosed at the same time and underwent Roux-en-Y laparoscopic-assisted radical gastrectomy (referred to as the “laparoscopic-assisted group”). A comparative analysis was conducted between these 2 groups. Both groups of patients were operated on by the same surgical team. Before each operation, the location, pathological type, depth of invasion, lymph node metastasis and distant metastasis of the tumor were determined by ultrasonography, pathological biopsy and computed tomography (CT). The TNM (tumor, node, metastasis) staging was based on the American Joint Committee on Cancer (AJCC) Eighth Edition 2017 gastric cancer staging standard. 13 The study was conducted in accordance with the Declaration of Helsinki(as was revised in 2013). The study was approved by Ethics Committee of Hubei Cancer Hospital (No.LLHBCH2017KY-010) and informed consent was taken from all the patients.
Inclusion criteria: 1. Clinical data were complete and gastric cancer was confirmed by pathological results. 2. Patient’s status was adequate to take the operation (no other severe diseases). 3. Had not taken neoadjuvant chemoradiotherapy before surgery. 4. Signed the informed consent. Exclusion criteria: 1. Patient’s status was not adequate to take the operation (had other severe diseases). 2. Imaging results showed that the tumor had invaded adjacent organs or had distant metastasis. 3. ≥2 cm of the lower esophagus was implicated. 4. Had massive hemorrhage or completely obstruction of the tumor
Preoperative Preparation and Trocar Position
The patient was placed in the supine position and the legs were separated from each other. General anesthesia was performed by tracheal intubation. The preoperative preparation was the same as for a routine laparotomy, with the the side of the head raised about 15°. The pneumoperitoneum needle was inserted under the umbilicus. Air was pumped into the abdominal cavity and the pressure was maintained at 12–15 mm Hg (1 mm Hg = 0.133 kPa). A 10 mm trocar was placed under the umbilicus as the observation hole. At 2 cm below the costal margin and on the left anterior axillary line, a 12 mm trocar was placed as the main operation hole. A 5 mm trocar was placed on the middle line of the left clavicle, 2 cm above the umbilicus, and a 5 mm trocar was placed in the corresponding position on the right. Routinely, the surgeon stands on the patient’s left side, while the first assistant stands on the right. The locations of the 5 trocars are shown in Figure 1.

Trocar location diagram.
Radical Gastrectomy
According to the principle of radical treatment of gastric cancer, gastrectomy and regional lymph node dissection were performed. 14
Mode of Anastomosis
The totally-MA group
First, the duodenum was cut off with a linear Endo-GIA stapler. (1) Modified Roux-en-Y anastomosis after distal gastrectomy: the upper part of the gastric body was cut off with a linear Endo-GIA stapler. The resected specimen was put into the bag, then the specimen bag was closed. The jejunum was cut off at a distance of 25 cm from the Treitz ligament with a linear Endo-GIA stapler. Small incisions were made at the greater curvature of the remnant stomach and the distal jejunum, then the linear Endo-GIA stapler was extended into the small incision to close the distal jejunum and the remnant stomach, and complete the side-to-side anastomosis of the gastrojejunum. The assistant lifted both ends of the common opening for gastrojejunostomy; closed the common opening with the linear Endo-GIA stapler; marked the proximal jejunum stump and the small intestine 40 cm from the gastrointestinal anastomotic opening with silk thread sutures; enlarged the incision of the trocar under the umbilicus by approximately 3 cm around the umbilicus; took out the specimen completely; took the segment of the small intestine marked with silk thread out of the body, and manually completed the end-to-side small intestine-small intestine anastomosis. (2) Modified Roux-en-Y anastomosis after total gastrectomy: A small incision was made in the subphrenic esophagus; the mesentery was dissected at a distance of 25 cm from the Treitz ligament; a small incision was made in the intestinal wall; then the linear Endo-GIA stapler was extended into the small incision to close the distal jejunum and the esophagus, to complete side-to-side esophagojejunostomy; the common opening was closed with the Endo-GIA stapler, and the whole stomach and proximal jejunum were cut off. The resected specimen was put into the specimen bag; the proximal jejunum stump and the small intestine 40 cm from the gastrointestinal anastomotic opening were sutured with silk threads and marks were made. The incision of the trocar under the umbilicus was enlarged by approximately 3 cm around the umbilicus; the specimen was taken out completely; the segment of the small intestine marked with silk thread was taken out of the body, and the end-to-side small intestine-small intestine anastomosis was manually completed (Figures 2 –4). To use the circular stapler in most cases of esophagojejunostomy in laparoscopic assisted gastrectomy, and the linear stapler is often used in totally laparoscopic gastrectomy.

Modified Rouxen-Y anastomosis after distal gastrectomy. A, Amputation of duodenum, (B) Amputation of the stomach, (C) Amputation of jejunum. D, Incising the distal jejunum. E, Incising the greater curvature of the stomach. F, Closing the remnant stomach and jejunum. G, Closing the common opening of the stomach and jejunum. H, Anastomosis between the stomach and jejunum. I, Suturing marked proximal jejunum and small intestine.

Modified Rouxen-Y anastomosis after total gastrectomy. A, Amputation of duodenum. B, Incising marked wall of esophagus. C, Incising jejunum. D, Closing the esophagus and jejunum. E, Closing the common opening of the esophagus and jejunum. F, Anastomosis between the esophagus and jejunum. G, Suturing marked proximal jejunum and small intestine.

Completing small intestine-small intestine end-to-side anastomosis out of the body.
Laparoscopic-assisted group
After laparoscopic distal gastric separation and lymph node dissection, a 5–7 cm longitudinal incision was made under the xiphoid process to reconstruct the digestive tract. The surgical procedure was the same at that of traditional open surgery. 15,16
Observation Indexes
Intraoperative Indexes
The time of anastomosis, the number of dissected lymph nodes, the volume of bleeding and the length of incision were recorded during the operation. In the laparoscopic-assisted group, the time of anastomosis was calculated from amputation of the duodenum to the end of anastomosis. In the totally-MA group, it was calculated from the amputation of the duodenum to the removal of the specimen, until the small intestine-small intestine end-to-side anastomosis was completed manually outside of the body.
Postoperative Indexes
After the operation, After the operation, the patient’s pain score on the first day post-surgery was recorded, as was the time needed before the patient was breathing unassisted, drinking fluids and getting out of bed. Complications such as anastomotic leakage, anastomotic stenosis or anastomotic bleeding were also observed (Figure 5). Pain was rated using a visual analog scale (VAS). In this method, the degree of pain is represented by 11 numbers from 0 to 10. No pain is represented by 0; less than 3 points, the patient has slight but bearable pain; 4–6 points, the patient has tolerable pain which affects sleep and clinical treatment should be given; 7–10 points, the patient has gradually intensifying, severe or intolerable pain; 10 points represents maximum pain.

Incision after the operation.
Statistical Analysis
Data were statistically analyzed using SPSS25.0 software. Measurement data were expressed as mean ± standard deviation (`x ± SD), and compared using independent sample t-test. Count data were compared using the Chi-square test or the Fisher exact probability test. P < 0.05 was considered statistically significant.
Results
Comparison of General Data Between 2 Groups of Patients
As shown in Table 1, the distribution differences in gender proportion, age, distribution of primary lesion, the number of operation cases and TNM stage between the 2 groups were not statistically significant (P > 0.05), while the number of dissected lymph nodes (25.4 ± 9.6 vs. 28.1 ± 7.6, P = 0.044) between the 2 groups were statistically significant.
Comparison of General Data Between 2 Groups of Patients.

Comparisons of Intraoperative Conditions Between 2 Groups of Patients
As shown in Table 2, all 170 cases were successfully operated on. The differences in the time of anastomosis (57.2 ± 9.0 min vs 55.1 ± 9.7 min, P = 0.147), and the number of dissected lymph nodes (25.4 ± 9.6 vs. 28.1 ± 7.6, P = 0.044) between the 2 groups were not statistically significant. In the totally-MA group, the amount of bleeding (113.0 ± 61.5 ml vs 178.2 ± 96.1 ml, P = 0.000) decreased significantly, and the length of incision (3.2 ± 0.4 cm vs 6.5 ± 0.9 cm, P = 0.000) also decreased significantly. Furthermore, there were 2 cases of double primary carcinoma in the totally-MA group. One had gastric cancer with right renal cancer; the other had gastric cancer with sigmoid colon cancer. Both successfully underwent a right renal cancer radical resection and a sigmoid colon cancer radical resection, respectively, by changing the body position.
Comparisons of Intraoperative Conditions Between 2 Groups of Patients.

Comparisons of Postoperative Conditions Between The 2 Groups of Patients
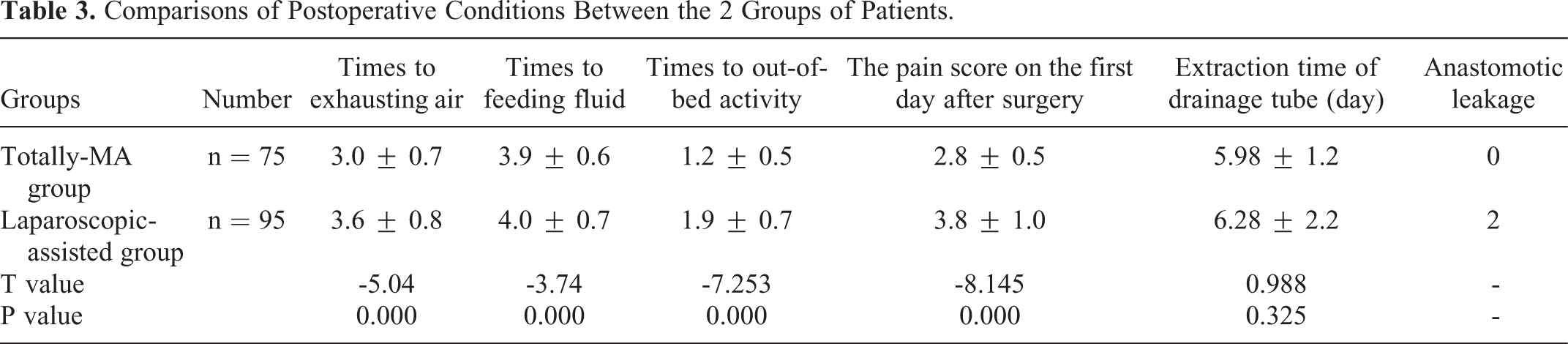
As shown in Table 3, in the totally-MA group, the times to exhausting air (3.0 ± 0.7 d vs 3.6 ± 0.8 d, P = 0.000), feeding fluid (3.9 ± 0.6 d vs 4.0 ± 0.7 d, P = 0.000), and out-of-bed activity (1.2 ± 0.5 d vs 1.9 ± 0.7 d, P = 0.000) were significantly shorter than those of the laparoscopic-assisted group, and the pain score on the first day after surgery was significantly lower than that of the laparoscopic-assisted group (2.8 ± 0.5 points vs 3.8 ± 1.0 points, P = 0.000).
Comparisons of Postoperative Conditions Between the 2 Groups of Patients.
In the laparoscopic-assisted group, the incidence of anastomosis-related complications was 2.1% (2 cases). One case of duodenal stump leakage healed after unobstructed drainage, and symptomatic and supportive treatment. One case of esophagojejunostomy leakage healed after inserting a jejunal nutrition tube, continuous negative pressure washing and drainage, and stronger anti-infection treatment. No complications such as anastomotic leakage, anastomotic stenosis or anastomotic bleeding occurred in the totally-MA group.
Discussion
In the present study, the clinical data of 75 patients who underwent totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis (the totally-MA group) and 95 patients who underwent laparoscopic-assisted radical gastrectomy with Roux-en-Y anastomosis (the laparoscopic-assisted group) in the same period and under the care of the same surgical team were compared and analyzed. The results revealed that the differences in the time of anastomosis and the number of dissected lymph nodes between the 2 groups were not statistically significant, but in the totally-MA group the volume of intraoperative bleeding and the length of incision were significantly decreased compared to those in the laparoscopic-assisted group (P < 0.05). The time to exhausting air, feeding fluid and out-of-bed activity were significantly shorter than those in the laparoscopic-assisted group (P < 0.05), and the pain score at the first day after surgery was significantly lower than that of the laparoscopic-assisted group (P < 0.05). The results suggest that, compared with laparoscopic-assisted gastrectomy, totally laparoscopic radical gastrectomy has the advantages of a smaller incision, less bleeding, a faster recovery and less pain. 17
During the study, in the laparoscopic-assisted group, 1 case of duodenal stump leakage healed after unobstructed drainage, and symptomatic and supportive treatment. One case of esophagojejunostomy leakage healed after indwelling a jejunal nutrition tube, continuous negative pressure washing and drainage, and stronger anti-infection treatment. The incision of anastomosis after radical gastrectomy is a grade II incision. In case of anastomotic leakage, the contents of the digestive tract easily contaminate the ruptured anastomosis, causing infection along the protracted course of the anastomosis. Statistics show that the incidence of anastomotic leakage after radical gastrectomy fluctuated within 0–26%. 18 Statistics from Japan revealed that the incidence of anastomotic leakage was 1.0–2.1% in laparoscopic-assisted radical gastrectomy. 19 The incidence of postoperative anastomoses in this study is consistent with the results of the aforementioned literature.
There were 2 cases of double primary carcinoma in the totally-MA group. One patient had gastric cancer with right renal cancer. Another had gastric cancer with sigmoid colon cancer. Both patients underwent right renal cancer radical resection and sigmoid colon cancer radical resection, respectively, under a laparoscope. Multiple primary cancer belongs to primary cancer. The treatment and prognosis are different from those of recurrent or metastatic cancer. Most primary cancers are treated comprehensively, mainly by surgery. The different types of cancer have different prognoses. The prognosis of multiple primary cancer is better than that of recurrent or metastatic cancer. Some scholars consider that the prognosis of multiple primary cancer is poorer than that of single primary cancer. 20,21 However, there are also reports of opposing views. LV et al. revealed in their study that the prognosis of multiple primary cancer was not poorer than that of single primary cancer. 22 It was reported in a previous literature that the 5-year survival rate of patients with gastric cancer (50.7%) was not statistically significantly different from those with single primary gastric cancer (51.6%), which was better than that of tumor recurrence and metastasis. 23 Therefore, for such patients, once the diagnosis is confirmed, active treatment including surgery should be given. In our study, 2 patients were found to have double primary cancer by preoperative examination. The diagnosis was confirmed by comprehensive examination, then the operation plan was formulated. Both of the patients completed totally laparoscopic right renal cancer radical resection and sigmoid colon cancer radical resection (respectively) by changing the body position, so as to prolong their survival period and improve their quality of life.
Compared with laparoscopic-assisted radical gastrectomy with Roux-en-Y anastomosis, totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis is a more technically difficult operation and carries the risk of secondary damage to the small intestine-small intestine end-to-side anastomosis. Therefore, it limits the clinical popularity of totally laparoscopic radical gastrectomy with reconstruction of the digestive tract. However, in the modified Roux-en-Y anastomosis in this study, the subumbilicus hole was enlarged to 3 cm, the specimen was taken out, then the proximal and distal ends of the small intestine were taken outside the abdominal wall through the subumbilicus hole to complete the small intestine-small intestine end-to-side anastomosis. This made the operation easier to perform, quicker, and also shortened the learning cycle. In addition, the incision was small and more cosmetically pleasing. The incision length of the subumbilicus hole was 3.2 ± 0.4 cm, significantly shorter than that of longitudinal incision (6.5 ± 0.9 cm) in the laparoscopic-assisted group. The subumbilicus hole was folded within the patient’s navel after healing, leaving little scarring. In addition, due to the reduced trauma, small abdominal incision and mild postoperative pain, patients could eat and undergo get out of bed quickly. The immune function recovered quickly after the operation; consequently, patients recovered quicker. Furthermore, manually completing the small intestine-small intestine end-to-side anastomosis through the subumbilicus hole saves on the cost of expensive materials such as anastomotic instruments, reducing the financial burden on patients, and is better suited to the economic conditions of patients in China. The limitations of this study were that sample size calculation was not done, and the sample size was not large enough.
Conclusion
Compared with laparoscopic-assisted radical gastrectomy, totally laparoscopic radical gastrectomy with modified Roux-en-Y anastomosis is safe and feasible. It has the advantages of being less traumatic, and resulting in less blood loss and pain, offering short-term benefits.
Footnotes
Acknowledgments
We are particularly grateful to all the people who have given us help on our article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
