Abstract
Objective:
Long non-coding RNAs (lncRNAs) play a critical role in tumorigenesis. Upregulation of lncRNA deleted in lymphocytic leukemia 1 (DLEU1) has been reported in endometrial cancer (EC) tissues. This prospective study aimed to determine the potential clinical significance of serum lncRNA DLEU1 in EC.
Methods:
The serum lncRNA DLEU1 level was detected in EC patients, patients with endometrial hyperplasia and healthy controls by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). Then its clinical value in EC was further evaluated.
Results:
Our results demonstrated that serum lncRNA DLEU1 levels were significantly increased in patients with EC, and serum lncRNA DLEU1 showed good performance for discriminating EC patients from patients with endometrial hyperplasia and healthy controls. In addition, EC patients with advanced clinicopathological features had higher circulating lncRNA DLEU1 level than those with favorable clinical characteristics. Moreover, EC patients in the high serum lncRNA DLEU1 group suffered worse overall survival and disease-free survival than those in the low serum lncRNA DLEU1 group. Furthermore, multivariate cox regression analysis displayed that the serum lncRNA DLEU1 served as an independent prognostic factor for EC.
Conclusions:
Collectively, our study suggests that serum lncRNA DLEU1 is a novel and promising biomarker for prognostic estimation of EC.
Introduction
Endometrial cancer (EC) is the second most common gynecological cancer in China and the fourth most prevalent malignancy among women. 1 In 2019, it is estimated that 61,880 newly diagnosed EC cases and approximately 12,160 deaths were reported in United States. 2 The primary therapeutic strategy for treating EC is surgery, followed by radiotherapy and chemotherapy. 3 Although the prognosis of EC patients is relatively favorable at the early stage, patients with advanced-stage EC often suffer a dismal median survival of less than 1 year. 4,5 Prognostic biomarkers can provide valuable information for identifying the cancer patients with different outcome risks, which will facilitate to tailor appropriate therapy for individual patients and significantly improve the clinical outcome. Therefore, it is of great importance to develop novel and robust biomarkers for accurately and effectively predicting the prognosis of EC.
Long non-coding RNAs (lncRNAs) are highly conserved RNA molecules with a length of more than 200 nucleotides that lack of protein encoding capacity. 6,7 LncRNAs are important for regulating a wide range of biological processes such as proliferation, migration, survival and apoptosis. 8,9 Accumulating evidence has suggested that abnormal expression of lncRNAs plays critical roles in the initiation and progression of human malignancies, including EC. 10,11 For instance, the expression level of lncRNA CHL1-AS1 was significantly upregulated in EC cells. In addition, downregulation of lncRNA CHL1-AS1 suppressed the proliferation and migration capacities of EC cells by sponging miR-6076. 12 Similarly, lncRNA TDRG1 was overexpressed in EC tissues. Ectopic expression of lncRNA TDRG1 promoted cell viability, invasion and migratory capacities as well as suppressed apoptosis in EC cells by regulating vascular endothelial growth factor A. 13
Aberrant expression of lncRNA deleted in lymphocytic leukemia 1 (DLEU1) has been found in many types of cancer such as EC, gastric cancer and glioblastoma. 14 -16 In addition, lncRNA DLEU1 is characterized as an onco-lncRNA involving in the progression of EC. 14
However, the potential prognostic value of circulating lncRNA DLEU1 in EC is unclear. In this study, the expression of serum lncRNA DLEU1 was examined in patients with EC Additionally, the clinical significance of serum lncRNA DLEU1 in EC was further investigated.
Materials and Methods
Patients Information
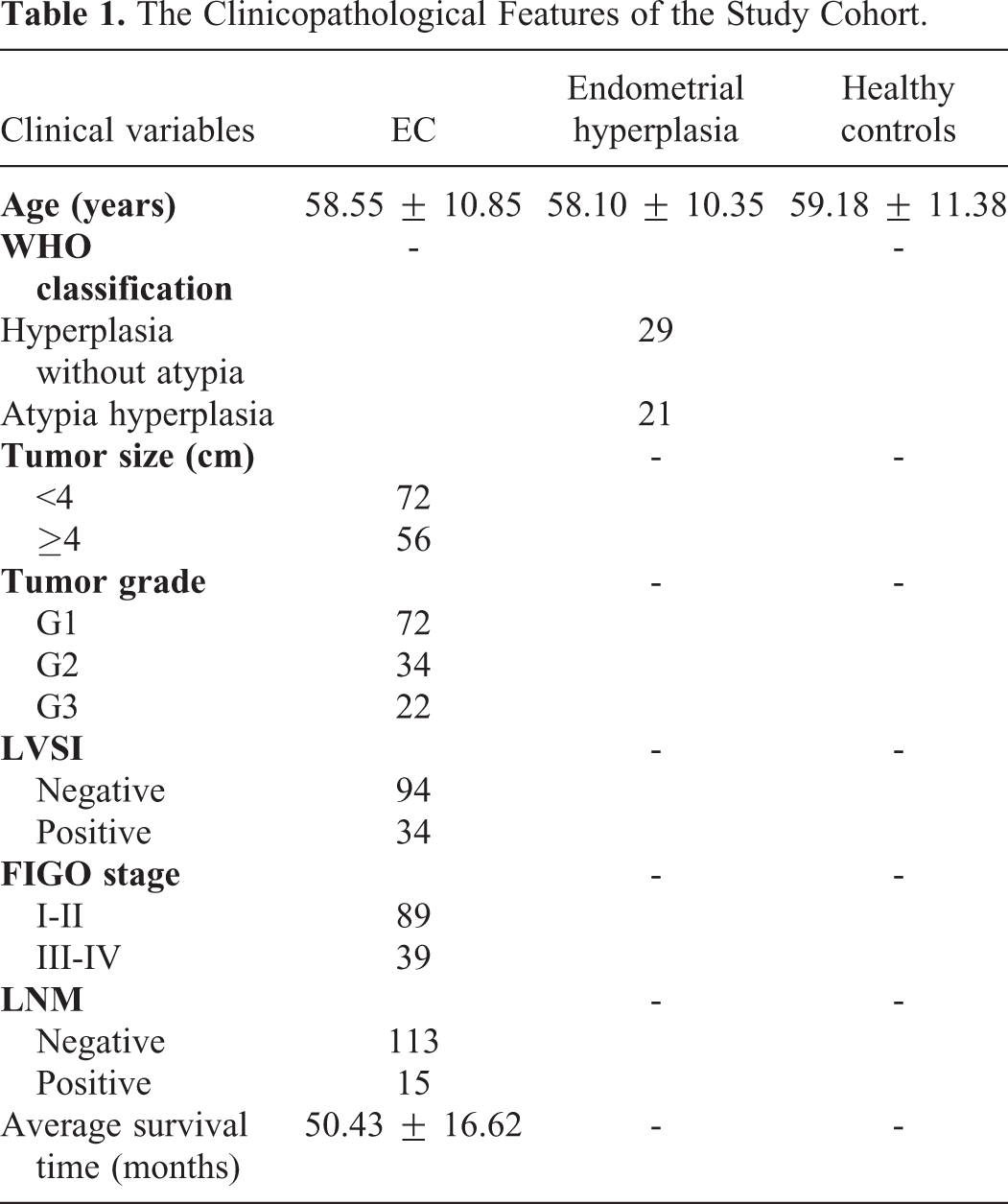
A total of 128 consecutively and newly diagnosed EC patients were recruited in this prospective study. The exclusion criteria were as follows: (1) EC patients unappropriated for surgical treatment, (2) presence of a secondary malignancy, (3) presence of systemic diseases such as multiple organ dysfunction syndrome and end stage renal disease, (4) patients who have received any treatment for the EC earlier. The EC was staged based on the International Federation of Gynecology and Obstetrics (FIGO) system criteria. For the histological subtypes, there were 107 cases with endometrioid carcinoma, 16 cases with serous carcinoma and 5 cases with clear cell carcinoma. Fifty patients with endometrial hyperplasia (29 cases with hyperplasia without atypia, and 21 cases with atypia hyperplasia) and 50 age matched healthy women were included as controls. The study was approved by Research and Ethical Committee of Shijiazhuang NO.1 Hospital. Written informed consent was obtained from all patients and healthy volunteers. The clinical characteristics of the study cohort were summarized in Table 1.
The Clinicopathological Features of the Study Cohort.
Sample Collection
Blood samples were collected from EC patients before receiving any treatment. To thoroughly remove cell debris, the blood samples from all participants were centrifuged at 3000 × g for 15 min at 4 °C, followed by 10,000 × g for 15 min at 4 °C. The serum samples were stored at -80 °C for further use.
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
Total RNA was extracted from serum samples using TRIzol LS Reagent (Invitrogen, Carlsbad, CA, USA). RNA was then reverse transcribed into complementary DNA (cDNA) using a PrimeScript® RT Reagent Kit (Takara, Dalian, China). The amplification and quantification of cDNA was performed with SYBR Green SuperMix reagent (Takara) on an ABI Prism 7500 sequence detection system (Applied Biosystems, Foster City, CA, USA). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the standard internal control. The relative level of circulating lncRNA DLEU1 was calculated using the 2−ΔΔCt method. The primers used for qPCR analyses were as follows: lncRNA DLEU1 forward primer 5′-TCAGAATGCCGACTCTATGCT-3′, reverse primer 5′-GGTGAGGACAGAGT-TAAACGC-3′; GAPDH forward primer, 5′-AGCCACATCGCTCAGACAC-3′ and reverse primer, 5′-GCCCAATACGACCAAATCC-3′.
Detection of Tumor Marker CA125
The expression levels of serum CA125 in EC patients were measured using a commercially available ELISA kit (Abcam, Cambridge, UK) according to the manufacturer’s protocol.
Statistical Analysis
Statistical analyses were performed with GraphPad Prism 8.0 (GraphPad Software, Inc., San Diego, CA, USA) and MedCalc (version 9.3.9.0; Mariakerke, Belgium). The expression levels of circulating lncRNA DLEU1 in different groups were compared using Mann-Whitney U test or Kruskal-Wallis test. Receiver operating characteristic (ROC) curve and the area under the ROC curve (AUC) was used for evaluating the diagnostic performance of circulating lncRNA DLEU1 for EC. The differences in 5-year overall survival (OS) and disease-free survival (DFS) were analyzed using the Kaplan-Meier method combined with a log-rank test. OS was defined as the duration of patient survival from the time of treatment initiation. DFS was defined as the time elapsed between treatment initiation and tumor progression or death from any cause. Univariate and multivariate analyses were performed to determine the independent prognostic factors affecting OS. All the statistical tests were 2 sided, and P < 0.05 was considered statistically significant.
Results
Serum lncRNA DLEU1 Was Significantly Increased in EC
As shown in Figure 1A, the levels of serum lncRNA DLEU1 were significantly increased in EC patients compared to healthy donors (***P < 0.001) as well as patients with endometrial hyperplasia (***P < 0.001). In addition, the expression level of circulating lncRNA DLEU1 was higher in patients with endometrial hyperplasia than in healthy controls (**P < 0.01). Interestingly, for the patients with endometrial hyperplasia, the cases with atypia hyperplasia had higher levels of serum lncRNA DLEU1 than those without atypia hyperplasia (**P < 0.01) (Figure 1B).
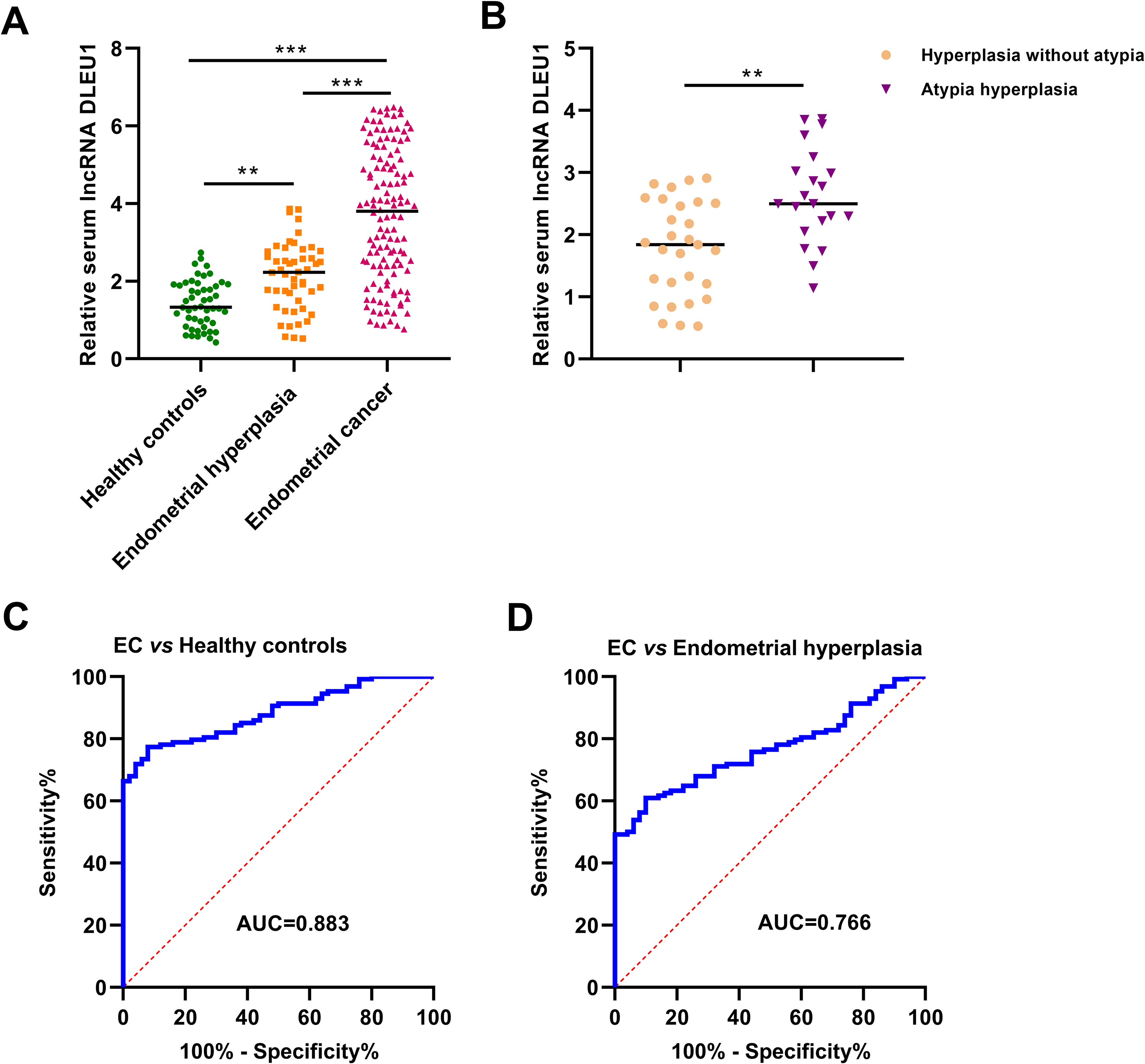
Serum lncRNA DLEU1 was significantly increased in EC. (A) The expression level of serum lncRNA DLEU1 was progressively increased from healthy controls, patients with endometrial hyperplasia to EC. (B) Serum lncRNA DLEU1 levels were higher in patients with atypia hyperplasia than in those with hyperplasia without atypia. (C) The diagnostic performance of serum lncRNA DLEU1 for identifying EC patients from healthy controls. (D) The diagnostic performance of serum lncRNA DLEU1 for identifying EC patients from patients with endometrial hyperplasia.
ROC curves were constructed to evaluate the diagnostic accuracy of circulating lncRNA DLEU1 for EC. The AUC value for discriminating EC patients from healthy controls was 0.883 (95% CI = 0.826-0.926). The sensitivity and specificity were 77.3% and 92.0%, respectively (Figure 1C). The AUC value for identifying EC patients from patients with endometrial hyperplasia was 0.766 (95% CI = 0.697-0.826), with 60.9% sensitivity and 90.0% specificity (Figure 1D).
The Association Between Serum lncRNA DLEU1 Levels and Clinicopathological Parameters of EC
No significant association was found between serum lncRNA DLEU1 level and age, tumor size as well as CA125 level (Figure 2A-C). As shown in Figure 2D to 2H, serum lncRNA DLEU1 levels were significantly higher in EC patients with serous carcinoma (serous carcinoma vs endometrioid carcinoma, P < 0.001), higher tumor grade (G3 vs G1, P < 0.001; G3 vs G2, P 0 .034), positive lymphovascular space invasion (LVSI) (P = 0.001), advanced FIGO stage (P < 0.001), and positive lymph node metastasis (LNM) (P < 0.001).

The association between serum lncRNA DLEU1 levels and clinicopathological parameters of EC. (AC) No significant correlation was found between serum lncRNA DLEU1 level and age, tumor size and CA125 level. (D-H) EC patients with aggressive clinical features (serous carcinoma, higher tumor grade, positive LVSI, positive FIGO stage, and positive LNM) had higher serum lncRNA DLEU1 levels than their respective controls.
EC Patients With Higher Serum lncRNA DLEU1 Levels Suffered Worse OS and DFS
The median value of serum lncRNA DLEU1 levels in EC patients was used as the cut-off point to divide the EC patients into high serum lncRNA DLEU1 group and low serum lncRNA DLEU1 group. The survival analysis showed that the EC patients in the high serum lncRNA DLEU1 group had significantly shorter OS than those in the low serum lncRNA DLEU1 group (P < 0.001) (Figure 3A). In addition, high levels of circulating lncRNA DLEU1 were strongly associated with shorter DFS (P < 0.001) (Figure 3B).

The association between serum lncRNA DLEU1 levels and survival of EC. (A) The EC patients in the high serum lncRNA DLEU1 group had worse OS than those in the low serum lncRNA DLEU1 group. (B) Similarly, the EC patients in the high serum lncRNA DLEU1 group had significantly shorter OS than those in the low serum lncRNA DLEU1 group.
Serum lncRNA DLEU1 Was an Independent Prognostic Factor for EC
Univariate analysis showed that tumor grade (P < 0.001, HR = 2.725, 95% CI = 1.879-3.951), LVSI (P < 0.001, HR = 5.088, 95% CI = 2.748-9.422), FIGO stage (P < 0.001, HR = 9.778, 95% CI = 4.958-19.284), lymph node metastasis (P < 0.001, HR = 4.844, 95% CI = 2.495-9.404) and serum lncRNA DLEU1 (P < 0.001, HR = 4.649, 95% CI = 2.222-9.727) were significantly associated with OS of EC (Figure 4A). Multivariate analysis revealed that FIGO stage (P < 0.001, HR = 6.051, 95% CI = 2.389-15.327) and serum lncRNA DLEU1 (P = 0.003, HR = 3.201, 95% CI = 1.496-6.848) were independent prognostic factors of EC (Figure 4B).

Serum lncRNA DLEU1 level was an independent prognostic factor for EC. (A) Univariate analysis showed that tumor grade, LVSI, FIGO stage, LNM and serum lncRNA DLEU1 level were strongly associated with OS of EC. (B) Multivariate analysis demonstrated that FIGO stage and serum lncRNA DLEU1 level were independent prognostic risk factors for EC.
Discussion
In this study, our results showed that the expression level of serum lncRNA DLEU1 was remarkably increased in patients with EC compared to patients with endometrial hyperplasia and healthy controls. Serum lncRNA DLEU1 accurately discriminated EC patients from endometrial hyperplasia patients and healthy individuals. In addition, EC patients with more aggressive clinicopathological parameters tended to have significantly higher circulating lncRNA DLEU1 levels than those with less aggressive clinical features. Moreover, EC patients in the high serum lncRNA DLEU1 group suffered dramatically worse OS and DFS than those in the low serum lncRNA DLEU1 group. Multivariate analysis showed that serum lncRNA DLEU1 was an independent prognostic factor for OS of EC. In summary, serum lncRNA DLEU1 might be a promising biomarker for predicting the development of precancerous conditions and prognosticating the clinical outcome of EC.
Accumulative evidence has demonstrated that lncRNAs are stably present in serum, plasma and other bodily fluids and protected from degradation by endogenous RNase. 17 These characteristics enable them to become promising biomarkers for the diagnosis and prognosis prediction of different types of malignancies as well as for monitoring therapeutic responses. For instance, circulating MALAT-1 level was markedly increased in patients with prostate cancer (PCa) and exhibited great potential for PCa detection. 18 Similarly, independent reports demonstrating consistent results that the expression level of circulating H19 was significantly higher in gastric cancer patients than in healthy controls. 19,20
To the best of our knowledge, this is the first study to explore the expression pattern and clinical significance of circulating lncRNA DLEU1 in EC. One possible reason accounting for the increased serum lncRNA DLEU1 levels in patients with EC is that the tumor cells derived from EC synthesize and secrete much more lncRNA DLEU1 into the circulatory system than the normal endometrial cells, leading to the upregulation of serum lncRNA DLEU1 in EC patients. It should be noted that a portion of the circulating lncRNA DLEU1 might be from extracellular vesicles or exosomes released by cells. However, currently, the mechanism responsible for the release of lncRNA into bodily fluids and extracellular environment remains poorly understood.
Consistent with our findings, lncRNA DLEU1 was highly expressed in EC tissues compared to normal endometrium. Overexpression of lncRNA DLEU1 significantly increased the proliferation and invasion of EC cells and suppressed cell apoptosis, indicating that lncRNA DLEU1 plays a tumor promoting role in EC. 14 Similarly, lncRNA DLEU1 was demonstrated to promote the EC progression by sponging miR-490 to regulate SP1 expression. 21 In addition to EC, lncRNA DLEU1 is also closely involving in the tumorigenesis of many cancer types. For instance, lncRNA DLEU1 was significantly increased in clear cell renal cell carcinoma (ccRCC) tissues compared to the adjacent normal tissues. Knockdown of lncRNA DLEU1 inhibited proliferative and migratory capacities of cancer cells. 22 Similarly, lncRNA DLEU1 was overexpressed in breast cancer. Overexpression of lncRNA DLEU1 promoted the malignant activities of breast cancer cells, and opposite trends of cellular behaviors were observed when lncRNA DLEU1 was suppressed. 23 LncRNA DLEU1 was highly expressed in most of the oral squamous cell carcinoma (OSCC) tissues, and high lncRNA DLEU1 expression was strongly associated with shorter overall survival. Additionally, downregulation of lncRNA DLEU1 inhibited the oncogenic behaviors of OSCC cells both in vitro and in vivo. 24 Interestingly, lncRNA DLEU1 might also acted as a tumor suppressor. Enforced expression of lncRNA DLEU1 suppressed the tumorigenicity of Burkitt lymphoma cells, and vice versa. 25 These opposite findings of lncRNA DLEU1 in different types of cancer suggests that the specific role of lncRNA DLEU1 in tumorigenesis might depend on the tumor type and the downstream targets it regulates.
One limitation of our current study was the relatively small sample size. Multicenter studies with larger sample size are warranted to confirm our findings. In addition, whether abnormally expressed circulating lncRNA DLEU1 is also observed in other malignancies or human diseases remains unknown. Circulating lncRNA DLEU1 was an independent prognostic factor for EC, indicating it is critical for the tumorigenesis of EC. Therefore, the underlying mechanisms for the tumor promoting role of lncRNA DLEU1 in EC needs deeper investigation, and lncRNA DLEU1 may be a potential therapeutic target for EC.
Conclusion
In this study, we have illustrated that serum lncRNA DLEU1 is progressively increased from healthy donors, patients with endometrial hyperplasia and EC patients, indicating that lncRNA DLEU1 might play critical role in transforming the premalignant endometrial lesion into EC. In addition, upregulation of serum lncRNA DLEU1 is obviously associated with unfavorable prognosis of EC. Taken together, serum lncRNA DLEU1 might serve as an effective biomarker for predicting the development of precancerous conditions and stratifying the EC patients with different risk outcome, and thus contribute to improving the patients’ management and treatment effect.
Supplemental Material
Supplemental Material, DFS_data - Upregulation of Serum lncRNA DLEU1 Predicts Progression of Premalignant Endometrial Lesion and Unfavorable Clinical Outcome of Endometrial Cancer
Supplemental Material, DFS_data for Upregulation of Serum lncRNA DLEU1 Predicts Progression of Premalignant Endometrial Lesion and Unfavorable Clinical Outcome of Endometrial Cancer by Lixia Shan, Tao Zhao and Yu Wang in Technology in Cancer Research & Treatment
Supplemental Material
Supplemental Material, Survival_data - Upregulation of Serum lncRNA DLEU1 Predicts Progression of Premalignant Endometrial Lesion and Unfavorable Clinical Outcome of Endometrial Cancer
Supplemental Material, Survival_data for Upregulation of Serum lncRNA DLEU1 Predicts Progression of Premalignant Endometrial Lesion and Unfavorable Clinical Outcome of Endometrial Cancer by Lixia Shan, Tao Zhao and Yu Wang in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
The study was approved by the Research and Ethical Committee of Shijiazhuang NO.1 Hospital (approval number: 20160055).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
