Abstract
Melanoma is the main death cause of human skin cancer. Increasing evidences demonstrate that microRNAs act as key roles in mediating tumor occurrence and progression. MiR-508-5p has proved to participate in the development of various types of human malignancies. However, the role of miR-508-5p in melanoma remained unclear. In in vitro study, miR-508-5p level in peripheral blood samples of patients with melanoma and human melanoma A375 cells was downregulated compared to that in normal peripheral blood samples or normal human epidermal melanocytes (MHEM). MiR-508-5p overexpression significantly inhibited the cell proliferation, migration and invasion in A375 cells, and thus inhibiting KIT expression at both gene and protein levels. Furthermore, western blot analysis showed miR-508-5p reduced cell proliferation by targeting KIT to modulate RAS/RAF/MEK/ERK pathway. Taken together, we speculated that miR-508-5p functioned as an important suppressor in human melanoma by targeting KIT, suggesting miR-508-5p might be a promising tumor suppressor gene for further target therapies from bench to clinic.
Introduction
Ranked as the leading cause of human skin cancer death worldwide, human melanoma is formed by malignant transformation of melanocytes, 1 accounting for about 5% of all skin cancers, with a mortality rate exceeded 80%. 2 Traditional therapeutic strategies targeting melanoma, such as surgery, radiation therapy and chemotherapy, could not obtain satisfied prognosis. Although great improvements have been achieved in immunotherapy and targeted therapy, current treatments are stymied due to drug resistance and occasional recurrences. 3 Therefore, it is urgent to investigate the molecular pathogenesis of melanoma and screen effective prognostic biomarkers for the treatment of melanoma.
Micro RNAs (miRNAs) are short non-coding RNA molecules containing ∼21-25 nucleotides in length. It is reported miRNAs play important roles in regulation of post-transcriptional gene expressions in animals and plants. 4 Mature miRNAs are actively involved in cell proliferation, migration, apoptosis, metabolism and other cellular responses of every subtype of human cancers, such as melanoma. 5 Therefore, it is promising to explore mechanisms of miRNAs with the aim to provide insights into the pathogenesis and to develop novel tumor targets for melanoma treatment. Recently, miR-508-5p was found to be associated with various diseases including glioma, 6 gastric cancer, 7 hepatocellular carcinoma 8 and chronic heart failure. 9 In addition, miR-508-5p has been proved to negatively modulate the tumor initiation, invasion and metastasis. 10 However, the specific functions and potential regulatory mechanisms of miR-508-5p in melanoma still require further investigations.
Also known as c-kit or CD117, KIT is a type III receptor tyrosine kinase (RTK) encoded by the KIT gene in human. 11 As a receptor tyrosine kinase, KIT functions in regulation processes of cell proliferation, migration, stem cell maintenance, differentiation as well as the occurrence of several cancers, such as leukemia, 12 melanoma, 13 breast cancer 14 and thyroid carcinoma, 15 suggesting KIT might be an important tumor-promoting factor that associated with metastasis and overall poor prognosis. It is reported that binding between KIT and corresponding target mRNAs, such as miR-137, 16 miR-221 17 and miR-155 18 would result in the suppression of growth and metastasis in various cancers. However, whether binding between KIT and miR-508-5p participates in progression of melanoma is still unclear.
In this study, we found miR-508-5p was down-regulated in melanoma cells and in peripheral blood from melanoma patients, which was correlated with clinical characteristics and prognosis of melanoma patients. Our results demonstrated KIT was a promising direct target of miR-508-5p in patients with melanoma and miR-508-5p overexpression reduced tumor proliferation, migration, and invasion by targeting KIT in vitro. Additionally, miR-508-5p was proved to inhibit the cell proliferation by targeting KIT via Ras-Raf-MEK-ERK signaling pathways. In conclusion, miR-508-5p functions in preventing the human melanoma progression by mediating KIT- Ras-Raf-MEK -ERK axis, shedding lights on miR-508-5p as a novel therapeutic target for human melanoma from bench to clinic.
Materials and Methods
Collection of Clinical Peripheral Blood Samples
The peripheral blood samples from healthy people (n = 26) and patients with melanoma (n = 68) were separately collected into pyrogen-free ethylene diamine tetraacetic acid (EDTA) tubes. All the participants involved in this project agreed to sign informed consent. The examination and the informed consents were approved by Ethics Committee of Shenzhen People’s Hospital (Approval no. LL-KY-201935), and there is no contrary to World Medical Association Declaration of Helsinki. Peripheral blood samples (3 mL) were collected into Paxgene RNA preparation tubes and stored at -80°C.
Cell Culture and Transfections
Normal human epidermal melanocytes (NHEM) and human melanoma cells (A375) were obtained from ATCC (American type culture collection, Manassas, VA, USA). NHEM and A375 cells were cultured in DMEM medium (Gibco, Rockville, MD, USA) supplemented with 10% of FBS (Gibco, NY, USA Bio-Rad, CA, USA) and 1% penicillin (100 μg/mL) / streptomycin (100 U/mL), and maintained at 37°C under an atmosphere of 5% CO2. MiR-508-5p mimic and negative control (NC mimic), as well as miR-508-5p inhibitor (miR-508-5p inh) and matched negative control (NC inh) were synthesized by Gene Pharmaceutic company in Shanghai, China. pcDNA3.1-KIT plasmid was constructed in this study by inserting ORF (open reading frame) of KIT onto a pcDNA3.1 empty plasmid. Transfections were performed and completed by using Lipofectamine 2000 reagents (Invitrogen, Carlsbad, CA, U.S.A.) according to standard instructions.
RNA Extraction and Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
Total RNAs were extracted from patients’ peripheral blood samples and cultured cell lines using TRIzol reagent (Thermo Fisher Scientific, Inc., MA, USA), and then converted into cDNA using RevertAid RT Reverse Transcription kit (Thermo Fisher Scientific, Inc., MA, USA) according to manufacturer’s instructions. mRNA levels were measured by qRT-PCR using the Standard SYBR-Green RT-PCR kit (Takara Bio, Inc., Otsu, Japan) in accordance with the standard protocols. GAPDH was used as an internal control. All miRNA-related reagents used in this study were purchased from RiboBio Co., Ltd (Guangzhou, China). Primers were designed (Sangon, Shanghai, China) as follows: miR-508-5p-forward: 5’-ACACTCCAGCTGGGTACTCCAGAGGGCGTC ACT-3’; miR-508-5p-reverse: 5’-TGGTGTCGTGGAGTCG-3’. KIT-forward: 5’-AACAACAAAGAGCAAATCCAGG-3’; KIT-reverse: 5’-GGAAGTTGCGTCGG GTCTAT-3’. GAPDH-forward: 5’-AGACACCATGGGGAAGGTGAA-3’; GAPDH-reverse: 5’-ATTGCTGATGATCTTGAGGCTG-3’. 2-ΔΔCT formula was employed to calculate the relative expression levels of target genes after normalization to GAPDH small nuclear RNA.
Cell Proliferation Assay
Cell proliferation rate was assessed by using 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay according to the standard protocol. MTT was purchased from Dojindo Chemical Reagent Company (Japan). 2*10 3 A375 cells were seeded in a 96-well plate and incubated for 24 hr. Then A375 cells were transfected with NC/NC inhibitor (NC inh)/miR-508-5p inhibitor (miR-508-5p inh)/NC mimic/miR-508-5p mimic as indicated. The blank control was set up using medium only. 48 hr post-transfection, cells were incubated with 20 µL of MTT (5 mg/mL) for 4 hr at 37°C, followed by the addition of 150 µL of DMSO and shaking for 15 min at room temperature to solubilize the crystals. The optical density (OD) was determined using a microplate reader (Model 680 [BIO-RAD]) at a wavelength of 490 nm.
Colony Formation Assay
5*10 2 A375 cells were separately seeded in each well in 6-well plates. After incubated for 14 days in DMEM medium containing 10% FBS in a 37°C atmosphere of 5% CO2, colonies were fixed and stained using methanol and 0.1% crystal violet in 20% methanol for 15 min. Finally, cell number was calculated under a light microscope (Laica, Germany).
Cell Migration and Invasion Assays
Cell migration was estimated by wound healing assay. A375 cells were seeded in 6-well plates with an amount of 5*10 5 per well. 24 hr post-seeding, a micropipette tip was used to remove the cell monolayer, followed by phosphate-buffered saline (PBS) wash.
Then a plastic scriber was applied to generate a wound. Cell monolayer was subsequently incubated in serum-free medium for 48 hr. Finally, gap distance was assessed under a light microscope (Laica, Germany). Each condition was repeated in triplet, and repeated 3 times.
Cell invasion ability was estimated by transwell assay. Firstly, melanoma cells were planted into the upper chamber of transwell chambers coated with/without Matrigel matrix (BD Biosciences, San Jose, CA, USA). RPMI1640 medium containing 10% FBS, as a chemoattractant, was added into the bottom chamber. Then, the chamber was removed, followed by 24 hr incubation in a 37°C atmosphere of 5% CO2. Matrix glue and cells from the upper chamber were gently removed with a wet cotton swab. Then cells were fixed by using 4% paraformaldehyde and stained with crystal violet. 5 visual fields were randomly selected under a light microscope to calculate the cell number.
Luciferase Reporter Assay
To confirm the binding between miR-508-5p and KIT, luciferase reporter assay was performed in HEK-293 T cells. pmirGLO-KIT-3′UTR wild type (KIT-WT) and pmirGLO-KIT-3′UTR mutant (KIT-MUT) were designed and constructed by Sangon (Shanghai, China). HEK-293 T cells were seeded in 12-well plates with an amount of 1.5*105 per well. Then, 30 nM miR-508-5p mimic or NC-mimic was co-transfected with 300 ng DNA (KIT-WT or KIT-MUT) into HEK-293 T cells using Lipofectamine 2000 (Invitrogen) in accordance with the manufacturer’s instructions. 24 hr post-transfection, relative luciferase activity was assessed with using Dual- Luciferase Reporter Assay System (Promega, Madison, WI, USA).
Western Blot Analysis
RIPA Buffer (9800, Cell Signaling, Danvers, MA) was used to lyse and extract total proteins from A375 cells. After the centrifugion, protein samples were separated by SDS-PAGE to isolate the supernatant fractions. Subsequently, proteins were transferred onto the polyvinylidene fluoride (PVDF) membranes and blocked with 5% milk for 1 hr. Next, PVDF membrane was incubated with the following primary immunoblotting antibodies: anti-KIT(1:1000 dilution), anti-K-Ras (1:1000 dilution), anti-c-Raf (1:1000 dilution), anti-p-MEK (1:1000 dilution), anti-MEK (1:1000 dilution), anti-p-ERK (1:1000 dilution), anti-ERK (1:1000 dilution) and anti-GAPDH (1:1000 dilution) (Abcam, Cambridge, UK) at 37°C for 2 hr. Then, PVDF membranes were washed for at least 3-5 times and co-incubated with HRP-conjugated secondary antibody for 1 hr at room temperature. ECL detection reagents from Amersham Pharmacia Biotech, (Tokyo, Japan) were used to visualize immunocomplexes. Image Pro software was applied to calculate the intensity of protein bands.
Statistical Analysis
All experiments performed in this project were repeated for 3 times and the data was calculated using average value. SPSS 19.0 statistical software was used to analyze the data and were presented as mean ±SD (standard deviation). Difference between the 2 groups was tested by independent sample t test as well as Pearson’s correlation coefficient was also used. A value of P < 0.05 was considered statistically significant in this study. (*P < 0.05, **P < 0.01, ***P < 0.001).
Results
Decreased miR-508-5p Level in Peripheral Blood of Melanoma Patients and Melanoma Cells
In this study, qRT-PCR was performed to assess the expression difference of miR-508-5p levels between melanoma patients and healthy people (Table 1). Results showed that miR-508-5p was significantly down-regulated in peripheral blood of melanoma patients compared to that in control group (Figure 1A, **P < 0.01). We further evaluated miR-508-5p expressions in normal human epidermal melanocytes (NHEM) and human melanoma cells (A375) by qRT-PCR assay. Consistent with results obtained from patients, miR-508-5p level in NHEM cells was obviously higher than that in A375 cells (Figure 1B), suggesting miR-508-5p was inhibited in melanoma in vivo and in vitro.
Relationship Between miR-508-5p Expression Level and Clinicopathologic Features of Melanoma.

P < 0.05 = *;P < 0.001 = **.

Low miR-508-5p level in peripheral blood sample from patients with melanoma and associated melanoma cells. (A) qRT-PCR analysis of expression difference of miR-508-5p between peripheral blood samples from 68 melanoma patients and peripheral blood samples from 26 normal people. (B) qRT-PCR analysis of expression difference of miR-508-5p in normal human epidermal melanocytes (MHEM) and human melanoma cells (A375). (mean ± SD; *P < 0.05, **P < 0.01).
miR-508-5p Overexpression Suppressed Cell Proliferation Ability
To investigate the effect of miR-508-5p on the cell proliferation in human melanoma cells, we generated A375 cell lines that stably expressing either miR-508-5p mimic or inhibitor. Firstly, qRT-PCR assay revealed that A375 cells expressing miR-508-5p mimic displayed higher miR-508-5p level than that of NC-mimic-transfected cells. Moreover, cells expressing miR-508-5p inhibitor exhibited lower miR-508-5p level than that of NC inh-transfected cells (Figure 2A). Interestingly, cell proliferation was subsequently assessed. MTT assay indicated cell proliferation rate decreased significantly in A375 cells expressing miR-508-5p mimic (*P < 0.05). On the contrary, cell proliferation rate could be enhanced by miR-508-5p inhibitor overexpression (miR-508-5p inh) (**P < 0.01) (Figure 2B). Colony formation assay demonstrated cells expressing miR-508-5p mimic showed a reduced colony number. Similarly, miR-508-5p inhibitor (miR-508-5p inh) boosted colony number compared to that of NC-inh group (Figure 2C), suggesting miR-508-5p possessed negative regulation ability in regulating cell proliferation in A375 cells.

miR-508-5p overexpression suppressed the proliferation of human melanoma cells in vitro. (A) qRT-PCR analysis the relative expression levels of miR-508-5p in A375 cells transfected with NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic. (B) MTT assay was used to evaluate the cell proliferation of A375 cells transfected with NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic. (C) Colony formation assays displayed miR-508-5p affected the cell proliferation of A375 cells transfected with NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic and the colony number was quantified after transfection. NC group represents untreated cells. (mean ± SD; *P < 0.05, # P < 0.05, **P < 0.01, ## P < 0.01).
miR-508-5p Inhibited the Cell Migration and Invasion
To identify the role of miR-508-5p in cell migration and invasion of melanoma cells, wound healing and transwell assay were subsequently conducted. Wound healing assay illustrated cells expressing miR-508-5p mimic showed significantly wider wound width. Similarly, miR-508-5p inhibitors increased would healing rate compared to A375 cells expressing NC inhibitor (Figure 3A), suggesting miR-508-5p overexpression decreased the cell migration ability in human melanoma cells. Additionally, cells expressing miR-508-5p mimic showed more invasive colonies. And colony numbers could be restored upon miR-508-5p inhibitor overexpression (Figure 3B), indicating miR-508-5p suppressed cell invasion in vitro.

miR-508-5p inhibited the migration and invasion of human melanoma cells in vitro. (A) Wound healing assays examined the cell migration of A375 cells transfected with NC / NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic. (B) Transwell assay determined the cell invasion in NC / NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic-transfected A375 cells. (mean ± SD; *P < 0.05, # P < 0.05, **P < 0.01, ## P < 0.01).
KIT Is the Direct Target of miR-508-5p in Melanoma
Based on bioinformatics predication using miRanda (http://www.microrna.org/microrna/getGeneForm.do) and TargetScan (http://www.targetscan.org/), we identified KIT gene as a potential target gene of miR-508-5p (Figure 4A). We then used luciferase reporter assays to explore the binding affinity between miR-508-5p and wild type of KIT 3’UTR (KIT-WT) in HEK-293 T cells. Luciferase activity in HEK-293 T cells was significantly upon miR-508-5p mimic expression. Interestingly, miR-508-5p overexpression could not alter the luciferase activity of mutant KIT 3’UTR (KIT-MUT) (Figure 4B). We speculated KIT was a direct target of miR-508-5p. To explore how miR-508-5p regulated KIT expression, qRT-PCR analysis was performed. Results showed KIT mRNA level was significantly increased upon miR-508-5p mimic treatment. Similarly, miR-508-5p inhibitor could remarkably elevated KIT expression in A375 cells, demonstrating miR-508-5p negatively regulated KIT mRNA expression (Figure 4C). Additionally, western blot assay showed KIT protein level was significantly upregulated upon miR-508-5p mimic treatment compared to NC-mimic. Furthermore, miR-508-5p inhibitor could also remarkably increased KIT expression in comparison to NC-inh (Figure 4D), suggesting miR-508-5p might negatively regulate the protein expression of KIT. Taken together, our results strongly suggested KIT might be a downstream target gene of miR-508-5p and the decreased miR-508-5p level directly elevates KIT level in melanoma.

KIT is the direct target of miR-508-5p. (A) Sequences of miR-508-5p binding sites complemented with KIT 3’UTR (1654-1660). KIT-WT represents the entire 3’UTR sequences of wild-type KIT, while KIT-MUT represented the binding sites were mutated. (B) Luciferase reporter assays was performed in A375 cells to detect the binding affinity of miR-508-5p to the KIT-WT or KIT-MUT. (C) qRT-PCR analysis the relative mRNA expression levels of KIT in NC / miR-508-5p NC / NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic-transfected A375 cells. (D) Western blot analysis the relative protein expression levels of KIT in NC / miR-508-5p NC / NC inhibitor (NC inh) / miR-508-5p inhibitor (miR-508-5p inh) / NC mimic / miR-508-5p mimic-transfected A375 cells. (mean ± SD; *P < 0.05, # P < 0.05, **P < 0.01, ## P < 0.01).
Interplay of miR-508-5p and KIT Regulated Proliferation, Migration and Invasion of Human Melanoma Cells
To explore the role of interaction between miR-508-5p and KIT in human melanoma, A375 cells that stably expressing KIT plasmid, miR-508-5p mimic or miR-508-5p inhibitor were generated. qRT-PCR analysis showed KIT mRNA level was significantly higher in A375 cells expressing KIT plasmids than that in untransfected A375 cells (**P< 0.01), and miR-508-5p mimic could significantly inhibit KIT expression (**P < 0.01). Furthermore, co-expression of miR-508-5p mimic and KIT boosted KIT expression in comparison to miR-508-5p mimic group. It is worth mentioning that KIT level in co-expression group was still lower than that in KIT overexpressing group (Figure 5A). Western blot assay displayed a similar trend compared to qRT-PCR results, illustrating miR-508-5p negatively regulated the mRNA and protein levels of KIT (Figure 5B). The interplay of miR-508-5p and KIT in regulating the cell proliferation was evalutaed by MTT and colony formation assay. MTT assay showed cell proliferation rate in cells expressing KIT was increased significantly compared to that in untreated group (*P < 0.05). On the contrary, cell proliferation rate in A375 cells expressing miR-508-5p mimic was significantly decreased (*P < 0.05). Co-expression of miR-508-5p mimic and KIT restored cell proliferation rate compared to miR-508-5p mimic treatment only. However, cell proliferation rate was still lower than that in cells expressing KIT group (Figure 5C). Combined, these results showed miR-508-5p negatively regulated cell proliferation in human melanoma A375 cells. Similarly, colony formation assay showed colony number in cells expressing KIT was significantly higher than that in untransfected group (**P < 0.01). Additionally, colonies in A375 cells expressing miR-508-5p mimic was significantly reduced (**P < 0.01). Furthermore, co-transfection of miR-508-5p mimic and KIT could elevated colony number compared to miR-508-5p mimic only, which was still less than that in KIT overexpression group (**P < 0.01) (Figure 5D). Wound healing assay implicated miR-508-5p overexpression significantly inhibited wound healing in A375 cells, which could be reversed by KIT expression, demonstrating miR-508-5p overexpression suppressed cell migration by targeting KIT (Figure 5E). Meanwhile, transwell assay displayed miR-508-5p overexpression significantly decreased the invasive cell number, which could also be restored by additional KIT treatment, suggesting miR-508-5p reduced melanoma cell migration and invasion in cell model by targeting KIT (Figure 5F). Taken together, we concluded that interaction between miR-508-5p and KIT regulated cell proliferation, migration and invasion of human melanoma cells in vitro.

The interplay of miR-508-5p and KIT in regulating the migration and invasion of human melanoma cells in vitro. (A) qRT-PCR analysis the relative mRNA expression levels of KIT in KIT mimic (KIT) and / or miR-508-5p mimic-transfected A375 cells. (B) Western blot analysis the relative protein expression levels of KIT in KIT mimic (KIT) and / or miR-508-5p mimic-transfected A375 cells. (C) MTT assay was used to evaluate the cell proliferation of A375 cells transfected with KIT mimic (KIT) and / or miR-508-5p mimic. (D) Colony formation assays displayed the cell proliferation of A375 cells transfected with (NC mimic + vector) / (NC mimic + KIT) / (miR-508-5p mimic + vector) / (miR-508-5p mimic + KIT) and the colony number was quantified after transfection. (E) Wound healing assays examined the cell migration of A375 cells transfected with (NC mimic + vector) / (NC mimic + KIT) / (miR-508-5p mimic + vector) / (miR-508-5p mimic + KIT). (F) Transwell assay determined the cell invasion in (NC mimic + vector) / (NC mimic + KIT) / (miR-508-5p mimic + vector) / (miR-508-5p mimic + KIT) -transfected A375 cells. (mean ± SD; *P < 0.05, # P < 0.05, **P < 0.01, ## P < 0.01).
miR-508-5p Inhibited Cell Proliferation by Targeting KIT to Modulate Ras-Raf-MEK-ERK Signaling
Ras-Raf-MEK-ERK signaling has been reported to play an important role in controlling the cell proliferation, migration, survival and differentiation in human melanoma. Previous study reported that microRNAs acted as inhibitors to suppress the tumor progression by blocking Ras-Raf-MEK-ERK signaling pathway. We then explored the relationship between miR-508-5p and Ras-Raf-MEK-ERK signaling. Western blot analysis revealed there were significant up-regulations of KIT, K-Ras, c-Raf, p-MEK and p-ERK in A375 cells expressing KIT plasmid. However, obvious down-regulations of KIT, K-Ras, c-Raf, p-MEK and p-ERK in A375 cells expressing miR-508-5p mimic were also observed. In addition, KIT, K-Ras, c-Raf, p-MEK and p-ERK levels were rescued by co-expression of KIT and miR-508-5p mimic (Figure 6). In summary, miR-508-5p suppressed the melanoma cell proliferation, migration and invasion by targeting KIT via Ras-Raf-MEK-ERK pathway.
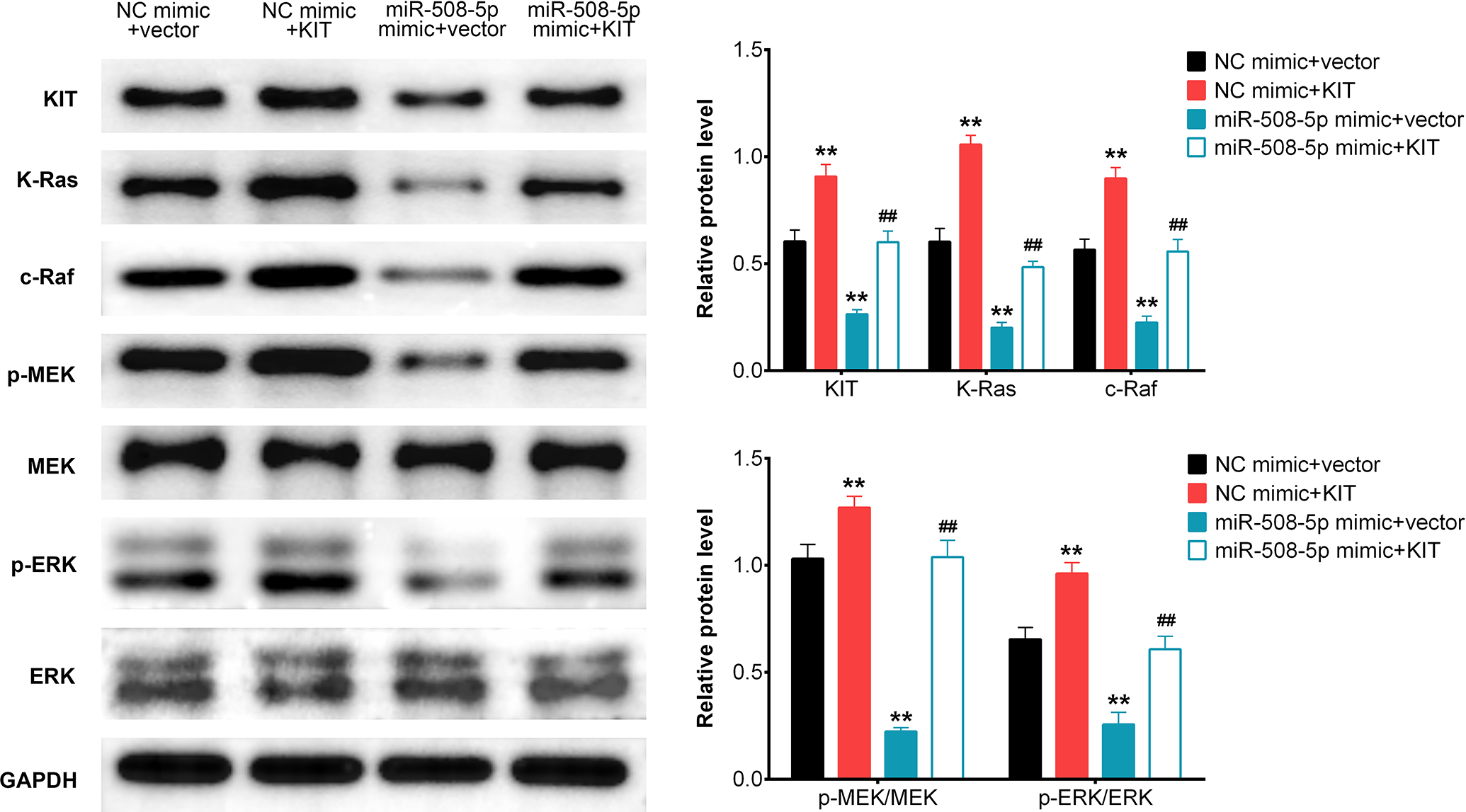
MiR-508-5p inhibited the cell proliferation by targeting KIT to modulate RAS/RAF/MEK/ERK signaling pathway. Western blot analysis the relative protein expression levels of KIT / K-Ras / c-Raf / p-MEK / MEK / p-ERK / ERK in A375 cells transfected with (NC mimic + vector) / (NC mimic + KIT) / (miR-508-5p mimic + vector) / (miR-508-5p mimic + KIT). (mean ± SD; *P < 0.05, # P < 0.05, **P < 0.01, ## P < 0.01).
Discussion
Melanoma is the most dangerous type of skin cancer. Despite the 5-year survival rate of patients with melanoma has improved greatly in recent years due to the combination of surgery, radiotherapy and chemotherapy, the prognosis of patients with invasive melanoma remains poor. 19 Due to lack of obvious early symptoms and precise tumor markers, the majority of patients with melanoma cannot be diagnosed until the middle or late stages. 20 Therefore, it is urgent to identify molecular targets as well as to further explore the pathogenesis of melanoma with the aim to develop novel therapeutic strategies.
Accumulating evidences have reported that aberrant expression of miRNAs is closely related to occurrence, diagnosis, development, and staging of various human diseases such as spinal cord injury, 21 cervical cancer 22 and human melanoma. 23 Previous studies showed the involvement of abnormally expressed miR-508-5p in regulating tumor progression in several types of cancer, including the melanoma, 24 indicating miR-508-5p could be a useful target for the diagnosis, prevention and treatment of melanoma. In this study, we found miR-508-5p levels were down-regulated in peripheral blood sample from patients with melanoma and in melanoma cell models, indicating miR-508-5p was correlated with the initiation and progression of patients with melanoma. It is worth mentioning that the precious molecular pathogenesis of miRNAs in modulating the cancer metastasis are quite different. Multiple evidences illustrated miR-508-5p regulated tumor cell growth and invasion by targeting downstream genes via different signaling pathways. However, the precise molecular pathogenesis of miR-508-5P as well as its direct target in human melanoma are not clear. In this study, qRT-PCR and western blot assays exhibited that miR-508-5p mRNA levels were down-regulated in peripheral blood samples from patients with melanoma and associated melanoma cells. We therefore speculated miR-508-5p was an important tumor suppressor gene for treating melanoma patients and its overexpression could inhibit tumor growth. We evaluated cell proliferation, migration and invasion separately using MTT, colony formation, wound healing and transwell assays with A375 melanoma cells. Taken the in vitro results together, we concluded miR-508-5p overexpression suppressed the melanoma cell growth, decreased the migrated and invasive cell numbers. All these phenotypes could be restored by miR-508-5p inhibitor, confirming the suppressive effects of miR-508-5p on cell proliferation, migration and invasion in human melanoma. All the in vitro results were consistent with previous studies regarding suppression role of miR-508-5p in tumor proliferation, migration and invasion.
KIT, a post-transcriptional regulator, is widely expressed in many types of cancers, such as liver cancer 25 and breast cancer, 26 which acts as a carcinogenic gene to accelerate the cell proliferation and invasion. Simultaneously, KIT is found to be regulated by a variety of miRNAs, and thus affecting the occurrence and development of tumors. For example, it is reported that downregulation of miR-664 suppressed the cervical cancer cells proliferation by directly targeting KIT. In this study, bioinformatics prediction revealed miR-508-5p might directly bind to the 3’UTR of wide type-KIT and its overexpression significantly inhibited KIT expression in vitro, which was consistent with previous results. In addition, qRT-PCR and western blot assays also proved either overexpression or inhibition of miR-508-5p resulted in the downregulation or upregulation of KIT respectively, therefore we strongly speculated KIT was a directly downstream target for miR-508-5p.
Previous studies have reported that miRNAs directly targeted Ras-Raf-MEK-ERK pathway to accelerate or suppress tumor cells proliferation and migration. For instance, miRNA-30a suppressed tumor progression by blocking Ras/Raf/MEK/ERK signaling pathway in hepatocellular carcinoma. 27 MiR-520c and miR-373 targeted mTOR and SIRT1, activated Ras/Raf/MEK/Erk pathway and NF-κB, along with up-regulation of MMP9 in human fibrosarcoma cell. 28 In this study, using western blot analysis, we observed miR-508-5p overexpression decreased the KIT expression and activated the downstream target genes (K-Ras, c-Raf, p-MEK, p-ERK), demonstrating miR-508-5p suppressed the melanoma cell proliferation, migration and apoptosis by targeting KIT via Ras-Raf-MEK-ERK pathway.
In summary, we found miR-508-5p level was reduced in peripheral blood of patients with melanoma and melanoma cell model. Our results further confirmed that miR-508-5p could suppress the cell proliferation, migration and invasion in melanoma A375 cells. Further in vitro studies revealed that miR-508-5p inhibited melanoma cell proliferation, migration and invasion by targeting KIT via Ras-Raf-MEK-ERK pathway. To sum up, we identified miR-508-5p as a novel tumor suppressor gene in human melanoma, shedding lights on understanding the molecular mechanisms of pathogenesis as well as developing therapeutic strategies from bench to clinic.
Footnotes
Abbreviations
Authors’ Note
SNF and LD conceived and designed the experiments, YW and CPS analyzed and interpreted the results of the experiments, MSL and ZJS performed the experiments. All data generated or analyzed during this study are included in this published article. All the participants involved in this project agreed to sign informed consent. The examination and the informed consents were approved by Ethics Committee of Shenzhen People’s Hospital (Approval no. LL-KY-201935), and there is no contrary to World Medical Association Declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Young and Middle-aged Scientific Research Backbone Cultivation Project of Shenzhen People’s Hospital (Grant No. Sykypy201914).
