Abstract
Purpose:
External beam radiotherapy is one of the treatment options for organ-confined prostate cancer. A total dose of 70 to 81 Gray (Gy) is given daily (1.8-2.5 Gy/d), with a dose rate of 3 to 6 Gy/min over 28 to 45 treatments during 8 to 9 weeks. We applied the latest technological development in linear accelerators for enabling a wide range of dose rates (from 0.2-21 Gy/min) to test the effect of different delivery dose rates on prostate tumor growth in an animal xenograft model.
Materials and Methods:
A prostate cancer xenograft model was established in CD1/nude mice by means of PC-3 and CL-1 cells. The animals were radiated by a TrueBeam linear accelerator that delivered 4 dose rates ranging from 0.6 to 14 Gy/min, and reaching a total dose of 20 Gy. The mice were weighed and monitored for tumor development twice weekly. A 2-way analysis of variance was used to compare statistical differences between the groups.
Results:
Tumor growth was inhibited by radiation at all 4 dose rates in the 20 study mice compared to no radiation (n = 5, controls). The most significant reduction in tumor volumes was observed when the same dose of radiation was delivered at a rate of 0.6 Gy/min (
Conclusions:
Delivery of radiation with a TrueBeam linear accelerator at the lowest possible rate was most effective in prostate cancer growth inhibition and might be considered a preferential treatment mode for localized prostate cancer.
Introduction
Prostate cancer is the second leading cause of cancer-related deaths among American men following heart disease (CDC, C.f.D.C.a.P. Cancer among men. 2015; Available from: https://gis.cdc.gov/cancer/USCS/DataViz.html). 1 The majority of patients are diagnosed with localized prostate cancer and successfully treated with radical prostatectomy or radiation therapy. Radiotherapy usually involves external beam radiotherapy or brachytherapy, 2 and photons are the main particles used in the external radiotherapy approach. 3 Recent breakthrough technology of external beam radiation, such as linear accelerators, can produce higher levels of energy and precision delivery of radiation to the tumors at a high delivery rate, while minimizing the damage to the surrounding tissues, reduce costs, and improve patient convenience. 3 These high-precision photon radiotherapies have improved treatment outcomes in various malignancies, including prostate cancer. 3
A wide range of dose rates has been used in radiotherapy, extending from a few Gray (Gy) per day to tens of Gy in a small fraction of a second. 4 For localized prostate cancer, daily small doses (1.8-2 Gy) of radiation at a rate of 4 Gy/min over 8 to 9 weeks have traditionally been delivered, with the total dose ranging between 70 and 81 Gy over 38 to 45 treatments. A predefined radiation dose can be delivered by employing variable time spans and modulating dose rates. The impact of dose rate on the extent of radiation-induced biological effects in cells has been studied in depth. Those studies showed that small dose rates are more effective in inducing genetic damage and cell death compared to high dose rates for the same magnitude of radiation dose. 5 -8 However, the issue of the impact of different delivery dose rates on tumor growth, particularly in prostate cancer, is still unresolved. The feasibility of delivering a wide range of dose rates by means of modern linear accelerators allowed us to study the effect of different delivery rates of the same dose on tumor growth in a prostate cancer xenograft model.
Materials and Methods
Cell Culture
Both PC-3 and CL-1 human prostate cancer cells were purchased from American Type Culture Collection. The cells were cultured in RPMI-1640 containing 10% fetal bovine serum and antibiotics, and incubated in a 5% CO2 humidified incubator at 37 °C.
Animal Studies
The animal procedures were carried out under institutional guidelines of the Tel Aviv Sourasky Medical Center (permission number 11-6-17). PC-3 and CL-1 cells were suspended in an equal volume of phosphate buffered solution and matrigel and kept on ice to prevent cell culturing. A total of 0.5 × 10 6 of PC-3 or 1 × 10 6 of CL-1 cells were injected subcutaneously by means of a 23G syringe needle into the right hind quarters of CD1/nude mice. When the tumor volume reached 100 mm3, the mice were randomized into 5 groups for each xenograft model (n = 5 per group) as follows: (1) no treatment (controls), (2) to (5) radiation treatment for a total dose of 2 Gy/d in dose rates of 0.6 Gy/min, 2 Gy/min, 4 Gy/min, and 14 Gy/min, respectively. Groups 2 to 5 were radiated with 2 Gy daily × 5 days/week for 2 weeks, yielding a total dose of 20 Gy. The no-radiation control group underwent the same handling without receiving radiation. Metal identification tags were attached to the animals’ ears in order to distinguish among them. The mice were weighed and monitored for tumor development twice weekly. Tumor variables were measured with calipers, and tumor volume was calculated by width2 × length × 0.5, which is an estimate derived from the formula for obtaining sphere volume. 9 The mice were sacrificed by CO2 inhalation one week after the last session of radiation.
Radiation
Radiation was carried out in a TrueBeam linear accelerator (Varian Medical System) with X-ray energy outputs of 6 MV and dose rates of 0.6 Gy/min, 2 Gy/min, 4 Gy/min, and 14 Gy/min. The mice were secured in plastic holders, and the collimated X-ray beam radiated an area of 24 × 24 mm2 at the tumor site, which was large enough to cover the entire area of the largest tumor.
Statistical Analysis
The statistical analysis of the results was performed with a 2-way analysis of variance (ANOVA) test. The results were reported as mean ± SE as indicated in the figures. Data were considered significant when
Results
Effect of Delivery of Radiation at Each Dose Rate on Prostate Tumor Growth
We established a xenograft model of CD1/nude mice in order to study the effect of 4 radiation dose rates on prostate cancer tumor growth. Following the implantation of prostate cancer cells (PC-3 and CL-1), the mice were divided into 5 groups (4 different dose rates and 1 control as specified in the Material and Method section) when the tumor volume had reached 100 mm3. The study mice underwent radiotherapy 5 days a week for 2 weeks, thus receiving a total dose of radiation of 20 Gy. As expected, tumors that were not radiated exhibited significantly increased tumor volume compared with tumors that were treated with any rate of radiation (Figures 1A and 2A for PC-3 and CL-1 cells, respectively). Moreover, the lower the dose rate of radiation treatment, the stronger the inhibition of the tumor growth, especially in tumors that were formed by PC-3 cells (Figure 1B). The significance of the differences between the groups is depicted in Table 1. CL-1 tumors had similar radiation responses compared to PC-3 tumors; however, the differences in tumor growth inhibition at the lower doses were less significant (Figure 2B and Table 2). Of note, the CL-1 tumors exhibited a more prolonged radiation response at the lowest radiation dose rate (Figure 2B). Also notable was that none of the dose rates had any effect on the animals’ weight compared with that of the controls (Figure 3). Delivery of the same dose at the lowest rate emerged as being the most effective in inhibiting prostate tumor growth among the 4 tested rates.

The effect of dose rates of radiation on PC-3 prostate tumor growth over time. A, Comparison between the control and the radiated groups. B, Comparison between the different dose rates among the radiated groups.

The effect of dose rates of radiation on CL-1 prostate tumor growth over time. A, Comparison between the control and the radiated groups. B, Comparison between the different dose rates among the radiated groups.
Comparison Between the Groups of Mice that Received Different Radiation Dose Rates for the Treatment of Prostate Tumor Established by PC-3 Cells.a

Abbreviations: ANOVA, analysis of variance; NS, not significant.
a The
Comparison Between the Groups of Mice That Received Different Radiation Dose Rates for the Treatment of Prostate Tumor Established by CL-1 Cells.a
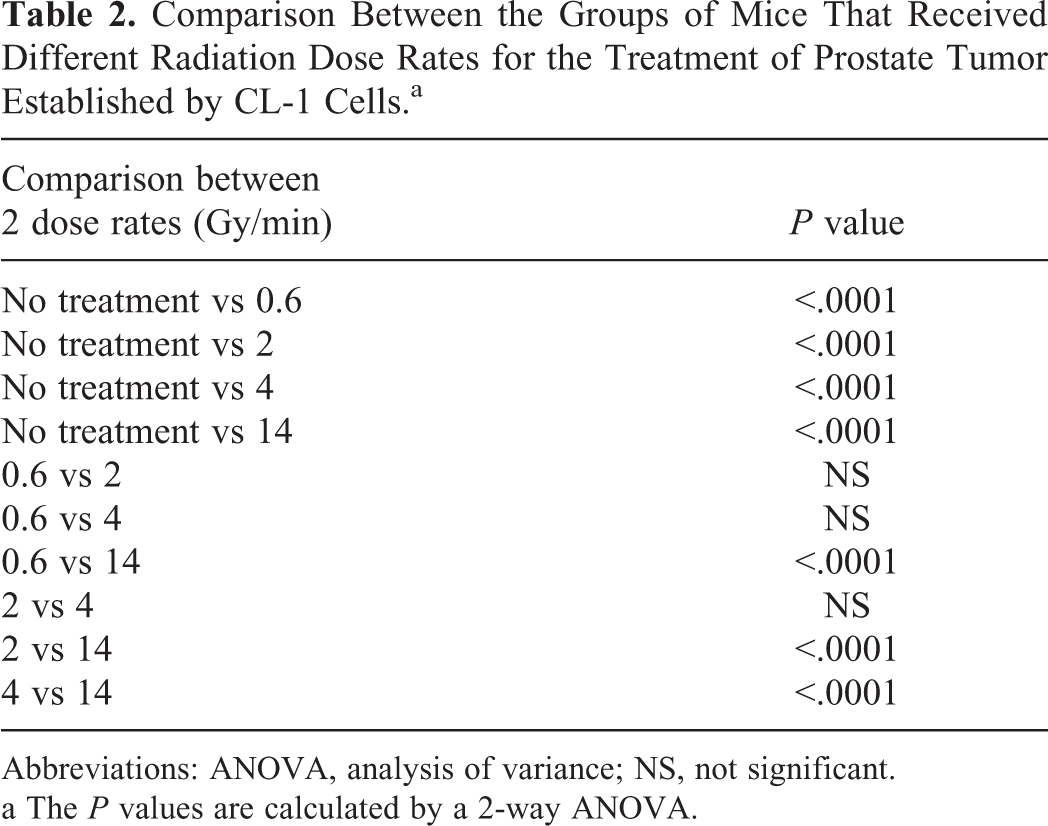
Abbreviations: ANOVA, analysis of variance; NS, not significant.
a The
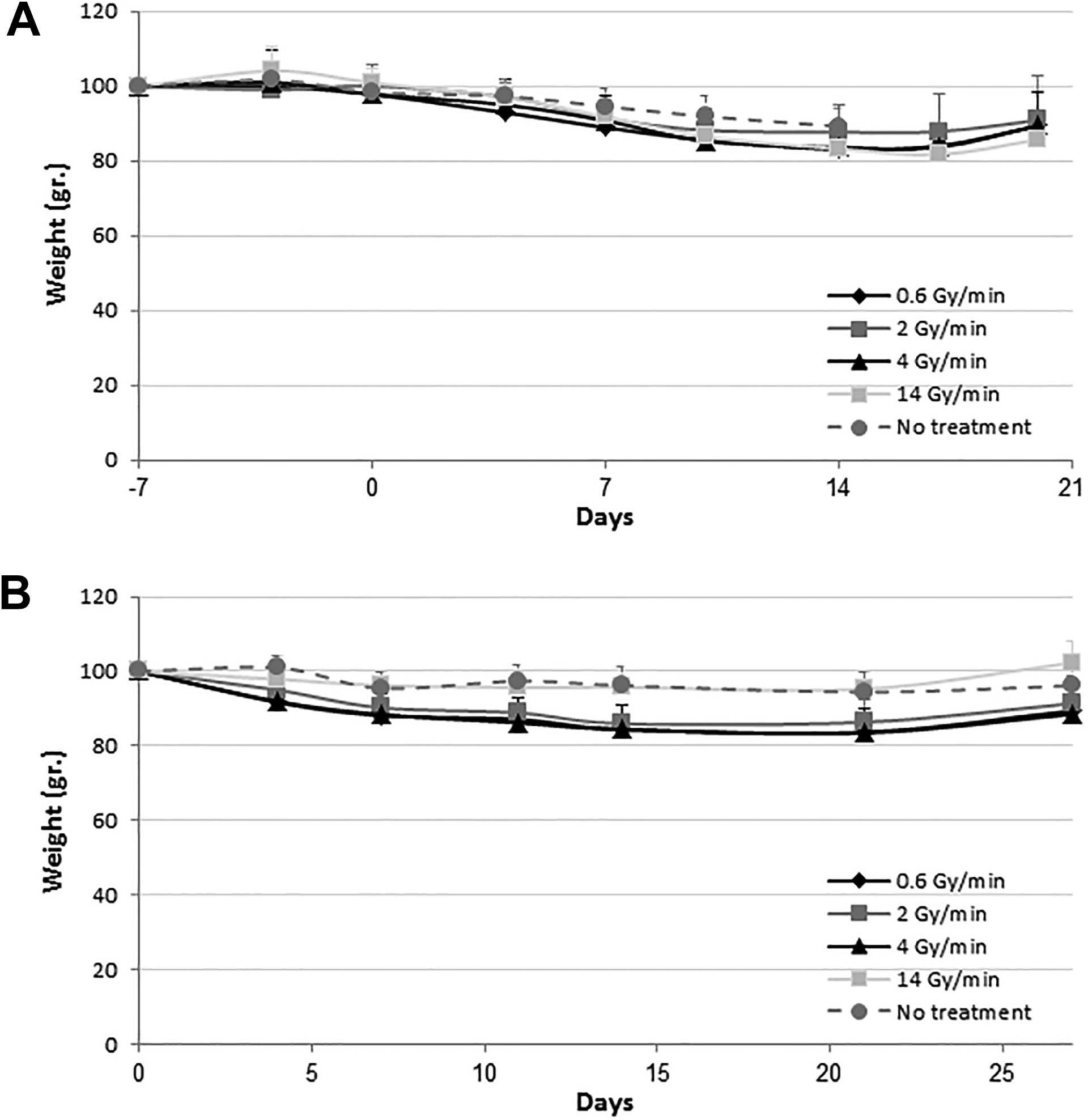
Lack of any effect of different dose rates of radiation on the animals’ weight. The same animals used in Figures 1 and 2 were also weighed twice weekly. The graphs display the weight of the no-treatment group and the daily effect of the different dose-rates in the 4 treatment groups. Day “0” refers to the first day of radiation.
Discussion
The results of this study demonstrated that the lowest delivery rate of the same dose of radiation had an inverse effect on tumor volume. Specifically, inhibition of the tumor growth increased as the radiation rate gradually decreased from 14 Gy/min to 0.6 Gy/min. The tumor volumes were significantly reduced compared to no-radiotherapy at lower radiation rates (Figure 1A and 2A). Furthermore, the radiation rate of 0.6 Gy/min had the strongest effect on tumor volume compared with the higher dose rates of 2, 4, and 14 Gy/min in the PC-3 tumors (Figure 1B and Table 1).
A vast proportion of the published data on the effect of dose rate of radiation was done
To date, radical prostatectomy (the main option) and external beam radiotherapy remain the most ubiquitous treatments for localized prostate cancer. Standard radiation has been among the most time-consuming radiation treatments for that malignancy, that is, 38 to 45 daily treatments of 1.8 to 2.5 Gy at a rate of 3 to 6 Gy/min for a total treatment duration of 2 months. This treatment protocol is long, costly, and inconvenient. Recent studies showing that prostate cancer cells had an effective response to higher daily radiation doses motivated the transition from the conventional fractionation to hypofractionation, 17 which is defined as the delivery of higher daily doses of radiation over a shorter number of days. This schedule would reduce costs and health care burden, and possibly also make treatment more accessible to patients who do not live near a treatment facility.
This advanced technology of hypofractionation affects the total dose and the dose per fraction without altering the radiation dose rate. Although our results do not support advantages with regard to hypofractionation, treatment costs, and patient inconvenience, we consider that the favorable treatment outcome outweighs the absence of those benefits. Our results in prostate cancer models are consistent with an “inverse dose rate effect” according to which some mammalian cell populations exhibit a marked increase in cell death under low dose exposure. The main theory for explaining this phenomenon is that low dose rates cause a failure to arrest in G2. 18
The current work reveals that the radiation delivery dose rate as being a new factor that warrants in-depth study. It will be interesting to further explore our model using other cancer cells and even lower radiation delivery dose rates, as well as seeking the explanation for why the delivery of the same amount of radiation at a very low rate is more effective than at higher delivery rates.
Conclusions
Our results indicate that the delivery of radiation at a low dose rate, such as 0.6 Gy/min, is feasible by means of modern linear accelerators, and that it is more effective in inhibiting prostate cancer growth compared to the tested higher dose rates.
Footnotes
Abbreviations
Authors’ Note
All procedures were done in compliance with the Tel Aviv Sourasky Medical Center Animal Care and Use Committee (permission number 11-6-17).
Acknowledgment
The authors would like to thank Hila Granot for her technical support in radiating the mice by the TrueBeam linear accelerator and to Ms. Esther Eshkol for editorial assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding through the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (AMRF).
