Abstract
Triple-negative breast cancer shows worse outcome compared with other subtypes of breast cancer. The discovery of dysregulated microRNAs and their roles in the progression of triple-negative breast cancer provide novel strategies for the treatment of patients with triple-negative breast cancer. In this study, we identified the significant reduction of miR-133 in triple-negative breast cancer tissues and cell lines. Ectopic overexpression of miR-133 suppressed the proliferation, colony formation, and upregulated the apoptosis of triple-negative breast cancer cells. Mechanism study revealed that the YES Proto-Oncogene 1 was a target of miR-133. miR-133 bound the 3′-untranslated region of YES Proto-Oncogene 1 and decreased the level of YES Proto-Oncogene 1 in triple-negative breast cancer cells. Consistent with miR-133 downregulation, YES1 was significantly increased in triple-negative breast cancer, which was inversely correlated with the level of miR-133. Restoration of YES Proto-Oncogene 1 attenuated the inhibitory effects of miR-133 on the proliferation and colony formation of triple-negative breast cancer cells. Consistent with the decreased expression of YES Proto-Oncogene 1, overexpression of miR-133 suppressed the phosphorylation of YAP1 in triple-negative breast cancer cells. Our results provided novel evidence for the role of miR-133/YES1 axis in the development of triple-negative breast cancer, which indicated miR-133 might be a potential therapeutic strategy for triple-negative breast cancer.
Introduction
Considering the strong invasiveness, local recurrence, and high distant metastasis, triple-negative breast cancer (TNBC) has been the dominant cause of cancer-related mortality among women worldwide. 1 -6 Triple-negative breast cancer is characterized by the absence of estrogen receptor, progesterone receptor, and epidermal growth factor receptor 2 (eGFR) . 4 The lack of specific diagnostic and prognostic biomarkers for TNBC results in only a minority (15%) of patients with TNBC responding well to chemotherapy, and eventually most of these patients will advance to tumor recurrence and metastasis. Therefore, exploring novel therapeutic targets against TNBC is urgently needed to improve the outcome of patients with TNBC.
MicroRNAs (miRNAs) are classified as single-stranded noncoding RNA molecules with a length of 20 to 24 nucleotides. 7,8 MicroRNAs act as negative regulators of protein-coding genes predominantly via binding to the 3′-untranslated region (UTR) of target messenger RNAs (mRNAs), which leads to mRNA degradation or translation repression. 9 Extensive miRNA research has revealed the involvement of miRNAs in numerous key cellular functions including cell proliferation, differentiation, and apoptosis. 10 Aberrant expression of miRNAs, which regulates the function of tumor suppressors and oncogenes, has been associated with the initiation and progression of TNBC. 11 -15 Given the overwhelming evidence that the pattern of miRNA expression was correlated with the cancer subtype, stage, and survival of patients with TNBC, miRNAs might be considered as potential novel biomarkers for the diagnosis and prognosis in cancer therapy.
MiR-133 has frequently been reported as downregulated in several cancers, including lung, gastric, bladder, and prostate cancer. 16 -20 Accordingly, overexpression of miR-133 inhibited the malignant behavior of cancer cells, suggesting miR-133 as a possible target to interrupt cancer progression. Downregulation of miR-133 predicted poor survival of patients with glioma. 21 It was also reported that overexpression of miR-133 sensitized non-small cell lung cancer cells to irradiation. 22 Much more attention has been drawn to the versatile molecular functions of miR-133, making it a promising therapeutic target in miRNA-based gene therapy. However, to our knowledge, the role and the underlying mechanism of miR-133 in the progression of TNBC remain largely unknown.
In the present study, we aimed to investigate the expression and function of miR-133 on the malignant behavior of TNBC cells and characterize the underlying mechanism. Our results found that miR-133 was significantly downregulated in TNBC tissues and cell lines. Decreased miR-133 expression was associated with the poor prognosis of patients with TNBC. Overexpressed miR-133 inhibited the proliferation and induced the apoptosis of TNBC cells. Further mechanism study revealed YES1 was a potential target of miR-133 and played a critical role in the suppressive function of miR-133 in TNBC. These findings provide novel insights into the biological significance of miR-133/YES1 axis in TNBC.
Materials and Methods
Tissue Specimens
A total of 50 tumor tissues and paired adjacent normal tissues were obtained from 50 patients with TNBC who underwent surgery at the Shanxi Provincial Cancer Hospital between April 2013 and September 2014. The normal tissues were collected from the distal resection margins (>2 cm away from the tumor tissue). All specimens were stored at −80 °C until the start of the experiments. None of these patients received chemotherapy or radiotherapy prior to surgery. This study was approved by the ethics committee of Shanxi Provincial Cancer Hospital (approval no. SXCH201503425). All patients provided written informed consent prior to enrollment in the study.
Cell Culture and Transfection
The human TNBC cell lines MDA-MB-231, BT-549, HCC-1937, MDA-MB-468, and normal cell MCF-10A were purchased from the American Type Culture Collection. Cells were cultured in RPMI-1640 medium with 10% fetal bovine serum (FBS) at 37 °C in a humidified 5% CO2 incubator. The miR-133 mimic, miR-NC, miR-133 inhibitor, and inhibitor-NC were commercially obtained from RiboBio Co, Ltd. The expression plasmid of pcDNA-Myc-YES1 was constructed by amplifying the complementary DNA (cDNA) fragment of YES1 via polymerase chain reaction (PCR) and inserted into the backbone of pcDNA-Myc vector. For the overexpression of miR-133, miR-133 mimic, or miR-133 inhibitor (50 nM) was mixed with Lipofectamine 2000 (Invitrogen) in OPTI-MEM for 15 minutes at room temperature (RT) when the cells reach to 70% confluence.
Quantitative Real-Time PCR
Total RNA was extracted from tissues and cells using the TRIzol reagent (Invitrogen). RNA concentration was measured with NanoDrop 2000 (Invitrogen). Complementary DNA was synthesized using the First-Strand cDNA Synthesis Kit (TakaRa Bio). Expression of miR-133 was determined by quantitative real-time PCR with the miScript SYBR Green PCR Kit (Applied Biosystems) according to manufacturer’s instructions. The fold change of miR-133 expression in TNBC cells or tissues was normalized to that of U6 RNA and compared with that of the controls using the 2 (−ΔΔCT) method. Therefore, the expression of miR-133 in normal cells and the adjacent normal tissues was considered as “1.” The sequences of primers were listed as follows: miR-133 forward primer: 5′-TTTGGTCCCCTTCAACC-3′ and reverse primer: 5′-GAGCAGGGTCCGAGGT-3′. U6 forward primer: 5′-TGCGGGTGCTCGCTTCGGCAGC-3′ and reverse primer: 5′-CCAGTGCAGGGTCCGAGGT-3′. The thermocycling conditions were set as follows: 94 °C for 5 minutes, followed by 40 cycles at 94 °C for 30 seconds and 58 °C for 45 seconds. The experiments were performed in triplicate.
Cell Proliferation Assay
Cells transfected with final concentration of 50 nM miR-133 mimics and control miRNA or miR-133 inhibitor and inhibitor-NC were preincubated on a 96-well plate with 1000 cells per well for 24 hours. Then, 10 µL of CCK-8 solution (Dojindo) were added into the medium at the indicated time points (days 1, 2, 3, 4, and 5) and incubated for additional 4 hours at 37 °C. The absorbance at 450 nm was measured with a microplate reader (Bio-Rad). For the chemotherapy-resistance assay, TNBC cells were treated with 25 μM cis-dichlorodiammine platinum (DDP) in the presence of miR-133 mimic or control miRNA. Cell counting kit-8 (CCK-8) reagent was added at the indicated time points and the cell proliferation was detected as described above. The experiments were performed in triplicates.
Western Blot
Total proteins were extracted from cells using NP-40 lysis buffer containing 1% protease inhibitor (Beyotime). Protein contents were measured using the BCA assay kit (Beyotime) according to manufacturer’s instructions. Equal amounts of protein (20 μg) were separated on 15% polyacrylamide gels and transferred onto nitrocellulose (Millipore Corporation). Membranes were blocked with 5% nonfat milk for 1 hour at RT and then incubated with primary antibodies against YES1 (ab109265; Abcam) or glyceraldehyde-3-phosphate dehydrogenase (GAPDH; ab181602, Abcam) at a dilution of 1:1000 at 4 °C overnight. After washing twice with tris-buffered saline with tween-20, membranes were incubated with goat anti-rabbit IgG (H + L)-horseradish peroxidase (HRP)–conjugated secondary antibody at a dilution of 1:10 000 for 1 hour at RT. The detected level of GAPDH served as the internal reference. Signals were visualized by Immobilon Western Chemiluminescent HRP Substrate (Millipore Corporation) using the American Imager 600 (GE Healthcare).
Dual-Luciferase Reporter Assay
The fragment of wild-type or mutant YES1 3′-UTR was ligated into the pmirGLO Dual-Luciferase miRNA target expression vector (Promega). Triple-negative breast cancer cells were co-transfected with the luciferase vector and miR-133 mimics or miR-NC (or miR-133 inhibitor or inhibitor-NC). After transfection for 48 hours, cells were harvested and the luciferase activities of firefly and renilla were measured using the Dual Luciferase Reporter Assay Kit (Promega) according to manufacturer’s instructions. The ratio of firefly to Renilla was used to normalize the firefly luciferase values. The experiments were performed in triplicate.
Colony Formation Assay
Triple-negative breast cancer cells expressing miR-133 mimics or miR-NC were suspended in fresh medium containing 10% FBS and 0.3% low-melting point agarose. A total of 1000 cells were seeded on a 12-well plate that was pre-coated with 0.5% agarose. Cells were cultured at 37 °C with 5% CO2 for 14 days, and then the colonies were stained with 0.1% crystal violet (Beyotime) and counted under the light microscopy.
Cell Apoptosis
The apoptosis analysis of TNBC cells carrying miR-133 mimics or miR-NC was conducted using the Annexin V-FITC Detection Kit (Dojindo) according to manufacturer’s instructions. Briefly, TNBC cells were seeded in a 6-well plate (5 × 105 cells per well) and cultured overnight. Cells were then transfected with 50 nM of miR-133 mimics or miR-NC using 5 µL of Lipofectamine 2000 when the cell confluence reached to ∼70%. After transfection for 48 hours, cells were trypsinized and the single cell suspension was incubated with 5-µL Annexin V-FITC followed by addition of 5 µL of propidium iodide solution and incubated in the dark for 15 minutes. Cells were then suspended with 400 µL of Annexin V-binding buffer and the apoptotic cells were analyzed using the BD Accuri C6 Plus flow cytometry (BD Biosciences).
Statistical Analysis
Data were presented as mean ± standard deviation and analyzed using the GraphPad Prism 6.01. Differences between groups were considered significant when P value <.05. Significance between groups was analyzed using Student t test or one-way analysis of variance followed by Tukey–Kramer post hoc test. The correlation between miR-133 and YES1 was determined by the Spearman correlation test.
Results
MiR-133 Was Downregulated in TNBC
To evaluate the expression level of miR-133 in TNBC, quantitative real-time PCR was performed on tissues from 50 patients with TNBC. MiR-133 was frequently and significantly downregulated in TNBC tissues compared with the adjacent normal tissues (Figure 1A). To further investigate the clinical significance of miR-133 in TNBC, the correlation between the expression of miR-133 with the clinical factors of patients with TNBC was analyzed. Those 50 patients were divided into miR-133-high and miR-133-low groups according to the mean value of miR-133. As presented in Table 1, lower miR-133 was significantly correlated with the larger tumor size, high histological grade, lymph node metastasis, and tumor necrosis metastasis (TNM) stage of patients. Additionally, the expression of miR-133 was determined in a panel of human TNBC cells lines, including MDA-MB-231, BT-549, HCC-1937, MDA-MB-468, and normal breast cell line MCF-10A. Real-time quantitative polymerase chain reaction (RT-qPCR) data showed that the expression of miR-133 in TNBC cells was significantly lower than that in normal cells (Figure 1B). These findings suggested the downregulation of miR-133 in TNBC.
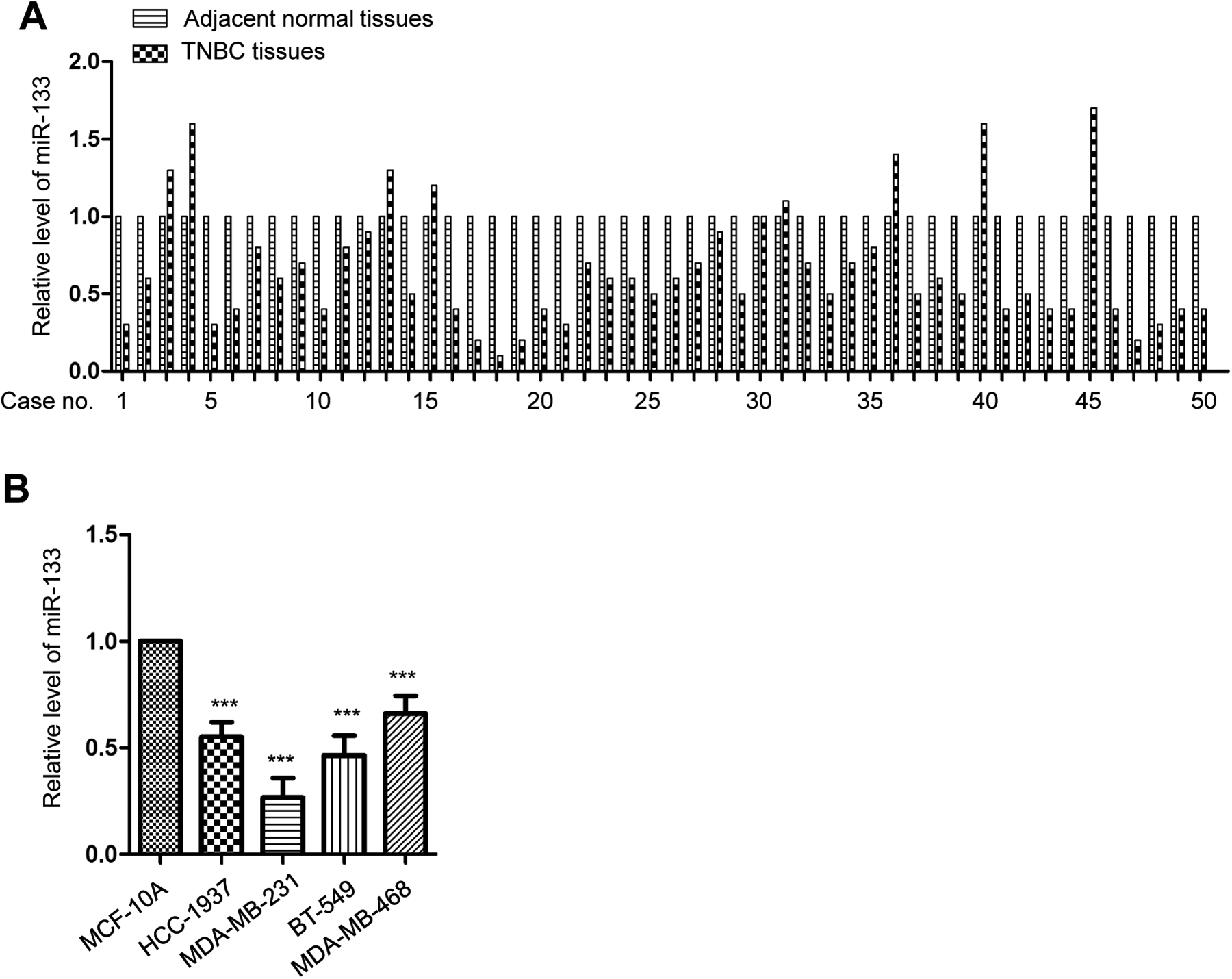
miR-133 was downregulated in TNBC. A, The level of miR-133 in TNBC tissues and paired adjacent normal tissues was detected by RT-qPCR. B, Expression of miR-133 in normal MCF-10A and TNBC cell lines (HCC-1937, MDA-MB-231, BT-549, and MDA-MB-468) was detected by RT-qPCR. ***P < .001. RT-qPCR, real-time quantitative polymerase chain reaction.
The Correlation Between the Expression of miR-133 and the Clinical Features of Patients With TNBC.

Abbreviation: TNBC, triple-negative breast cancer.
MiR-133 Suppressed the Proliferation of TNBC Cells
To determine whether the aberrant expression of miR-133 affected the proliferation of TNBC cells, both MDA-MAN-231 and BT-549 cells were transfected with miR-133 mimics or miR-NC using Lipofectamine 2000. The overexpression of miR-133 was validated by RT-qPCR (Figure 2A). The regulation of miR-133 on the proliferation of TNBC cells was assessed by the 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide (MTT) assay. As shown in Figure 2B and C, compared with that of the control cells, overexpression of miR-133 significantly suppressed the proliferation ability of TNBC cells. The suppressive function of miR-133 in TNBC was also determined by evaluating the apoptosis of cells. The results indicated that overexpression of miR-133 dramatically increased TNBC cell apoptosis compared with that of cells expressing the negative control (Figure 2D). The colony formation assay demonstrated that transfection of miR-133 showed decreased colony formation capacity compared with that of cells carrying miR-NC (Figure 2E). These results suggested a tumor suppressive role for miR-133 in the progression of TNBC.

miR-133 suppressed the growth of TNBC cells. A, Cells were transfected with miR-133 mimic or miR-NC, and the expression of miR-133 was validated by RT-qPCR. B and C, The proliferation of MDA-MB-231 and BT-549 cells was detected by CCK-8 assay. D, The apoptosis of TNBC cells with miR-133 mimic or miR-NC transfection was examined by flow cytometry analysis. E, Colony formation assay showed miR-133 overexpression reduced the number of colonies. ***P < .001. CCK-8 indicates cell counting kit-8; RT-qPCR, real-time quantitative polymerase chain reaction.
YES1 Was a Target of miR-133 in TNBC
To further understand the functional mechanism of miR-133 in TNBC, the targets of miR-133 were predicted using the miRDB database (http://mirdb.org/). Given the inhibitory effects of miR-133 in TNBC, YES1 was identified as a potential target of miR-133 as it carries a complementary binding site for miR-133 in the 3′-UTR (Figure 3A). To support this prediction, the expression of YES1 in TNBC tissues and paired adjacent normal tissues was determined by RT-qPCR. The results showed that compared with the non-cancer tissues, the level of YES1 in TNBC samples was frequently significantly upregulated (Figure 3B). Additionally, the higher expression of YES1 was significantly correlated with the advanced progression of patients with TNBC (Table 2). The correlation between the abundance of miR-133 and YES1 was examined with the Spearman test. As presented in Figure 3C, significant negative correlation was observed between the levels of miR-133 and YES1 in TNBC tissues. To further confirm the negative regulation of YES1 by miR-133, the luciferase reporter assay was performed by co-transfecting miR-133 mimics and luciferase vector carrying wild-type or mutant 3’-UTR of YES1. The data showed that overexpression of miR-133 decreased the luciferase activity on both MDA-MB-231 and BT-549 cells that expressed WT, but not MT 3’-UTR of YES1 (Figure 3D and E), which suggested the specific binding of miR-133 with the 3’-UTR of YES1.

YES1 was a target of miR-133. A, Predicted binding sites of miR-133 at the 3′-UTR of YES1. B, Expression of YES1 in TNBC tissues and paired adjacent normal tissues was detected by RT-qPCR. C, Correlation between miR-133 and YES1 was evaluated by the Spearman test. D and E, Luciferase reporter assay showed miR-133 suppressed the luciferase activity of WT 3′-UTR of YES1. F and G, Overexpression of miR-133 decreased the mRNA and protein levels of YES1 in TNBC cells. ***P < .001. mRNA indicates messenger RNA; RT-qPCR, real-time quantitative polymerase chain reaction; TNBC, triple-negative breast cancer; 3′-UTR, 3′-untranslated region.
The Correlation Between the Expression of YES1 and the Clinical Features of Patients With TNBC.

Abbreviation: TNBC, triple-negative breast cancer.
To determine whether miR-133 binding affected the mRNA stability of YES1, the expression of YES1 was detected by RT-qPCR analysis. The data showed that overexpression of miR-133 significantly decreased the mRNA level of YES1 in both MDA-MB-231 and BT-549 cells (Figure 3F). Consistently, compared with cells harboring a negative control, highly expressed miR-133 downregulated the protein abundance of YES1 in TNBC cells (Figure 3G). Collectively, these results demonstrated that miR-133 targeted YES1 and negatively modulated the abundance of YES1 in TNBC cells.
Downregulation of miR-133 Promoted the Proliferation of TNBC Cells and Increased Expression of YES1
To further characterize the function of miR-133, both MDA-MB-231 and BT-549 cells were transfected with miR-133 inhibitor or inhibitor NC. The downregulation of miR-133 was detected by RT-qPCR (Figure 4A). Then, the effects of miR-133 depletion on the proliferation of TNBC cells were investigated by CCK-8 assay. As shown in Figure 4B and C, knockdown of miR-133 significantly promoted the proliferation of TNBC cells compared with cell expressing inhibitor NC. Additionally, the luciferase reporter assay also showed that downregulation of miR-133 obviously increased the luciferase activity of WT but not the mutant 3′-UTR of YES1 (Figure 4D and E). Consistent with these results, depletion of miR-133 increased both the mRNA and protein levels of YES1 in both MDA-MB-231 and BT-549 cells (Figure 4F and G). Collectively, these results demonstrated that downregulation of miR-133 promoted the proliferation of TNBC cells and increased expression of YES1.

Depletion of miR-133 promoted the proliferation and inhibited the expression of YES1 in TNBC cells. A, MDA-MB-231 and BT-549 cells were transfected with miR-133 inhibitor or inhibitor-NC, and the knockdown efficiency of miR-133 was confirmed by RT-qPCR. B and C, Depletion of miR-133 significantly promoted the proliferation of TNBC cells compared with that of cells expressing inhibitor-NC. D and E, Downregulation of miR-133 increased the luciferase activity of TNBC cells expressing WT but not mutant 3′-UTR of YES1. F and G, Depletion of miR-133 increased both the mRNA and protein levels of YES1 in both MDA-MB-231 and BT-549 cells. ***P < .001. mRNA indicates messenger; ns, no significance; RT-qPCR, real-time quantitative polymerase chain reaction; TNBC, triple-negative breast cancer; WT, wild-type; 3′-UTR, 3′-untranslated region.
Reintroducing YES1 Abrogated the Suppressive Role of miR-133 in the Malignancy of TNBC
To investigate whether miR-133 repressed the growth of TNBC cells via targeting YES1, YES1 was reintroduced into MDA-MB-231 and BT-549 cells by transfecting them with pcDNA-Myc-YES1. Overexpression of YES1 was validated by Western blot as indicated in Figure 5A. The MTT assay showed that TNBC cells transfected with miR-133 presented decreased cell proliferation. However, the proliferation properties of MDA-MB-231 and BT-549 cells were significantly upregulated after co-transfection with YES1 (Figure 5B and C). In addition, for the colony formation assay, TNBC cells carrying miR-133 and Myc-YES1 exhibited increased growth patterns during the colony formation compared with cells only expressing miR-133 (Figure 5D). These findings revealed that YES1 plays an important role in miR-133-induced growth of TNBC cells.
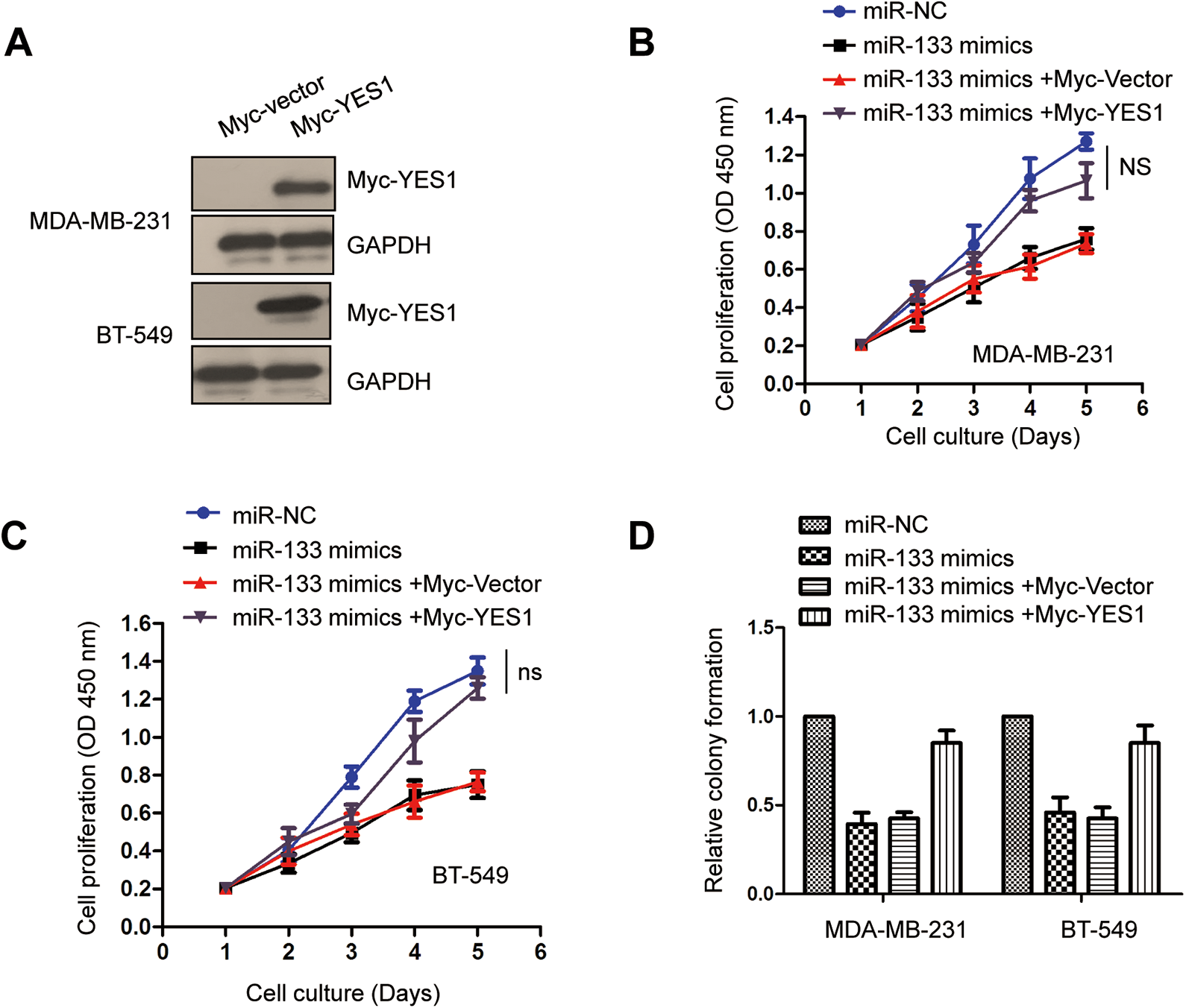
Expression of YES1 attenuated the inhibition of cell growth by miR-133 in TNBC cells. A, Cells were transfected with the overexpression plasmid of pcDNA-3Myc-YES1, and the ectopic expression of YES1 was confirmed by Western blot with anti-Myc antibody. B and C, Inhibition of miR-133 in the proliferation of TNBC cells was reversed with YES1 overexpression. D, Restoration of YES1 attenuated the suppressive role of miR-133 in the colony formation of TNBC cells. ns indicates no significance; TNBC, triple-negative breast cancer.
Downregulation of YES1 by miR-133 Inhibited the Phosphorylation of YAP1
To further understand how YES1 inactivation by miR-133 affected the progression of TNBC, the downstream signals of YES1 were investigated. YAP1, which was originally identified as a binding protein of YES1, acts in the Hippo pathway and facilitates tumor development. Previous researches demonstrated that YES1 directly phosphorylated YAP1 at tyrosine 357 and regulated YAP1-dependent transcriptional activity. To test whether the reduced expression of YES1 by miR-133 regulated the activity of YAP1, the phosphorylation of YAP1 at tyrosine 357 was detected within MDA-MB-231 and BT-549 cells. The data showed that upon decreasing the level of YES1, overexpression of miR-133 also reduced the phosphorylation of YAP1 without affecting the total protein level of YAP1 in TNBC cells (Figure 6A). Meanwhile, reintroduction of YES1 reversed the inhibitory effects of miR-133 on the phosphorylation of YAP1 (Figure 6B). These results suggested that miR-133 downregulated YES1 and consequently suppressed the activation of YAP1 in TNBC cells.
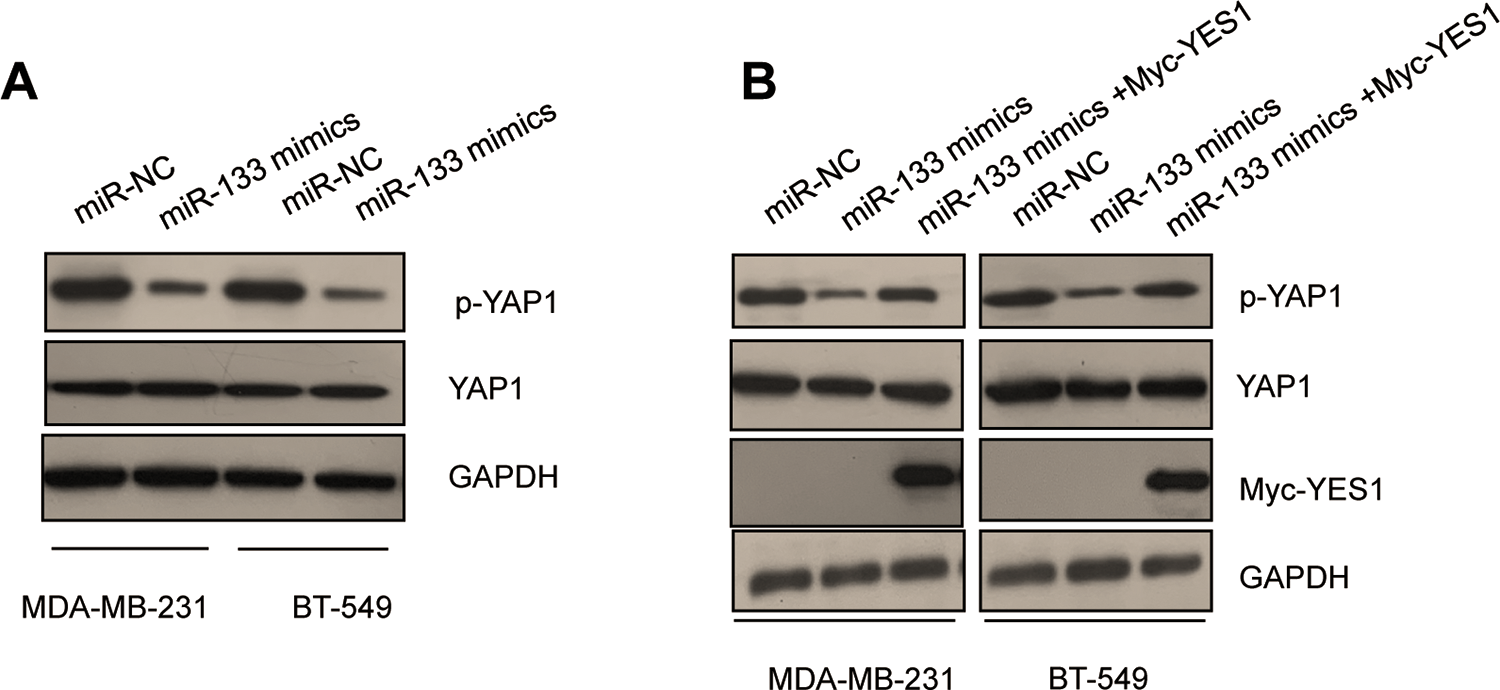
miR-133 inhibited the phosphorylation of YAP1. A, Overexpression of miR-133 in TNBC cells decreased the phosphorylation of YAP1. B, Inhibition of YAP1 phosphorylation induced by miR-133 was attenuated with overexpression of YES1 in both MDA-MB-231 and BT-549 cell. TNBC indicates triple-negative breast cancer.
Discussion
Tumorigenesis is considered a synergetic intricate process accompanied with dysfunction of oncogenes and tumor suppressors. In this study, decreased expression of miR-133 was observed in TNBC tissues. Overexpression of miR-133 caused suppression of proliferation in TNBC cells. Mechanistically, miR-133 targeted YES1 and eliminated the expression of YES1. Reintroduction of YES1 neutralized the suppressive effects of miR-133 on the malignancy of TNBC cells. Our findings suggested an antitumor role for miR-133 in TNBC by targeting YES1.
The dysregulation of miR-133 has been validated as an important driver for cancer development. 16 -19 Overexpression of miR-133 inhibited gastric cancer cell growth and migration and epithelial-mesenchymal transition processes, suggesting a critical role for miR-133 in the development and clinical therapy of gastric cancer. 18 High concentration of miR-133 was identified as a potential marker for the diagnosis of lymphoma-associated hemophagocytic syndrome. 23 Decreased expression of miR-133 predicted poor overall survival of patients with glioma. 21 Recent study also showed that overexpressed miR-133 sensitized non-small cell lung cancer cells to irradiation, indicating the promising application of miR-133 in the development of anticancer therapeutics. 22 In this study, overexpression of miR-133 significantly inhibited the proliferation of TNBC cells, in vivo xenograft mice model is needed to further confirm the potential therapeutic significance of miR-133 in TNBC. Notably, chemotherapy-resistance and metastasis are the big challenges in the treatment of TNBC. Given the suppressive roles of miR-133 in the proliferation of TNBC cells, it would be interesting to address the effects of miR-133 on the chemotherapy-resistance or metastasis of TNBC cells. In the present study, MDA-MB-231 and BT-549 cells were exposed to DDP (25 μM) treatment with or without the overexpression of miR-133. The CCK-8 assay showed that overexpressed miR-133 increased the sensitivity of TNBC cells to DDP exposure (Supplementary Figure 1A and 1B), suggesting the potential role of miR-133 in the chemotherapy-resistance of TNBC. More evidence is needed to further support this observation in future study. Additionally, the effects of miR-133/YES1/YAP1 in the progression of other breast cancers also remain to be addressed.
YES1, belonging to the SRC family, has various important cellular functions, including cell proliferation, differentiation, and adhesion. 24 Gene amplification of YES1 is present in lung, head and neck, bladder, and other cancers, suggesting inhibition of YES1 could act as a potential anticancer drug target. 24,25 YAP1 played a role in the downstream of YES1 and contributed greatly to the tumor-promoting effects of YES1. 26 Mechanically, YES1 modulated the transcription activity of YAP1 via regulating the serine phosphorylation and nuclear translocation. 27 Notably, an increasing body of evidence has demonstrated the negative regulation of YES1 by miRNAs in the progression of cancers. 28 -32 For example, miR-140-5p suppressed the malignancy of gastric cancer by targeting YES1. 30 MiR-210-mediated YES1 inhibition was the underlying mechanism of miR-210 action in the progression of hepatocellular carcinoma. 33 Additionally, YES1 was targeted by miR-17-5p and caused suppression of cell proliferation of ovarian cancer. 29 In this study, miR-133 was found to bind the 3′-UTR of YES1 and reduce the expression of YES1 in TNBC cells. The level of YES1 in TNBC tissues was inversely correlated with miR-133. Reintroducing YES1 abrogated the suppression of miR-133 on the proliferation of TNBC cells. Consistent with the decreased expression of YES1, overexpression of miR-133 inhibited phosphorylation of YAP1, suggesting defects in the function of YAP1. These findings indicated a novel mechanism for miR-133 in TNBC by downregulating YES1. As reported by previous studies, miR-133 inhibited the growth, metastasis, and sensitized lung cancer cells to irradiation via regulating the expression of different targets, such as the eGFR, the flotillin-2 and FOXQ1. 17,34,35 In bladder cancer and prostate cancer, miR-133 suppressed the proliferation, migration, and invasion of cancer cells by targeting the eGFR. 16,20 These reports demonstrated the tumor suppressive function of miR-133 in cancers, which is similar to our finding in TNBC. Differently, we identified YES1 as an important target of miR-133 in TNBC. The other targets, such as eGFR as reported in lung cancer, bladder cancer, and prostate cancer, in the suppressive role of miR-133 in TNBC is also interesting to be addressed.
In conclusion, the results of the present study demonstrated that miR-133 played a tumor suppressive role in TNBC by downregulating YES1. Downregulation of miR-133 was observed in TNBC tissues and cell lines. To evaluate the clinical significance of miR-133 in TNBC, further investigation should collect the clinical data of a larger sample size and examine the correlation between miR-133 and the overall survival of patients with TNBC. Additionally, it would be useful to detect and compare the level of circulating miR-133 in the blood of patients with TNBC and normal population, which would benefit the usage of miR-133 as an indication for TNBC. Considering the suppressive effects of miR-133 on the growth of TNBC cells, more evidence is needed to explore how to deliver the miR-133 to the tumor sites and evaluate the side effects induced by miR-133 in vivo.
Supplemental Material
Supplementary_Figure_1 - MiR-133 Targets YES1 and Inhibits the Growth of Triple-Negative Breast Cancer Cells
Supplementary_Figure_1 for MiR-133 Targets YES1 and Inhibits the Growth of Triple-Negative Breast Cancer Cells by Guochen Zhang, Junlan Wang, Ruilin Zheng, Beibei Song, Li Huang, Yujiang Liu, Yating Hao and Xiangdong Bai in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
This study was approved by the Ethics Committee of Shanxi Provincial Cancer Hospital. Written informed consents were obtained from all the participants.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
