Abstract
The alteration of biomass into simpler molecular remnants is relevant for the search for ancient and extraterrestrial life, where identifying recurrent taphonomic pathways is crucial for the attribution of biogenicity to otherwise nonbiological molecules. This work evaluates the alteration of lipids—recalcitrant biomarkers derived from cell membranes—across a lithification gradient, from a biologically active microbial mat, through a lithifying mat, to a fully lithified microbialite. Lipids from these samples, obtained from the high-altitude, hypersaline lake of Pozo Bravo (Argentinean Andes), were analyzed at molecular and isotopic levels to reconstruct biological sources and assess preservation along a bio-to-geo transition. Lipids from the lithifying mat and microbialite retained molecular features from the soft microbial mat (e.g., cyano- and purple sulfur bacteria), albeit at lower concentrations and diversity. Moreover, our analysis revealed preferential alteration of labile structural features such as unsaturations, methyl-, and pentacyclic structures, which decreased by ≥91% from soft to lithifying mat and ≥68% from lithifying mat to microbialite. Saturated and linear chains were more resistant, decreasing by ≥64% and ≥29%, respectively. These findings highlight how lipid preservation varies during lithification; thus, they provide valuable insights for biogenicity assessments and can help guide future efforts aimed at detecting ancient life.
Introduction
Investigating the earliest microorganisms that populated Earth represents an ambitious yet crucial scientific endeavor to explore the origins of life on our planet. Such a pursuit involves the necessity to probe into the distant past to uncover ancient biosignatures that offer key insights into biochemical evolution and survival under extreme conditions. The complexity of this task, however, is reflected in the multiple challenges that must be addressed in order to succesfully glimpse into the past, including the selection of suitable preservation matrices, the implementation of robust extraction protocols, and the application of appropriate analytical techniques. In addition, the billion-year timescales impose a challenging scenario where most biosignatures are likely to degrade and vanish over time. Notably, these intricacies hold an added importance for astrobiology. Mars is one of the most extensively studied extraterrestrial targets in the Solar System and once exhibited environmental conditions analogous to those of early Earth. Under those conditions, ancient life may have once existed on Mars. Ongoing and future attempts to detect evidence of this possible ancient life emphasize the importance of understanding the way biosignatures degrade and preserve.
The degradation of complex organic matter can lead to simpler molecular remnants through diagenesis, which is defined as a combination of biological, chemical, and physical processes that transform organic matter in terms of quantity and composition into more stable fossil forms, typically after losing the more labile features (Berner, 1980; Henrichs, 1992). Over long timescales, molecular markers of life—namely biomarkers—will be at the mercy of a wide range of mechanisms of degradation (Broz, 2020) that range from postdepositional processes (early diagenesis) to thermal maturation, compaction, and lithification (late diagenesis). After the more labile characteristics within a given biomarker are lost, the more recalcitrant skeletons (i.e., diagenetic products) may persist longer in the sedimentary record. However, this persistence might come at the cost of their biomarker potential, as their biogenicity may no longer be recognizable. Therefore, in the search for ancient life, it is crucial to go beyond conventional biomarker detection and analysis in order to deepen our understanding of the processes involved in the capture, alteration, preservation, and especially degradation of organic matter, collectively referred to as taphonomy (Efremov, 1940; Behrensmeyer and Kidwell, 1985; Allison and Pye, 1994; Shkolyar and Farmer, 2018).
In light of these challenges, lipids emerge as strong biomarker candidates for three major reasons. First, lipids are a diverse group of organic compounds that are integral and universal components of the biological cell membrane (Georgiou and Deamer, 2014). Second, in addition to their ubiquity on Earth, lipids possess a degree of specificity that often enables taxonomic assignments below the domain level (Brocks and Summons, 2003); this provides a broad yet valuable framework for biological origin compared with other organic molecules such as nucleic acids or peptides. Third, and most pertinent to this study, the recalcitrant hydrocarbon cores of lipids confer remarkable stability, which allows these molecules to retain their diagnostic value over geological timescales (Vinnichenko et al., 2020). This resilience is particularly prominent when lipids are entrapped and protected within mineral matrices in the geological record (Brocks and Summons, 2003; Sánchez-García et al., 2021, 2018; Finkel et al., 2023). In the search for ancient life on Earth or elsewhere in the Solar System, these properties must be complemented by detailed taphonomic studies to understand (i) how lipids degrade in relevant environmental contexts; (ii) which features are the most prone to be preserved, and, by association; (iii) which diagenetic products are likely to remain. Elucidating lipid degradation pathways would enable a more accurate inference of biogenicity from molecular patterns that might otherwise be attributed to abiotic processes.
In this work, we evaluate how lipid biomarkers from a soft microbial mat undergo transformation during (early) diagenesis, contributing to the organic molecular inventory preserved in the geological record, with the final objective of assessing the chemical and physical traits that are preserved in the geosphere in the long term. To analyze this bio-to-geo transition, we investigated the lipid profile of three samples: a soft mat representing a biological end-member, a microbialite serving as the geological end-member of the lithification gradient, and a lithifying microbial mat representing the transitional phase from the bio- to the geosphere (i.e., the process of mineralization while still growing, but to a lesser extent). The environmental samples were collected from Pozo Bravo, a high-altitude Andean lake subjected to low oxygen pressure, high ultraviolet (UV) irradiation, conditions of aridity and hypersalinity, daily temperature fluctuations, and strong winds (Vignale et al., 2022; Della Vedova et al., 2023). Some of these extreme conditions position Pozo Bravo not only as a suitable primitive Earth analog (Dundas, 1998; Sauterey et al., 2020; Lyons et al., 2024; Vignale et al., 2025) but also as an analog to Noachian Mars, where saline lagoons are believed to have existed (Cabrol and Grin, 2001). These factors contribute to an extreme environment with restricted predation and competition (Vignale et al., 2025) that fosters extensive microbial ecosystems with various degrees of mineralization, including biofilms, soft (growing) microbial mats, gradually lithifying mats, and fully lithified microbialites (Supplementary Fig. S1).
Overall, this study presents a qualitative and quantitative assessment of the taphonomy of lipid biomarkers across a gradient of lithification in a primitive Earth and Mars analog environment. By integrating molecular and stable-carbon isotopic analyses, we have gained insights into the predominant biological sources and metabolic pathways that drive the growth of a soft microbial mat, and we have identified those that have better resisted the transition to the geological record. Furthermore, we have identified and quantified the loss of intramolecular and functional features during this transition, which is vital to constrain likely mechanisms of degradation and preservation in this specific set of samples. Furthering our knowledge of lipid taphonomy provides a deeper understanding of the processes involved in the alteration of organic matter, not necessarily by probing the most ancient samples to assess lipid preservation, where most diagenetic alteration has already occurred, but by observing the process of alteration itself at different stages of lithification. These findings underscore the importance of considering taphonomic pathways in the search for ancient life on Earth and especially on Mars, where lipid-related organics have been detected (Freissinet et al., 2025).
Methodology
Site description
In the northwestern province of Catamarca, Argentina (Fig. 1A), lies Salar de Antofalla (Fig. 1B), a large (500 km2) salt pan that extends up to 136 km in length and 40 km in width. This confined depocenter has an internal drainage system characterized by its closed basin nature and high evaporative rates. Water sources in the region are scarce, limited to a mean annual precipitation of 82.1 mm, occasional surface runoff from surrounding highlands, and mostly infiltration from an underground water table (Lencina et al., 2021). Consequently, the salt pan harbors small hypersaline lagoons where salt crusts interact with local wetlands. At the northern end of the pan, near the native community of Antofalla, lies a long (500 m) and narrow (52 m) lake called Pozo Bravo (25°30′58.20′′ S, 67°34′41.33 W). This lake is situated at an altitude of 3330 meters above sea level (masl), flanked to the west by basaltic lava deposits and to the east by the predominant halite facies of the salt pan (Fig. 1C).
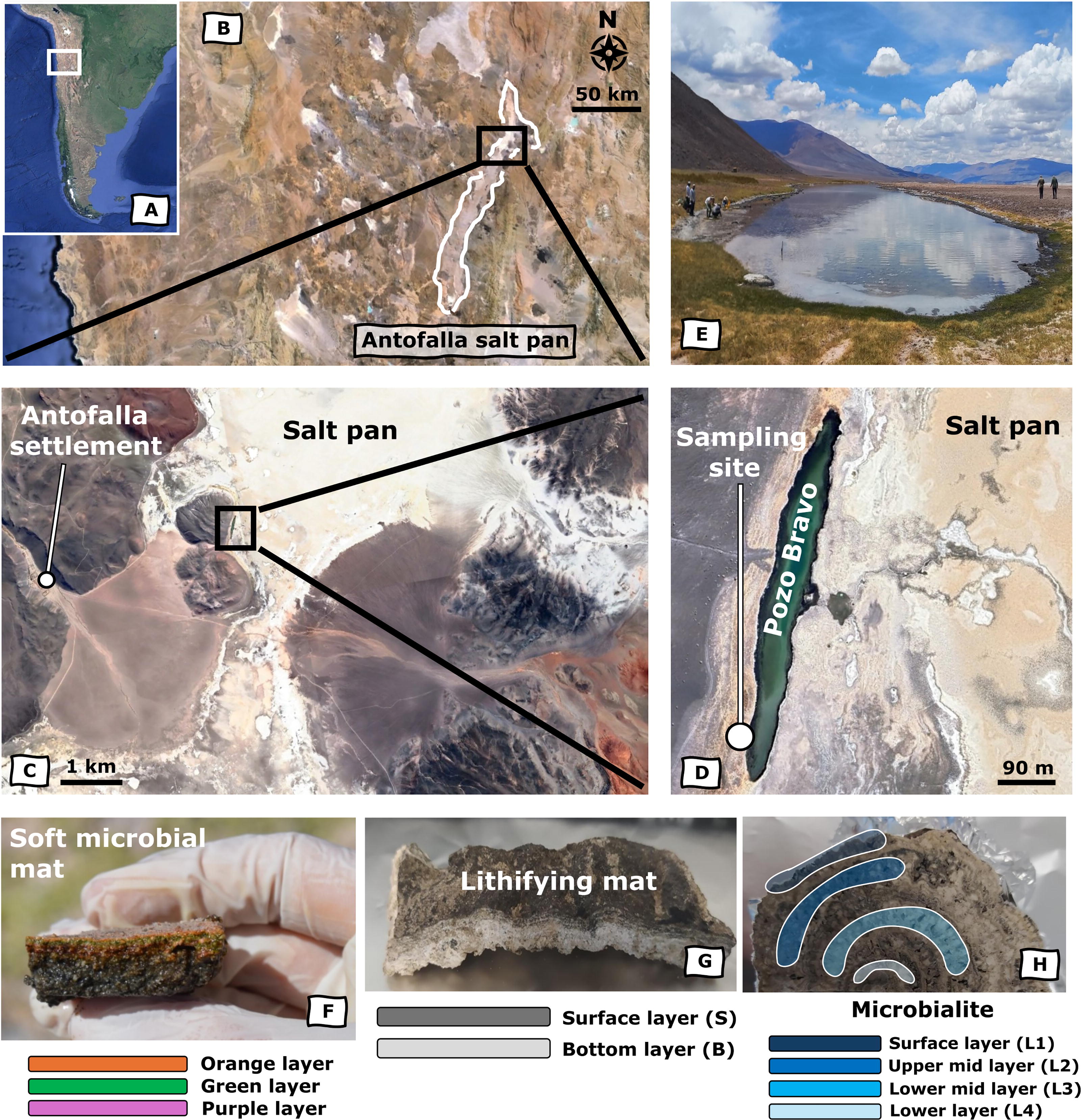
Geographical location of Pozo Bravo, sampling area, and sample types.
The physicochemical parameters of the water in Pozo Bravo fluctuate significantly between summer and winter (Lencina et al., 2021; Vignale et al., 2025). Here, we report the summer parameters, recorded in a study by Vignale and colleagues (2025). The surface temperature of the lake reaches 23°C in January, which closely matches the maximum average temperatures of the surrounding environment (26°C) in that same month (Rojas, 2016). Sodium (57,500 mg/L) and chloride (100,620 mg/L) are the most concentrated ionic species, followed by sulfate (1840 mg/L). This results in an elevated surface conductivity of 175.2 mS/cm. The water is slightly alkaline (pH ∼8) and reports a hardness of 10,250 mg CaCO3/L, influenced by groundwater discharge in contact with lacustrine carbonate deposits along the edges of the salt pan and by the active microbial metabolic activity of the area (Lencina et al., 2021).
The shallow waters (∼2.5 m depth) and shores of Pozo Bravo are home to a reef of modern microbialites that vary widely in sizes and structures (Lencina et al., 2021; Vignale et al., 2025). The external morphology of these microbialites is largely controlled by environmental factors such as high evaporation rates and hypersalinity, water-level fluctuations, and lake-bottom topography (Lencina et al., 2021). Moreover, the high solar irradiance delivers an instantaneous UV-B flux of 17 W/m2 (roughly half of what is experienced at equatorial Mars; Farías et al., 2011), which, along with hypersalinity, restricts predation and competition (Vignale et al., 2025), allowing for extensive microbial mat coverage. Intense evaporation and wet-dry cycles that respond to a frequently oscillating water column promote the lithification of these mats. In addition, the intrusion of fresh to brackish water from vegas in the north, south, and west margins of Pozo Bravo (Lencina et al., 2021) favors the growth of microbial mats and provides calcium carbonate and bicarbonate ions that participate in the process of mineralization (Warden et al., 2019). As a result, the Pozo Bravo area exhibits distinct organosedimentary structures that trace microbial growth from organic soft mats to increasingly lithifying mats and finally to fully lithified microbialites. Despite their varied mesostructure (i.e., domal, discoidal, tabular, or horseshoe-like bioherms; Lencina et al., 2021), most microbialites in Pozo Bravo exhibit similar microbial community structures (Lencina et al., 2021). Vegetation in the area is concentrated in marshes as grass covers near water bodies, with sparse thorny bushes (“tolares”) and grass clumps (“pajonales”) of various genera (Moreno, 2012) across the landscape. Small reptiles, mammals such as rodents, and larger animals such as vicuña constitute the predominant vertebrate input near the lake, alongside humans from the nearby Antofalla community (Moreno, 2012).
A field campaign was carried out in Pozo Bravo during January 2019 (summer season) to collect samples from the western shore of the lake (Fig. 1D and E). After a preliminary inspection of the biological and sedimentary structures of the shore and shallow waters, three sets of samples were collected at a maximum depth of ∼10 cm, all in close proximity to each other (Fig. 1D). The samples aimed to cover a bio-to-geo gradient; they included a soft (growing) microbial mat, a lithifying mat, and a completely lithified structure (i.e., microbialite). The soft mat, picked from under the water layer, was vertically arranged in three colored layers that lay on top of dark sediment (Fig. 1F): an orange superficial layer rich in extracellular polymeric substances (EPSs) as typically secreted in high-altitude systems for protection against UV and desiccation (Gomez et al., 2014; Lencina et al., 2021), an intermediate green layer, and a dark pink/purple layer. The three colored layers were sampled separately on-site with a scoop and a spatula previously rinsed with 96% ethanol. The lithifying mat was picked farther away from the water—in drier shore conditions—and was comprised of two apparent layers, one visibly darker exposed to the atmosphere (surface or S layer) and a lighter bottom (B) layer (Fig. 1G). The microbialite (Fig. 1H) was also picked up from the dry edge of the shore. All samples were stored at 4°C in solvent-cleaned polypropylene containers until delivered to the laboratory, where they were kept frozen at −20°C prior to further downstream analyses.
At the laboratory, the lithifying and microbialite samples were separated in their corresponding layers with the aid of a high-speed Dremel 300 corded Multi-Tool equipped with a shaping metal mandrel (Dremel Mandrel 402) and thoroughly rinsed with 96% ethanol before each use. For that, the microbialite was first cut in half with a Struers Secotom-20 metallographic cutting machine (previously sterilized with 96% ethanol) to observe the inner structure. Prior to the layer separation, the lithifying mat and the microbialite cross-section were externally cleaned with methanol (MeOH) to remove any contamination from handling. Four different layers were sampled in the microbialite: the outer surface (L1), a middle layer near the outer surface (L2), a deeper, middle layer (L3), and the lowermost layer (L4), as shown in Figure 1H. The three soft mat layers and the powder from the lithifying mat and microbialite layers were lyophilized using a Lyomicron freeze drier (Coolvacuum Technologies) and ground using an agate mortar.
Mineralogical and bulk geochemical analyses
After freeze-drying, the mineralogical composition of the S and B layers from the lithifying mat and the four microbialite layers was determined by X-ray diffraction using a Bruker X-ray diffractometer (Eco D8 Advance) equipped with a Cu X-ray source (Cu Kα1,2, λ = 1.54056 Å) operating at 40 kV and 40 mA. For analysis, samples were mounted on a polymethyl methacrylate specimen holder and scanned in the 2·θ-diffraction angle from 5° to 60° employing a scanning step size of 0.02° and 1 s. The PDT-22002 database from the DIFFRAC-EVA software was used to identify the diffraction patterns.
Prior to further geochemical analyses, the soft mat and lithifying mat layers, and only two microbialite layers (L1 and L2 + L3 due to limitations in the sample amount) were treated with 2N hydrochloric acid to remove inorganic carbon from carbonate minerals. After 72 h of equilibration due to the high carbonate content in the samples, pH was adjusted to neutral values with ultrapure milliQ water; then, the samples were dried in a 50°C oven for 3 days until they attained an unvarying weight. The stable isotopic composition of the bulk organic carbon (δ13C) and total nitrogen (δ15N) in each sample was measured by isotope ratio mass spectrometry (IRMS) using a MAT 253 (Thermo Fisher Scientific, Waltham, MA, USA) following analytical methods by the US Geological Survey (Révész et al., 2012) as described in Carrizo et al. (2019). Results were reported in the standard per mill notation (‰) relative to three certified standards (USGS41, IAEA-600, and USGS40), with an analytical precision of 0.1‰. The total organic carbon (TOC%) and total nitrogen (TN%) content of the samples was determined using an elemental analyzer (HT Flash, Thermo Fisher Scientific, Waltham, MA, USA) in parallel to bulk stable isotope measurements.
Extraction, fractionation, and analysis of lipid biomarkers
Freeze-dried aliquots of ∼0.5 g (fresh mat) and ∼1.5 g (lithifying mat and microbialite) were extracted using a mixture of dichloromethane (DCM) and MeOH in a 3:1 volumetric proportion (DCM:MeOH) via ultrasound extraction in a series of three 15 min cycles. Prior to ultrasound extraction, samples doped with three internal standards (i.e., deuterated tetracosane-D50, deuterated myristic acid-D27, and 2-hexadecanol) were respectively used for the quantification of hydrocarbons, fatty acids, and alcohols. The concentrated and desulfurized lipid extracts were then hydrolyzed with potassium hydroxide (6% MeOH) and left overnight at room temperature for subsequent separation into a neutral and an acidic fraction via liquid–liquid extraction with n-hexane (details in Megevand et al., 2022). Further separation of the neutral fraction into a nonpolar (hydrocarbons) and a polar (alcohols) fraction was conducted according to a methodology broadly explained elsewhere (Sánchez-García et al., 2020b). Then, the acidic and alcohol fractions were subjected to derivatization in methanolic BF3 to generate fatty-acid methyl esters (FAMEs) and N,O-Bis(trimethylsilyl)trifluoroacetamide to generate trimethylsilyl alcohols as detailed by Sánchez-García et al. (2020a). The apolar fraction that contained hydrocarbons did not require derivatization for analysis. Procedural blanks were generated and carried through the entire extraction process to ensure that the detected lipids were indigenous to the samples.
All three fractions were analyzed via gas chromatography coupled to mass spectrometry (GC-MS) using an 8860 GC system interfaced with a 5977 BCE mass selective single-axis detector (Agilent Technologies) operating with electron ionization at 70 eV and scanning from m/z 50 to 650. Aliquots of 1 μL of each fraction were injected in splitless mode onto a 30 m DB-5MS fused-silica column (30 m × 0.25 mm internal diameter and film thickness of 0.25 μm). The GC oven was programmed to increase from 50°C at the moment of injection (held for 1.5 min) to 160°C at 20°C/min and, after isothermal hold of 5 min, increase again to 315°C at 5°C/min (held for 10 min). The injector, transfer line, and MS source temperatures were set at 290, 300, and 230°C, respectively.
Compounds were identified according to retention times and by comparing mass spectra to reference materials both in the literature and in the NIST library, provided by the MSD ChemStation software (v. 01.03.2357, Agilent Technologies). Quantification was based on external calibration curves of normal (n)-alkanes (i.e., linear and saturated alkyl chains) of length C10 to C40, n-FAMEs (even chains C8 to C24), and n-alkanols (C14, C16, C18, and C20 chains). All chemicals and standards were provided by Sigma Aldrich (St. Louis, MO, USA). The average recovery of the internal standards added prior to the organic solvent extraction was measured to be 69 ± 21%.
Compound-specific isotopic analysis
The carbon isotopic composition of individual lipid compounds was determined by GC-MS (Trace GC 1310–Ultra and ISQ QD-MS) coupled to an IRMS system (MAT 253, Thermo Fisher Scientific). For the GC analysis, the oven temperature program was set to gradually increase from 70°C to 130°C at a rate of 20°C/min and to 300°C at a rate of 10°C/min (held for 15 min). For IRMS analysis, electron ionization was set to 100 eV; Faraday cup collectors are m/z 44, 45, and 46; and the temperature of the CuO/NiO combustion interface was set to 1000°C. Samples were injected into a programmable temperature vaporizer injector in splitless mode, with an inlet temperature of 50°C, and an increasing temperature ramp of 2.5°C/min until reaching 320°C (held for 2.5 min), using helium as a carrier gas at a constant flow of 1.1 mL/min. The carbon isotopic ratio of individual lipids separated by GC was measured by comparing carbon dioxide spikes of known isotopic composition, introduced directly into the MS source, three times at the beginning and three times at the end of each run.
Reference mixtures (Indiana University, USA) of known isotopic composition of n-alkanes (A6) and FAMEs (F8) were run every three samples to validate the accuracy of the isotopic ratio determined by GC-MS-IRMS. The stable-carbon isotopic composition was expressed as the δ13C ratio, in per mill notation (‰), relative to the Vienna Pee Dee Belemnite standard. For the alkanoic acids, the δ13C values were calculated from the FAME data and corrected, accounting for the extra carbon atom added during methanolysis. The same correction procedure was applied to the alcohols but accounting for the three carbons incorporated by trimethylsilylation.
Results
Mineralogy and bulk organic composition in the Pozo Bravo mats
Mineralogical composition was only determined for the two lithifying mat samples S and B, and for the L1–L4 microbialite samples. Identified minerals included carbonates (low-magnesium [Mg] calcite, calcite, and hydrotalcite), silicates (quartz and andesine), feldspars (albite, anorthite, and labradorite), and illite, which was the only detected clay (Fig. 2). In both sample types, carbonates were the dominant minerals, accounting for 74–92% of their composition. Low-Mg (0.03%) calcite was the most abundant carbonate in the lithifying mat, whereas calcite was the most abundant in the microbialite (Fig. 2). Illite was only detected in the microbialite samples in minor abundances (<6%).
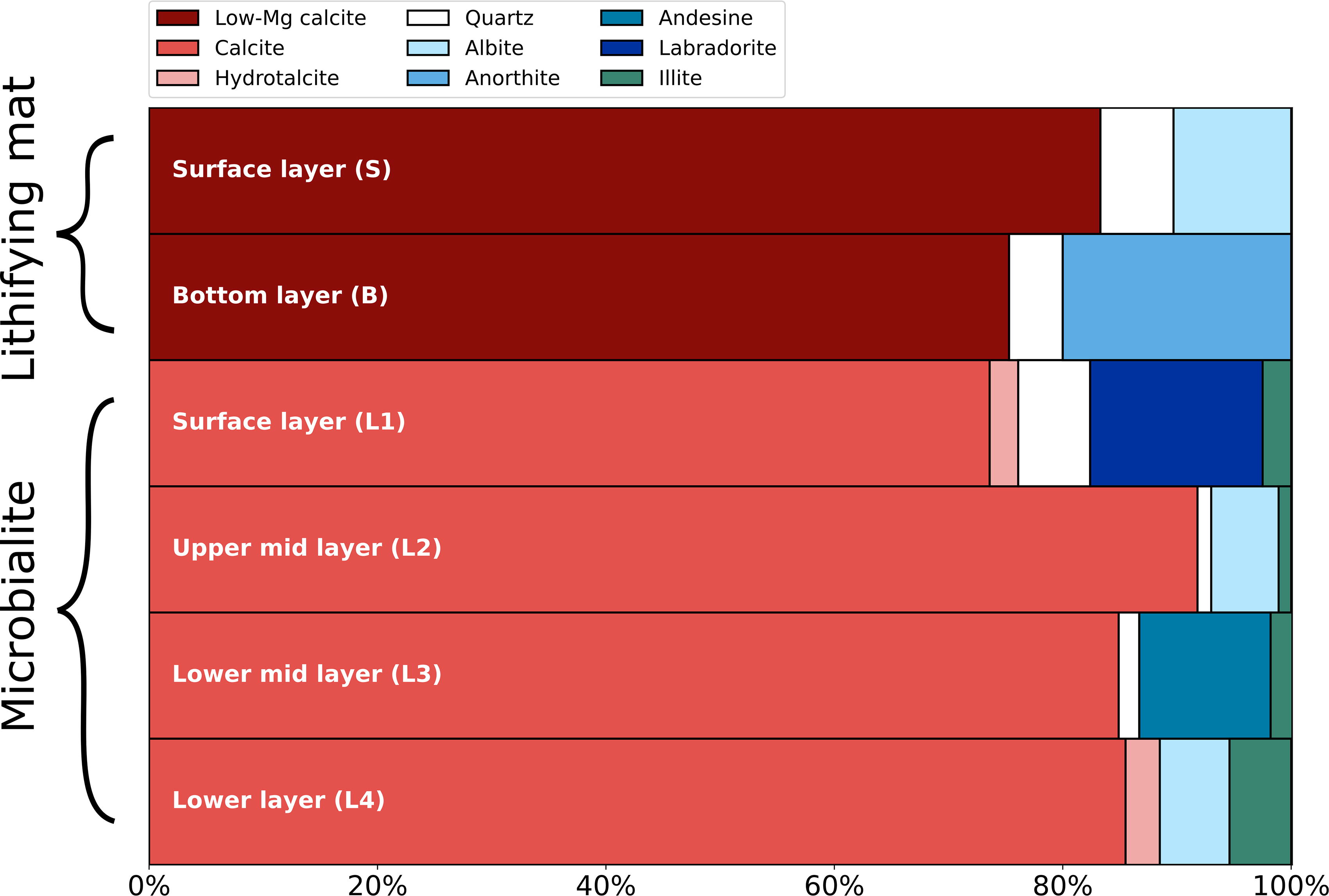
Relative abundance of minerals in the lithifying microbial mat and microbialite measured by XRD. Low-magnesium (Mg—0.03%) calcite, calcite, and hydrotalcite are grouped in red tones as carbonates. Albite, anorthite, andesine, and labradorite are all plagioclases and are depicted in blue tones. Illite was the only detected clay mineral. XRD, X-ray diffraction.
The TOC content in the Pozo Bravo samples was measured to range from 5.1% to 8.4% of dry weight (dw) in the soft mat in descending order (from the upper orange to the lower pink layer), from 1.4% to 2.2% in the lithifying mat (S and B layers, respectively), and from 0.39% to 0.71% in the microbialite (L1 and L2+L3, respectively), as shown in Table 1. TN contents oscillated between 0.06% and 1.3% across all samples (Table 1).
Bulk Organic Geochemical Parameters (TOC, TN, δ13C, and δ15N) in Samples from Pozo Bravo
In the microbialite, the two inner layers (L2 and L3) were merged into one (representing the inside of the microbialite) to ensure sufficient sample for bulk geochemical analysis. In L4, there was insufficient sample for any measurement. N/A = the measurement was not applicable.
Total organic carbon, percentage relative to total dry weight.
Total nitrogen, percentage relative to total dry weight.
Stable isotopic composition of organic carbon.
Stable isotopic composition of total nitrogen.
The bulk δ13C ratio ranged from −17 to −18‰ in the soft microbial mat, from −18 to −21‰ in the lithifying mat, and from −11 to −13‰ in the microbialite (Table 1). The bulk δ15N ratio varied from 0.57 to 1.4‰ in the soft mat, from 0.92 to 2.2‰ in the lithifying mat, and from 8.2 to 9.4‰ in the microbialite layers (Table 1).
The molecular analysis by GC-MS of the organic extracts from the Pozo Bravo samples detected the presence of several lipidic families that included linear n-alkanes, n-alkanols, and n-alkanoic acids, as well as methylated alkanes, alkenes, isoprenoids, polycyclic triterpenoids, sterols, and stanols. The diversity and concentration of lipid biomarkers varied in the three sample types depending on their soft/lithification state (Table 2).
Concentrations and Geochemical Proxies of Major Lipid Families (Alkanes, Alkanoic Acids, and Alkanols) in the Soft and Lithified Samples from Pozo Bravo
Concentrations and Geochemical Proxies of Major Lipid Families (Alkanes, Alkanoic Acids, and Alkanols) in the Soft and Lithified Samples from Pozo Bravo
Includes sterols and stanols.
Carbon preference index = ½∑(X i + X i +2 + … + X n )/∑(X i −1 + X i +1 + … + X n −1) + ½∑(X i + X i +2 + … + X n )/∑(X i +1 + X i +3 + … + X n +1) for the C23–C33 range, where X is the concentration and i is the number of carbons in the chain; Van Dongen et al., 2008.
The soft mat was the sample where lipids were detected at the highest concentration (597–777 μg·g−1 dry weight, or dw; Table 2), with compounds from the apolar fraction less abundant (65–108 μg·g−1 dw; Supplementary Table S1) than those in the polar (214–309 μg·g−1 dw) and acidic (302–385 μg·g−1 dw) fractions (Supplementary Tables S4 and S7, respectively). In the apolar fraction, n-alkanes from 15 to 31 carbons (C15–C31) showed total concentrations of 8.8 (purple layer) to 22 μg·g−1 dw (orange layer) of dw (Table 2). The dominant n-alkanes were n-C15, n-C17, n-C18, and, in the orange layer, n-C29 (Fig. 3A, left panels). Among the isoprenoids, squalene dominated in the three layers together with neophytadiene in the orange and phytene in the purple layers (Fig. 3A, left panels). Altogether, the isoprenoids accounted for 29 μg·g−1 dw within all layers of the fresh microbial mat. The polycyclic triterpenoids oleanadiene, norursatriene, serratene, and diploptene were only present in the soft microbial mat and were particularly dominant in the orange layer (Fig. 3A, left panels). Other hydrocarbons such as the monomethyl alkane 7-methyl hexadecane (7-Me C16) exhibited a relatively higher abundance in the green and purple layers. In contrast, two unsaturated alkanes (i.e., alkenes), the isomers 3- and 8-heptadecene (i.e., 17:1ω3 and 17:1ω8), dominated all three layers, with concentrations of the 17:1ω8 being the highest of all hydrocarbons detected in all layers (14–57 μg·g−1 dw) of the fresh microbial mat (Fig. 2A, right panels).
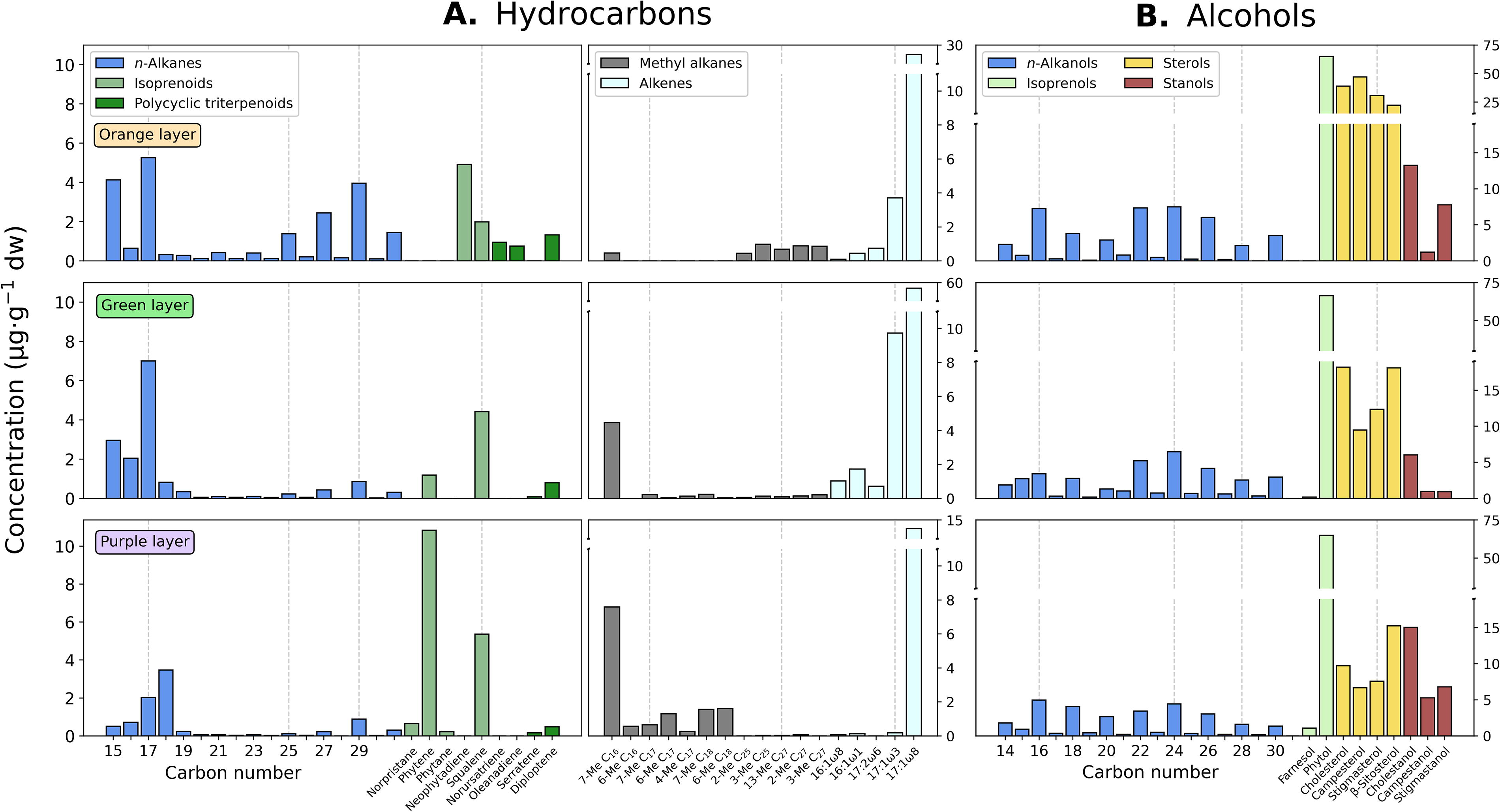
Molecular distribution of organic compounds in three different layers of the soft microbial mat.
In the polar fraction, a suite of n-alkanols (C14–C31) was detected in total concentrations that ranged from 31 to 46 μg·g−1 dw (Table 2) with a dominance of even-over-odd compounds and peaks at C16, C22, and C24 (Fig. 3B). Besides n-alkanols, the acyclic diterpenol phytol was the most abundant alcohol detected in the polar fraction (Fig. 3B); it exhibited similar concentrations across all three layers from the soft mat (65–66 μg·g−1 dw; Supplementary Table S4). The sesquiterpene farnesol, conversely, was mostly present in the purple layer (Fig. 3B) in a maximum concentration of 1.1 μg·g−1 dw (Supplementary Table S4). In addition, a series of sterols and stanols were also detected, with the former significantly more abundant than the latter in the (mostly) orange and green layers (Fig. 3B). In the purple layer, sterols and stanols were in similar proportion, with β-sitosterol and cholestanol (∼15 μg·g−1 dw) the most abundant (Supplementary Table S4).
The acidic fraction of the soft mat contained n-alkanoic acids (C12–C30) in total concentrations from 62 to 110 μg·g−1 dw (Table 2) with peaks at C14 (only in the orange layer), C16, C18, and, to a lesser extent, C24 (Fig. 4A). The n-alkanoic acids represented the most abundant lipid compounds of saturated and linear configuration. In addition, the monounsaturated fatty acid (MUFA) 16:1ω9 was also abundant in all layers, particularly in the orange one (87 μg/g dw; Supplementary Table S4), as well as 18:1ω9 in the purple layer (Fig. 4B). Other detected acidic compounds were cyclopropyl (C19), methoxy (Fig. 4A), and terminally branched (iso/anteiso) fatty acids (Fig. 4B).
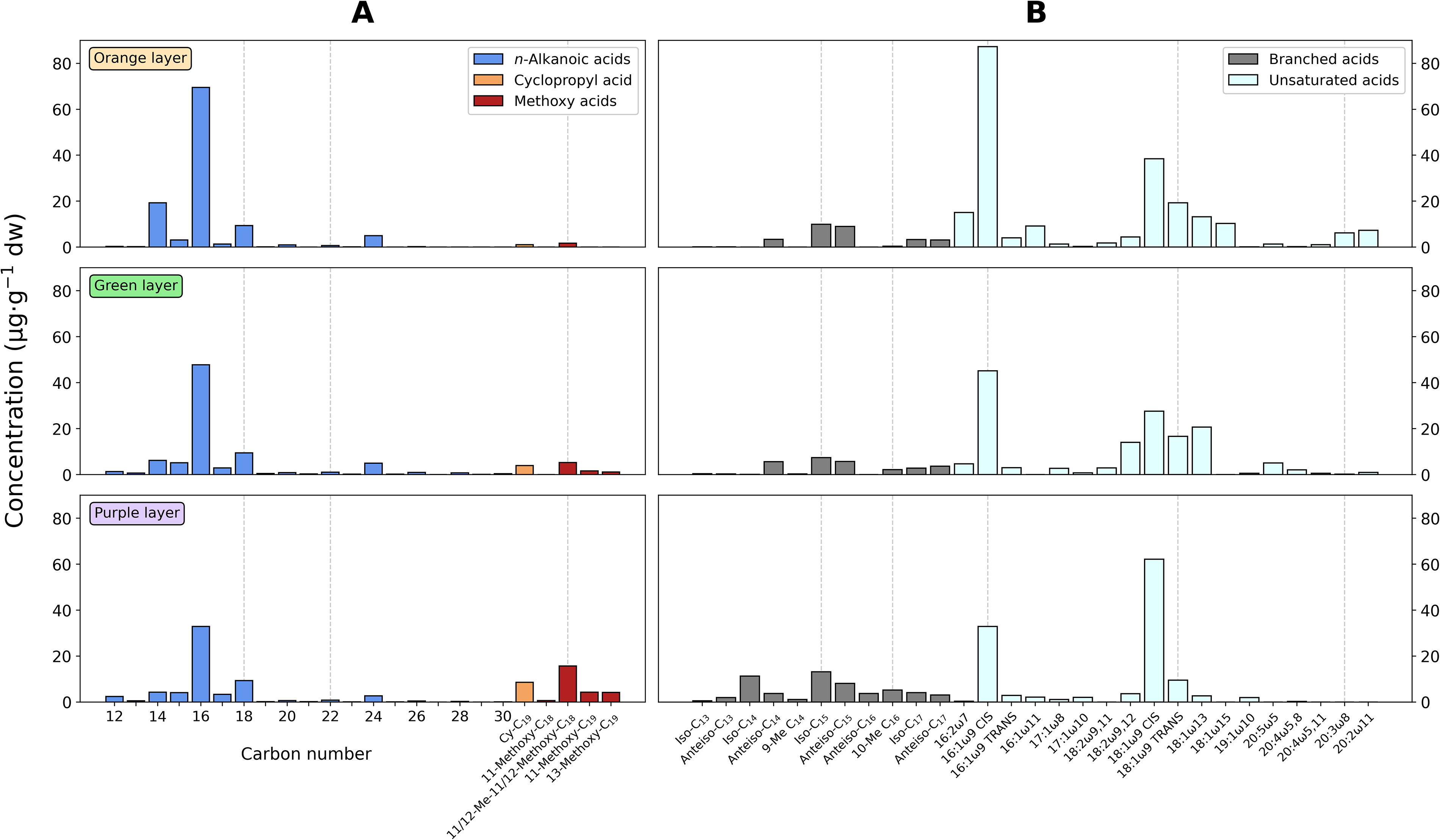
Molecular distribution of alkanoic acids in three different layers of the soft microbial mat.
The concentration of lipid biomarkers in the lithifying mat was one order of magnitude lower (44–61 μg·g−1 dw) than in the soft microbial mat (Table 2). In the apolar fraction, the total concentration of n-alkanes (C15–C31) in the S and B layers was similar (1.0 and 1.1 μg·g−1 dw, respectively) but showed different relative abundance of long versus short chains (Fig. 5A). In the S layer, the long chains dominated, and n-C29 was the most abundant congener; in the B layer, the short chains were instead more abundant and displayed a prominent peak at n-C17 (Fig. 5A, left panels). The isoprenoids squalene, pristane, and phytane were present in both samples, but pristane and phytane were considerably more abundant in the B layer (Fig. 5A, left panels). As in the soft microbial mat, the alkene 17:1ω8 displayed high abundance, particularly in the B layer (Fig. 5A, right panels).
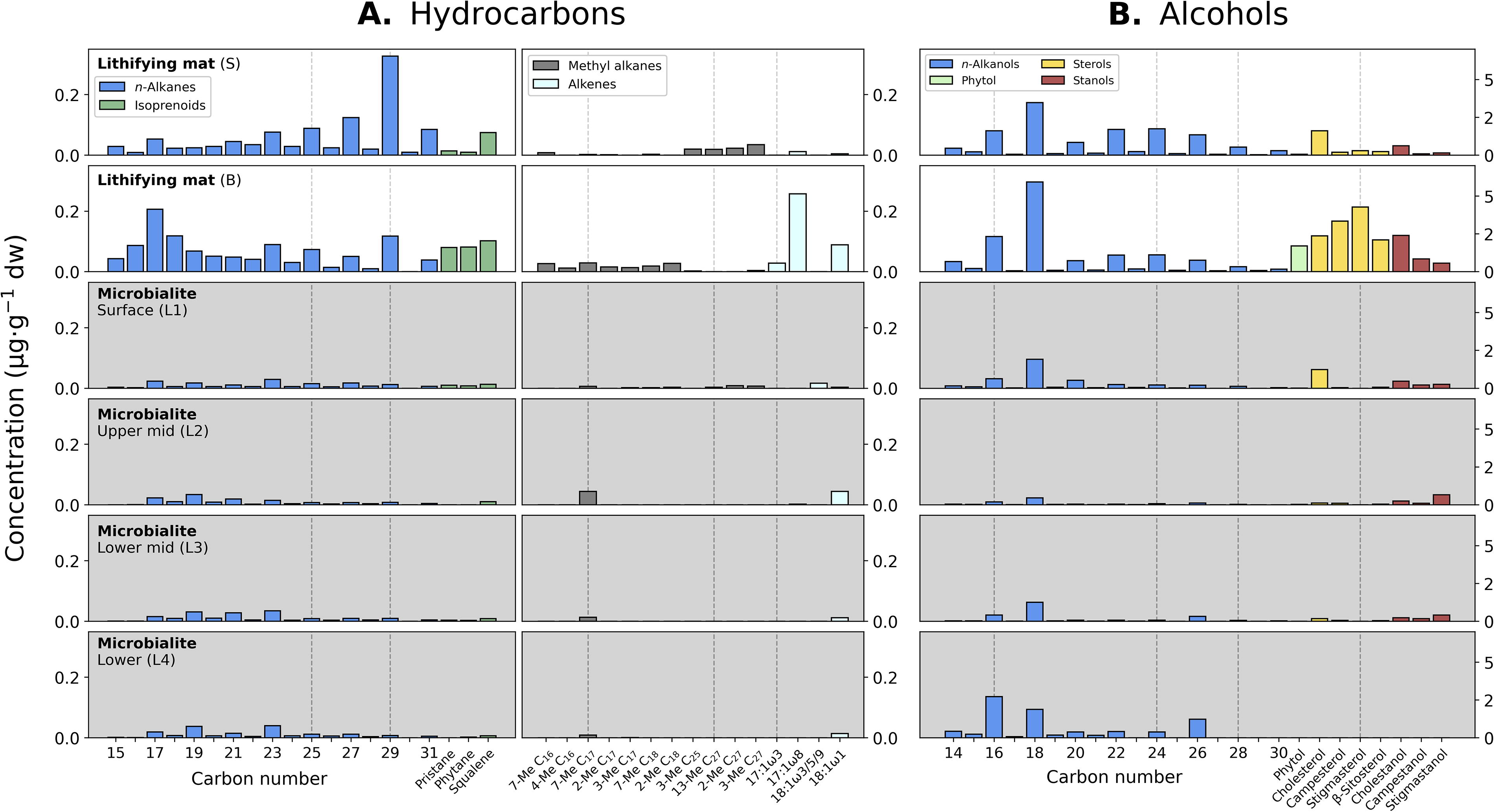
Molecular distribution of organic compounds in the lithifying mat (white background) and in the microbialite (gray background).
In the polar fraction, the n-alkanols (C14–C30) accounted for total concentrations of 13 and 15 μg/g dw in the S and B layers, respectively (Table 2). With an even-over-odd carbon chain preference, the short-chained n-C16 and n-C18 were relatively more abundant than the longer-chained n-C22, n-C24, and n-C26 (Fig. 5B, white background). Cholesterol was the most abundant sterol in the S layer, and in general, phytol, sterols, and stanols were clearly more abundant in the B layer (Fig. 5B, white background).
The acidic fraction of the lithifying mat contained n-alkanoic acids from C11 to C26 with total concentrations of 12 μg·g−1 dw (layer S) and 8.9 μg·g−1 dw (layer B; Table 2) and particularly high abundance of n-C16 and n-C18 in both layers (Fig. 6A). Other detected fatty acids, particularly in the S layer, were the cyclopropyl acid C19, a methoxy acid, the terminally branched anteiso-C15, and the MUFAs 16:1ω9 and 18:1ω9 (Fig. 6A).
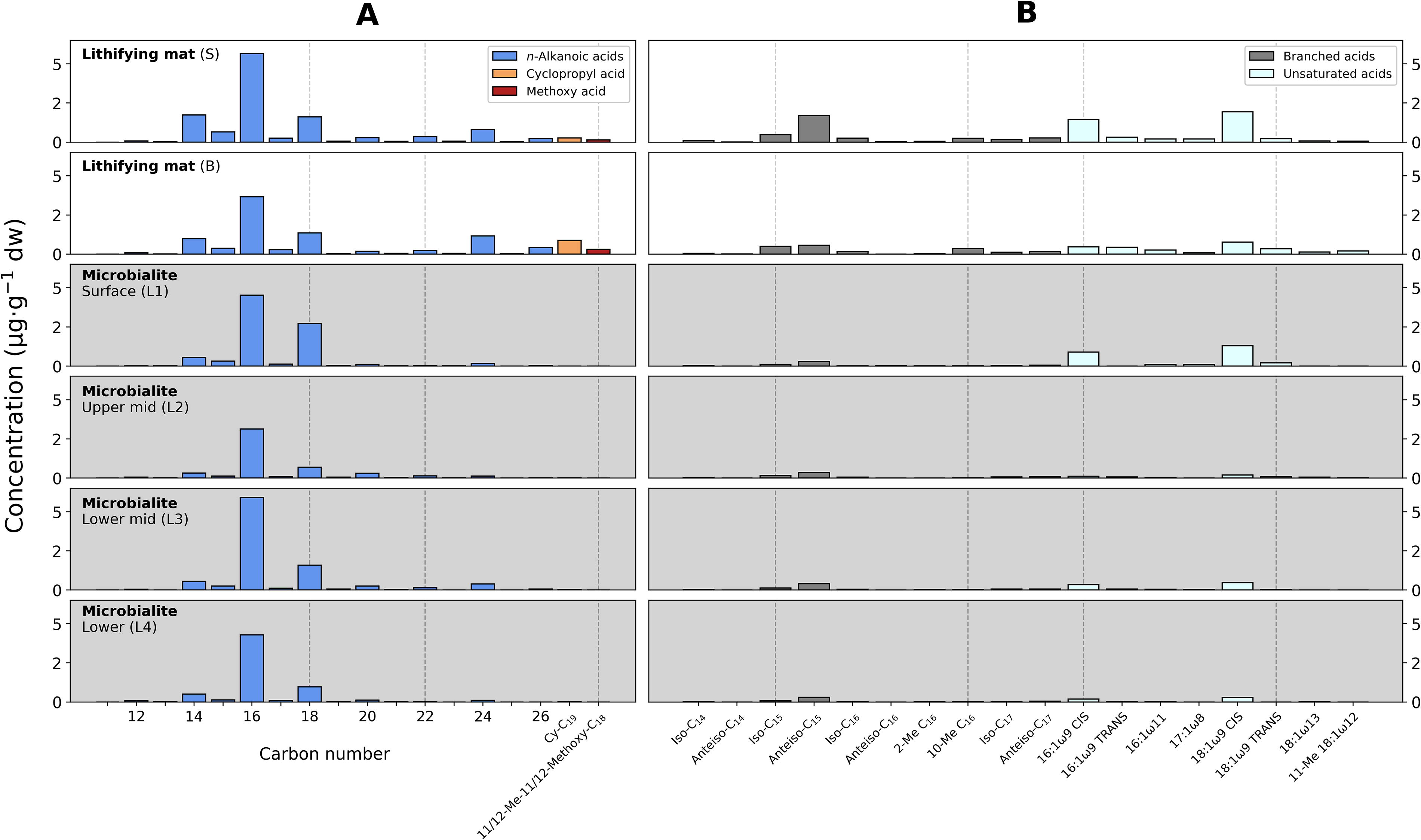
Molecular distribution of organic acids in the lithifying mat (white background) and in the microbialite (gray background).
The abundance (25–32 μg·g−1 dw) and variety (Supplementary Tables S1–S9) of lipids in the microbialite were the lowest of all three samples (Figs. 5 and 6, shaded panels). Total concentrations of n-alkanes in all microbialite layers were even lower (0.15–0.19 μg·g−1 dw) than in the lithifying mat (Table 2), isoprenoids were virtually absent (Fig. 5A, left panels), and the only other somewhat abundant hydrocarbons were 7-Me C17 and the alkene 18:1ω1 (Fig. 5A, right panels). In the polar fraction, the total concentration of the n-alkanols was also lower (1.1–12 μg·g−1 dw) than in the lithifying and (mostly) soft mat (Table 2), and excluding a distinguished cholesterol peak in L1, stanols were consistently more abundant than sterols (Fig. 5B, shaded panels). In the lowermost L4, only n-alkanols were detectable.
In the acidic fraction, total concentrations of n-alkanoic acids per layer were generally lower (5.1–9.4 μg·g−1 dw) than those of the lithifying mat (Table 2), yet the same n-alkanoic acid peaks prevailed (Fig. 6A, shaded panels). Similarly, the anteiso-C15, 16:1ω9, and 18:1ω9 acids were also detected in the microbialite in similar relative abundances as in the lithifying mat (Fig. 6B).
Stable-carbon isotopic composition of lipid biomarkers in all sample types
The stable-carbon isotopic composition of the lipids (δ13C) was measured in all samples and layers. However, lipids from the apolar and polar fractions in all layers of the microbialite and those from the acidic fraction in layer L1 of the microbialite were not abundant enough for isotopic determination, so no δ13C is reported for them. Hydrocarbons from the soft and lithifying mats showed δ13C values from −13.5 to −32.5‰ (Supplementary Table S3), with the lithifying mat generally containing more 13C-depleted compounds (i.e., more negative δ13C) than the soft mat (Fig. 7A). In the latter, the green layer had the most 13C-enriched (i.e., less negative δ13C) hydrocarbons (squalene δ13C: −13.5‰), whereas the purple layer harbored the most 13C-depleted (n-C29 δ13C: −27.4‰). In the lithifying mat, both layers S and B exhibited similar isotopic composition (Fig. 7A).
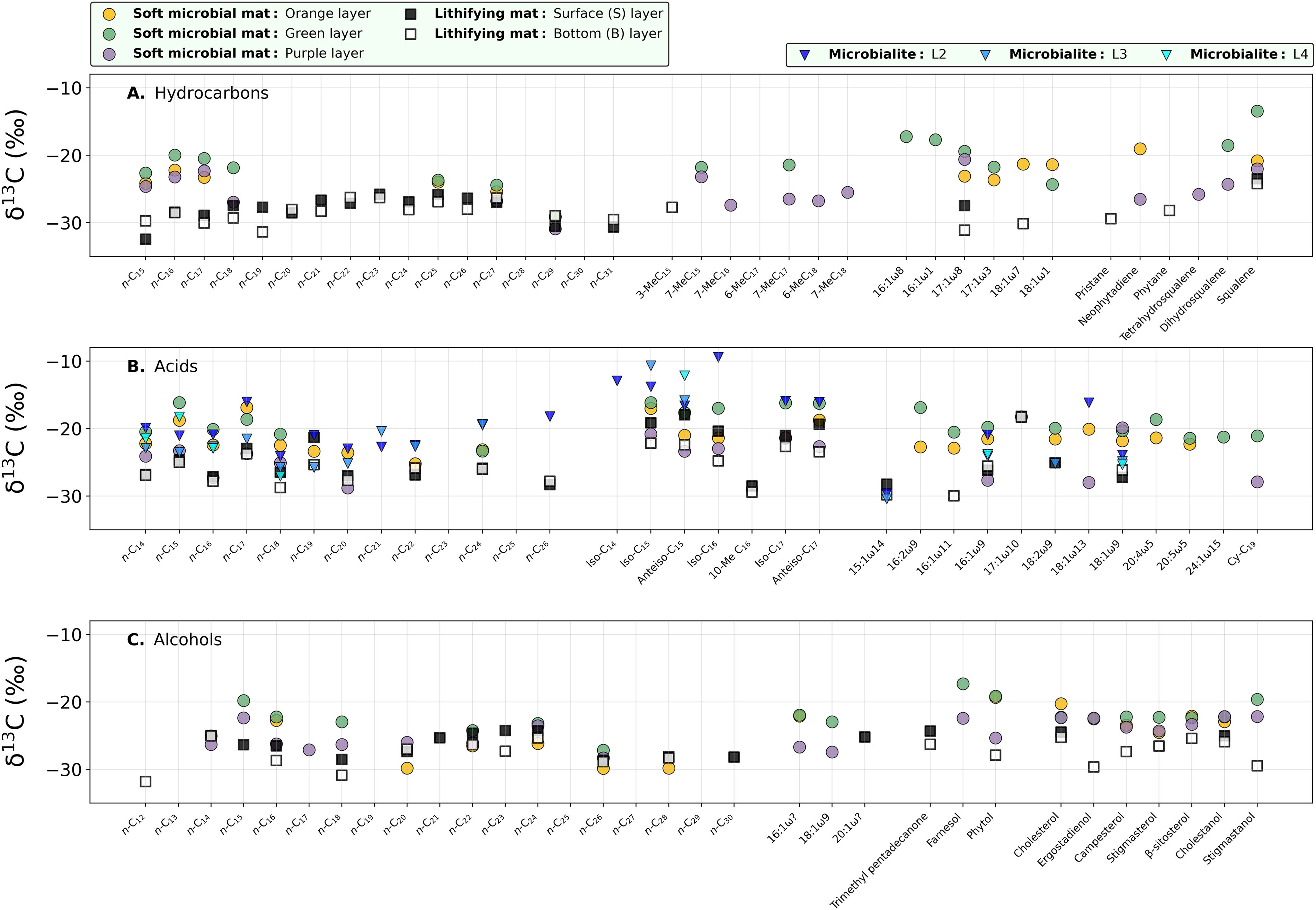
Stable-carbon isotopic composition (δ13C ratio) of the organic compounds in the three Pozo Bravo samples.
Compared with the hydrocarbons, the variation range of δ13C was wider among the alkanoic acids (−9.4 to −30.4‰; Supplementary Table S6), and like the hydrocarbons, the acids were overall more 13C-depleted in the lithifying than in the soft mat (Fig. 7B). In the microbialite, the δ13C values were generally more similar to those in the soft mat or even higher. In this sample, the most 13C-enriched acids were those of monomethylated chains, mostly iso-C16, as well as the long-chain n-alkanoic acids (Fig. 7B). Finally, the alcohols were the compounds that exhibited the narrowest δ13C range (−17.3 to −31.8‰; Supplementary Table S9). Again, the lithifying mat displayed δ13C values generally lower than those from the soft mat (Fig. 7C).
Biological sources and metabolic traits in Pozo Bravo samples
The molecular and compound-specific isotopic analyses of the lipids extracted from the Pozo Bravo samples enabled the identification of different biological sources in the lake system. In the soft mat, typical cyanobacterial hydrocarbons such as n-C17, unsaturated C17 isomers (17:1ω3 and 17:1ω8), and monomethyl alkanes ≤ C18 (Ladygina et al., 2006; Coates et al., 2014; Plet et al., 2018) were detected across all layers but with varying relative abundances between them (Fig. 3A, right panels). The particularly abundant 7-Me C16 has been frequently reported to derive from Microcoleus vaginatus (Dembitsky et al., 2001, 2000; He et al., 2015). Although M. vaginatus has not been explicitly reported in Pozo Bravo, it is a plausible candidate source due to its well-documented EPS production (contributing to the mucilaginous texture of the orange layer), halotolerance, xerotolerance, and UV resistance (Chen et al., 2009, 2003). Cyanobacterial genera that have been revealed by metagenomic analysis in Pozo Bravo include Coleofasciculus and Synechococcus, in addition to the two halotolerant genera Halothece and Halomicronema (Vignale et al., 2025). Moreover, another halotolerant genus, namely Rivularia (Nostocales order), has been previously reported in other Andean aqueous environments comparable to Pozo Bravo (Boidi et al., 2020).
Apart from the hydrocarbons, other compounds prolifically detected in all layers, such as the unsaturated acids 16:1ω9 and 18:1ω9 (Fig. 4B), were considered to be associated with cyanobacterial sources (Cohen et al., 1995; Allen et al., 2010; Coates et al., 2014), although other biological sources (e.g., green algae, diatoms, or dinoflagellates) cannot be excluded as they are highly ubiquitous acids (Derrien et al., 2017; Carvalho de and Caramujo, 2018). The presence of phototrophic microorganisms in the soft mat was supported by isotopic signatures (Fig. 7) generally representative of the Calvin cycle (i.e., δ13C values from −18 to −34‰; Hayes, 2001).
Another abundant lipid found in the soft mat was phytol, which is the esterifying component of chlorophyll α and certain bacteriochlorophylls, and in general a universal biomarker for phototrophs (De Souza and Nes, 1969; De Rosa et al., 1971; Didyk et al., 1978; Schouten et al., 2008). Its elevated concentrations in all three layers (Fig. 3B) denote a general abundance of phototrophs in the soft mat that includes cyanobacteria, as suggested by the hydrocarbon profile, as well as microalgae, whose presence in Pozo Bravo has been confirmed elsewhere (Gomez et al., 2018; Vignale et al., 2025). In addition, the considerably lower δ13C signature of phytol in the purple layer (−25.4‰) compared with the orange (−19.3‰) and green (−19.2‰) layers (Fig. 7C) suggested the additional presence of purple sulfur bacteria (PSB; Hartgers et al., 2000) in the former layer. These anoxygenic phototrophs with similarly depleted δ13C values have been reported in comparable lacustrine systems such as hypersaline ponds in Antofalla, near Pozo Bravo (Carrizo et al., 2022), in sulfidic lakes in Spain (Hartgers et al., 2000) and the Swiss Alps (Putschew et al., 1996), and in one saline basin in Nebraska, USA (Fang et al., 2006).
Other PSB biomarkers detected in the green and purple layers were the sesquiterpene farnesol (Bühring et al., 2009) and the cyclopropyl-C19 fatty acid (Grimalt et al., 1992; Thiemann and Imhoff, 1996; Fang et al., 2006), whose δ13C values were consistently more 13C-depleted in the purple layer (−22.4 and −27.9‰, respectively) than in the green layer (−17.3 and −21.1‰; Fig. 7B and C). In fact, most lipids in the purple layer were more depleted in 13C than in the orange or green layers (Fig. 7), altogether pointing toward a relevant contribution of PSB in the lowermost layer of the soft mat. Vignale and colleagues (2025) have indeed shown that the family Chromatiaceae (Halochromatium genus) is the most likely PSB representative in the Pozo Bravo soft mats. Also, methoxy-fatty acids were relatively more prominent in the purple layer (Fig. 4A). These acids have been detected in Rhizobium (Orgambide et al., 1993), Helicobacter (Inamoto et al., 1995), and Thiobacillus (Kerger et al., 1986) isolates. Still, it should be noted that the methoxy acids could also be derived from cyclopropane-containing acids by chemical alteration during the transesterification reaction to convert the fatty acids into the corresponding methyl ester derivatives (Orgambide et al., 1993).
Biomarkers related to other sulfur metabolisms such as sulfate reduction (e.g., iso/anteiso fatty acids or the 10-Me C16) were also detected in the three soft mat layers (Fig. 4B). Overall, all these biomarkers were relatively more concentrated in the purple layer, especially 10-Me C16 and the iso-C14 congener (Fig. 4B). The anoxic conditions in this layer (Vignale et al., 2025) are favorable for anaerobic metabolisms by sulfate-reducing bacteria (SRB) such as Desulfobacter (Dowling et al., 1986; Könneke and Widdel, 2003). These biomarkers exhibited isotopic signatures that were generally more enriched in 13C than the rest of the biomarkers (Fig. 7B), denoting the participation of additional carbon metabolic routes (e.g., the reverse tricarboxylic acid pathway, with δ13C values usually ranging from −8 to −18‰; Preuß et al., 1989) that are complementary to the generally predominant Calvin cycle.
Supplementary evidence for SRB presence in the purple layer includes the detection of the isoprenoid phytene (Fig. 3A, left panels), a degradation product of phytol produced anaerobically (Grossi et al., 1998) and associated with SRB in hypersaline systems (e.g., Schulze et al., 2001), including Pozo Bravo (Vignale et al., 2025) and nearby water bodies in La Puna (Carrizo et al., 2022). Squalene, common in microbial mats (Dobson et al., 1988; Rontani and Volkman, 2005; Scherf and Rullkötter, 2009; Allen et al., 2010; Słowakiewicz et al., 2016), was detected in all layers (Fig. 3A, left panels). Although squalene is concurrent in archaea in hypersaline settings (Tornabene et al., 1979, 1969; Brassell et al., 1981), it can be quite common across biological systems (Brocks and Summons, 2003; Micera et al., 2020). The faint detection of its unsaturated homologues dihydrosqualene and tetrahydrosqualene, typical in archaea (Rontani and Volkman, 2005; Stiehl et al., 2005), minimizes the possibility of squalene from the purple layer being derived from archaea. This agrees with the metagenomic survey of Pozo Bravo soft mats where archaeal presence was reported to be minimal (Vignale et al., 2025).
Besides prokaryotes, other eukaryotic biological sources also contributed to the lipid profile of the soft microbial mat. For instance, long-chain n-alkanes (≥n-C25) with a general preference for odd carbon chains and n-alkanols with preference for even chains were detected mostly in the orange layer (Fig. 3A and B). They are considered ubiquitous components of land plants and leaf epicuticular waxes (Rieley et al., 1991; Ficken et al., 2000; Killops and Killops, 2005; Derrien et al., 2017) and are more or less specific of higher plants such as shrubs (n-C27 and n-C29), among others (Zech et al., 2013). Also, phytosterols such as campesterol, stigmasterol, and β-sitosterol were considerably more abundant in the orange layer than in the green or purple layers (Fig. 3B). Although they can also be found in microalgae and cyanobacteria (Volkman et al., 1998), these sterols are largely produced by terrestrial plants (Wen-Yen and Meinschein, 1976; Volkman, 1986; Volkman et al., 1987) and are thus considered plant biomarkers. Similarly, diploptenes are a pentacyclic triterpene common in membranes from bacterial phototrophs (De Rosa et al., 1971; Sakata et al., 1997) but also reported in algae (Venkatesan, 1988; Yamada et al., 1997), macrophytes (Ageta and Arai, 1983), ferns (Ageta and Arai, 1983), and mosses (Huang et al., 2010). Finally, the terrigenous and in particular higher plant-derived biomass accumulated in the orange layer was further supported by the detection of the pentacyclic triterpenes norursatriene, oleanadiene, and serratene mostly associated with plant sources (Arie et al., 2020; Boonya-udtayan et al., 2019; ten Haven and Rullkötter, 1988; Fig. 3A, left panels).
The relative enrichment of diploptene, together with the three phytosterols and the long-chain n-alkanes in the uppermost orange layer, points to a common allochthonous origin (i.e., land plants) for these biomarkers. Material from the abundant shrubs and/or marsh-forming herbaceous plants that surround the lake (Moreno, 2012) can be easily introduced via aeolian action into the lake and deposited on the mat surfaces. Accordingly, the concentration of the aforementioned terrestrial biomarkers decreased downward through the soft mat layers as the contribution of land plants dilutes among relatively more prominent sources (i.e., microbial) in the green and purple layers (Fig. 3A, left panels). Finally, animal contribution can be inferred from the abundant presence of cholesterol (Volkman, 1986) in all layers but especially in the orange layer (Fig. 3B). Different animal sources including crustaceans, insects, or other arthropods, as well as mammals such as the autochthonous vicuña or abundant small rodents, could contribute to the presence of cholesterol in the lake.
Overall, the identification of biological sources based on lipid biomarkers was generally in agreement with those derived from microscopic or metagenomic studies conducted by others on the soft mats from Pozo Bravo. In contrast, scanning electron microscopy of said mats revealed copious diatoms in the EPS layer of a Pozo Bravo soft mat (Lencina et al., 2021; Vignale et al., 2025), which is compatible with our detection in the orange layer of abundant phytol and related isoprenoids (neophytadiene and diploptene; Fig. 3A, left panels), eukaryotic sterols (Fig. 3B), and microalgae-associated polyunsaturated fatty acids (Fig. 4B). In contrast, metagenomic analysis of a whole soft mat in Pozo Bravo revealed not only various genera of eukaryotes inhabiting the orange EPS layer, including kinetoplastid flagellates (Rynchomonas) and phototrophic diatoms (Phaeodactylum, Amphora, Nitzschia), but also several other phyla of free-living amoebae (Discosia) and oxygenic green and red algae (Vignale et al., 2025). The presence of these organisms was supported by the higher abundance of sterols (Volkman, 2005, 1986) that were observed in this layer (Fig. 3B).
In addition, Vignale et al. (2025) also reported various cyanobacterial genera coexisting with other aerobic and facultative guilds in the green phototrophic layers, consistent with our detection of cyanobacterial biomarkers (e.g., n-C17 and unsaturated counterparts such as 17:1 and 17:2; Fig. 3A). Furthermore, our detection in the purple layer of PSB biosignatures (i.e., moderately δ13C-depleted phytol or the cyclopropyl C19 fatty acid) and SRB biomarkers (i.e., 10-Me C16 and iso/anteiso fatty acids) is consistent with the presence of 10 different PSB genera and 15 different SRB genera reported by Vignale et al. (2025), altogether attesting to the coexistence in this predominantly anoxic layer of sulfur oxidizers and reducers.
Most lipids in the soft mat were also found in the lithifying mat and microbialite at lower concentrations; this follows a trend of decreasing lipid biomarker diversity and 13C-enrichment from soft mat to microbialite (Table 2 and Fig. 7B). In the lithifying mat and microbialite, the presence of land plant biomarkers (odd long-chain n-alkanes) was more prominent in the surface layers (S in the lithifying mat and L1 in the microbialite) than in the rest of their vertical profiles (Fig. 5A). In contrast, in the lower layers of both samples, the proportion of microbial biomarkers (short-chain n-alkanes, alkenes, isoprenoids, phytosterols, and the cyclopropyl C19 acid) of marked phototrophic nature (phytol and derived pristane and phytane) prevailed (Figs. 5 and 6). Overall, the microbialite was the most impoverished in lipid biomarkers, with a virtual absence in some of its layers of certain compounds detected in the soft and lithifying mats (i.e., photosynthesis-related isoprenoids, some alkenes and unsaturated acids, sterols, cyclopropyl C19, or methoxy acids; Figs. 5A and 6).
Degradation of lipid biomarkers in Pozo Bravo samples in the transition to the geological record
Labile features that suffered the most degradation in the transition to the geosphere were double bonds (C = C) or unsaturations. Unsaturated compounds such as alkenes (Fig. 8B) and unsaturated fatty acids (Fig. 9B) exhibited some of the greatest decreases in concentration between the soft and the lithified samples. The average concentration of alkenes in the lithifying mat decreased 99% relative to the soft mat, and the microbialite was further depleted by 86% relative to the lithifying mat (Fig. 8B). Similarly, 98% of the unsaturated fatty acids were lost in the transition from soft to lithifying mats and an additional 68% to the microbialite (Fig. 9B).
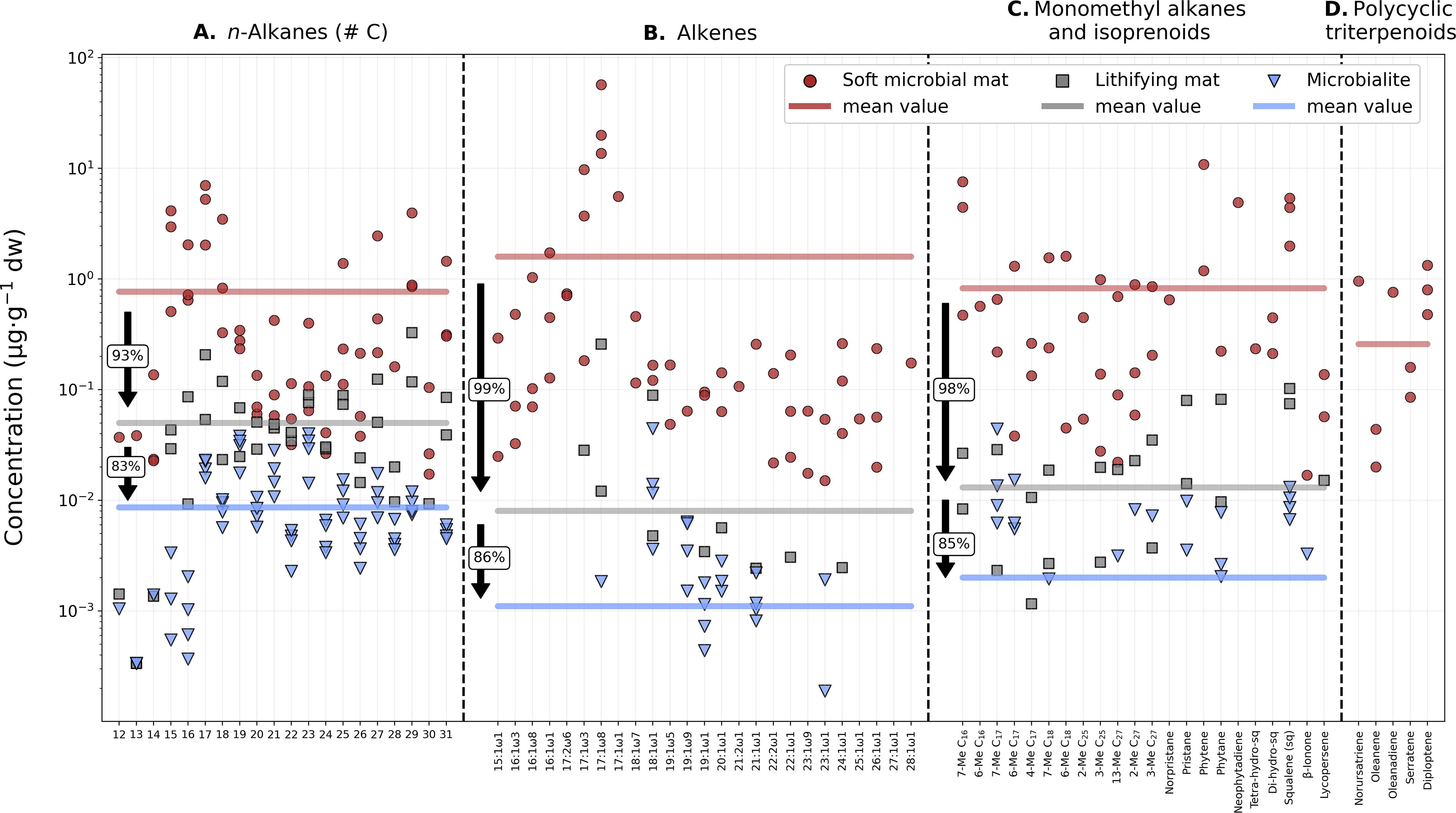
Decreasing concentration of hydrocarbons from the soft mat to the microbialite.
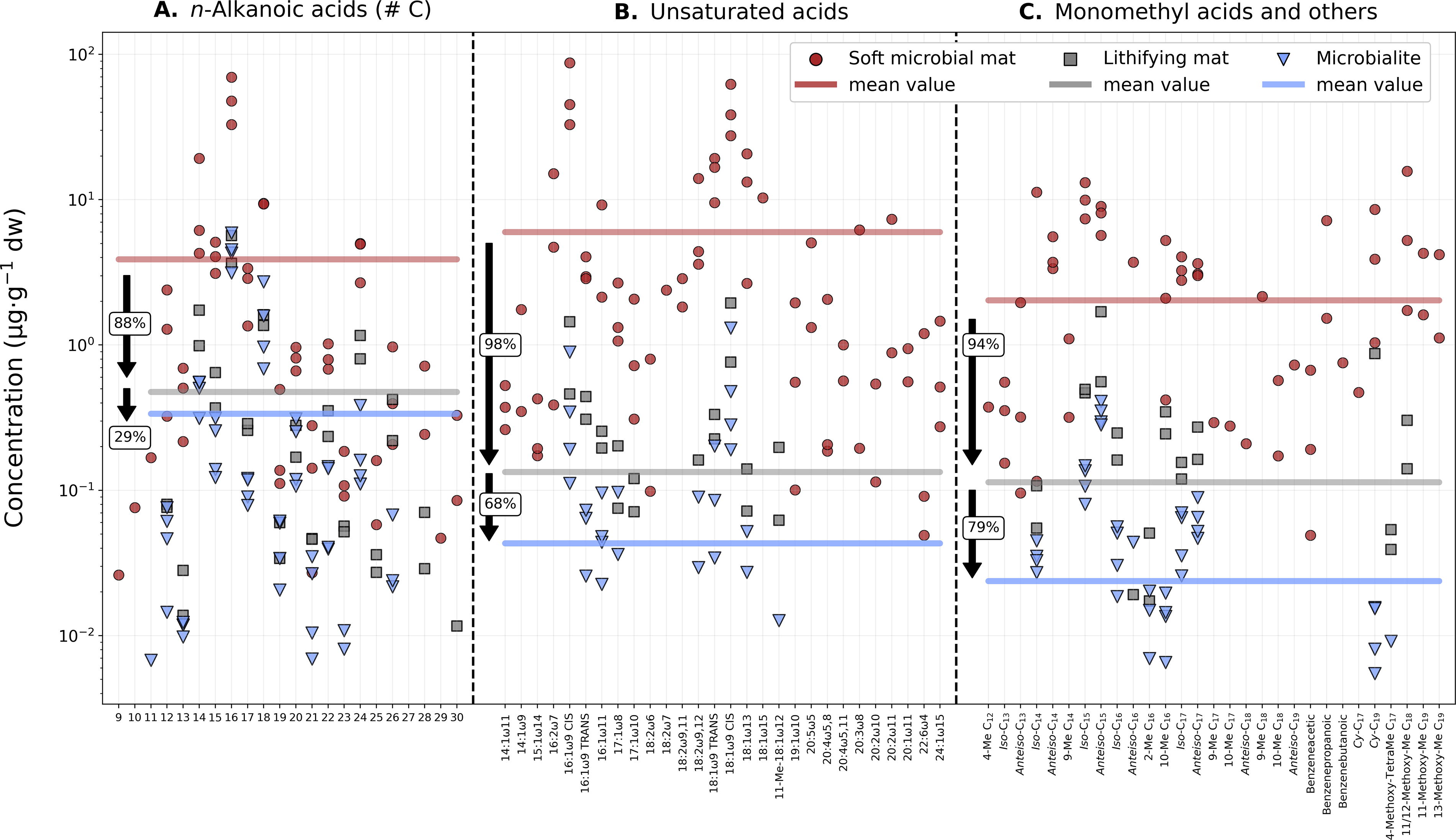
Decreasing concentration of organic acids from the soft mat to the microbialite.
Double bonds are of biological relevance for four reasons: (i) anaerobic and aerobic biosynthesis of unsaturated lipids is widespread (Marrakchi et al., 2002; Feng and Cronan, 2011); (ii) their presence may affect membrane fluidity and permeability, allowing adaptation to extreme conditions (Georgiou and Deamer, 2014); (iii) they allow for distinctive biological structures such as geometrical isomerism (cis/trans configurations) or archaeal isoprenoids; and (iv) they stabilize molecules via electron delocalization (conjugation and/or resonance), which is key for the correct functioning of biological molecules (e.g., aromaticity, pigmentation, and signal transduction). The loss of double bonds in lipid compounds is a common example of early diagenesis in progress. This has been reported in lipids subjected to silica sinter entombment (Pancost et al., 2005; Williams et al., 2021; Sánchez-García et al., 2023), encapsulation in ferrihydrite (Tan and Sephton, 2021), artificial thermal stress (Salter et al., 2022), entrapment in deep-sea carbonates (Birgel et al., 2006), or in a freshwater microbialite (Johnson et al., 2018).
The processes responsible for double bond loss in early diagenesis are varied and depend on the environment in which lipids are preserved. Microbial selective reduction of hydrocarbons (hydrogenation; Ohta et al., 1985) and oxidation (Hou, 1995) are major factors that lead to the loss of double bonds. Moreover, early abiotic transformations that involve reactions approaching intramolecular thermodynamical stability (De Leeuw et al., 1989) can also result in double bond loss. For example, hydrogenation of double bonds via low temperature disproportionation processes is a widespread scenario where common carbon compounds (e.g., formic acid and isopropanol) act as reductants (H donors) that disassemble the C = C bond (Watts and Maxwell, 1977; Walters et al., 2010; McGlynn et al., 2020), often catalyzed by mineral surfaces. In late diagenesis and catagenesis, high temperatures (50–150°C) may supply sufficient energy to break double bonds (homolytic cleavage), although such conditions usually involve deep burial within the million-year scale and therefore have not influenced our Pozo Bravo samples.
In the Pozo Bravo high-altitude context, however, it is important to consider the effects of UV-B light on unsaturated molecules. The dissociation energy necessary to dissociate a carbon double bond within a stable alkyl chain is roughly equal to 728 kJ/mol (Blanksby and Ellison, 2003). In terms of photon energy, we can use the Planck equation (
Instead, UV light largely affects organic molecules indirectly through photo-oxidation. This process, to some extent, is dependent on photosensitizing agents, which are molecules (e.g., pigments and aromatic hydrocarbons) or mineral surfaces (e.g., illite clays—Fig. 2) that are efficient at absorbing UV light. Upon UV light absorption, photosensitizing agents in the surrounding environment transfer the absorbed energy to molecular oxygen (Foote, 1976; Rontani and Belt, 2020). This process triggers the formation of reactive oxygen species (ROS) such as singlet oxygen, as well as the hydroxyl radical, which will preferably oxidize the unsaturated sites of a given molecule. When ROS reacts with the carbon double bond, the oxygen can be inserted into the chain to form new functional groups (e.g., hydroxyl). Additionally, the molecule can be cleaved at the C = C bond site (oxidative scission) into carbonyl products (e.g., ketones, aldehydes, and carboxylic acids). In either case, the C = C bond is lost in most instances; therefore, the amount of unsaturated compounds is expected to decline over time, especially in a high-altitude environment subjected to high UV flux. While photo-oxidation could theoretically play a role in degrading the organic matter within our samples, our data do not provide direct evidence for this mechanism as the primary driver of double bond loss in the form of new generation of ketones or aldehydes or concentration increase of existing alkanoic acids or alcohols (Figs. 8B and 9B). Instead, other degradation pathways may also be involved as the samples progress in the gradient of lithification.
While the unsaturated chains dilute over time, saturated molecules remain at higher concentrations, since they are less labile and not as prone to electrophilic attack as their unsaturated counterparts (Tobias and Soffer, 1985). Saturated chains, by definition, contain only single bonds whose cleavage requires further energetic inputs. Nonetheless, structural elements such as methyl groups (CH3) bound as “branches” by a single bond to the aliphatic chain might still serve as weak links of the chain, as the associated tertiary CH bonds are weaker and thus easier to oxidize than those in the methylene (CH2) units (Clayden et al., 2001). Methyl branches in hydrocarbon chains, fatty acids, or fatty alcohols provide a significant biological identity to the lipid in question, as they are added through enzymatic action in the form of side branches, in contrast to the linear growth characteristic of abiotic pathways such as Fischer–Tropsch reactions (McCollom et al., 1999; Holm and Charlou, 2001). For that reason, regular methyl branching along a chain or within a specific compound carries additional biogenic value upon long-term preservation.
In this study, we observed a 98% decrease in the average concentration of monomethylated alkanes in the lithifying mat relative to the soft mat (Fig. 8C and Supplementary Fig. S2) and an 85% decrease in the microbialite relative to the lithifying mat (Fig. 8C). Monomethylated fatty acids in the lithifying mat exhibited a 94% decrease in concentration compared with the soft mat, with the microbialite showing a further 79% depletion relative to the lithifying mat (Fig. 9C). In the case of the methylated alkanols, the decrease in methyl groups relative to the soft mat was 98% in the lithifying mat, with only an additional 9.9% decrease in the microbialite (Fig. 10B). The elevated amounts of methyl-hexacosanol in the microbialite (Me-C26; methyl position unknown) may explain the notable similarity in average concentrations between the lithifying mat and the microbialite (Fig. 10B).
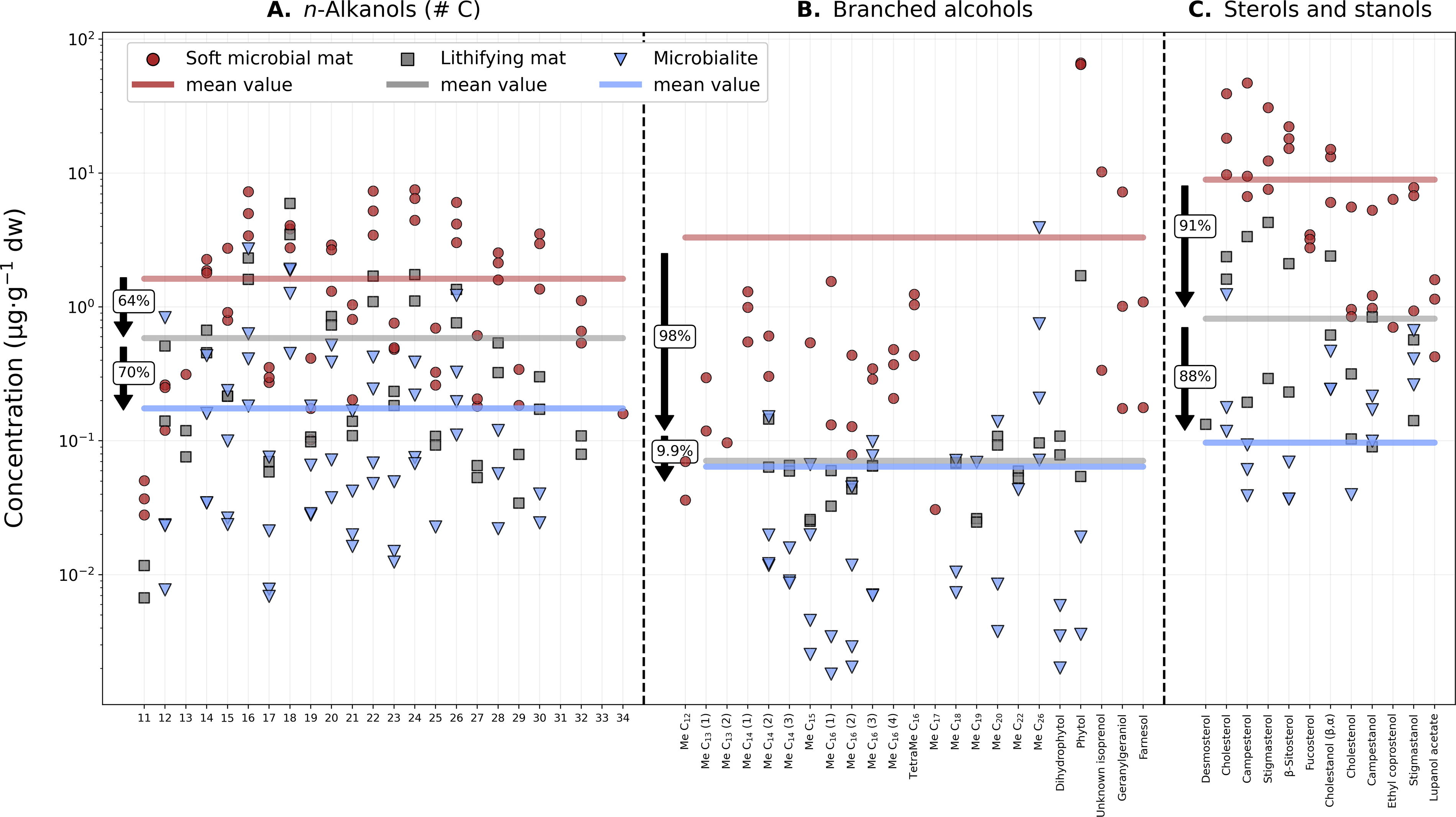
Decreasing concentration of alcohols from the soft mat to the microbialite.
Consistent with our findings, a decrease in methylated lipid chains has also been reported when comparing a fresh biofilm with its silicified version in studies aimed at elucidating key taphonomic pathways. In these studies, other structural features such as cyclopropyl side chains were also shown to degrade (Sánchez-García et al., 2023). In our samples, the Cy-C19 fatty acid (but not its congener Cy-C17) was still detectable both in the lithifying mat and microbialite although at very low concentrations in the latter (Fig. 9C). For individual compounds such as squalene and phytol, the decay observed along the lithification gradient (Figs. 8C and 10C) may be attributed to the combined presence of labile structural features including methyl branching and double bonds.
The pentacyclic triterpenoids (diploptene, norursatriene, oleanadiene, and serratene) did not survive the transition from the soft mat to the lithifying mat and microbialite (Fig. 8D). In fact, none of the typical early diagenetic products of diploptene—referred to as geo-hopanes (e.g., norhopanes, diashopanes, and desmethylhopanes; Simoneit et al., 1998)—were detected in the lithified samples. Despite being considered as highly recalcitrant biomarkers (Summons et al., 1999), the absence of pentacyclic hydrocarbons in both of our lithified samples could be largely explained by their relatively low abundance in the system (i.e., the soft mat; Fig. 3A, left panels), which is determinant, as the slightest degradation they may have undergone during lithification could have been sufficient to degrade these congeners until undetectable. Three factors can explain the low abundance of pentacyclic triterpenoids in the soft mat: (i) pentacyclic triterpenoids are generally biosynthesized in minor quantities (Simoneit et al., 1998; Tritz et al., 1999), (ii) microbial or early diagenetic demethylation of pentacyclic triterpenes in metabolically active mats is well documented (He et al., 2018), and (iii) the elevated humidity at the high-altitude lake combined with intense UV-B radiation can lead to the formation of water-derived oxidizing radicals that indirectly photolyze and degrade these molecules in the matrix and thus prevent their accumulation. This has been proven in a laboratory using gamma irradiation in shales with more or less interlayer water content, the former being a more hostile matrix for hopanoid survival (Roussel et al., 2022).
Among the polycyclic terpenoids, sterols and stanols showed the greatest concentration decrease during lithification, with a 91% drop from the soft to the lithifying mat and an additional 88% drop from the lithifying mat to the microbialite (Fig. 10C). This is probably due to the presence of multiple labile features (double bonds, methyl branches, and hydroxyl groups) that make these molecules prone to degradation.
Linear and saturated (normal) chains also exhibited noteworthy concentration decays, although less substantial than those displayed by their unsaturated and branched counterparts. For instance, n-alkanes from the lithifying mat showed an average 93% decrease relative to the soft microbial mat, whereas microbialite-derived n-alkanes experienced a further 83% reduction compared with the lithifying mat (Fig. 8A). These hydrocarbons were the most recalcitrant during the bio-to-geo transition (Fig. 8), reflecting the greater potential of the simplest hydrocarbon skeletons for long-term preservation in the geological record (Brocks and Summons, 2003; Brocks et al., 2005; Brocks and Banfield, 2009).
Similar to the n-alkanes, n-alkanoic acids were the organic acids that best resisted the transition from the biosphere to the geosphere (Fig. 9). Their average concentration decreased by 88% from the soft to the lithifying mat and by another 29% in the microbialite (Fig. 9A). The third family of normal compounds, the n-alkanols, displayed the smallest decrease in their average concentration from the soft to the lithifying mat (64%), followed by a further 70% decrease in the microbialite (Fig. 10A). Compared with the n-alkanes, both n-alkanoic acids and n-alkanols are relatively more labile lipid families due to the presence of O-containing carboxyl and hydroxyl groups that render these molecules more susceptible to degradation than their n-alkane counterparts (Bordenave, 1993; Volkman, 2005). However, their terminal functional groups also confer valuable biomarker identity to acids and alcohols, which offers crucial insight into past biogenic activity when detected in the geological record (Sánchez-García et al., 2023).
A study that employed a similar workflow to ours, which compared fresh biofilm samples with silicified samples (preserved in siliceous sinter), reported an average concentration decay in n-alkanoic or fatty acids of 83% from fresh to silicified samples (Sánchez-García et al., 2023). This closely aligns with our observed 88% decay from soft to lithifying mat. The study by Sánchez-García and colleagues reported a degradation rate that ranged from 0.01 to 84 μg·g−1/year, which translates to a maximum preservation span of up to 1.2 Gyr (Sánchez-García et al., 2023) and rivals the billion-year old resilience of n-alkanes (Brocks et al., 2005). In the context of Mars exploration, fatty acids have demonstrated notable preservation in hyperarid (Wilhelm et al., 2017) and iron sulfur-rich environments (Tan et al., 2018). Additionally, experiments that assessed lipid preservation in progressively heated ferrihydrite matrices found that fatty acids were the most resistant compounds, which strengthens their suitability for life-detection missions (Tan and Sephton, 2021). While Mars has been a primary focus, icy moons such as Europa and Enceladus are also of significant interest in the search for life. In this context, laboratory hydrothermal processing of microorganisms at high temperatures (300°C) has shown that fatty acids can succesfully retain their biogenic fidelity (Salter et al., 2022), which is a valuable property not only in light of the upcoming exploration missions to Europa but also for the thermal processing of analytical instruments (e.g., mass spectrometers) in future Mars missions.
Comparing lipids that have retained their molecular integrity through lithification (preservation in a lithifying mat) and through their entry into the geological record (microbialite) with those that have undergone evident degradation allows us to draw relevant conclusions regarding lipid preservation in carbonate matrices. This also offers preliminary insights applicable to the search for ancient, extinct life on Earth and Mars. Our results highlight that unsaturated or methylated structures are the most vulnerable to degradation along the bio-to-geo gradient, mostly due to their higher lability. In contrast, more recalcitrant, saturated chains (n-alkanes, n-alkanoic acids, and n-alkanols) were less affected by degradation. This is largely due to their lower lability and greater relative abundance, both in this lacustrine system and in the biosphere in general. Therefore, linear saturated chains stand out as the most reliable biomarkers for long-term preservation in appropriate mineral matrices (i.e., the most representative “geolipids”). In contrast, more labile compounds such as unsaturated fatty acids, isoprenoids, or pentacyclic triterpenoids might be better candidates from a diagnostic standpoint (i.e., unambiguous biological origin), even though they may be limited by a shorter time window of applicability due to their greater susceptibility to degradation.
Two key factors influence the detection of certain biomarkers in the geological record: the initial biomass concentration and the geochemical properties that favor the preservation of specific molecular structures. Both significantly affect the time window to detect traces of biomarkers over extended periods. To accurately assess preservation, understanding the physicochemical environment (Seong-Joo et al., 2000) that governs the Antofalla and Pozo Bravo regions, along with the characteristics of their microbial consortia, is crucial. Factors such as UV-B incidence, temperature shifts, water chemistry (and flow rate), and alkalinity shape the steep chemical gradients that influence the growth of microbial communities (Stolz, 2000). These extreme conditions are particularly significant in the high-altitude, hypersaline environment of the Antofalla region (Carrizo et al., 2022), where soft mats exhibit a characteristic structure that includes a protective surface layer of EPS, an upper phototrophic layer, and deeper anoxic layers (Stolz, 2000), which is consistent with previous observations in Andean hypersaline lakes (Gomez et al., 2014), including Pozo Bravo (Lencina et al., 2021; Vignale et al., 2025).
A key process that drives the preservation of biomarkers is lithification. Under favorable conditions, soft microbial mats undergo mineralization, initiating gradual lithification, which is essential for the long-term preservation of organics. Based on the inferred biological sources that dominate the different layers of our soft mat, along with what is described in the literature in comparable environments, a likely mechanism driving the precipitation of calcium carbonate (
Interestingly, while the process of alkalinization favors the presence of bicarbonate (
As lithifying mats trap sediment and precipitate minerals, they gradually form a laminated structure that characterizes microbialites (Seong-Joo et al., 2000). In this process, the geochemical properties of the carbonate matrix play a crucial role in biomarker preservation, often favoring certain lipid families. The structure of our microbialite from Pozo Bravo mainly consists of amorphous micritic laminae with irregular thick layers (Fig. 1H), where nanoscale carbonate crystals encapsulate organic matter during precipitation (Gomez et al., 2014). Lipids physically entrapped within these carbonate microcrystals likely benefit from enhanced protection against UV-B radiation and oxidizing agents; thus, the microcrystals offer some defense against degradation. ever, early diagenesis can partially compromise this intracrystalline protection. Processes such as micritization or recrystallization of the carbonate matrix may alter the encapsulated lipids’ integrity, which may explain the considerably lower concentrations of distinct lipid biomarkers observed in the microbialite compared with the lithifying and soft microbial mats.
Another potential mechanism for biomarker preservation during lithification involves the interaction between organic molecules and the carbonate matrix at an intercrystalline level. These interactions can occur through physical entrapment in less protective, noncrystalline pore spaces or through adsorption onto the mineral phase via forces such as van der Waals interactions, H bonding, electrostatic attractions, and, more rarely, covalent bonding. In covalent interactions, certain lipids can form organomineral complexes, where hydroxyl groups establish H bonds with dissolved carbonate ions, or where carboxyl groups ionically bind
Overall, various factors influence the effectiveness of organic matter preservation, emphasizing the importance of understanding the (bio)geochemical and physicochemical dynamics of the microenvironment. This understanding not only enhances the ability to predict biomarkers that represent ancient life but also aids in reconstructing past microbial communities. Additionally, recognizing which chemical interactions are most favorable for successful preservation between the geochemical setting, the mineral matrix, and certain biomarkers is vital for astrobiological missions to Mars. Selecting regions where biomarker preservation is likely will maximize the chances of capturing evidence of past life and will extend the time window through which the past can be explored.
Conclusion
Understanding the intricacies of taphonomy in lipid biomarkers and how they transform over time as they transition into the geological record is key to accurately interpreting and distinguishing the molecular fingerprints of ancient life. These, however, are elusive signatures that can be easily altered or obscured by diagenesis. The work presented here is of particular relevance for origin of life studies and for the search for extinct life on Mars, where distinguishing potential biomarker fragments from a pool of simple compounds of unknown origin can mean the difference between making a groundbreaking discovery or encountering false negatives.
Here, we have assessed the alteration and degradation of lipid biomarkers across a lithification gradient, from a fresh microbial mat, through a lithifying mat, to a completely lithified microbialite (the geological end-member). Lipids that harbored unsaturations, methyl groups, and/or polycyclic ring structures exhibited the least resistance to this bio-to-geo transition and thus lost most of said features. In particular, the transition from the soft, metabolically active mat to the lithifying mat showed the most intense degradation, with an average concentration decrease of over 90%. While further degradation toward the microbialite was observed, it was less severe and more variable between lipid families. Conversely, linear and saturated chains (n-alkanes, n-alkanoic acids, and n-alkanols) exhibited the least degradation within their respective families (hydrocarbons, acids, and alcohols) and were the most abundant in all three samples. Furthermore, the carbon isotopic analysis revealed the maintenance of 13C-depleted signatures of the organic compounds throughout the bio-to-geo transition, which ensured the preservation of their biogenic character also in the form of an isotopic signature. In conclusion, the combined molecular and isotopic analyses revealed which are the most resilient structural features of lipid biomarkers most likely to be preserved along a lithification gradient.
The effective preservation of certain lipid biomarkers in the transition from the biosphere to the geosphere provides critical insights into which organic compounds have the potential to retain their structural identity over geological timescales. In this study, we detected saturated chains with functional groups in notable abundances in a fully lithified microbialite, meaning that certain structural features with relevant diagnosis value for biogenicity (i.e., carboxyl and hydroxyl groups) have been effectively retained during lithification. All these observations highlight the importance of focusing resources to collectively assess the preservation and detection of similar compounds of 13C-depleted signatures, particularly in ancient terrestrial samples, or within the first 2 m of regolith at sites such as Oxia Planum. While this study focused on the effects of relatively early diagenesis, further investigations are needed to assess how these biomarkers behave over much longer timescales relevant to potential Noachian life on Mars. Additionally, to refine our understanding of lipid taphonomy, future studies should explore the effects of other possible degradation factors (e.g., harmful radiation) on lipid biomarkers in controlled laboratory conditions. This would provide clearer insights into the degradative processes that affect these compounds under the extreme conditions of Mars or other planetary environments.
Footnotes
Acknowledgments
The authors thank their guide and driver, Luis Ahumada, and the native community of Antofalla for their invaluable support during field work and sample retrieval. They also thank the Secretaría de Medio Ambiente de la Provincia de Catamarca for processing and providing their research permit (Resolución S.E.A. y D.S. No.: 053/2017). Finally, they express their gratitude to Paloma Martínez Sarmiento and Miguel Ángel Lominchar for their assistance with isotopic analysis and to María Teresa Fernández Sampedro for her assistance with X-ray diffraction.
Authors’ Contributions
P.L.F. wrote the original draft with input from all coauthors, conducted the analytical tasks, worked on software, data curation, and visualization. D.C. collected the samples, reviewed and edited the article, conceptualized the study, was involved in sample processing, and conducted analytical tasks. V.P. reviewed and edited the article. F.A.V. and M.E.F. designed and coordinated field work, obtained the sampling permits, collected samples, and reviewed and edited the article. L.S.-G. supervised the elaboration, reviewing and editing of the article, conceptualized the study, and conducted analytical tasks. D.C., F.A.V., M.E.F., and L.S.-G. acquired funding for the project.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research has been funded by the Spanish Ministry of Science and Innovation/State Agency of Research MCIN/AEI/10.13039/501100011033 and by “ERDF A way of making Europe,” through the grant numbers PRE2020-093795 (P.L.F.), PID2022-140180OB-C21 (D.C.), PID2021-126746NB-I00 (V.P. and L.S.-G.), and RYC2018-023943-I (L.S.-G.). Additionally, funds were granted by the CABANA project, by UKRI-BBSRC on behalf of the Global Challenges Research Fund (BB/P027849/1—funds to F.A.V.), and by the Argentinian National Council for Scientific and Technological Research (CONICET—funds to F.A.V. and M.E.F.).
Supplementary Material
Supplementary Figures
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
Supplementary Table S8
Supplementary Table S9
Associate Editor: Mary Beth Wilhelm
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

 )
)