Abstract
Future missions dedicated to the search for extant life on Mars will require a clear understanding of the organic biosignature degradation processes in the shallow icy subsurface. Galactic and solar cosmic rays constantly bombard the martian surface and transform and degrade organic biomolecules over time, eventually destroying chemical evidence of life. We conducted radiolysis experiments by exposing individual amino acids in H2O-ice and silicate matrices and amino acids from dead Escherichia coli microorganisms in H2O-ice to gamma radiation as a proxy for cosmic ray exposure on the martian surface. The rates of amino acid radiolytic degradation were determined. We found that amino acids in the surface ice on Mars would survive over 50 million years of cosmic ray exposure, which is far greater than the expected age of the current surface ice deposits on Mars. Amino acids from dead E. coli organic matter in H2O-ice and isolated pure amino acids dissolved in H2O-ice tend to degrade at similar rates. We found that amino acid radiolytic degradation rates increased with increasing ice temperature in both abiotic and biological amino acids. Montmorillonite did not provide additional protection against gamma radiation to amino acids. Based on our experiments, locations with pure ice or ice-dominated permafrost would be the best places to look for recently deposited amino acids on Mars and, thus, should be considered as a target sampling location for future Mars missions searching for extant life.
Key Points
Gamma irradiation of individual amino acids in H2O-ice and silicate matrices and amino acids from dead Escherichia coli microorganisms in H2O-ice was conducted at Mars-like temperatures to simulate the radiolytic destruction of biomolecules in the martian permafrost. We estimate that amino acids in the surface ice on Mars would survive over 50 million years of cosmic ray exposure, which is far greater than the expected age of the current surface ice deposits on Mars. Thus, pure ice or ice-dominated permafrost should be considered for sampling in future life search Mars missions.
Introduction
Detection of organic biomarkers is a crucial step in NASA’s strategy for searching for extraterrestrial life in our solar system. Due to the thin CO2 atmosphere and weak magnetic field of Mars, martian surface rocks and regolith have been continuously exposed to ultraviolet (UV) radiation as well as galactic cosmic rays (GCRs) and solar cosmic rays (SCRs) particles for billions of years. Both UV radiation and energetic particles can alter or destroy organic molecules (Kminek and Bada, 2006; Noblet et al., 2012; Orzechowska et al., 2007; Pavlov et al., 2022). However, a fundamental difference between UV radiation and cosmic ray exposure on the martian surface lies in the depth of radiation penetration. All UV radiation is absorbed within the top 1 mm of surface rocks or icy regolith (Cockell and Raven, 2004), while energetic particles can cause ionization several meters deep into the rocky or icy surface (Dartnell et al., 2007; Pavlov et al., 2012). Hence, the effects of UV exposure on organic degradation are relatively easy to overcome by minimal drilling or scooping the very top layer of the surface. In contrast, exposure to GCRs and SCRs has been recognized as a major factor that could “erase” organic biomarkers over long timescales at meter depths within ancient rocks on Mars (Ertem et al., 2021; Kminek and Bada, 2006; Pavlov et al., 2022; Roussel et al., 2024). Therefore, cosmic ray (CR) exposure should be carefully evaluated when future sampling locations are selected.
Future in situ life search missions to Mars (e.g., Mars Life Explorer [MLE] [National Academies of Sciences, Engineering, and Medicine, 2023]) will focus on the search for extant life (active or dormant). Active life, as we know it, requires water. Thus, unlike Curiosity, Perseverance, and eventually Rosalyn Franklin, a MLE-type mission will consider locations with ice and icy permafrost among the prime candidates for investigation (Mellon et al., 2024). A previous study by Pavlov et al. (2022) found that the radiolytic destruction of amino acids in silicate samples with 10% of added water under Mars-like temperatures (218–223 K) was much more effective than the degradation of amino acids in dry (∼1% water) silicates under similar Mars-like conditions. Specifically, in hydrated silica samples, most amino acids were destroyed after just 0.2 MGy of gamma radiation exposure (see Fig. 6, Pavlov et al., 2022), which is equivalent to ∼1.25–3 million terrestrial years of ice exposure on the martian surface and shallow subsurface (Dartnell et al., 2007; Ehresmann et al., 2023; Pavlov et al., 2012). Rapid degradation of biomolecules can be a serious challenge to the sampling strategies in shallow icy soils, which can be several million years old (Head et al., 2003; Mellon et al., 2024). Since the increase in water content in silicates to 10 wt % increased the rate of radiolytic destruction (Pavlov et al., 2022), it is not clear if intact amino acids from extant life can survive in ice-dominated permafrost on million-year timescales.
To address this question, we conducted gamma irradiation experiments—used as a proxy for CR exposure—at Mars-like temperatures on pure amino acids in H2O-ice, as well as on amino acids derived from dead microorganisms dissolved in ice. We also irradiated amino acids mixed with montmorillonite powder, a common clay on Mars (Clark et al., 2007; Du et al., 2023), to validate the previous results, which showed that silicates alone increased the rate of radiolysis (Pavlov et al., 2022).
Methods
We conducted radiolysis experiments by exposing individual amino acids to gamma radiation to simulate CR exposure of the martian near-surface. Three types of samples were irradiated: individual amino acids mixed with ice, amino acids mixed with silicates (fused silica and montmorillonite clay), and dead Escherichia coli biomass dissolved in ice. We compared the abundance of amino acids present after irradiation (up to 2 MGy) to the abundance of amino acids before irradiation to determine their degradation and survival rates.
Sample preparation procedure
Stock solutions of individual amino acids and mixtures (1 × 10−3 M) were prepared by dissolving solid amino acid standards (97–99% purity from Sigma-Aldrich) in Millipore Direct Q3 UV (18.2 MΩ, < 3 ppb total organic carbon) ultrapure water. The FS-120 fused silica was manufactured by H.P. Technical Ceramics (Sheffield, UK). Solid FS-120 was crushed using a porcelain mortar and pestle. The crushed FS-120 was passed through a 150 µm sieve and then baked at 500°C in air overnight to remove organic contaminants. All samples were prepared in 13 mm diameter borosilicate glass test tubes that were wrapped in aluminum foil and then heated in a furnace at 500°C in air overnight. The samples were prepared in an ISO 5 HEPA laminar flow bench with a procedural blank prepared in parallel with each sample type.
The E. coli was cultivated in 1 L of Luria-Bertani (LB) broth media divided into four 1 L-sized glass Erlenmeyer flasks. Cells were grown to late-log phase at 37°C and then harvested by centrifugation at 8000 RPM for 10–15 min to make a wet cell pellet. The pellet was washed three times with filtered distilled water. The cell pellet was then autoclaved and freeze-dried to produce a powder of dead cellular material for preparation of the various experimental conditions.
Samples were prepared at room temperature and then sealed under vacuum. The sample sets were prepared as shown in Table 1 and described as follows: Amino acid and ice solutions were prepared by dissolving solid amino acid standards (97–99% purity from Sigma-Aldrich) in Millipore Direct Q3 UV (18.2 MΩ, <3 ppb total organic carbon) ultrapure water.
The glycine and fused silica samples were prepared by weighing out 5010.7 mg of FS-120 fused silica in a 20 mm test tube, followed by the addition of 1 mL of 1 mM glycine. The mixture was vortexed to ensure equal mixing. The sample was then dried overnight under vacuum at 30°C using a Labconco CentriVap benchtop centrifuge vacuum concentrator to remove as much water as possible. To prevent water adsorption upon air exposure, the dried sample was immediately capped. After drying, the sample was vortexed to ensure thorough mixing of glycine and FS-120. The FS-120 and glycine mixture was then divided into five test tubes, with approximately 850 mg of the mixture per test tube, and sealed within an hour of preparation. See precise amounts in Table 1.
The glycine plus montmorillonite clay samples were prepared by weighing out 5071.2 mg of montmorillonite clay in a 15 mL centrifuge tube, followed by adding 4 mL of 1 mM glycine and 2 mL of ultrapure water. The mixture was vortexed to ensure equal mixing. The sample was then dried overnight under vacuum at 30°C using a Labconco CentriVap benchtop centrifuge vacuum concentrator to remove as much water as possible. To prevent water adsorption upon air exposure, the dried sample was immediately capped. After drying, the sample was vortexed to ensure thorough mixing. To ensure reproducibility, three sets of glycine-montmorillonite clay mixture were extracted to verify consistency across subsamples. The final mixture was then portioned into five test tubes, each containing ∼850 mg. See precise amounts in Table 1.
Each test tube was connected via a stainless steel 0.5-inch Ultra-Torr fitting to a vacuum glass line with a liquid nitrogen trap and a turbo drag pump. To reduce adsorbed water and oxygen in the sample tubes, each tube was evacuated until the pressure inside the tube reached ∼20 mTorr and was then flame sealed. For the tubes that contained liquid water, the tubes were first frozen in liquid nitrogen before being evacuated and subjected to three freeze-pump-thaw cycles to remove dissolved air from the samples prior to flame sealing.
GCRs and SCRs that impact the martian surface are primarily high-energy protons with energies that exceed ∼100 MeV (Pavlov et al., 2012). However, previous studies (e.g., Bonner et al., 1985; Ertem et al., 2021; Kminek and Bada, 2006; Pavlov et al., 2022; Quinn et al., 2013) have used gamma irradiation as a proxy for CR exposure on the martian surface. There are two main reasons for this approach.
First, exposing samples to high-energy protons is significantly more expensive than using gamma irradiation to deliver the same radiation dose. Second, the composition of the ionizing radiation field on Mars changes with depth in rocks or ice (Dartnell et al., 2007), as primary CR protons generate cascades of secondary electrons, gamma photons, and other particles that, in turn, degrade and destroy organic molecules. Even within the top meter of the surface, a significant fraction of the energy from incident GCRs is deposited via secondary electrons (Dartnell et al., 2007, their Fig. 6).
A comparative study of organic degradation by gamma photons versus electrons in air concluded that, for the same radiation dose, both types of radiation cause similar molecular damage (Blanco et al., 2018). Thus, gamma irradiation serves as a reasonable proxy for CR exposure of rocks and regolith on Mars, although a direct comparative study of the effects of gamma photons versus protons should be conducted in the future.
In this study, we used the same 60Co gamma-ray source and setup at the Radiation Science & Engineering Center (RSEC) facility at Pennsylvania State University as was used in the study by Pavlov et al. (2022). During irradiation, samples in flame-sealed glass tubes were cooled with dry ice (replenished daily), and the sample temperature was maintained within a range of 218–223 K. Samples were exposed to accumulated gamma dosages of 0.5, 1.0, 1.5, and 2.0 MGy. The three dosages (0.5, 1, and 2 MGy) were chosen for a direct comparison with our previous studies (Pavlov et al., 2022, 2024) and correspond to ∼3, 6, and 12 million terrestrial years of exposure in the martian surface ice, respectively (Dartnell et al., 2007).
Sample extraction
After irradiation, each test tube was examined for leaks by placing the sealed 13 mm test tube in a 20 mm tube and submerging the test tubes with samples in milli-Q ultrapure water to find gas leaks. The irradiated samples along with their nonirradiated controls were then scored with a diamond tipped glass scorer and opened for subsequent analysis. An aliquot of each of the samples was removed for the amino acid analysis. All remaining samples were removed from the original irradiated test tubes and stored in glass vials in a −80°C freezer for future analyses.
For the amino acids plus ice samples, three aliquots of 50 µL each were hot water extracted at 100°C for 24 h. For the amino acids plus fused silica samples, ∼250 mg was hot water extracted at 100°C for 24 h. For the amino acids plus montmorillonite clay samples, ∼250 mg was hot water extracted at 100°C for 24 h. Hot water extraction is employed due to the high solubility of amino acids in water, with heating enhancing their dissolution for efficient recovery from clay surfaces. While pure amino acid samples may not require this method, it is indispensable for extracting amino acids from clays, biological matrices, and extraterrestrial materials, where complex interactions with the surrounding substrate necessitate more rigorous extraction techniques. Given its extensive application in meteorite studies, E. coli cultures, and other extraterrestrial samples, this approach ensures direct comparability across datasets. Furthermore, hot water extraction optimizes sample integrity for liquid chromatography mass spectrometry (LC-MS) analysis by mitigating interference from harsh solvents and aggressive extraction protocols. To maintain methodological consistency and preserve the fidelity of intersample comparisons, uniform extraction and preparation protocols are imperative across all experiments in this study. Ultimately, hot water extraction provides a robust, precise, and scientifically validated means of studying amino acids while preserving their native chemical characteristics.
After hot water extraction, all samples were centrifuged at 3000 rpm for 3 min, and the supernatants were drawn off to be analyzed using the AccQ·Tag derivatization technique described in Section 2.4 and the Supplementary Data.
LC-MS: Derivatization and analysis
Samples were derivatized with Waters AccQ•Tag reagents according to the manufacturer’s protocol (Boogers et al., 2008). This is a robust technique frequently used to detect amino acids in meteorites and prebiotic samples (e.g., Aponte et al., 2020; Dworkin et al., 2018; Giese et al., 2022; Vinogradoff et al., 2020). The AccQ•Tag derivatization process is as follows: 10 μL of the standard or sample extract was mixed with 70 μL borate buffer and 20 μL of AccQ•Tag reagent. The reaction was allowed to proceed for 10 min at 55°C. Samples were then analyzed with a Waters Xevo G2 XS time-of-flight mass spectrometer using electrospray ionization in positive sensitivity mode.
Sample analysis began and ended with a concentration series of a derivatized amino acid standard, with samples analyzed in between the standards. Amino acid products were confirmed by comparing two chromatographic retention times to the amino acid standard, including one UV fluorescence chromatogram and one selected ion chromatogram. The amino acid products were quantified using the peak areas from the selected ion chromatogram. A linear least-square model was fit to each amino acid in the standard calibration set, and these calibration curves were used to quantify the analytes in the samples. Triplicate runs were averaged for the final value, and the calculated standard error is the reported uncertainty.
Additional LC-MS methods, including the instrument method configuration, are included in the Supplementary Data. An example set of LC-MS chromatograms is provided in Supplementary Figure S1, and the LC-MS retention times and exact masses used to identify the amino acids in this study are provided in Supplementary Tables S1 and S2.
Results
We studied radiolytic degradation of the individual amino acids (glycine, alanine, and isovaline) in icy mixtures (Fig. 1). The glycine concentration did not exhibit a degradation correlation with radiation dose. We observed a 20% decrease in alanine abundance and ∼40% decrease in isovaline abundance at 2 MGy dosage. The 2 MGy dosage is equivalent to ∼12.5 million terrestrial years of exposure in the martian surface ice or icy regolith (Dartnell et al., 2007).

Normalized abundances of individual amino acids + H2O-ice irradiated up to 2 MGy. All samples were irradiated in 218–223 K temperature range. The amount of irradiation for each sample is defined as follows:  for 0.5 MGy,
for 0.5 MGy,  for 1 MGy,
for 1 MGy,  for 1.5 MGy, and
for 1.5 MGy, and  for 2 MGy. H2O:amino acids ratio in control samples was 55560:1 similar to that from Pavlov et al. (2024).
for 2 MGy. H2O:amino acids ratio in control samples was 55560:1 similar to that from Pavlov et al. (2024).
The absence of a clear degradation trend in glycine at 2 MGy may be attributed either to partial reformation of glycine following initial fragmentation or to the relatively large error bars in the measurements. Specifically, glycine—like any organic molecule exposed to ionizing radiation—has some probability of spontaneously reforming from its radiolysis products and radicals. Due to its simple molecular structure, glycine may reform more readily than more complex amino acids. In our previous study (Pavlov et al., 2022), pure dry glycine also showed no measurable degradation. However, in both the current study and the study by Pavlov et al. (2022), we did not analyze the specific products of amino acid radiolysis—this will be the focus of a future investigation. Consequently, we cannot yet determine the exact pathway by which glycine might reform. Notably, glycine can be produced by various forms of ionizing radiation in water–ice mixtures such as H2O/CO2/N2 or H2O/CO/NH3 (Kobayashi et al., 1995, 2008). In Kobayashi’s experiments, the production rate of alanine was 5–10 times lower than that of glycine. Alternatively, the lack of a clear degradation trend could simply reflect the relatively large uncertainties in the glycine measurements.
It is convenient to compare radiolytic degradation efficiency using the standard exponential equation:
Based on observed data (Fig. 1), the “alanine plus ice” radiolytic constant k alanine (ice) is 0.10 ± 0.01 MGy−1, which is similar to the radiolysis constants for pure alanine in the work by Kminek and Bada (2006; Table 1) as well as by Pavlov et al. (2022; Fig. 3).
Isovaline in ice exhibits gradual degradation with dosage increase. Kminek and Bada (2006) found that the radiolysis constants of pure amino acids (k) are linearly dependent on their molecular weight (M): k = 0.0014 × M − 0.0223. Thus, the predicted value of the isovaline’s radiolysis constant should have been ∼0.14 MGy−1. However, based on our data, k isovaline (ice) is 0.25 ± 0.02 MGy−1. Therefore, isovaline’s faster radiolytic degradation rate compared to alanine’s might not be solely due to the larger molecular weight.
We found that the radiolytic degradation of amino acids in pure ices was significantly slower than the radiolysis of amino acids in silicates under Mars-like temperatures (Fig. 2). The “glycine plus fused silica” samples showed a gradual decrease in glycine abundance with a dosage increase. By 2 MGy of exposure, only 20% of the original glycine was left, which is in excellent agreement with our previous study (see Fig. 5 in Pavlov et al., 2022). Radiolytically produced oxidants from the fused silica matrix (e.g., oxygen radicals) lead to an increase in the amino acid degradation rate.

Normalized abundances of glycine plus fused silica and glycine plus montmorillonite irradiated up to 2 MGy. All samples were irradiated in 218–223 K temperature range. The abundances measured after irradiation have been normalized against the control (unirradiated) samples. The amount of irradiation for each sample is defined as follows:  for 0.5 MGy,
for 0.5 MGy,  for 1 MGy,
for 1 MGy,  for 1.5 MGy, and
for 1.5 MGy, and  for 2 MGy.
for 2 MGy.
The “glycine plus montmorillonite” samples (Fig. 2) exhibited a slightly greater rate of glycine degradation, possibly due to the radiolytic production of the hydroxyl radicals, which would contribute to glycine destruction.
Figure 3 presents normalized amino acid concentrations in E. coli samples exposed to increasing doses of irradiation that ranged from 0.5 to 2.0 MGy, compared to a control group set at 1.00. The observed trends suggest that the side-chain acid dissociation constant (pKa) values (Supplementary Table S1) may play a role in determining each amino acid’s stability under irradiation. Amino acids with lower side-chain pKa values, such as aspartic acid (pKa 3.9) and glutamic acid (pKa 4.3), exhibit significant reductions in concentration as irradiation increases. These acidic residues might be more prone to ionization and subsequent degradation due to their tendency to lose protons under different environmental conditions. Conversely, some amino acids with higher side-chain pKa values, such as lysine (pKa 10.5), display greater stability across doses.

Normalized abundances of amino acid extracted from E. coli + ice samples. This figure shows the amino acids grouped by functional group, and then arranged in ascending order according to molecular weight. Uncertainties (δx) were determined as the standard error (δx = σx (n) −1/2), whereby the uncertainties were based on the standard deviation (σx) of the average value of triplicate measurements (n = 3). The amount of irradiation for each sample is defined as follows: Gray  for the control, dots
for the control, dots  for 0.5 MGy, stripes
for 0.5 MGy, stripes  for 1 MGy, blank
for 1 MGy, blank  for 1.5 MGy, and white
for 1.5 MGy, and white  for 2 MGy.
for 2 MGy.
All irradiated E. coli + ice samples (Fig. 3, Supplementary Table S3) showed decreased levels of amino acids compared to the unirradiated control. As expected, amino acids in 0.5, 1, and 1.5 MGy samples showed a gradual decrease in amino acid abundance with the dosage increase. However, Figure 3 shows that in the 2 MGy E. coli sample (which represents a single tube), the abundance of each amino acid was higher compared to the abundance of the respective amino acids in 1 and 1.5 MGy E. coli samples. We do not have a clear explanation for this result. However, it is possible that for this single tube, the initial mixture of dead E. coli cell material and water had a small clump of undissolved cell material. If such a clump ended up in one of the samples before irradiation and then later extracted after irradiation, the amino acids could appear to be artificially high. Assuming such a clump was present once and that there were five samples in the E. coli + ice set, the probability that a clump of E. coli would occur only in the 2 MGy sample is about 20%. Alternatively, it is reasonable to consider that some amino acids remain bound in nonirradiated E. coli samples. At a 2 MGy dosage, radiation exposure could lead to the breakdown of cellular structures, which would facilitate the release of these previously bound amino acids and potentially explain the observed increase. Further experiments with larger radiation dosages can prove or disprove this hypothesis.
Given the uncertainty with the 2 MGy E. coli + ice sample, we calculated two radiolysis constants—including and excluding the 2 MGy datapoint (Supplementary Table S4). For example, k valine (E. coli-ice) with all dosage datapoints is 0.17 ± 0.07 MGy−1. The same constant k valine (E. coli-ice) with the excluded 2 MGy datapoint would be 0.3 ± 0.04 MGy−1.
Comparison to previous studies
There have been only two previously reported experiments of amino acid radiolysis in ices at Mars-relevant temperatures. Specifically, Gerakines and Hudson (2013, 2015) studied the degradation of glycine in H2O and CO2 ices under 0.8 MeV proton irradiation in a vacuum. They discovered that glycine degrades more effectively in CO2-ice samples compared to H2O-ice samples. It is difficult to make a direct comparison between their study and ours due to different irradiation conditions. Specifically, we used a different type of ionizing radiation (gamma rays), and their irradiation temperature was kept significantly lower than in our study (theirs 15–140 K vs. ours 218–223 K sample temperatures). Gerakines and Hudson (2015) reported the glycine radiolysis constant 0.05 MGy−1 for 100 K irradiation and a 300:1 H2O/glycine ratio in samples. In contrast, we did not observe a noticeable decline in glycine abundance after 2 MGy of exposure. However, if glycine were to degrade in our experiments with the radiolysis constant of 0.05 MGy−1, then by 2 MGy, glycine abundance would have decreased by ∼10%. A 10% decline in glycine abundance at 2 MGy would be within 1-sigma error bars for glycine (Fig. 1).
Also, it is worthwhile to compare the rates of degradation in previous experiments on the amino acid decay in ices under UV radiation (e.g., Johnson et al., 2012; Orzechowska et al., 2007) and in our gamma irradiation experiments. As noted earlier in the Introduction, UV radiation is absorbed within the top 1 mm layer of the surface. However, we can express the degradation in UV experiments as a function of absorbed energy, estimate the rate of decay constant for amino acids as a function of absorbed energy, and compare with the radiolysis constants for amino acids in our study. Orzechowska et al. (2007) conducted experiments on the degradation of various amino acids in ices under UV irradiation at 100K. They used an argon mini-arc lamp with the following characteristics—average photon 2 × 1014 photons/cm2/s in the wavelength range of 130–335 nm. For the comparison, we chose their experiment on the 0.28 mm ice layer sample. Assuming most of the lamp’s energy was absorbed in 0.28 mm of ice layer [Orzechowska et al. (2007), their Fig. 5] and the average photon of lamp’s energy to be ∼7 eV [based Orzechowska et al. (2007), their Fig. 3], the absorbed energy rate in such a thin ice layer would be ∼13 Gy/s. Orzechowska et al. (2007) reported the half-life of aspartic acid and glutamic acid to be ∼5 h. That means that after exposure to 5 h or 0.165 MGy of UV radiation, half of the amino acids were destroyed. Using D 1/2 = ln(2)/K decay, we find that K rad for aspartic acid and glutamic acid should be K decay = 4.8 MGy−1. That is 13–20 times larger than the K rad values (0.23–0.36 MGy−1) for aspartic (in E. coli), glutamic (in E. coli), and isovaline acids in our current gamma irradiation study (see Supplementary Table S4).
Why is UV irradiation significantly more efficient at degrading organic molecules than gamma radiation, even when the same total energy is deposited into irradiated ice samples? The reason likely lies in the different ways UV and gamma photons deposit energy into the sample. The gamma photons used in our study have energies of ∼1 MeV—several orders of magnitude greater than the molecular bond energies or ionization potentials of atoms and molecules in the H2O–amino acid matrix. As a result, gamma photons ionize both water and organic molecules indiscriminately. Consequently, the absorbed energy is distributed between water radiolysis and the degradation of organic compounds. The reactive products of water radiolysis (e.g., OH radicals) may or may not contribute to organic degradation, depending on their mobility in the matrix.
In contrast, UV photons in the >190 nm range primarily photolyze amino acids in the H2O–amino acid mixture. For these wavelengths, the photolysis cross section of H2O is relatively small (∼10−20 to 10−21 cm2/molecule; Heays et al., 2017; Hróðmarsson and van Dishoeck, 2023), whereas the cross sections for amino acids are significantly higher (∼10−17 to 10−18 cm2/molecule; Ehrenfreund et al., 2001; ten Kate et al., 2005). Thus, UV photons deposit their energy in the H2O–amino acid mixtures more selectively into breaking down amino acids, rather than producing water-derived radicals. This selective energy absorption may explain why, for the same total absorbed dose, UV irradiation results in significantly higher degradation rates of amino acids compared to gamma irradiation.
The difference in amino acid degradation between UV and gamma irradiation is not limited to ice samples. For example, dos Santos et al. (2016) conducted UV irradiation experiments on various minerals spiked with amino acids and demonstrated that clay minerals (e.g., montmorillonite) are particularly effective at preserving amino acids. This protective effect is attributed to the high specific surface area and small average pore size of clays, which likely allows a greater proportion of adsorbed amino acids to be shielded from direct UV radiation.
In our study, we exposed amino acids mixed with montmorillonite to gamma radiation under Mars-like conditions (Fig. 2). Our objective was to confirm the increased radiolytic degradation of amino acids in silicate mixtures—previously observed with fused silica (FS-120) by Pavlov et al. (2022)—using a natural mineral (montmorillonite). We found that glycine degraded even faster when mixed with montmorillonite than with fused silica (Fig. 2). In contrast to the UV experiments, montmorillonite did not protect amino acids from degradation by gamma radiation. This is likely because 1 MeV gamma rays can penetrate tens of centimeters into clays or other minerals, rendering any shielding effect from surface area or small pores negligible.
Temperature effect on radiolysis rates
Irradiation experiments were conducted on samples similar to those used by Pavlov et al. (2024) on radiolytic degradation of the amino acids in Europa- and Enceladus-like ices. Since both studies were performed using the same gamma source and followed identical amino acid extraction and measurement procedures, we were able to isolate the effect of temperature on radiolysis rates (Fig. 4a,b). We found that Mars-like temperatures (218–223 K) accelerated the rate of radiolytic degradation of the amino acids in both silicates (Fig. 4a) and ices (Fig. 4b), compared to the radiolytic degradation of the same amino acids at Europa-like temperatures (77 K). This increase in radiolysis is consistent with the enhanced mobility of radiolytically produced oxidants at higher temperatures, as discussed by Pavlov et al. (2022). Consequently, the higher temperatures led to increased reaction rates between amino acids and these oxidants. The radiolysis constant for glycine in silicates k glycine(SiO2) increased from 0.36 ± 0.04 MGy−1 (Pavlov et al., 2024) to 0.72 ± 0.03 MGy−1 (this study), while the radiolysis constant for isovaline in ices k isovaline (ice) increased from 0.13 ± 0.01 MGy−1 (Pavlov et al., 2024) to 0.25 ± 0.02 MGy−1 (this study). Interestingly, despite the entirely different matrices (solid ice vs. porous silicate powder), the radiolysis constants for amino acids increased by a factor of ∼2 under Mars-like temperatures.
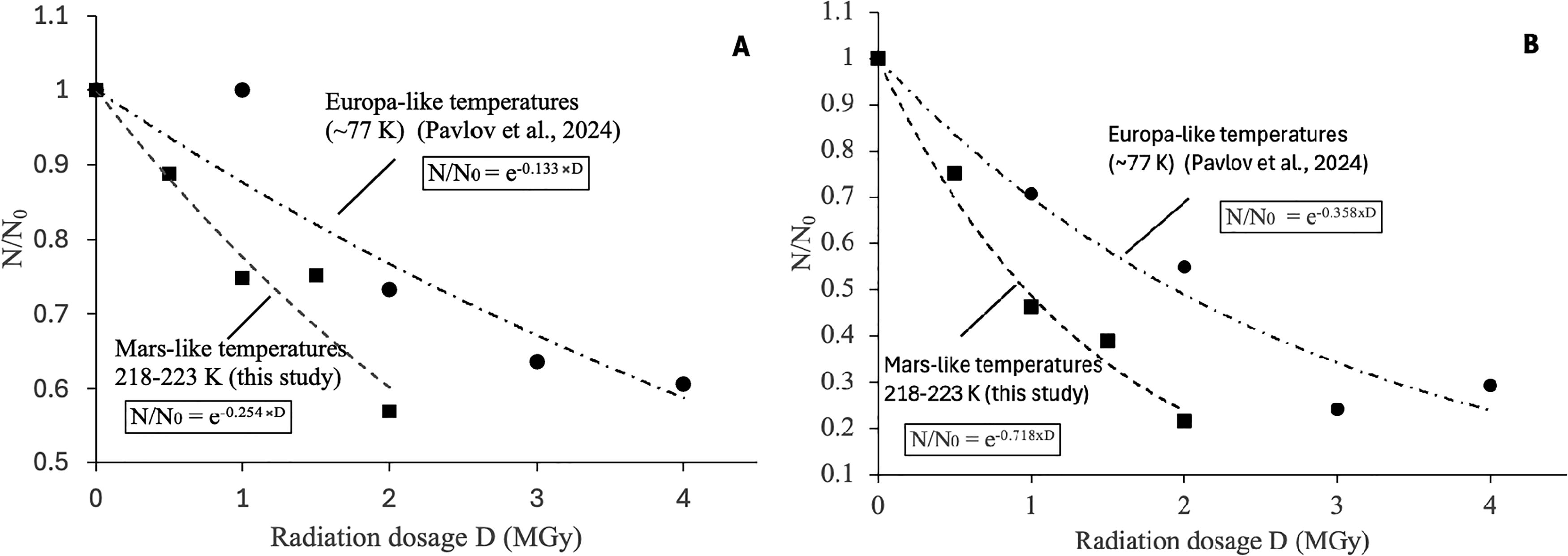
Radiolysis of amino acids in E. coli also accelerated in ice samples at Mars-like temperatures compared to Europa-like temperatures. Specifically, proline’s radiolysis constant, k proline (E. coli-ice), increased from 0.056 ± 0.04 MGy−1 to 0.24 ± 0.04 MGy−1, glycine’s constant, k glycine (E. coli-ice), from 0.033 ± 0.05 to 0.13 ± 0.05 MGy−1, and valine’s constant, k valine (E. coli-ice), from 0.027 ± 0.02 MGy−1 to 0.17 ± 0.07 MGy−1.
Our results on the temperature dependence of amino acid radiolysis in H2O-ice require further experimental confirmation through irradiation of ice samples at higher temperatures.
In the previous study, we found that hydrated silicates, with 10% of H2O by weight, greatly increased the rates of radiolytic amino acid destruction compared to radiolysis rates in dry silicates (see Figs. 5, 6 in Pavlov et al., 2022). We suggested that the increase in the amino acid destruction rates is due to the production of oxidative radicals from H2O radiolysis. Therefore, it was unexpected that when the same amino acids (glycine and isovaline) were gamma-irradiated under the same temperatures (218–223 K) in pure ice (100% H2O) in this study, the observed amino acids’ radiolysis rates were much lower (Fig. 1). Radiolysis of amino acids in pure ice was slower not only compared to radiolysis in hydrated silica samples but also slower than radiolysis in dry silicates (Fig. 2). Thus, our original hypothesis—higher water abundance in irradiated samples increases the rate of radiolysis of the organic molecules (Pavlov et al., 2022)—must be adjusted. We hypothesize that the mobility of the radiolytically produced oxidative radicals is more important for amino acids’ destruction than the total production of oxidative radicals from the H2O radiolysis. Specifically, although more OH and other radicals are likely formed in gamma-irradiated ice samples, the smaller number of radicals gamma-generated in hydrated silica samples may be more mobile—and therefore more effective at degrading amino acids.
The basis for our hypothesis comes from Pearson and Derbyshire (1974), who demonstrated that adsorption water films on various silicates can remain 2D liquid-like at temperatures as low as −196°C. The reason for such behavior is that the water molecules bond more strongly to mineral (silicate) surfaces rather than to the ice matrix. Recognizing such a phenomenon, Mohlmann (2004) pointed out that the presence of a few percent of adsorbed mobile water in the martian topsoil is the reason for the production of OH radicals through photo-Fenton reaction, which proceeds in the acidic liquid films. Mohlmann (2004) suggested that those OH radicals could be responsible for the lack of organics of meteoritic origin in martian soil. Liquid films should be expected in silicate samples with 10% adsorbed H2O at Mars-like temperatures (Pavlov et al., 2022) as well. Thus, radiolytically produced oxidants could migrate and react with amino acids even if the radiolysis of H2O did not occur in the immediate vicinity of amino acids. In “dry” silicate samples (Fig. 2), about 1% of the residual H2O was present, and some liquid films could still form. Therefore, migration of radiolytically produced oxidants through liquid films would still be possible, albeit at a slower rate, and result in relatively slower amino acid degradation rates. In contrast, radiolytically produced oxidants in pure ice samples could not diffuse effectively due to the lack of mobile water at 218–223 K, and the rates of amino acid degradation are, therefore, much lower.
Our hypothesis—liquid films of water greatly increase the rate of organic destruction from radiolysis—should be tested in future experiments on samples with various proportions of silicates and H2O, as well as on soils with various porosity and grain sizes.
In this study, we did not explore the radiolysis of biomolecules in icy samples with added salts. Our previous studies (Pavlov et al., 2022) showed that the addition of perchlorates to silicates greatly increases the rate of amino acid degradation because the radiolysis of perchlorates produces ClO−, O− radicals (eventually forming O2 gas), and other radicals (Quinn et al., 2013), which cause additional degradation of the amino acids. However, in icy samples, perchlorates would not only provide an extra source of oxidants due to radiolysis but also would lower the eutectic point of water. Thus, liquid films of water would exist not only on the silicate ice boundary but also throughout the ice-rich samples. Therefore, it is plausible that abundant liquid films of water will increase the transport of radiolytically produced oxidants throughout irradiated samples and increase the overall rates of organic destruction even further. Future irradiation experiments of icy samples with various amounts of salts are needed. Future irradiation experiments of icy samples with different salt compositions and concentrations are needed to better understand the effect that salt has on the radiolysis of organic molecules in salty martian permafrost.
Implications for Mars
Using radiolysis constants from our experiments (Supplementary Table S4) and the radiation accumulation rates from Dartnell et al. (2007), we calculated the timescales to accumulate dosage for 10% amino acid survival (D 10) (Table 2).
Radiation rates were taken from Figure 3 in Dartnell et al. (2007).
Timescale for valine (E. coli) is given as a range due to the uncertainty in the radiolysis constants for valine (E. coli) + ice (0.17–0.3 MGy−1); see Supplementary Table S4.
Our calculations indicate that a significant fraction of amino acids in the surface ice on Mars would survive over 50 million years of exposure. Amino acids from dead biological material (E. coli) dissolved in ice would survive even longer. Because ice has a relatively low density, the radiolysis rates in ice do not change significantly with depth (Dartnell et al., 2007; Pavlov et al., 2012). Thus, there is no significant advantage in drilling the surface ice (or ice-dominated regolith) to sample unaltered organic molecules. Amino acids degrade faster in dry silicates (Table 2). Yet, even in silicates, it would take ∼20 million years to achieve D 10 dosages at the martian surface. Since silicates have higher densities than ice, the degradation of organic molecules will be much slower with depth—D 10 dosages would be accumulated in ∼100 Myr at 1 m depth.
Surface ice distribution is constantly evolving on Mars due to changes in planetary obliquity. Head et al. (2003) estimated that the last ice age on Mars occurred 2.1–0.4 million years ago. Ice deposits have sublimated and continue to sublimate from low and mid-latitudes and redeposit in the polar regions ever since. Thus, any surface ice was exposed to ionizing radiation for a few million years. Therefore, if an abiotic or biological amino acid were deposited in the surface ice matrix on Mars somehow (e.g., spores or interplanetary dust particles), it would have a high chance of survival. In contrast, amino acids deposited in the silicate-dominated regolith would degrade faster, particularly if silicates are hydrated or have a high perchlorate content (Pavlov et al., 2022).
Based on the results of this study, locations with pure ice and ice-dominated permafrost should be the best places to look for recently deposited amino acids on Mars and, thus, should make a good target for future MLE-type missions. Amino acids in the surface ice matrix would survive over 50 million years of CR exposure, which is far greater than the expected age of the current surface ice deposits on Mars. Biological amino acids (mostly peptide bound) in H2O-ice at 218–223 K temperatures tend to radiolytically degrade at similar rates as isolated free amino acids dissolved in H2O-ice. Amino acid radiolytic degradation rates increase with increasing ice temperature in both abiotic and biological amino acids. Montmorillonite did not provide additional protection to amino acids against gamma radiation. It is likely that the formation of liquid water films in hydrated silica (permafrost with a large SiO2/H2O ratio) or due to the presence of perchlorate salts greatly increases the rates of amino acid radiolysis on Mars.
Footnotes
Author Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Information
This work was supported by NASA’s Planetary Science Division Internal Scientist Funding Program through the Fundamental Laboratory Research (FLaRe) work package at Goddard Space Flight Center. K.K.F. and H.L.M. were supported by the Center for Research and Exploration in Space Science and Technology II cooperative agreement with NASA and the University of Maryland, Baltimore County, and Catholic University of America, under award number 80GSFC24M0006.
Supplementary Material
Supplementary Data
Abbreviations Used
Associate Editor: Lewis Dartnell
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
