Abstract
Background:
The Manuel Antonio National Park (MANP) and its surrounding areas in Costa Rica are home to endangered non-human primates (NHPs), including Saimiri oerstedii citrinellus and Cebus imitator, that coexist in close contact with humans and domestic animals. Such interactions may facilitate the transmission of vector-borne zoonotic diseases. This study aimed to investigate the presence of selected vector-transmitted pathogens of zoonotic relevance in NHPs from this region.
Methods:
Between 2021 and 2022, 40 free-ranging NHPs (19 C. imitator, 19 S. oerstedii citrinellus, 2 Alouatta palliata) were sampled in MANP and surrounding areas. Blood samples were tested for Trypanosoma spp., Plasmodium spp., Mycoplasma spp., and arboviruses including dengue virus (DENV), chikungunya virus (CHIKV), Zika virus (ZIKV), Venezuelan equine encephalitis virus (VEEV), and West Nile virus (WNV). Molecular analyses were conducted using PCR and RT-qPCR protocols, with sequencing for species confirmation.
Results:
Eight of 40 NHPs (20.0%) tested positive for at least one pathogen. Trypanosoma minasense was confirmed by sequencing in three C. imitator (16.7%). Plasmodium spp. was detected in one C. imitator and one A. palliata (5.0%), though species-level identification was unsuccessful. One C. imitator tested positive for CHIKV (Ct 36.8), representing the first PCR-confirmed case of CHIKV in NHPs in Central America. Two C. imitator were positive for Mycoplasma spp. (11.1%). No co-infections were observed. All animals were negative for DENV, ZIKV, VEEV, and WNV.
Conclusions:
This study provides the first molecular evidence of CHIKV infection in NHPs in Costa Rica and highlights the circulation of Trypanosoma spp., Plasmodium spp., and Mycoplasma spp. in wild primates from MANP. These findings emphasize the need for a One Health approach to surveillance, particularly in regions where human–wildlife interactions are frequent. Longitudinal and serological studies are recommended to assess the potential reservoir role of NHPs and their implications for public health and conservation.
Keywords
Introduction
The Central Pacific region of Costa Rica, specifically the Manuel Antonio National Park (MANP) and its surrounding areas, represents one of the most important areas for the conservation of the titi, or gray-crowned Central American squirrel monkey (S. oerstedii citrinellus), and the white-faced capuchin monkey (Cebus imitator). It is the only state-protected wildlife region where the gray-crowned Central American squirrel monkey subspecies is conserved worldwide. These species are threatened by a series of anthropogenic factors, including habitat loss and fragmentation, as well as electrocutions and vehicle collisions, which have led to its population being in a declining state and at risk of extinction (Solano-Rojas, 2020).
Nonhuman primates (NHP) are one of the emblematic species of Costa Rica, and their conservation can bring benefits to both the ecosystem and society through ecotourism (Johnson et al., 2023). Tourism is one of the country’s main industries, especially tourism associated with protected areas and biodiversity. Public protected areas are supervised by the National Park System, and public and private protected natural areas are available to tourists (Villalobos-Céspedes et al., 2009). Manuel Antonio is a clear example of a community whose development and livelihood depend largely on tourism. The MANP is one of the most visited national sites, with a daily visiting capacity of up to 1,300 people (Presidencia de la República de Costa Rica, 2020). One of the main attractions of MANP is the observation of wildlife, including the titi and white-faced monkeys.
However, the close contact and proximity between wildlife and tourists, especially in the surrounding areas outside the park, represent a potential negative relationship from a public health and conservation perspective. In these areas, anthropogenic development mixes with wildlife, increasing the frequency of contact and probability of disease transmission between humans and animals (Daszak et al., 2000). Given the behavioral characteristics of primates, as well as of some tourists and the environment in which they interact, it is necessary to evaluate infectious agents that may be present in this population of primates. However, information about infectious agents present in primates in MANP is limited (Dolz et al., 2019; Fuentes-Ramírez et al., 2017). For this reason, it is essential to generate information to understand the dynamics between wild species, humans, and domestic animals. Due to this knowledge gap, it is intended to determine the presence of vector diseases with zoonotic potential (Trypanosoma spp. and Plasmodium spp.), dengue virus (DENV), chikungunya virus (CHIKV), Zika virus (ZIKV), Venezuelan equine encephalitis virus (VEEV), West Nile virus (WNV), and Mycoplasma spp. in individuals of nonhuman primates in the MANP and surrounding areas.
Materials and Methods
Anesthesia, physical examination, and sampling of nonhuman primates
Three species of primates, the gray-crowned Central American squirrel monkey (S. oerstedii spp. citrinellus), the Panamanian white-faced capuchin (C. imitator), and the mantled howler monkey (Alouatta palliata), were tested in the MANP and surrounding areas between 2021 and 2022. S. oerstedii citrinellus were captured using Tomahawk tramps (Tomahawk, WI, USA) with fruits as bait. C. imitator was captured using darts syringes (TeleDart GMBH & CO. KG, Germany) and a blowpipe. Alouatta palliata samples were collected from necropsies requested by the park rangers. These A. palliata died from electrocution while the study was carried on. Animal capture and sampling were done during 06:00–10:00 and 15:00–17:00 h, avoiding crowded and dangerous places for sedated animals (rivers, electric wires, and roads), extreme weather conditions like precipitation and environmental temperatures over 30°C. Infants, pregnant females, lactating animals, or those carrying their young were excluded from the investigation. A combination of Ketamine (KET-A-100®, Agrovet Market, Lima, Perú, 5 mg/kg, IM)—Medetomidine (Sedator®, Dechra, Montigny-le-Bretonnneux, France, 0.04–0.06 mg/kg, IM) was used for the anesthesia. Once the procedure finished (∼30 min), animals were antagonized with atipamezole (Atipam®, Dechra, Montigny-le-Bretonnneux, France, 5x the dose of medetomidine, IM) to significantly shorten recovery time (Calle and Ott Joslin, 2015). The targeting site for the injections was the femoral muscle. Information about gender and age (juvenile and adult) was collected. Each animal was physically evaluated and kept under observation with monitoring devices and care until it regained consciousness. Blood samples (1 mL) were taken from the femoral or cephalic veins (Calle and Ott Joslin, 2015) in EDTA tubes. Once the animals were fully recovered, they were released to a safe place in the same area where they were captured. There were no problems with the animals during the capture, anesthesia process, and release back to nature. A total of 100 µL of the blood sample was immediately transferred to RNA later (RNAlater™ Solution, Invitrogen, Vilnius, Lithuania). The samples were stored at 4°C and transported to the laboratory, where they were stored at −80°C until processing.
Molecular analysis
Blood samples underwent RNA and DNA extraction using commercial kits, QIAamp® Viral RNA and DNeasy® Blood & Tissue Kit, respectively (Qiagen, Hilden, Germany), following the instructions of the manufacturer and were analyzed using the polymerase chain reaction (PCR). To detect Plasmodium falciparum, Plasmodium vivax, and Plasmodium malariae, a nested PCR amplifying a sequence of the small ribosomal DNA subunit was used (Rubio et al., 2002). To detect Trypanosomatidae, a region of the small ribosomal RNA subunit (SSU-rRNA) gene was amplified as described by Uliana et al. (1994). Reverse transcriptase real-time polymerase chain reaction (RT-qPCR) following the protocol described by Waggoner et al. (2016) was used to amplify the segments that encode the 5' untranslated region of the DENV capsid gene, the CHIKV nonstructural protein 2 (NSP2), and the nonstructural protein 4B (NS4B) of ZIKV. For VEEV, EEEV, and WEEV, detection of the E2 region was carried out (Linssen et al., 2000), and for the detection of WNV, a region of the envelope gene was amplified (Lanciotti et al., 2000). To identify the presence of Mycoplasma species, a fragment of the 16S rRNA gene was amplified (Kong et al., 2001; van Kuppeveld et al., 1994). The positive controls were donated or purchased; free nuclease water was used as a negative control in all protocols. All samples that amplified in the RT-qPCR with a growth curve exceeding the threshold Ct (calculated automatically by the equipment) between the first 45 cycles were considered positive. The RT-PCR and PCR products were visualized by 2% agarose gel electrophoresis and stained with gel red. The samples that showed bands with the expected size (Trypanosomatidae 520 bp, P. falciparum: 395 bp, P. vivax: 499 bp, P. malariae: 269 bp, VEEV: 194 bp, WNV: 400 bp, Mycoplasma spp.: 280 bp) were considered positive. The amplicons were purified using the QIAquick® PCR Purification kit (QIAGEN, Hilden, Germany), following the manufacturer’s instructions, and were sent to Macrogen (Seoul, Korea) for sequencing. The sequences were aligned with the option ClustalW from BioEdit (Hall, 1999) and compared with other sequences deposited in the NCBI database (National Center for Biotechnological Information) using the BLASTn algorithm.
Results
A total of 40 primates were sampled (19 C. imitator, 19 S. oerstedii citrinellus, and 2 A. palliata). Of these, 32 were adults and 8 juveniles. In terms of sex, 11 were females and 29 were males. According to the general physical evaluation, none of these animals showed any significant physical alterations or clinical signs of disease. A total of eight (20.0%) nonhuman primates were found positive for at least one pathogen; no coinfections were detected in the positive animals. Three (16.7%) white-faced monkeys were positive for Trypanosoma spp. Sequencing determined 98.8% (494/500 bp) nucleotide identity with Trypanosoma minasense obtained from a tamarin (Sanguinus midas) from Japan (AB362411.1). The three sequences were deposited in GenBank (PQ524303, PQ530293, PQ530294). Additionally, Plasmodium spp. was determined in two (5.0%) animals, one white-faced monkey and one howler monkey (Table 1); however, sequencing of the amplicons was not possible. One white-faced monkey (2.5%) was found positive for CHIKV (Ct36.8), and two white-faced monkeys (11.1%) were positive for Mycoplasma spp. (Table 1). Most positives (87.5%) were detected by April 2022. All NHPs were negative for DENV, ZIKV, VEEV, and WNV.
Capture Date, Species, Sex, Age, and Results of Tests for Infectious Agents Detected in 40 Primates in the Manuel Antonio National Park and Surrounding Areas
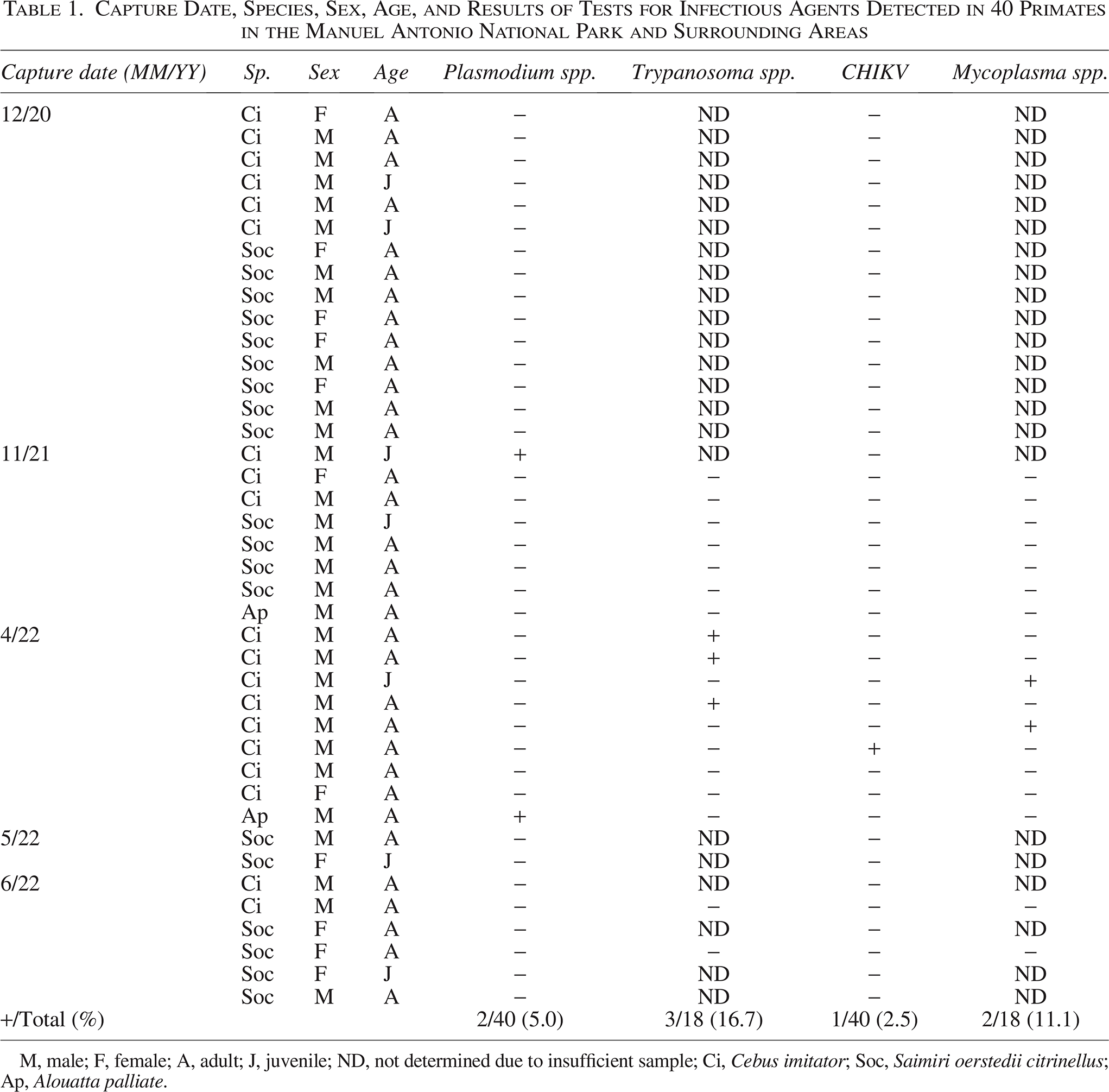
M, male; F, female; A, adult; J, juvenile; ND, not determined due to insufficient sample; Ci, Cebus imitator; Soc, Saimiri oerstedii citrinellus; Ap, Alouatta palliate.
Discussion
To the authors’ knowledge, this is the first molecular report of CHIKV in Costa Rica and Central America in nonhuman primates. Information is limited, and little is known about the presence of this virus in primates and animals in this region. In other Asian and African regions, urban and wild cycles of CHIKV were reported, and it seems that the virus could circulate among monkeys without showing clinical signs (Mongkol et al., 2022). The viremia is short, so detection by PCR may be limited, and seroprevalence studies are generally performed in primates (Tongthainan et al., 2020). The detection of CHIKV in blood suggests that viremia may occur in white-faced capuchin monkeys and generates hypotheses on the role of nonhuman primates as reservoirs and amplification hosts of this virus. Although a positive PCR result does not demonstrate that the species acts as a reservoir, detection of viral RNA reflects current or recent exposure to the pathogen but does not establish if the animal is capable of sustaining viremia at levels necessary for vector transmission, nor whether it can contribute to maintaining the virus in the ecosystem. A recent study documented natural exposure to CHIKV in golden-headed lion tamarins (Leontopithecus chrysomelas) in nonprotected areas of Bahia, Brazil. The animals tested seropositive for CHIKV, suggesting contact with the virus in anthropogenically altered environments, but did not demonstrate active infection or vector-mediated transmission. Further research is necessary to determine whether CHIKV is maintained in a sylvatic cycle in MANP and whether NHPs in Central America play any role in its amplification or transmission (Bernal-Valle et al., 2025). CHIKV emerged in 2014 in Costa Rica with the potential to become endemic and generate epidemics that threaten the national health system (Corrales-Aguilar et al., 2020). In 2022, the year in which this virus was identified in primates, the National Virological Reference Center processed 1,148 human samples by PCR, all of which were negative. Thus, from 2017 to 2022, no endemic case of CHIKV has been confirmed by PCR in Costa Rica by this laboratory, a pattern also observed in other countries in the region (González, 2022). The detection of this virus in a free-living primate in an area with a high diversity of wildlife and people, including tourists, living at the human-animal interface, should alert surveillance systems to the presence of the virus in both humans and animals. Although spillover events require several factors to align for an infectious agent to pass from a wild animal to humans or vice versa (Plowright et al., 2017), prevention measures must be maintained to mitigate the risk of contagion. Surveillance to detect a possible epidemic early can generate savings in the Costa Rican health system, as well as prevent disease outbreaks. Given that the surveillance of this agent is not decentralized in the country (González, 2022), it is important to consider this in the future for this pathogen and other arboviruses for diagnostic and epidemiological surveillance purposes.
Across the world, the Plasmodium species from nonhuman primates exhibit a significant degree of diversification (Coatney et al., 1971). In New World NHPs, infections with Plasmodium malariae, P. falciparum, and P. vivax have been documented, primarily through molecular detection. However, reports of clinical disease manifestations in these primates are scarce, and most infections appear to be asymptomatic. P. malariae/brasilianum was detected in captive monkeys (Callithrix jacchus) in this area (Fuentes-Ramírez et al., 2017), the results of the present investigation show that the cycle was still active in November 2021 and April 2022. During this period, the number of total cases reported in people has exceeded previous years. There was also an active outbreak reported in this region. P. falciparum was the species with the highest circulation in the country (89.0% predominance), followed by P. vivax (11.0%). However, in the Central Pacific Region, where the study was conducted, P. malariae was found in humans (Ministerio de Salud Costa Rica, 2022). Although attempts were made to characterize this Plasmodium sample, the National Parasitological Reference Laboratory was unable to do so, leaving a void of information about the Plasmodium genus that may be circulating in PNMA. Costa Rica is a leading country in the Mesoamerican region in trying to eliminate malaria in people (Chaves et al., 2020). However, to achieve this objective, a One Health approach must be considered, including epidemiological surveillance efforts and action plans that include wildlife, especially nonhuman primates. This is due to the potential that these animals maintain the sylvatic cycle of this parasite. Community education and outreach including social scientists and communicators can be beneficial to help reduce the risk of exposure to vectors (Anopheles spp.) of this parasite.
Moreover, T. minasense is a highly specific hemoparasite to nonhuman primates considered as nonpathogenic and transmitted primarily by infected triatomine bugs (also known as kissing bugs) (Ferreira et al., 2015). There is no evidence of infection in humans (Coimbra et al., 2020). This parasite has been reported in Costa Rica in the free-living species A. paliatta (Chinchilla et al., 2005) and identified by PCR in all NHP species of the country (C. imitator, A. palliata, Ateles geoffroyi, S. oerstedii), being the first molecular report in Costa Rica (Solórzano-Morales et al., 2019). Other study groups have described T. minasense in C. imitator from Panama and suggest that primates of the genus Cebus are good hosts for Trypanosoma, including T. minasense (Sousa et al., 1974). Still little is known about the transmission cycle of this parasite and its potential vectors (Martínez et al., 2016). More detailed studies about the life cycle and repercussions on animal health should be carried out in Costa Rica.
Although it was not possible to establish the Mycoplasma species infecting NHP in the present study, Mycoplasma salivarium was found in three oral samples from captive NHP (C. imitator, Ateles geoffroyi, S. oerstedii) without clinical signs of infection in 2015 in the same area of this study (Varela-Amador et al., 2023). M. salivarium has been isolated from nonhuman primates (Neto et al., 2013), but no reports link them to disease in these species. More studies are needed to understand its role in primate health and its potential zoonotic implications. This species is also found in humans. To date, there are no public reports documenting the specific detection of M. salivarium in humans in Costa Rica. However, this bacterium is part of the normal oral microbiota in approximately 97% of healthy individuals worldwide. Although generally considered a harmless commensal, M. salivarium has been associated with certain clinical conditions (periodontitis, septic arthritis, and brain abscesses in immunocompromised individuals) (Watanabe et al., 1986). The presence of Mycoplasma species, particularly those also found in humans, signals a need for studies to evaluate the impact of these microorganisms on the health of NHPs and the possibility of cross-species transmission between NHPs and humans as permanent or temporary reservoirs.
Although there was no PCR detection of DENV, VEEV, and WNV, it could not be ruled out that these animals were exposed to these viruses. A previous study carried out in this area obtained a similar result in which there was no detection of WNV and DENV by PCR, but antibodies to arboviruses were detected (Chaves et al., 2021; Dolz et al., 2019). Serological or molecular positive results of NHP don’t mean that they play a role in the transmission or act as reservoirs of arboviruses. More evidence is needed, for example, establishing long-lasting viremias or transmission to other animals or vectors (Aedes aegypti and A. albopictus) (Haydon et al., 2002; Plowright et al., 2017). In Costa Rica, arboviruses circulate actively; however, their sylvatic cycles continue to be a poorly studied topic and require further research to elucidate doubts about which species may be involved (Barrantes-Murillo et al., 2022). Given the potential of these primates to develop viremias, it is important to carry out studies that allow a better understanding of the seasonality of these viruses to explore if these cycles coincide with disease cycles present in humans. Given the significant mortality of animals in these regions due to car hit trauma, electrocution, and other human-related activities, these deceased animals could be utilized for epidemiological surveillance. By analyzing these specimens, valuable data could be gathered to monitor the impact of human activities on wildlife health, identify disease patterns, and improve our understanding of environmental risks. Community engagement and outreach, particularly with younger generations, have been central to our research efforts in this region. We have made it a priority to ensure that the outcomes of our research directly contribute to the health and well-being of the community. In the future, establishing a multidisciplinary network that focuses on field data and sample collection to analyze the presence of antigens and antibodies to address seasonal and ecological analysis would bring beneficial information about the diseases in this region for both human and animal populations. This One Health approach will require a strong committee involving governmental and nongovernmental institutions, community leaders, academia, local businesses, as well as other important stakeholders.
Conclusions
This study represents the first report of CHIKV in nonhuman primates of Costa Rica confirmed by PCR. The control of this virus and other arboviruses in public health programs must essentially include the study of NHP in its transmission cycles, and epidemiological surveillance should not be neglected, especially in regions where outbreaks in humans have occurred. The detection of Plasmodium spp. in blood samples from Manuel Antonio NHPs suggests the presence of the cycles in November 2021 and April 2022 in the MANP and surrounding areas. It is recommended to investigate the wild and urban cycles of vector-transmitted agents and their interconnection. Also, studies of the impact of Mycoplasma species in humans’ and NHPs’ health are recommended. To prevent future outbreaks of infectious diseases, surveillance of interactions between people and wildlife is essential. It is important to carry out longitudinal studies for arboviruses and malaria, using molecular and serological techniques, to establish acute and past infections, determine the seasonality and geographical distribution of these agents in primates, and compare them with human cases in the area. Also, capture, classification of mosquitoes and determining the presence of these agents and blood source will offer information and elucidate transmission between humans and nonhuman primates and bridging vectors.
Authors’ Contributions
E.R.-S.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, project administration. M.M.-M.: Methodology, investigation, writing—review, visualization. J.C.-B.: Methodology, investigation, writing—original draft, writing—review, visualization. M.J.Z.-M.: Methodology, investigation, writing—review, visualization. A.S.-M.: Methodology and investigation. K.V.-B.: Methodology and investigation. M.J.-S.: Conceptualization, methodology, investigation, resources, supervision, project administration, and funding. G.D.: Conceptualization, methodology, investigation, resources, writing—original draft, writing—review, supervision, project administration, and funding.
Footnotes
Acknowledgment
Thanks to SINAC and CONAGEBIO for granting the research permit (R-CM-UNA-009-2021-OT-CONAGEBIO). Thanks to all the students and assistants of the Small Animal and Wildlife Hospital (HEMS) who supported the sampling. To park rangers and administrative personnel of the Manuel Antonio National Park.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This project was financed by project SIA-UNA 0048-18 “Diagnosis, Disease Control, and Management of Wild Animals of the Hospital de Especies Menores y Silvestres (HEMS), & by FUNDAUNA project “Diagnóstico e Investigación en Medicina Poblacional y Ecosistémica.” The open access publication of this article was funded by Vicerrectoría de Investigación, Universidad Nacional de Costa Rica.
Institutional Review Board Statement
This study was approved by the National Ministry of Environment and Energy (MINAE) and the National System of Conservation Areas (SINAC), under the research permit N° M-PC- SINAC-PNI-ACOPAC-021–2019 and executed according to the regulations of the National Committee of Biodiversity Management (CONAGEBIO) and approved in permit R-CM-UNA-008-2021-OT-CONAGEBIO. Informed consent was obtained from the National System of Conservation Areas—Pacific Central Office (SINAC-ACOPAC).
