Abstract
Background
Polyamines are involved in several cellular processes and inhibiting their synthesis affects chikungunya virus (CHIKV) replication and translation, and, therefore, reduces the quantity of infectious viral particles produced. In this study, we evaluated the inhibition of CHIKV replication by N-ω-chloroacetyl-L-ornithine (NCAO), a competitive inhibitor of ornithine decarboxylase, an enzyme which is key in the biosynthesis of polyamines (PAs).
Methods
The cytotoxicity of NCAO was evaluated by MTT in cell culture. The inhibitory effect of CHIKV replication by NCAO was evaluated in Vero and C6/36 cells. The intracellular polyamines were quantified by HPLC in CHIKV-infected cells. We evaluated the yield of CHIKV in titres via the addition of PAs in Vero, C6/36 cells and human fibroblast BJ treated with NCAO.
Results
We found that NCAO inhibits the replication of CHIKV in Vero and C6/36 cells in a dose-dependent manner, causing a decrease in the PFU/mL of at least 4 logarithms (p < 0.01) in both cell lines. Viral yields were restored by the addition of exogenous polyamines, mainly putrescine. The HPLC analyses showed that NCAO decreases the content of intracellular PAs, even though it is predominantly spermidines and spermines which are present in infected cells. Inhibition of CHIKV replication was observed in human fibroblast BJ treated with 100 μM NCAO 24 h before and 48 h after the infection at a MOI 1.
Conclusions
NCAO inhibits CHIKV replication by depleting the intracellular polyamines in Vero, C6/36 cells and human fibroblast BJ, suggesting that this compound is a possible antiviral agent for CHIKV.
Introduction
Chikungunya fever is a tropical vector-borne disease that has been spreading around the world increasingly rapidly during recent years. It is caused by the Alphavirus chikungunya (CHIKV), transmitted by mosquitoes of the genus Aedes.[1] Chikungunya vaccines are not currently included in national immunization systems, and contemporary therapies are based on the administration of analgesics, antipyretics and anti-inflammatory drugs to relieve symptoms.[2] Many studies have shown that polyamines (PAs) are important in the replication cycles of diverse viruses. Polyamines are small, positively charged molecules derived from arginine that are involved in multiple cellular processes, such as cell proliferation,[3] DNA and RNA stabilization,[4,5] cellular stress,[6] cellular signalling[7] and other functions in eukaryotic cells. Studies on the Semliki Forest virus showed that PAs are not present in the viral capsid, but when there was a depletion of PAs, there was a parallel marked reduction in the activity of viral RNA polymerase from the cells infected with these viruses.[8] It has been shown that the continuous synthesis of PAs is a requirement for some viruses, such as Vaccinia and Cytomegalovirus; when the biosynthetic pathways of polyamines are interrupted by the inhibitor of ornithine decarboxylase difluoromethylornithine (DFMO), viral dynamics are seriously affected.[9] Moreover, the depletion of PAs in cells infected with chikungunya and dengue viruses due to treatments with DFMO or DENSpm (diethylnorspermine), an analogue of PA that induces the expression of spermidine–spermine acetyltransferase (SSAT), affects translation and viral replication and limits the expression of non-structural proteins.[10,11] Recently, Mason et al. showed that polyamines facilitate coronavirus replication and the depletion of polyamines with DFMO and significantly reduce coronavirus replication.[12] Thus, the pathway of PA biosynthesis could be a target for the development of possible antivirals against chikungunya and other RNA viruses. Therefore, in this work, we studied whether N-ω-chloroacetyl-L-ornithine (NCAO) can inhibit the replication of chikungunya virus (CHIKV) because NCAO is a competitive inhibitor of ornithine decarboxylase (ODC). Rodríguez-Paéz et al. demonstrated the inhibition properties of this compound, which causes cytotoxic and antiproliferative effects in human cancer cell lines that overexpress ODC and showed minimal cytotoxic effects in non-cancer control cells.[13,14] We demonstrated that N-ω-chloroacetyl-L-ornithine (NCAO) inhibits the replication of the chikungunya virus in mammalian Vero, insect C6/36 and human fibroblast BJ cells in a dose-dependent manner by depleting the three intracellular polyamines.
Materials and methods
Cell culture and chikungunya virus
Vero and human fibroblast BJ cells were maintained in a Dulbecco’s modified Eagle’s medium/F-12 nutrient mixture (DMEM-F12; Sigma-Aldrich) supplemented with fetal bovine serum and penicillin–streptomycin, incubated at 37°C and 5% CO2. The C6/36 cells were maintained in a minimal essential medium (MEM, Sigma-Aldrich) supplemented with fetal bovine serum and penicillin–streptomycin and incubated at 34°C with an absence of CO2. Chikungunya virus was isolated from a confirmed sample from a Mexican patient by consecutive passages in C6/36 cells.
Viability tests
Vero and C6/36 cells were cultured and treated with 10, 100, 200, 300, 400, 500 and 1000 μM NCAO. Human fibroblast BJ were treated with 100, 200, 300, 500y and 1000 μM NCAO. NCAO was diluted with DMEM-F12 or MEM without supplementation. Cell viability was determined at 24, 48 and 72 h by reduction in the tetrazolium ((3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, MTT) salt assay. Absorbance was measured at 540 nm. Dose–response curves and EC50 were determined.
Inhibitory effect of CHIKV replication by NCAO
Vero and C6/36 cells were cultured and treated with NCAO 100 and 500 μM for 24 h. After the incubation time, the medium was removed and they were infected with CHIKV at 0.1, 0.25, 0.5 and 1 MOI. BJ cells were cultured and treated with NCAO 100 μM for 24 h. After the incubation time, the medium was removed, and they were infected with CHIKV at 1 MOI. NCAO was added after the infection at the same concentrations. The inhibitory effect was tested at 100, 200, 200, 400, 500 and 1000 μM of NCAO in Vero and C6/36 cells infected with CHIKV at 0.5 and 1 MOI. Titration of the supernatants was performed at 24 hpi for Vero and C6/36 cells. Supernatant titration for BJ cells was at 24 and 48 hpi. The tests were carried out in triplicate and duplicate respectively.
Viral titration
Viral titration on Vero cells was performed on 24-well plates, and 90, 000 cells/well were cultured. Serial dilutions were prepared to a 10−6 dilution of virus for infection. The plates were incubated at 37°C with 5% CO2, with rocking performed every 10 min for 1 h. After the absorption period, an overlay (2% carboxymethylcellulose (CMC) diluted in culture medium) was added and incubated at 37°C with 5% CO2 until the presence of lytic plaques was observed. The cells were then fixed with 5% formaldehyde for 15 min and stained with crystal violet–ethanol at 0.5% for 30 min. Then, the viral titre was determined by counting the wells present between 10 and 100 lytic plates above the 10−3 dilution.
Quantification of intracellular polyamines by HPLC in CHIKV-infected cells
The quantification of intracellular PAs was performed in the cellular debris from the tests to determine the inhibition effect on CHIKV replication by NCAO by HPLC. Sample pretreatments were performed with 50% trichloroacetic acid (Sigma-Aldrich) and 1 mM 1, 8 diaminooctane (Sigma-Aldrich). Benzoylation of the polyamine standards and samples was carried out with NaOH 2 M (Sigma-Aldrich) and 50% benzoyl chloride (Sigma-Aldrich). The extraction was carried out with chloroform–HPLC (Sigma-Aldrich), and the compounds were evaporated with a stream of nitrogen. The HPLC analysis was performed using a C18 reverse phase column, Agilent 5 μm that was 250 x 4.6 mm. The mobile phase was a mixture of methanol and water (60:40), eluted with a linear gradient at a speed flow rate of 0.4 mL/min. Detection was performed at 229 nm at room temperature using Agilent 1260 equipment.
Yield of CHIKV in titres via the addition of PAs in Vero and C6/36 cells treated with NCAO
Vero and C6/36 cells were pretreated with 300 μM NCAO in 24-well plates. The media were removed, and cells were infected with CHIKV at 0.5 and 1 MOI in the presence of NCAO; exogenous PAs (putrescine, spermine and spermidine, Sigma-Aldrich) were added at the same time, individually, at 10 μM. Supernatants were collected at 24 hpi and the viral yield of CHIKV was quantified.
Results
Viability of Vero and C6/36 cells treated with different concentrations of NCAO
Viability assays in Vero cells treated with NCAO showed that 24 h after exposure to the compound there was no cytotoxic effect (Figure 1A); however, at 48 h after exposure, the cell viability was 80% at 300 μM. This decrease was statistically significant compared to the control, 10, 100 and 200 μM samples; however, with the 400 μM concentration, viability was reduced to 40% (Figure 1B). At 72 h after exposure with NCAO, at concentrations of 10 μM up to 300 μM, viability decreased to between 60 and 80%; however, at a concentration of 400 μM, viability decreased to 20%, which was statistically significant (ANOVA *p < 0.01) (Figure 1C). There were no statistically significant reductions in the viability of C6/36 cells treated with NCAO at 24 and 48 h after exposure at 10–1000 μM (Figure 1D and 1E). However, 72 h after treatment with NCAO, a viability of 70–80% was observed from the 100 μM concentration: this decrease was statistically significant (ANOVA *p < 0.01) compared to the control and the 10 μM concentration samples (Figure 1F). The dose–response curve with Vero cells treated with NCAO exhibited an EC50 value of 385.12 μM at 48 h after exposure and an EC50 value of 323.8 μM after 72 h (Figure 2). Viability of Vero and C6/36 cells treated with different concentrations of N-ω-chloroacetyl-Lornithine. Vero cells were treated with concentrations of NCAO from 10 to 1000 μM. Viability was determined at 24 h (A), 48 h (B) and 72 h (C). C6/36 cells were treated with concentrations of NCAO from 10 to 1000 μM. Viability was determined at 24 h (D), 48 h (E) and 72 h (F). Statistical significance was determined using ANOVA tests, p < 0.05, n = 9; different letters between groups indicate significant differences. Dose–response curve of Vero cells treated with NCAO. Vero cells were treated with concentrations of NCAO from 1 to 1000 μM. The dose–response curves show EC50 values of 385.12 μM at 48 h (left) and 323.80 μM at 72 h (right).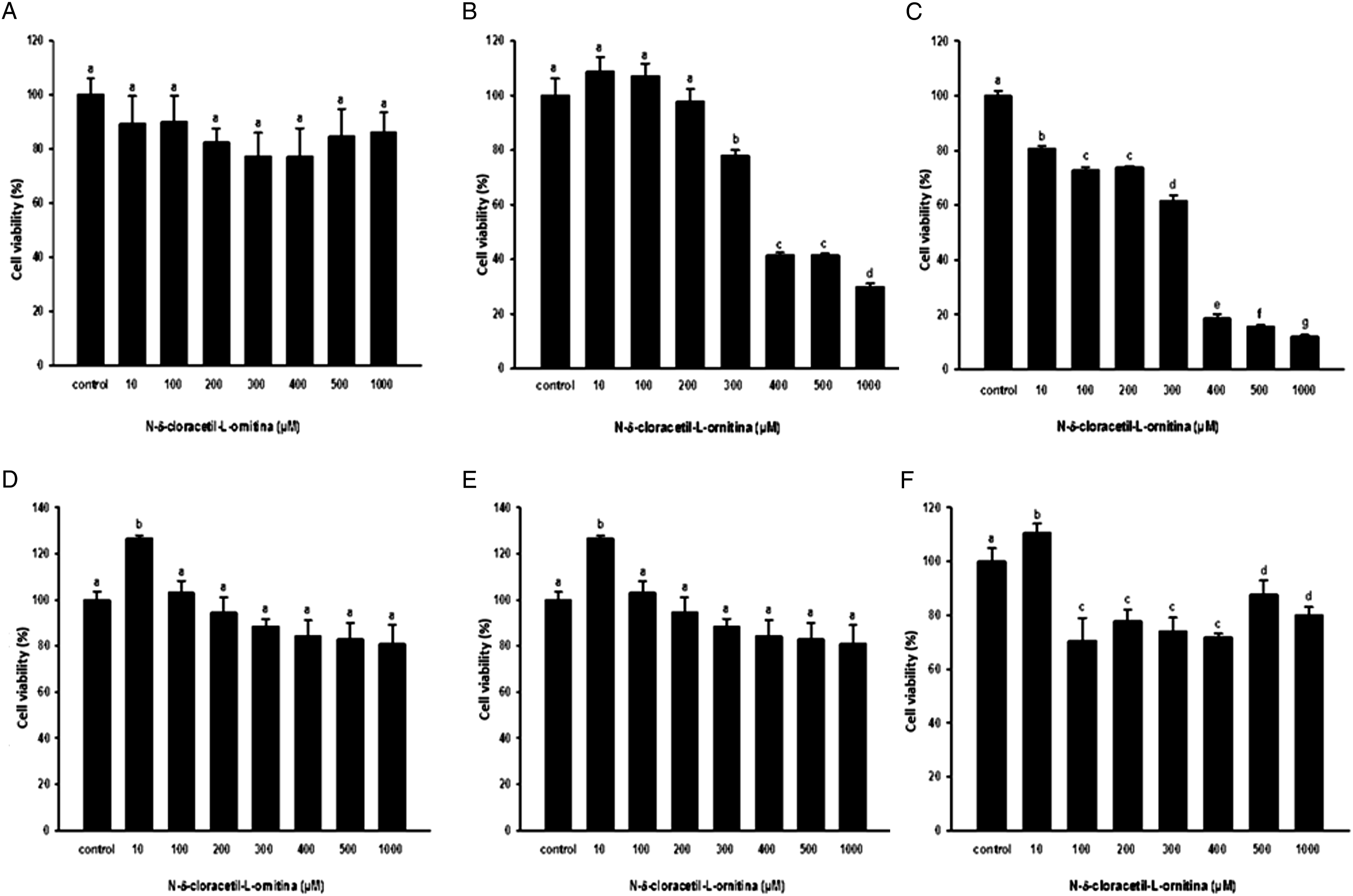

Inhibition of CHIKV replication by NCAO in C6/36 and Vero cells
Inhibition of chikungunya virus replication by NCAO exhibited a significant reduction (ANOVA *p < 0.01) of PFUs in Vero cells infected at 0.1, 0.25, 0.5 and 1 MOI and pretreated with 500 μM NCAO for 24 h, with respect to control and 100 μM NCAO cells pretreated and infected in the same conditions (Figure 3A). Interestingly, a significant reduction (ANOVA *p < 0.01) was observed in the viral titres of C6/36 cells pretreated with 100 and 500 μM NCAO and infected with 0.1, 0.25, 0.5 and 1 MOI, compared to the control (Figure 3B). The inhibitory effect of chikungunya virus replication by NCAO in Vero cells pretreated at 100, 200, 300, 400, 500 and 1000 μM, and infected with 0.5 and 1 MOI, showed a significant reduction (ANOVA test, p < 0.01) in PFUs, from 200 to 1000 μM (Figure 3C); C6/36 cells treated at the same conditions also exhibited a significant reduction, although from 100 to 1000 μM (Figure 3D). The decreases in the viral titres were dose-dependent in both cell lines (ANOVA p < 0.01) (Figure 3C and 3D). Inhibition of chikungunya virus replication by N-ω-Chloroacetyl-L-Ornithine in C6/36 and Vero cells. Vero and C6/36 cells were pretreated with NCAO concentrations of 100 and 500 μM for 24 h and subsequently infected with 0.1, 0.25, 0.5, and 1 MOI CHIKV. Titres were determined at 24 hpi for Vero (A) and C6/36 cells (B). Vero and C6/36 cells were pretreated at concentrations of 100, 200, 300, 400, 500 and 1000 μM NCAO before and during infection with CHIKV at 0.5 and 1 MOI. Titres were determined at 24 hpi for Vero (C) and C6/36 cells (D). Statistical significance was determined using the ANOVA test: *p < 0.01, n = 3, ns, no significant difference.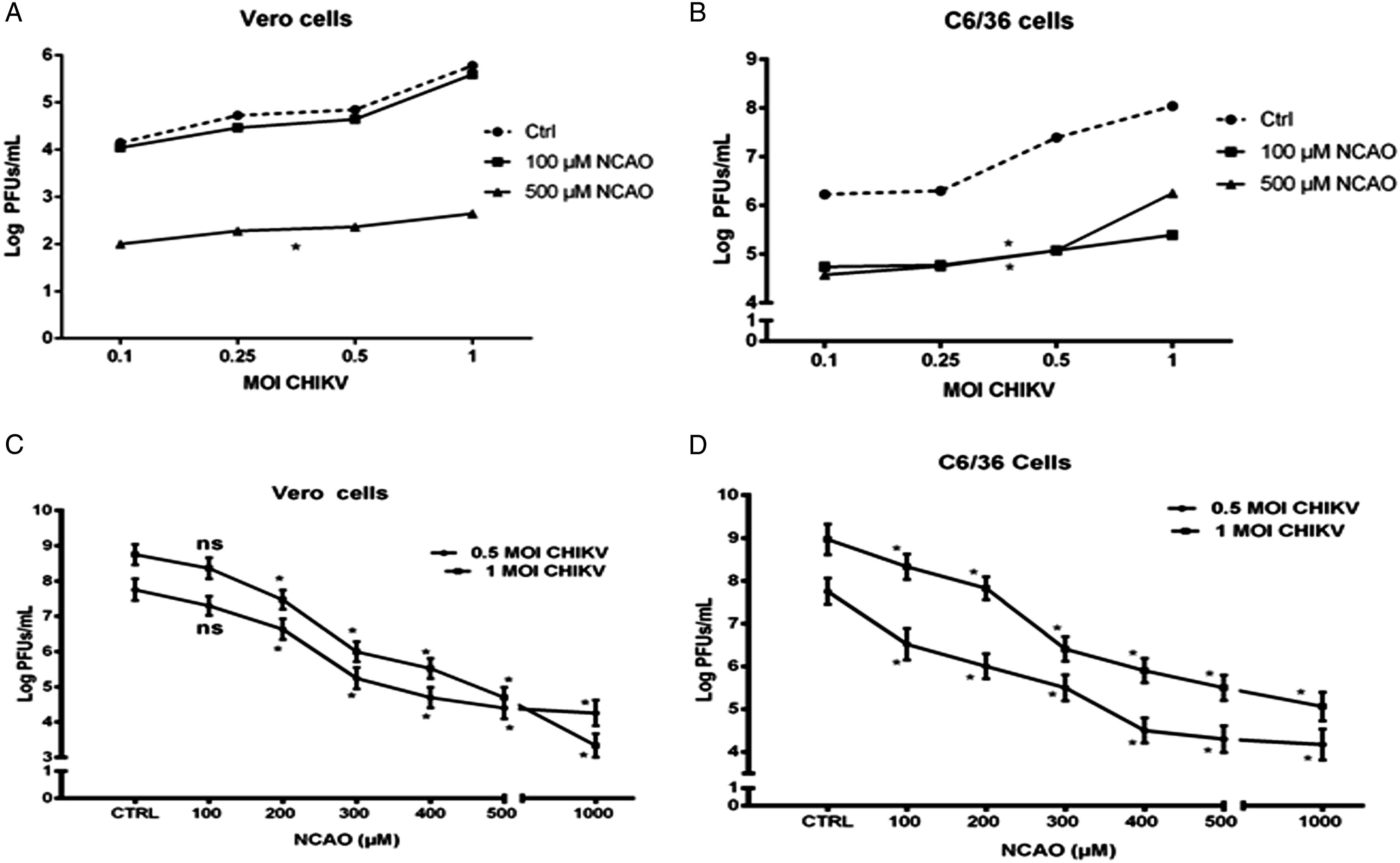
Yield of CHIKV in titres due to the addition of PAs in Vero and C6/36 cells
Pretreatment with 300 μM NCAO in Vero cells and subsequent infection with CHIKV at concentrations of 0.1 and 0.5 MOI showed a decrease in the viral titre of almost 2 logarithms of PFUs (Figure 4A). In the case of C6/36 cells, the same pretreatment process was performed, and there was a reduction in the viral titre by almost 3 logarithms of PFUs in cells infected at 0.5 MOI, and by 2 logarithms for those infected at 0.1 MOI with CHIKV (Figure 4B). By adding 10 μM PAs, a statistically significant increase (ANOVA p < 0.01) in the viral titre was observed in the Vero cells, like the control cells (Figure 4A). This increase was greater in cells to which putrescine was added (Figure 4A). The same effect was observed in infected cells at 0.1 and 0.5 MOI (Figure 4A). In C6/36 cells, a statistically significant increase (ANOVA p < 0.01) in the viral titre was also observed after the PAs were added, mainly in those cells treated with putrescine and spermidine and infected at 0.1 MOI (Figure 4B). Yield of CHIKV from the titres via the addition of PAs in Vero and C6/36 cells. Vero cells and C6/36 cells were pretreated with a concentration of 300 μM NCAO; then, 10 μM of the PAs were added during infection with CHIKV at concentrations of 0.5 and 1 MOI. Titres were determined at 24 hpi for Vero (A) and C6/36 cells (B). Statistical significance was determined using the ANOVA test: *p < 0.01, n = 3, ns, no significant difference.
Intracellular PAs in Vero and C6/36 cells
The concentration of intracellular PAs in the Vero and C6/36 cells exhibited a statistically significant decrease in the presence of NCAO during the infection at 0.5 and 1 MOI and was more evident when Vero cells were pretreated with NCAO before the infection with CHIKV (ANOVA *p < 0.01) (Figure 5A and 5B). Moreover, the content of intracellular PAs was higher in Vero cells than in C6/36 cells; the polyamine observed in greatest quantity was putrescine, even in the presence of NCAO, which decreased significantly in each of the conditions tested. The contents of spermidine and spermine decreased even in the presence of putrescine, the precursor PA. Interestingly, a significant decrease in spermidine content was also observed during infection with CHIKV in both cell lines (ANOVA *p < 0.01) (Figure 5A and 5B). Intracellular PAs in Vero and C6/36 cells. The Vero and C6/36 cells were pretreated with 300 μM NCAO and infected with 0.5 and 1 MOI CHIKV. Quantification of intracellular PA content was performed with HPLC for Vero (A) and C6/36 cells (B). Statistical significance was determined using the ANOVA test: *p < 0.05 n = 3, ns, no significant difference.
Viability in human fibroblast BJ with different concentrations of NCAO
Viability assays in human fibroblast BJ treated with 100 μM NCAO 24 and 48 h after exposure showed that there was no cytotoxic effect (Figure 6); however, at 72 h after exposure the cell viability decreased to 77%. This decrease was statistically significant compared to the control (p < 0.0001). The results showed that at concentrations higher than 100 μM there is a statistically significant (p < 0.0001) reduction in viability with respect to the control after 24 h of exposure to NCAO. We observed that the EC50 was not reached at concentration of 1000 μM (Figure 6). Cytotoxic effect of NCAO in human BJ fibroblast at 24, 48 and 72 h. Each point represents the average of 3 technical replicates and 3 biological replicates (n = 9) with the standard deviation in each point. Δ represents the points for p > 0.05 (one way ANOVA) compared to the control group in 24 h and O for 48 h.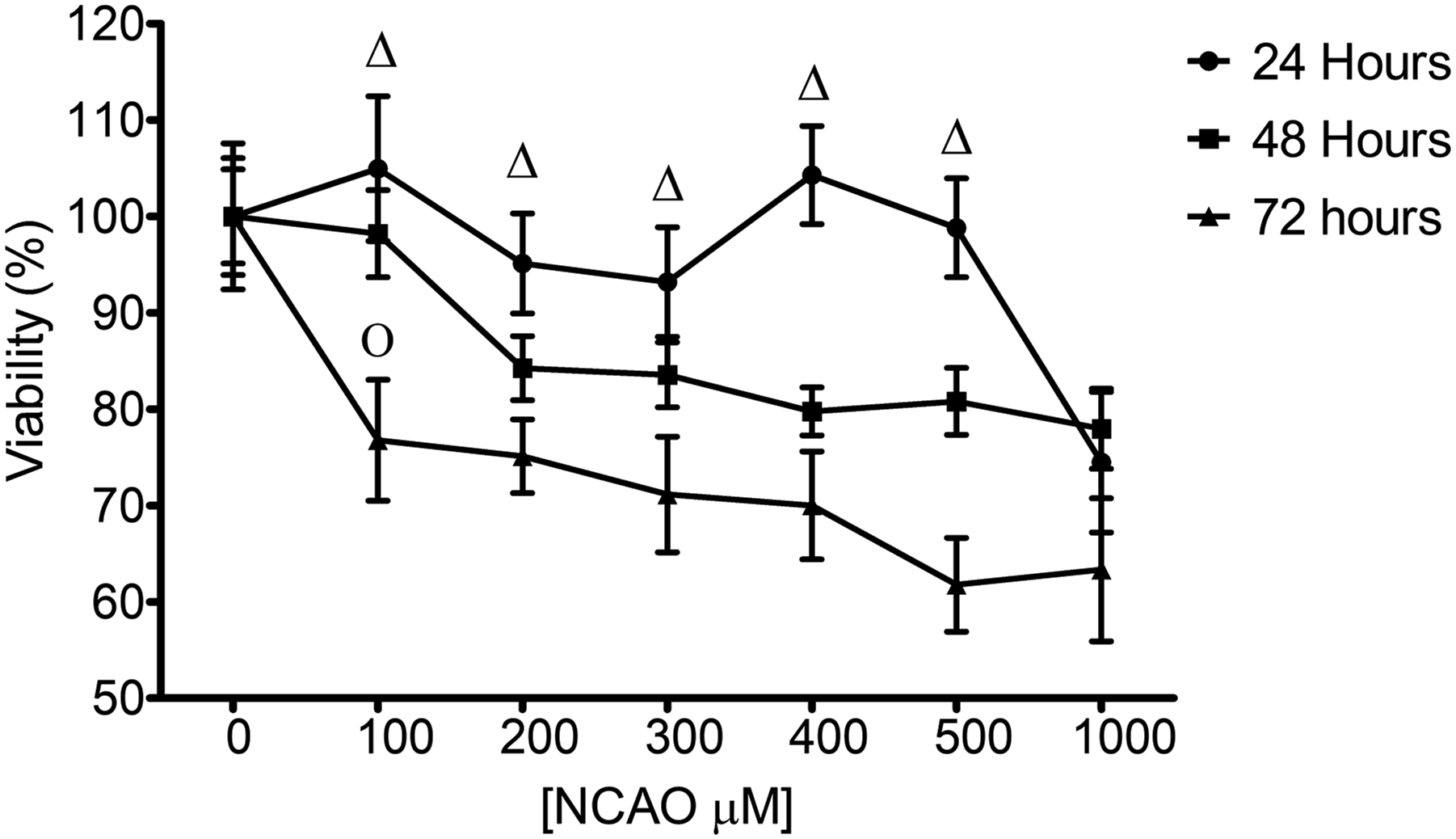
Inhibition of CHIKV replication by NCAO in human fibroblast BJ
Inhibition of chikungunya virus replication was observed in human fibroblast BJ treated with 100 μM NCAO 24 h before and 48 h after the infection with CHIKV at a MOI 1. There was a reduction from 2.6 log PFU/mL to 1.6 log PFU/mL, this difference was statistically significant p < 0.0001 (Figure 7). However, no significant decrease in CHIKV titre was observed in fibroblasts treated with 100 μM NCAO at 24 h before and after infection (Figure 7). Effect of NCAO in CHIKV replication in human fibroblast BJ at 24 and 48 h. Each bar represents the mean with SD of 2 technical replicates and 2 biological replicates. Δ and O represents the points where there was not significant difference, p > 0.05 (one way ANOVA).
Discussion
CHIKV infections are a public health problem due to the increases in the number of cases spread across several continents [15]. The widespread dissemination of CHIKV, its associated high morbidity rate, the lack of available treatments and the limited knowledge about the molecular mechanisms involved in the replication process provide an opportunity for investigation of these topics to facilitate the development of antiviral agents or effective vaccines for control of the infection [2]. Several compounds have been proposed, including chloroquine [16], ribavirin [17], arbidol [18], harringtonine [19], suramin [20] and silymarin [21], which act in different stages of the viral replication cycle. However, these options have certain disadvantages that make them ineffective for use in CHIKV infections [22]. Mounce et al. showed that the PAs putrescine, spermidine and spermine are important for the replication of a various RNA viruses, including chikungunya, and participate in other molecular processes, such as packaging of the viral genome [10,11]. In DNA viruses, polyamines stabilize the negatively charged genome within the virion particle, for example, the bacteriophage T7 cytomegalovirus (CMV) virions [23] and vaccinia virus [24]. Therefore, we evaluated whether N-ω-chloroacetyl-L-ornithine has antiviral effects by inhibiting the replication of CHIKV, due to NCAO is a competitive inhibitor of ornithine decarboxylase and decrease the biosynthesis of PAs [14]. We demonstrated, through kinetic inhibition assays, that NCAO inhibits the replication of CHIKV in Vero and C6/36 cells in a dose-dependent manner (Figure 3A and 3B) and that this inhibition is due to a depletion of the intracellular PAs in both cell lines; this was demonstrated by the PA HPLC quantification (Figure 5). Moreover, the inhibition increased when more NCAO was added to maintain the depletion of PAs (Figure 3C and 3D). Thus, there is a relationship between the levels of PAs and viral replication. All assays were performed 24 h after exposure to NCAO because NCAO did not significantly affect the cellular viability in all concentrations examined. To confirm that the inhibition of viral replication was not due to the possible cytotoxicity of NCAO or a decrease in the viability of Vero cells, these cells were subjected to different concentrations of NCAO; after 24 h, they were washed, identically to the cultures infected with CHIKV. Then, NCAO was added at the original concentration (Supplementary S1). After another 24 h, the viability was measured, and the recovery viability was confirmed to be due to the nutrients in the added culture medium. Therefore, we determined that the observed inhibition was due to the effect of NCAO on polyamines and not due to cytotoxicity. These results match those described by Mounce et al., who demonstrated that a decrease in viral replication occurs due to the depletion of polyamines in the cell lines C6/36 and BHK21 and Vero cells induced by inhibitors of PA synthesis [10,11]. The suicide inhibitor of ODC (difluoro-methyl ornithine (DFMO)) and diethylnorspermine (DENSpm; an activator of spermidine/spermine N1-acetyltransferase (SAT1)) induce the exhaustion of spermidine and spermine via the induction of SAT1 [10,11]. The results also indicated that the decrease in PAs generated by these compounds exerts a negative effect in vivo and in vitro on the replication of various RNA viruses, including chikungunya [25,26]. However, the great potential of DFMO for combatting viral infections is overshadowed by the adverse effects that would occur under high doses of this compound for counteracting the replacement of the ODC [27]. Moreover, the administration of type I interferon to induce STAT1 activity could represent a high cost.
In addition, inhibition of CHIKV replication was evaluated in human BJ skin fibroblasts (normal cells), since fibroblasts are susceptible to CHIKV infection following mosquito bite. Dermal fibroblasts induce the activation of innate immune responses such as IFNβ secretion, this has been described in HFF-1, HDF cell lines infected with dengue and Zika arboviruses [28–30] and in HT-1080 and HS 633T infected with CHIKV [31]. When treated with 100 μM NCAO, a decrease in viral replication was observed at 48 hpi (Figure 7), like what was observed in Vero and C6/36 cells.
CHIKV replication in fibroblasts at 24 hpi with and without 100 μM NCAO can be explained by the inhibitory effect of Nsp2 and Nsp3 on the IFNβ pathway [32,33]; however, at 48 hpi intracellular antiviral activity decreases viral replication (Figure 7), which is enhanced (up to 1 log) by exposure to 100 μM NCAO. This suggests that NCAO-induced polyamine depletion could have a synergistic effect with IFNβ in inhibiting viral replication in BJ dermal fibroblasts at 48 hpi.
Therefore, NCAO could be an alternative antiviral, the usefulness of which is being assessed in this study and studies of the antiviral effect and possible adverse effects in a C57BL/6 murine model of subacute CHIKV infection can be performed later [34].
In the present work, we have demonstrated that CHIKV requires polyamines for replication. When adding exogenous PAs, CHIKV was yielded in titres from both cell lines pretreated with NCAO (Figure 4). Therefore, NCAO inhibits viral replication by blocking the PA biosynthesis pathway, which agrees with Mounce’s findings [25,26] on the yield of viral titres in different viruses such as chikungunya when biogenic PAs are added after pretreatment with DFMO [10,11]. To demonstrate which of the PAs has the best effect, putrescine, spermidine and spermine were individually added; the best yield of the viral titres was observed with putrescine. This suggests that putrescine is more easily captured by the cells and used as a precursor for the synthesis of the other polyamines, which could be demonstrated by quantifying spermidine and spermine in the cells exposed to putrescine.
To rule out the possibility that viral replication was diminished by low viability, the viability of Vero or C6/36 cells in the presence of NCAO was determined. It was observed that NCAO did not exert cytotoxic effects during treatment (Figure 1D, 1E, and 1F). However, the viability in Vero cells decreased (Figure 1A, 1B and 1C) at concentrations above EC50 at 48 and 72 h (Figure 2). Interestingly, the intracellular PAs had higher concentration in the Vero cells than in the C6/36 cells (Figure 5), which raises questions about the efficiency of replication, because this virus requires PAs and replicates more efficiently in C6/36 cells. Furthermore, it has been described that the enzymes involved in the synthesis and catabolism of PAs are conserved throughout various animal species. Recent studies of polyamines in the mosquito Aedes aegypti showed that the content of PAs likely depends on the enzyme aaNAT5b, which is an acetyltransferase (SAT). SAT can act as a viral restriction factor by decreasing intracellular PAs [35]. However, there are likely to be other mechanisms in cells that allow the virus to replicate successfully in the absence of polyamines.
Regarding the concentration of PAs in Vero and C6/36 cells during infection with CHIKV (Figure 5), a decrease in the presence of NCAO was observed at the two MOIs tested (Figure 5A and 5B), which was expected because the NCAO decreased the pools of intracellular PAs by inhibiting the ODC [14]. Moreover, spermidine and spermine were diminished because the virus consumed these molecules during replication. In several experimental studies, it has been demonstrated that the activity of RNA-dependent RNA polymerases (RdRPs) of CHIKV [25] and Semliki Forest viruses (SFVs) [9] are diminished in the absence of polyamines, and their activity is restored by adding spermidine [25,26]. Similarly, it has been shown that spermidine is important in one of the most critical steps of viral replication–translation because the depletion of PA limits the synthesis of non-structural proteins, including viral polymerase [10,11], because spermidine plays an important role in the initiation and lengthening of translation through hypofunctioning the 5A initiation factor (elF5A) [36]. Indeed, bunyaviruses and alphaviruses replicate under the supplementation of 100 nM polyamines in polyamine-depleted cells. Thus, these viruses may have evolved mechanisms to take advantage of polyamines, even at low concentrations [37].
In this study, high quantities of putrescine were observed, even in the presence of NCAO, which suggests that the cells use other mechanisms to ensure the content of this PA. Yet, the mechanism by which PA is transported has not been fully elucidated. However, it is known that the transport of putrescine to the interior of the cell could occur through the exporter SLC3A2, which is present in the cell membrane [38]. Interestingly, in the treatment with NCAO, the spermidine and spermine content decreased even in the presence of putrescine, the precursor PA. This result suggests that NCAO could interact with some other enzymes of the biosynthetic route, in addition to ODC. N-ω-chloroacetyl-L-ornithine (NCAO) inhibited CHIKV replication in a dose-dependent manner in Vero and C6/36 cells by depleting the intracellular polyamines. Spermidine may have a more complex role in the process of translation and transcription of the virus. Firpo et al. suggest that polyamines are critical to coronavirus replication and represent a highly promising drug target for the contemporary, and any future, coronavirus outbreaks [37]. Subsequent studies should provide more information about the possible uses of this compound, or its related derivatives, as antiviral agents for RNA viruses.
Supplemental Material
Supplemental Material - Inhibition of chikungunya virus replication by N-ω-chloroacetyl-L-Ornithine in C6/36, Vero cells and human fibroblast BJ
Supplemental Material for Inhibition of chikungunya virus replication by N-ω-chloroacetyl-L-Ornithine in C6/36, Vero cells and human fibroblast BJ by Lucero Rojas Luna, Araceli Posadas-Modragón, Amanda M Avila-Trejo, Verónica Alcántara-Farfán, Lorena I Rodríguez-Páez, José Angel Santiago-Cruz, Marvin O Pastor-Alonso and J Leopoldo Aguilar Faisal in Antiviral Therapy
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto Politécnico Nacional (IPN) SIP 20195289.
Ethical Approval
The project was reviewed and approved by the biosafety committee of the Escuela Superior de Medicina, IPN.
ORCID iDs
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
