Abstract
Nylon 6 fiber was among the first synthetic fibers to be industrialized and is widely utilized across textiles, automotive, construction, and electronics due to its excellent mechanical and chemical properties. However, its inherent flammability and tendency to drip when melted limit its application in fire-sensitive environments. In response, significant efforts have been made to improve its flame-retardant performance. This review focuses on recent advances in halogen-free flame-retardant strategies specifically developed for Nylon 6 fibers. Flame retardants based on phosphorus, nitrogen, and nanoscale materials have been incorporated into the polymer matrix through methods such as blending, in situ polymerization, and copolymerization. These modifications aim to enhance flame retardancy by increasing the limiting oxygen index, reducing heat release, and improving thermal stability, while maintaining mechanical strength and spinnability. Despite substantial progress, challenges remain, including the toxicity of combustion byproducts, limited environmental compatibility of some additives, and negative effects on processing performance. This review emphasizes the structure–property–performance relationships that govern flame-retardant efficiency and highlights multifunctional designs offering improved processability, thermal protection, and environmental safety. By critically evaluating current materials and methods, the review provides technical insight into the development of high-efficiency, halogen-free flame-retardant systems for Nylon 6 fibers and outlines potential directions for future research.
Keywords
Introduction
Nylon 6 fiber is widely used in electrical engineering, machinery manufacturing, transportation, textiles, and apparel due to its excellent comprehensive performance.
1
In addition to Nylon 6, other commonly used synthetic fibers such as Nylon 66 and polyester (PET) also play vital roles in industrial and consumer applications. Nylon 66 features higher crystallinity and better thermal resistance than Nylon 6, while polyester fibers are favored for their mechanical strength and cost-effectiveness. However, similar to Nylon 6, both Nylon 66 and polyester are inherently flammable, which poses challenges in fire-sensitive applications. A comparative discussion of their flame-retardant performance is provided in Table 4. However, Nylon 6 fibers, in particular, are more susceptible to ignition due to their unique morphological structure. These fibers possess a high surface area and relatively loose molecular arrangement, allowing for rapid heat accumulation and facilitating ignition. The alignment of polymer chains and the presence of crystalline regions further contribute to this flammability, especially under heat or flame exposure.
2
Figure 1
3
presents an SEM image of Nylon 6 fibers, illustrating the surface morphology at a specific magnification. The fiber surfaces appear smooth and regular, with a tightly packed structure that contributes to dimensional stability and mechanical strength. However, even at this magnification, subtle surface irregularities and microstructural features are visible, which could potentially impact heat dissipation. These microscopic features may lead to localized heat accumulation, causing the fiber surface to reach ignition temperatures more quickly under high-temperature conditions. The smooth surface of the fibers inhibits efficient heat dissipation, causing the surface to heat up rapidly, thus increasing the likelihood of ignition and subsequent combustion. As a thermoplastic material, Nylon 6 has a relatively low melting point (around 220°C), which leads to melting and decomposition at elevated temperatures, releasing combustible gases that can accelerate fire spread. Furthermore, the dense packing of the fibers, while enhancing mechanical performance, may contribute to the retention of heat and gases during combustion, further facilitating the fire propagation process. Consequently, Nylon 6 fibers exhibit high flammability when exposed to high-temperature environments. The structure of Nylon 6 consists of amide groups (-CONH-) connected by carbon atoms along the polymer backbone, as shown in the following chemical formula: [-NH(CH2)5CO-]n. This structure imparts both thermal stability and mechanical strength to the material. However, it also makes Nylon 6 susceptible to decomposition at elevated temperatures, releasing flammable gases during combustion. Additionally, the alignment of polymer chains and the crystalline regions in Nylon 6 fibers contribute to increased heat accumulation and facilitate ignition, particularly in fire-prone environments. SEM image of Nylon 6 fiber.
3

Additionally, during combustion, Nylon 6 fibers are more likely to produce melting drops, which spread the fire, greatly endangering human life and property safety. This behavior also limits the application of Nylon 6 fibers in certain specialized fields, such as automotive, construction, and electronics, where fire resistance is critical. 4 The significance of enhancing flame retardancy in Nylon 6 fibers is well-supported by several fire-related accidents involving Nylon 6 products, which have resulted in substantial economic losses. For instance, in the textile industry, fires caused by the flammability of Nylon 6 fibers have led to severe property damage and human casualties. A notable case occurred in 2016 when a textile factory experienced a large-scale fire due to the combustibility of Nylon 6 fibers, resulting in significant damage to both materials and equipment, with estimated losses exceeding several million USD. Similarly, the automotive industry has reported fire incidents linked to the use of Nylon 6 in interior components. In one such case, a fire broke out in a vehicle's interior, where Nylon 6 materials were involved. Although no fatalities occurred, the damage to the vehicle was severe, leading to compensation and recall costs amounting to millions of dollars. According to industry estimates, fire-related accidents involving synthetic polymer materials—including Nylon 6—result in global economic losses exceeding USD 10 billion annually. 5 These statistics underscore the urgent need for effective flame retardant solutions, particularly in fire-sensitive sectors such as textiles and automotive manufacturing, to mitigate fire hazards and enhance safety.
Extensive research has therefore been dedicated to the development of flame-retardant Nylon 6 fibers to address these challenges, with various flame retardants such as halogens, nitrogen, phosphorus, silicon, and nanoscale additives being introduced into the base material via methods like “blending,” “in-situ polymerization,” and “copolymerization”.6–8 However, halogenated flame retardants pose environmental concerns, including the large amounts of halogen acid produced during combustion, which can cause “secondary poisoning” and damage to the environment. As a result, halogen-free flame retardants have become an inevitable trend in the research and development of flame-retardant Nylon 6 fibers. In this context, it is essential to understand the molecular structure of flame retardant systems, particularly how the rational allocation of flame retardant elements—such as phosphorus and nitrogen—can significantly enhance flame retardant performances. The interaction between flame retardant molecules and the polymer matrix plays a critical role in the material’s flame resistance, and optimizing this molecular design can help achieve better performance without compromising the mechanical properties of the fibers.9–11 In addition, global environmental policies—such as the European Union’s REACH regulation and RoHS directive—have accelerated the transition toward halogen-free alternatives by limiting the use of hazardous substances. These regulatory drivers have catalyzed significant advancements in the field. However, despite these developments, no comprehensive review has yet been published focusing exclusively on halogen-free flame-retardant Nylon 6 fibers. This review aims to fill that gap by summarizing the progress over the past 15 years.
This work provides a systematic overview of the latest strategies for enhancing the flame retardancy of Nylon 6 fibers, including blending, in situ polymerization, and copolymerization techniques (as illustrated in Figure 2). It will discuss the effects of these flame retardants on the flame retardancy, thermal stability, and mechanical properties of Nylon 6 fibers, along with their respective flame-retardant mechanisms. Furthermore, this review will cover the various applications of flame-retardant Nylon 6 fibers and provide insights into the key challenges and future research opportunities associated with these flame-retardant approaches. The advancements and trends shown in the timeline figure underscore the ongoing efforts to enhance the flame retardancy of Nylon 6 fibers through innovative strategies and materials. Progress roadmap for Halogen-free flame-retardant nylon 6 fiber.
Halogen-free flame retardant nylon 6 fiber research progress
In the field of research on halogen-free flame-retardant nylon 6 fibers, the commonly employed flame retardancy methods can primarily be categorized into three types 12 : (1) Introducing flame retardancy by blending (2) Introducing Flame retardancy through in situ polymerization (3) Introducing Flame retardancy through copolymerization.
Introducing flame retardancy by blending
Comparison of different flame retardants used in nylon 6 fiber blending.
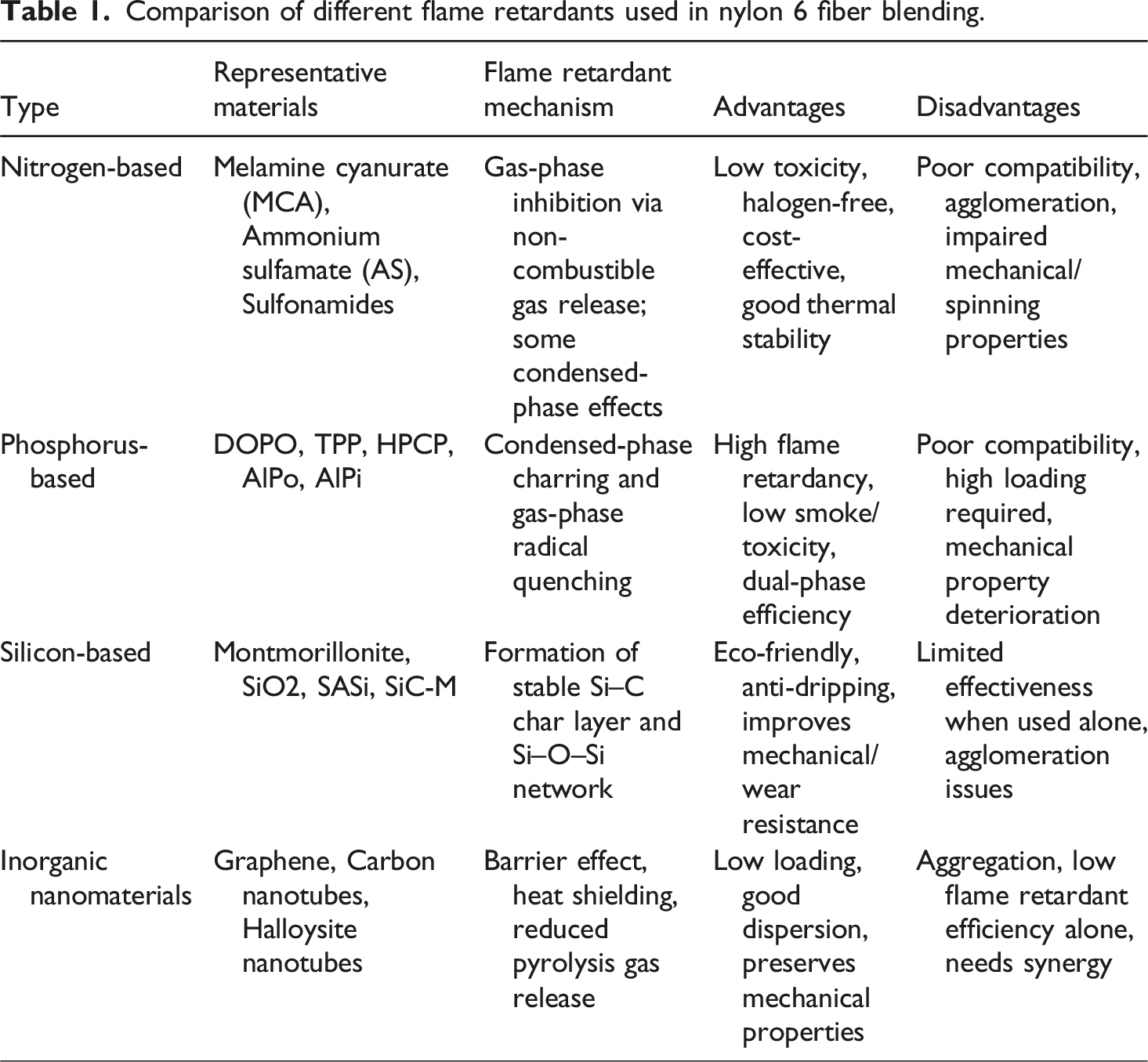
Nitrogen-based flame retardants, which are low-toxic, halogen-free, thermally stable, and cost-effective, are among the most commonly used halogen-free flame retardants for blended flame-retardant nylon 6 fibers.
13
Among them, melamine and its derivatives, especially melamine cyanurate (MCA), due to their carbon-nitrogen triazine ring structure, release a large amount of non-combustible gases during combustion, effectively suppressing the combustion of the substrate, making them typical representatives of nitrogen-based flame retardants used in nylon 6 fibers. The synthesis route of the nitrogen-containing flame retardant N, N-Dimethylphosphonomethylamine (DPMA) is illustrated in Figure 3.
14
Synthetic route of DPMA.
14

However, MCA typically has poor compatibility with nylon 6, prone to agglomeration, which greatly affects the spinnability of nylon 6. To address this issue, Šehić et al. 15 co-incorporated carbon black (CB) and MCA into nylon 6, demonstrating that under the influence of CB, the compatibility of MCA with nylon 6 was effectively enhanced, improving the flame retardancy of the resultant nylon 6 fibers. Moreover, sulfonamide-based flame retardants, capable of acting in both gas-phase and condensed-phase flame retardancy, are also commonly used nitrogen-based flame retardants for preparing flame-retardant nylon 6 fibers. Research by Coquelle et al. 16 found that introducing ammonium sulfamate (AS) into the nylon 6 matrix, with a 7 wt% AS content, resulted in a 30% reduction in the peak heat release rate (pHRR) of the obtained flame-retardant nylon 6 fibers, though with inferior mechanical properties. Xiang et al. 17 also blended α-zirconium phosphate (α-ZrP) and AS with nylon 6 to produce flame-retardant nylon 6 materials. When the contents of α-ZrP and AS were 3 wt% and 2 wt%, respectively, the LOI value of flame-retardant nylon 6 increased to 38.7%, achieving a UL94 V-0 rating, with the tensile strength and elongation at break of the fibers reaching 1.9 ± 0.07 cN/dtex and 70.4 ± 3.45%, respectively. Sun et al. 18 produced MWNT-GAS by modifying carbon nanotubes with guanidine sulfamate (GAS), and upon adding 3% MWNT-GAS to the nylon 6 matrix, the LOI value of the resultant flame-retardant nylon 6 fibers increased to 24.1%.
Phosphorus-based flame retardants are widely recognized for their high efficiency, low smoke emission, and low toxicity. They represent the most extensively studied category of halogen-free flame retardants for nylon 6 fibers.
During combustion, these flame retardants decompose to generate oxyacids such as phosphoric acid, phosphorous acid, and polyphosphoric acid. These oxyacids promote the dehydration of the nylon 6 matrix, leading to the formation of a stable, phosphorus-rich char layer. This char acts as a protective barrier, isolating the material from external oxygen and reducing heat transfer, thereby delivering excellent condensed-phase flame retardancy.
In addition to their condensed-phase action, phosphorus-based flame retardants also exhibit gas-phase flame retardant mechanisms. Specifically, they release PO· radicals during thermal degradation, which scavenge highly reactive H· and OH· radicals produced by the combustion of nylon 6. This radical quenching interrupts the propagation of the combustion chain reactions and contributes to the release of flame-retardant or non-combustible gases. Together, these effects significantly inhibit flame spread and improve the overall flame retardancy of the nylon 6 matrix.19,20
Liu et al.
21
introduced 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) into the nylon 6 matrix, reaching an LOI value of 31.5% with a 12wt% addition of DOPO. Shi et al.
22
blended triphenyl phosphate (TPP) and MCA with nylon 6. With 0.1wt% TPP and 9wt% MCA added, the flame-retardant nylon 6 fibers achieved a UL94 V-0 rating. Liu et al.
23
utilized aluminum phenylphosphinate as the primary flame-retardant component and combined it with melamine cyanurate (MC) to synthesize a phosphinate-based flame retardant system. This formulation was mechanically blended with glass fiber and polyamide 6 (PA6) at a weight ratio of 18:30:52 using a twin-screw extruder to obtain a homogeneous molten mixture. The resulting blend was pelletized into flame-retardant PA6 chips, which were subsequently used to prepare modified PA6 materials. The modified PA6 exhibited significantly enhanced flame-retardant performance. Thermogravimetric analysis (TGA) indicated a slight reduction in the initial decomposition temperature, suggesting a shift in the thermal degradation pathway. However, the degradation process proceeded more slowly at elevated temperatures, and a notable increase in char residue was observed, implying improved condensed-phase flame-retardant behavior. In addition, the material’s Limiting Oxygen Index (LOI) increased from 22.7% to 32%, and it successfully achieved a UL-94 V-0 rating in vertical burning tests. These results confirm the efficacy of the phosphinate-based system in enhancing the thermal stability and flame resistance of PA6. Gao et al.
24
successfully prepared flame-retardant nylon 6 (PA6) fibers by incorporating Hexaphenoxycyclotriphosphazene (HPCP) as a phosphorus-based flame retardant. With the addition of 15 wt% HPCP, the limiting oxygen index (LOI) of the resulting fibers reached 29.8%, indicating excellent flame retardancy. The composite fibers were fabricated via a melt spinning process, schematically illustrated in Figure 4. In this process, PA6 chips and HPCP were melt blended and pelletized into masterbatches. The masterbatches were then introduced into a single-screw extruder, where they were melted and homogenized. The molten polymer was filtered through a 35 µm filter to eliminate aggregates and extruded through a spinneret with a diameter of 0.35 mm. The extrudate was cooled and solidified in a cooling shaft and subsequently collected on a winding drum at a take-up speed of 850 m/min. The formulations used in this process are summarized in Table 2. Schematic illustration of the melt spinning process used to prepare pure PA6 and PA6/HPCP composite fibers.
24
Composition (wt%) of different spinning formulations. Adapted with permission from Journal of Industrial Textiles, 2021, 51 (4): 577–593. © SAGE Publications.

However, phosphorus-based flame retardants have drawbacks such as poor compatibility with the matrix and significant impact on the mechanical properties of the matrix, usually requiring the use of reinforcing materials to mitigate these issues.25,26 Hai Vothi and colleagues synthesized an aluminum methyl methoxyphosphonate (AlPo)-based flame retardant (FR). Thermal degradation and flame retardancy of nylon 6 (PA6)/AlPo composites were examined and compared with PA6/commercial aluminum diethylphosphinate (AlPi) composites. The PA6/AlPo composite achieved a V-0 rating at 20 wt% loading during the UL-94 test, and it exhibited the formation of a charred layer that protected the polymer from burning and reduced the release of gases during the combustion of PA6. AlPo demonstrated exceptional performance in gaseous and condensed phases in the PA6 matrix, whereas AlPi only worked in the gaseous phase. 27
Silicon-based flame retardants, recognized for their high flame retardant efficiency, low toxicity, anti-dripping properties, and environmental friendliness, represent a promising class of halogen-free condensed-phase flame-retardant materialsl.28,29 These retardants are generally classified into organic and inorganic types. Organic silicon-based flame retardants commonly used in nylon 6 include organosiloxanes (e.g., polydimethylsiloxane), silicone oils (e.g., methylphenyl silicone oil), silicone rubbers, and cage-like polysilsesquioxanes (e.g., polyhedral oligomeric silsesquioxane, POSS). In contrast, inorganic silicon-based flame retardants primarily consist of silicates, such as montmorillonite and hectorite, as well as nanoscale silicon dioxide (SiO2) particles.30,31 These compounds contribute to flame retardancy mainly by promoting the formation of a thermally stable, silica-rich char layer during combustion, which acts as a physical barrier to heat and mass transfer. Alisa 32 incorporated sodium silicate (SASi) into the nylon 6 matrix. Nylon 6 fibers with a 4wt% addition of SASi achieved a char yield of 17.6%, maintaining good mechanical properties. Wu 33 blended montmorillonite, aluminum organophosphate,and styrene-ethylene-butylene-styrene grafted with maleic anhydride (SEBS-g-MA) into nylon 6, where just 5wt% montmorillonite was required to reduce the peak heat release rate (pHRR) of the flame-retardant fibers by 7.8% and increase the elongation at break to 127%. Tian 34 introduced phosphorus amide-modified montmorillonite into nylon 6, resulting in flame-retardant nylon 6 fibers with an LOI value of 28.6%, with minimal loss in mechanical properties, maintaining superior spinnability. Wu et al. 35 combined montmorillonite and organic hypophosphite; with the introduction of 5wt% montmorillonite and 20wt% organic hypophosphite, the pHRR of flame-retardant nylon 6 fibers decreased by more than 50% compared to pure nylon 6 fibers, achieving a UL94 V-0 rating. Sun et al. 36 utilized the silane coupling agent KH-560 to graft melamine polyphosphate (MPP) onto the surface of silicon carbide (SiC) particles, producing modified silicon carbide (SiC-M). This SiC-M was then melt-blended with nylon 6 (PA6) to prepare silicon carbide modified PA6 composite materials (PA6/SiC-M). When the mass fraction of SiC-M was 10%, the composite material achieved a UL94 V-0 rating, with a limiting oxygen index of 29.8%. At this same mass fraction of 10% SiC-M, the composite material exhibited the highest coefficient of friction. With a SiC-M mass fraction of 4%, the composite material reached a bending strength of 285 MPa and a tensile strength of 51 MPa. The addition of SiC-M enhances the flame retardancy, wear resistance, and mechanical properties of the composite material. In summary, silicon-based flame retardants form a dense and stable silicon-carbon (Si-C) char layer and an interpenetrating Si-O-Si network structure upon combustion, endowing the nylon 6 matrix with excellent anti-dripping performance. However, when used alone and in low quantities, silicon-based flame retardants generally cannot achieve an ideal level of flame retardancy, necessitating combination with other gas-phase flame retardants for enhanced effectiveness.
Inorganic nanomaterial flame retardants have emerged as a promising class of flame-retardant additives owing to their nanoscale dimensions, non-toxic nature, environmental sustainability, excellent thermal stability, and low required loading levels, which help mitigate the deterioration of mechanical properties in polymer composites. Commonly used nanomaterials include halloysite nanotubes, graphene, and carbon nanotubes.37,38 However, achieving uniform dispersion of these nanomaterials within the polymer matrix remains a significant challenge due to their strong tendency to agglomerate, stemming from high surface energy and van der Waals interactions. This aggregation not only reduces the effectiveness of the flame retardant but also compromises the mechanical integrity of the composite. To address these issues, various strategies have been employed, such as surface functionalization of nanoparticles, incorporation of compatibilizers, and utilization of high-shear mixing or in situ polymerization techniques, all of which enhance interfacial compatibility and promote homogeneous dispersion. Kashiwagi and colleagues39,40 studied the flame retardant modification of polyamide 6 (PA6) by blending with nanoclay, with the addition amount of nanoclay being in the range of 2% to 5% by mass and a particle size of 1.3 to 1.4 nm. Using thermogravimetric analysis, electron microscopy, and X-ray diffraction, they analyzed the flame retardant mechanism of the nanocomposites. They discovered that during combustion, clay particles in the system are pushed to the surface by bubbles generated from degradation, forming a protective layer with a ‘sea-island’ flocculent structure, which reduces the heat release. Sun
41
employed a synergistic approach by integrating halloysite nanotubes (HNTs) with melamine polyphosphate (MPP), resulting in nylon 6 fibers that, while only achieving a Limiting Oxygen Index (LOI) value of 24%, demonstrated exceptional spinnability and mechanical properties. Zhang incorporated boron acid modified graphene oxide (BAC-GO) into nylon 6, and with a mere 0.9wt% inclusion of BAC-GO in the flame-retardant nylon 6 fibers, an increase in LOI to 25.5% was observed, alongside significant reductions in peak heat release rate (pHRR) and total heat release (THR). Figure 5
42
presents a schematic representation of graphene oxide modified through covalent bonding. Schematic diagram of covalently bonded modified graphene oxide.
42

Inorganic nanomaterials, during combustion, effectively act as barriers to heat transfer while also inhibiting the ingress of oxygen and the release of pyrolytic byproducts. Nevertheless, nanomaterial-based flame retardants also face challenges, such as proneness to agglomeration and limited flame-retardant efficacy. Moreover, within the research domain of flame retardants for nylon 6 fibers, nanomaterial flame retardants typically cannot function independently. They require synergistic application with nitrogen and phosphorus flame retardants to enhance their practical flame retardancy effectively. Liu et al. 43 modified Nylon 6 by melt blending with maleic anhydride grafted polyethylene (PE-g-MAH) or nano-clay reinforced Nylon (NYC). Their study found that when the content of PE-g-MAH was approximately 8 wt% or the content of NYC was around 0.5 wt%, the elongation and tensile properties reached their optimal values at the optimized stretching temperature of 120°C.
Blending flame-retardant additives with nylon 6 fibers is widely recognized for its operational simplicity, low cost, and ease of scale-up for industrial production. This approach offers broad applicability and compatibility with existing manufacturing infrastructure. However, it suffers from significant limitations. The use of flame retardants with large particle sizes often necessitates high loading levels, which increases the risk of agglomeration, migration, and leaching to the fiber surface. These issues are further exacerbated by poor dispersibility within the polymer matrix, ultimately compromising the mechanical integrity and spinnability of the resulting fibers. Such drawbacks present persistent challenges, particularly in high-performance applications where uniform dispersion and long-term stability are critical. Additionally, the environmental implications of additive leaching and the lack of permanence in flame retardancy raise concerns regarding product safety and sustainability. Addressing these limitations calls for innovative formulations or dispersion techniques that can improve compatibility between flame retardants and the polymer without undermining processing performance or mechanical properties.
Introducing flame retardancy through in situ polymerization
In situ polymerization flame retardancy, which involves incorporating additive flame retardants during the hydrolytic ring-opening polymerization of nylon 6, offers a significant advantage over conventional blending methods by promoting superior dispersion of flame retardants within the polymer matrix. This improved dispersion enhances flame-retardant efficiency and facilitates better integration of the flame retardant at the molecular level. 44 Commonly employed flame retardants in this method include organic phosphorus-based, inorganic nitrogen-based, inorganic nanomaterials, and bio-based flame retardants.
Notably, in situ polymerization is particularly effective in mitigating the adverse effects of organic phosphorus-based flame retardants on the mechanical properties of nylon 6, which are more pronounced when these additives are introduced via blending. However, despite these advantages, this method presents several limitations that should be considered. The process is typically more complex and requires stricter control over reaction conditions, such as temperature, pH, and monomer-to-initiator ratios. Additionally, the higher cost of specialized reactants and equipment, as well as the potential scalability challenges in industrial production, may limit its broader application. Therefore, while in situ polymerization is a promising strategy for achieving enhanced flame retardancy with minimal compromise to mechanical integrity, further optimization is necessary to improve its economic feasibility and industrial adaptability.
Jelena 45 synthesized flame-retardant nylon 6 fibers by introducing a DOPO derivative, specifically 6,6′-(1-phenylethane-1,2-diyl)bis (dibenzo [c,e][1,2] oxaphosphinine-6-oxide) (PHED), into the nylon 6 matrix via in situ polymerization. The findings indicated that the nanodispersion of PHED within the nylon 6 matrix contributed to an effective flame retardant outcome, enabling nylon 6/15wt%PHED to achieve a Limiting Oxygen Index (LOI) of 27.8% and a UL94 V-0 rating, with the absence of dripping observed. Beyond organic phosphorus-based retardants, organic nitrogen-based flame retardants are similarly favored in the in situ polymerization of flame-retardant nylon 6 fibers. Li 46 achieved flame-retardant nylon 6 fibers through the in situ molecular self-assembly reaction, creating melamine cyanurate (MCA) within the nylon 6 matrix by combining adipic acid-melamine salt with cyanuric acid-hexamethylene diamine salt and subsequently proceeding with melt spinning. This resulted in a composite material that, with an MCA content of 8wt%, reached a UL94 V-0 rating and an LOI of 29.3%, while also exhibiting superior spinnability. The MCA formed in situ demonstrated strong interfacial coupling with the nylon 6 macromolecules, restraining deformation under tension and reducing the composite material’s elongation at break. Xu 47 generated flame-retardant nylon 6 fibers by incorporating MCA via in situ polymerization into the hydrolytic ring-opening system of caprolactam, yielding fibers with a 7.2wt% MCA content, an LOI exceeding 35%, and maintaining excellent mechanical properties. Song et al. 48 synthesized and characterized a phosphorus and nitrogen-containing monomer, acryloyloxy phenylphosphoryl diethanolamine ester (AEPPA), and subsequently produced poly (styrene-co-AEPPA)/alpha-zirconium phosphate (α-ZrP) nanocomposite materials with different α-ZrP loadings through in situ free radical copolymerization. Thermogravimetric analysis (TGA) revealed enhancements in the thermal stability and char residue of both the copolymers and the nanocomposite materials. The inclusion of AEPPA notably reduced the flammability of polystyrene (PS), with further reductions observed upon the addition of α-ZrP, attributed to a decrease in the maximum mass loss rate and an increase in char residue of the nanocomposite materials involved in thermal degradation.
Beyond the previously discussed organic phosphorus and nitrogen-based flame retardants, inorganic nanomaterial flame retardants and bio-based flame retardants are also pivotal. Hou
49
achieved functionalization of multi-walled carbon nanotubes (MWCNTs) by grafting amino groups onto their surfaces, producing amino-functionalized MWCNTs (F-MWCNTs), which were then integrated into nylon 6 fibers through in situ polymerization. The inclusion of F-MWCNTs notably enhanced the thermal stability of the composite fibers; with a mere 0.5wt% addition of F-MWCNTs, the onset temperature for composite fiber decomposition was elevated to 388°C. Additionally, F-MWCNTs demonstrated a potent ability to adsorb and neutralize free radicals generated during the thermal decomposition of nylon 6, thereby significantly slowing down the degradation process of the composite fibers. This free radical scavenging effect plays a crucial role in interrupting the combustion chain reactions in the gas phase, effectively reducing the intensity and propagation of the flame. Moreover, the presence of F-MWCNTs contributes to the formation of a more compact and thermally stable char layer in the condensed phase, which acts as a physical barrier that insulates the underlying material from heat and oxygen. This dual mechanism—gas-phase radical suppression combined with condensed-phase protective barrier formation—results in a pronounced synergistic flame-retardant effect. Compared to pure nylon 6 fibers, the 0.5 wt% F-MWCNTs/nylon 6 composite fibers exhibited remarkable mechanical enhancement, with tensile strength and Young’s modulus increasing by 45% and 208%, respectively. These improvements not only surpass those achieved by conventional melt blending methods but also highlight the multifunctional role of F-MWCNTs in simultaneously enhancing both flame retardancy and mechanical performance. Jelena
50
successfully incorporated graphene nanoplatelets (GnPs) into the nylon 6 matrix through in situ polymerization and subsequently produced nylon 6/GN fibers via melt spinning. This resulted in fibers with exceptional thermal stability and significantly improved anti-dripping properties. Zhai
51
and co-workers introduced the bio-based flame retardant, D-pyranose glucose, into the nylon 6 matrix to manufacture flame-retardant nylon 6/GI fibers via in situ polymerization. Their findings revealed that nylon 6/GI fibers containing 2wt% D-pyranose glucose achieved an LOI value of 29.4% and met the UL-94 V-0 rating criteria, offering a novel research direction for developing halogen-free flame-retardant solutions for nylon 6 fibers. Zhang Xudong et al
52
incorporated perylene-3,4,9,10-tetracarboxylic dianhydride (PTCDA) ranging from 0.01 to 2.5% weight into caprolactam for in situ polymerization, creating flame-retardant polyamide 6 (PA6)/PTCDA composites. Their research indicated that 2.5 wt% PTCDA inhibited the crystallization of PA6 chains, elevating the composite’s LOI to 29.5. Furthermore, the introduction of PTCDA facilitated the formation of a char layer during combustion, shielding flammable components from volatilization and bolstering the composite’s flame retardancy. This highlights that PA6/PTCDA composites fabricated through in situ polymerization exhibit commendable flame retardant characteristics. Feng et al. prepared CGF/PA6 composites via in-situ polymerization, effectively addressing the challenge of poor impregnation of continuous fibers caused by the high viscosity of molten thermoplastic resins. Moreover, by modifying the surface of the continuous fibers, the mechanical properties of the composites were significantly enhanced. This study provides a theoretical basis and technical reference for the engineering applications of continuous fiber-reinforced composites in fields such as automotive lightweighting and aerospace. The Figure 6
53
below illustrates the preparation process of the CGF/PA6 composites. Flowchart illustrating the preparation process of CGF/PA6 composites.
52

Overall, the in situ polymerization approach for flame retardancy alleviates challenges such as non-uniform dispersion, agglomeration, and microphase separation of flame retardants within the matrix material, diminishing their adverse effects on the dimensional stability, mechanical, thermal, and processing attributes of flame-retardant nylon 6. By incorporating flame retardants directly into the polymer chain during polymerization, the additives are more uniformly distributed, leading to better flame retardant performance and less negative impact on the physical properties of the polymer. However, this methodology does not fundamentally address the intrinsic combustibility issues associated with nylon 6 upon thermal decomposition. In-situ polymerization offers a promising alternative by enhancing the dispersion of flame retardants and promoting strong interfacial interactions with the nylon 6 matrix. However, this method presents its own set of challenges. The polymerization conditions—such as reaction temperature, duration, and catalyst selection—play a pivotal role in determining the molecular architecture of the resulting polymer. These parameters directly influence key characteristics, including thermal stability, crystallinity, and crosslinking density, which in turn affect flame retardant efficiency and the mechanical behavior of the material. For example, elevated polymerization temperatures can lead to excessive crosslinking that enhances flame resistance but reduces ductility and fiber spinnability. Conversely, insufficient reaction temperatures may lead to incomplete polymerization, resulting in suboptimal flame retardant performance. Moreover, the economic and technical viability of this method remains a concern for large-scale industrial adoption. The need for precise control over reaction parameters, along with the potential use of costly catalysts or specialized equipment, can increase production complexity and costs. These limitations underscore the importance of optimizing polymerization protocols to strike a balance between performance enhancement and manufacturability. Furthermore, long-term durability, processing stability, and environmental compatibility must be systematically evaluated to support the practical implementation of in-situ polymerization strategies for flame-retardant nylon 6 fibers.
Introducing flame retardancy through copolymerization
Copolymerization flame retardancy involves chemically integrating flame retardant moieties as comonomers into the polymer backbone during the hydrolytic ring-opening polymerization of nylon 6, thereby producing inherently flame-retardant nylon 6 materials.54,55 This technique offers several advantages, including durable flame retardancy, high efficiency, and the preservation of favorable mechanical properties in the resulting fibers. 56 In practical applications, copolymerized flame-retardant nylon 6 has been employed in industries requiring stringent fire safety standards. For instance, in the automotive sector, such materials are used in the manufacturing of interior components like seat fabrics and electrical cable sheathing, where long-term flame resistance and mechanical durability are critical. Similarly, in the aerospace industry, copolymerized flame-retardant nylon 6 is utilized in aircraft cabin linings and insulation materials due to its ability to maintain flame retardancy under harsh environmental conditions without compromising structural integrity. These examples highlight the practical relevance and industrial potential of copolymerization as a flame-retardant strategy.
In the study of copolymerization flame retardancy, organic phosphorus-based flame retardants are commonly used in nylon 6 fibers due to their active groups that readily react with caprolactam. Colovic
57
chemically bonded DOPO to the main chain of nylon 6 through copolymerization. Initially, amino-functionalized caprolactam was synthesized, which then reacted with DOPO to create DOPO-functionalized caprolactam (DOPO-A-CLM). This product was further copolymerized with caprolactam to form an inherently flame-retardant nylon 6 copolymer (nylon 6-DC), which was successfully melt-spun. The results demonstrated that compared to pure nylon 6, nylon 6-DC fibers could self-extinguish within 1 s without producing any drips, though the tensile strength of nylon 6-10DC decreased by 54.8%. The incorporation of bulky phosphorus-containing flame-retardant monomers such as DOPO-A-CLM into the ε-caprolactam ring-opening polymerization system has been reported to influence polymerization kinetics. Specifically, these monomers may sterically hinder the propagation step or alter the hydrogen bonding interactions in the reaction medium, leading to a reduced polymerization rate. As a result, the final polymer may exhibit a lower degree of polymerization and shorter average chain length compared to conventional nylon 6, which can negatively affect mechanical properties. This effect was reflected in the observed reduction in tensile strength, suggesting that the inclusion of reactive flame-retardant monomers not only imparts flame retardancy but also modifies the polymerization behavior and molecular structure of the final product. Liu
58
introduced 10-(2,5-dicarboxyphenoxy)-10-hydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPODP) into the main chain of nylon 6, producing PA6-DOPODP copolymers. When synergized with molybdenum disulfide (MoS2) and zinc sulfide (ZnS), the resultant flame-retardant nylon 6 fibers achieved an LOI value of over 30% and a UL94 V-0 rating. Liu
59
also chemically bonded [(6-oxido-6H-dibenz [c,e][1,2] oxaphosphorin-6-yl)methyl]succinic acid (DDP) to the main chain of nylon 6 via salification. With a 5wt% addition of DDP, the LOI value of flame-retardant nylon 6 fibers increased to 28.4%. Sun
60
further enhanced the flame retardancy by synergizing DDP with polydiphenylsiloxane (PDPS) chains, introducing them into the nylon 6 main chain, which elevated the binary flame-retardant nylon 6 fibers’ LOI value to 26.3%, achieving a UL94 V-0 rating without drip formation. Cai
61
also introduced DDP chains into nylon 6 via copolymerization, reaching fiber LOI values up to 29.1%, which increased to 30.1% after washing. Zhang
62
applied transesterification in the copolymer modification of nylon 6 to integrate DDP, synthesizing flame-retardant nylon 6 copolymers and achieving successful melt spinning. As shown in Figure 7,
62
the detailed preparation process is presented. Schematic illustration of the preparation process.
62

With a 10wt% inclusion of EDE, the copolymer’s LOI value ascended to 31.1%, meeting a UL94 V-0 standard. Beyond DOPO and DDP. Mourgas 63 chemically bonded 3-hydroxyphenylphosphinyl propanoic acid (3-HPP) to the main chain of nylon 6, successfully producing copolymerized flame-retardant nylon 6 fibers. With a phosphorus content of 0.15wt% from 3-HPP, the fibers’ LOI value increased to 35.6%, achieving a UL94 V-0 rating while maintaining good mechanical properties. Simonetti 64 conducted Michael addition reactions between unsaturated phenyl bis(diallylphosphonate) (DVPPO) and polyamide 6, with piperazine acting as the nucleophilic agent, to synthesize a nylon 6/DVPPO/piperazine compound. Successful spinning was achieved by lowering the extrusion temperature during the spinning process or using a high-viscosity carrier polymer. The results demonstrated that the compound could achieve a Limiting Oxygen Index (LOI) of up to 30.2%, and significantly reduced combustion time. Bazheva and colleagues 65 synthesized a copolycarbonates based on 4,4′-dioxydiphenylpropane and 1,1-dichloro-2,2-di (3,5-dibromo-4-oxyphenyl) ethylene were synthesised, and their main properties were studied. The relationships governing the synthesis of copolymers by acceptor-catalytic polycondensation were determined, and the relationship between their composition, structure, and properties was established. It was shown that copolycarbonates possess a high softening point and high heat and flame resistance. Xiao et al. 66 developed a method for creating permanently flame-retardant nylon 6 materials, particularly focusing on a phosphorus-containing reactive flame retardant. This method involves a two-step polymerization process to produce block copolymer flame-retardant nylon 6 materials. Initially, the flame retardant reacts with diamines or diols to form a flame retardant prepolymer, which then copolymerizes with the prepolymer of nylon 6 to yield permanently flame-retardant nylon 6 materials. This approach results in flame-retardant nylon 6 materials characterized by low flame retardant loading, enduring flame retardancy, with a Limiting Oxygen Index (LOI) of over 30%, achieving a UL94V-0 rating in vertical burning tests, and possessing excellent mechanical properties. This type of nylon 6 material can be directly injection molded or processed into films, and can also be spun into fibers for further manufacturing into flame-retardant fabrics. Chen et al. 67 invented a flame-retardant and antistatic nylon and its manufacturing method. By selecting carbon black with a specific DBP value mixed in a particular ratio with carbon nanotubes and graphene to form a nano-composite structure with good conductivity, and then copolymerizing it with caprolactam followed by melt spinning, they produced composite nylon fibers that exhibit both flame retardancy and antistatic properties. This product is characterized by good spinnability, stable performance, low cost, and ease of industrialization, presenting significant practical value. Shuo et al. 68 used a laboratory-synthesized reactive silicone-based flame retardant as a copolymer unit, successfully incorporating it into the main chain of PA6 molecules through chemical copolymerization, resulting in inherently flame-retardant PA6 copolymers (FR-PA6). Thermal gravimetric analysis showed that FR-PA6 has good thermal stability, with significantly enhanced char formation capability compared to PA6. Comparing the combustion properties of pure PA6 and FR-PA6, it is evident that the fire hazard of FR-PA6 is substantially reduced, with FR-PA6-3 exhibiting an LOI of 26.3%, achieving a V-0 rating in vertical burning tests, and demonstrating dripping behavior during combustion. Wang et al. 69 used maleic anhydride grafted ethylene-octene copolymer (POE-g-MAH) to toughen and modify two types of nylon 6 resins. With the increase in the toughening agent content, the toughness of nylon 6 was significantly improved, while the strength decreased. Moreover, as the content of the toughening agent increased, effective toughening agent aggregates were formed in the nylon matrix, which played a role in improving the toughness.
In conclusion, the copolymerization flame retardancy modification method, which chemically bonds flame-retardant functional segments onto the main chains of nylon 6, produces flame-retardant nylon 6 fibers with outstanding flame retardancy and durability. This represents a promising direction for the flame retardant modification of nylon 6 fibers. However, due to the complexity of the process and stringent requirements for flame retardants, industrial-scale production is currently challenging, necessitating further research.
Comparative analysis of modification techniques for halogen-free flame retardant nylon 6 fibers
To enhance the flame retardancy of nylon 6 fibers while maintaining acceptable mechanical properties, various halogen-free flame retardant (FR) strategies have been developed. These include blending, in-situ polymerization, and copolymerization techniques. Each method offers distinct advantages and challenges, which are crucial for their practical application in fiber production.
Blending is the most commonly employed method due to its simplicity and cost-effectiveness. However, it often suffers from poor dispersion of flame retardants within the polymer matrix, leading to compromised flame retardancy and mechanical uniformity. In contrast, in-situ polymerization allows for better integration of flame retardant agents, resulting in improved thermal stability and compatibility with the polymer chain. The copolymerization approach, on the other hand, enables the chemical bonding of flame retardant units into the backbone of nylon 6, offering the best combination of mechanical strength and flame retardancy among the three.
Comparison of different flame-retardant modification methods and their effects on the mechanical and flame-retardant properties of nylon 6 fibres.

As illustrated in the table, the copolymerization strategy exhibits the most promising performance profile for flame retardant nylon 6 fibers, achieving high tensile strength (55–65 MPa), excellent flame retardancy (LOI up to 40%, UL-94 V-0), and enhanced elongation. These characteristics make it a competitive choice for high-performance textile applications requiring stringent flame retardant standards without halogen content.
Mechanisms of combustion and flame retardancy of nylon 6 fiber
The combustion process of polymers can be summarized into five steps: decomposition, ignition, flame propagation, stable combustion, and decay. 70 Additionally, the combustion of polymers typically involves three modes: vapor phase combustion, thermal decomposition, and solid surface combustion. 71
Research on flame retardant mechanism
It is widely recognized that heat sources, gas sources, combustibles, and free radical reactions are the key factors in sustaining the combustion of polymers. Therefore, disrupting one or more of these conditions could achieve flame retardancy of materials. 72 Consequently, based on the combustion elements suppressed by the flame retardant, the flame retardant mechanisms of nylon 6 fibers can generally be classified into gas-phase flame retardancy, condensed-phase flame retardancy, and thermal exchange interruption flame retardancy. 73
Gas-phase flame retardancy involves the action of flame retardants at the gas phase level to suppress or interrupt the combustion of polymers. There are mainly two forms of this retardancy: one is where the flame retardant releases active groups during combustion to capture free radicals, thus interrupting the free radical chain reaction to achieve flame retardancy; the other is where the flame retardant releases inert gases during combustion to dilute the gas source, thereby enhancing the flame retardancy of nylon 6 fibers.74,75
Condensed-phase flame retardancy primarily slows down or interrupts the thermo-oxidative decomposition of flame-retardant materials at the condensed phase level, i.e., the formation of a char layer during the combustion of nylon 6 macromolecular chains. This char layer effectively isolates the gas source, preventing further combustion of the material. Moreover, due to the poor thermal conductivity of the char layer, the transfer of external heat into the material is reduced, thereby slowing down the thermal degradation and suppressing combustion. 76
The mechanism of interrupting thermal exchange for flame retardancy mainly achieves its effect by removing the heat generated during the combustion of nylon 6 fibers. Typically, this involves the formation of molten droplets that carry away some of the heat, preventing thermal degradation and interrupting combustion. 74
Combustion mechanism of nylon 6
The enhancement of flame retardancy in Nylon 6 has become increasingly critical due to its widespread application in fields where fire safety is paramount, such as automotive components, electrical housings, and textile industries. Nylon 6 is inherently combustible due to its aliphatic polyamide structure, which makes it susceptible to rapid thermal degradation and flame propagation when exposed to high temperatures. Upon heating, Nylon 6 undergoes a two-stage combustion process. The first stage involves the breaking of amide bonds, leading to the release of volatile compounds such as ammonia and hydrocarbons. The second stage involves the formation of a molten phase, which accelerates flame spread due to its low viscosity and tendency to drip, spreading the fire. The reaction rate constant k for the thermal degradation of Nylon 6 has been reported to be in the range of 1.2 × 10−3 to 1.8 × 10−3 min−1, with an activation energy Ea of approximately 125 kJ/mol. 77 These values indicate the susceptibility of Nylon 6 to rapid degradation and the importance of developing effective flame retardants to mitigate the fire risks associated with its use.
The demand for flame-retardant Nylon 6 is further driven by stringent regulatory requirements and growing societal emphasis on fire safety. Many international standards now mandate the use of materials with enhanced flame resistance in consumer and industrial products, underscoring the critical need for effective flame retardant treatments. These treatments not only reduce the likelihood of ignition but also minimize the intensity and spread of flames, providing essential time for evacuation and fire suppression efforts. Standards such as UL 94 V-0, IEC 60335-1, and EN 45545-2 assess flame resistance based on parameters like combustion rate, smoke density, and toxic gas emissions. The incorporation of flame retardant additives has been shown to improve the Limiting Oxygen Index (LOI) of Nylon 6 to above 30%, a significant increase from its baseline value of approximately 24%. 78 To mitigate the flammability of Nylon 6, various flame retardant strategies have been employed. These include the incorporation of halogenated compounds, phosphorus-based additives, and intumescent systems. Halogenated flame retardants function by promoting the formation of a protective char layer during combustion, which acts as a barrier to heat and prevents further degradation of the underlying polymer. Phosphorus-based additives, on the other hand, enhance the char formation and promote the release of non-flammable gases, effectively diluting the flammable species in the combustion zone. The interaction between flame retardants and the polymer matrix can also influence the thermal stability and mechanical properties of Nylon 6, as these additives can either reinforce or weaken the polymer’s inherent characteristics. For instance, the addition of phosphorus-based flame retardants has been shown to increase the char yield by up to 30%, which significantly improves the material’s resistance to flame propagation. 79 Recent studies have highlighted the importance of synergistic effects between different flame retardants, suggesting that a combination of additives may provide enhanced protection compared to individual compounds. For example, a blend of phosphorus and nitrogen-based flame retardants can lead to a reduction in peak heat release rate (pHRR) by as much as 40%, compared to the use of each compound alone. Additionally, the incorporation of nanoscale flame retardants, such as nano-clays, has demonstrated improved flame retardancy by enhancing the physical barrier properties of the char layer formed during combustion. 80 Moreover, the flame retardant effectiveness is also influenced by the presence of impurities and additives. For example, the presence of plasticizers or moisture in the polymer matrix can alter the combustion kinetics, leading to a reduction in the overall flame retardant efficiency. Therefore, a comprehensive understanding of how these impurities affect the combustion process is critical for optimizing flame retardancy in practical applications.
The flame retardant strategies and combustion mechanisms of Nylon 6 are significantly different from those of other common polymers such as polyethylene and polypropylene, which exhibit less charring and more volatile degradation products. The formation of a protective char layer in Nylon 6 is crucial for its flame resistance, a characteristic that is less prominent in other polyolefin-based materials. This difference is essential when comparing the flame retardant performance of Nylon 6 with other materials in various applications, especially in fire-sensitive environments such as automotive and electrical industries. Recent experimental data suggest that microstructural changes during combustion play a crucial role in the overall flame retardancy of Nylon 6. Specifically, the formation of a dense char layer, which acts as a physical barrier to heat and flame propagation, is a key factor in improving fire resistance. These microstructural changes can be influenced by the type and amount of flame retardants incorporated, as well as the processing conditions used during the polymer’s manufacturing.
Flame retardant requirements and environmental advantages of halogen-free flame retardancy
With the widespread application of nylon 6 in industries such as electronics, electrical, automotive, and construction, its safety requirements, especially in terms of flame retardancy, have become increasingly stringent. To meet safety standards and regulatory requirements, nylon 6 must possess effective flame retardant properties to reduce fire risks and delay the spread of fire. These requirements are typically dictated by international and regional safety standards such as UL 94, 1 IEC 60335-1, 2 EN 45545-2, 3 among others, with the UL 94 V-0 rating becoming a fundamental requirement for many applications. These standards assess the performance of materials under heat or combustion conditions, including combustion rate, smoke density, and toxic gas emissions. Under the UL 94 standard, both PA6 and PA66 are considered flammable materials with a V-2 flame retardant rating and a Limiting Oxygen Index (LOI) of approximately 24%.81–84 PA6 and PA66 are both types of polyamides (commonly known as nylon) but differ in their chemical structure. PA6, or Nylon 6, is a polymer made from a single monomer, caprolactam, while PA66, or Nylon 66, is derived from two monomers, hexamethylenediamine and adipic acid. The key difference between PA6 and PA66 lies in their molecular structure, which affects their thermal properties and mechanical strength. PA66 generally exhibits higher melting points and better chemical resistance compared to PA6, but PA6 offers better processability and lower production costs. Despite these differences, both materials are widely used in similar applications, and their flame retardant behaviors under combustion conditions are quite comparable. 85
Flame retardant challenges, strategies, and shared characteristics of PA6, PA66, and polyester.

Halogenated flame retardants (such as compounds containing bromine and chlorine) may have good flame-retardant effects, but they can release toxic gases and persistent organic pollutants (POPs) during combustion, posing severe threats to the environment and human health. In contrast, halogen-free flame retardants are increasingly regarded as a more sustainable alternative due to their reduced ecological footprint and lower toxicity. International and regional regulatory frameworks—such as the European Union’s Restriction of Hazardous Substances (RoHS) Directive and Registration, Evaluation, Authorisation, and Restriction of Chemicals (REACH) regulation—have imposed stringent limitations on the use of halogenated flame retardants, thereby driving the advancement of halogen-free flame retardant technologies. Additionally, growing consumer awareness and demand for environmentally friendly materials have further propelled the adoption of non-halogenated flame retardants across various sectors. This shift reflects not only regulatory pressure but also an evolving preference for safer and more sustainable materials, particularly in fire-sensitive applications.
In specific end-use industries, such as the automotive sector, flame retardants must maintain long-term stability under conditions of elevated temperature, mechanical vibration, and chemical exposure. These factors can significantly compromise the durability of flame-retardant systems, necessitating the design of robust formulations tailored to such demanding environments. Therefore, optimizing flame retardant performance under operational stressors remains a critical area for future research.
To holistically evaluate the environmental impact of flame retardants, it is essential to incorporate life cycle assessment (LCA) methodologies. LCA provides a comprehensive framework for quantifying environmental burdens associated with raw material extraction, synthesis, processing, use-phase performance, and end-of-life disposal or recycling of flame retardant materials. Such assessments are crucial for guiding the development of truly sustainable flame-retardant solutions, enabling stakeholders to make informed decisions regarding material selection and regulatory compliance. Future research should prioritize not only the technical efficacy of halogen-free flame retardants but also their environmental sustainability across the entire product life cycle.
Demand for nylon 6 fibers in specific applications
As shown in the Figure 8, Nylon 6 fibers exhibit a wide range of applications across diverse sectors due to their excellent flame retardancy, mechanical strength, chemical resistance, and environmental compatibility. These fibers are extensively used in the electronics and electrical industries for the production of connectors, insulators, and component casings; in the automotive field for interior components, engine covers, and wiring systems; and in the textile industry for protective clothing, curtains, and bedding. Moreover, Nylon 6 fibers are finding increasing relevance in aerospace applications such as aircraft cable insulation and seating, owing to their high thermal stability and safety performance. Their non-halogenated flame-retardant formulation ensures reduced toxicity and environmental impact, making them a promising choice for sustainable material solutions. In such applications, materials are often exposed to high humidity, oxidative atmospheres, and prolonged thermal cycling. Therefore, the hydrolytic stability and long-term thermal aging resistance of flame-retardant Nylon 6 fibers become critical performance parameters. Recent studies on copolymerized flame-retardant systems have shown that while reactive phosphorus- or nitrogen-based flame retardants can be covalently bonded into the nylon backbone to enhance durability, their inclusion may also alter the polymer’s susceptibility to hydrolytic degradation or oxidative chain scission. Evaluating the retention of mechanical and flame-retardant properties after exposure to humid or thermally oxidative environments is essential for validating their applicability in aerospace and automotive contexts. These factors must be considered in future studies to ensure the long-term reliability of copolymerized flame-retardant Nylon 6 fibers in demanding service conditions. In the electronics and electrical fields, non-halogenated flame-retardant Nylon 6 fibers are primarily used in the manufacturing of various connectors, switches, insulators, and component casings, among others. These applications have stringent requirements for the material’s flame retardancy, electrical insulation properties, as well as physical and chemical stability.
93
Industrial and technical applications of flame-retardant Nylon 6 fibers.
In the automotive field, non-halogenated flame-retardant Nylon 6 is used to manufacture interior components, engine covers, wires, cables, and other critical parts. These applications require not only good flame retardancy in the material but also consider factors such as lightweight, heat resistance, and chemical resistance.
in theTextiles field:Flame retardant Nylon 6 fibers are primarily used in various textile applications where fire safety is a significant concern, such as in protective clothing, automotive fabrics, and home textiles. The incorporation of flame retardants into Nylon 6 fibers can be achieved through several methods, including blending, in-situ polymerization, and copolymerization. Each of these approaches has distinct effects on the performance of the resulting textiles. 94
Blending: In the textile industry, blending flame retardant Nylon 6 fibers with other fibers, such as cotton or polyester, is a common practice for enhancing the flame retardancy of fabrics. Blended fibers are often used in applications like firefighter uniforms, industrial workwear, and protective clothing for hazardous environments. The main advantage of blending is that it combines the inherent flame resistance of Nylon 6 with the comfort and breathability of other fibers. However, challenges remain in achieving uniform dispersion of flame retardants within the blend and maintaining the mechanical properties of the fabric.
In-situ Polymerization: The in-situ polymerization technique involves introducing flame-retardant monomers during the polymerization of Nylon 6, resulting in a homogeneous distribution of flame retardants within the fiber. Textiles made from in-situ polymerized flame retardant Nylon 6 fibers exhibit enhanced flame resistance without compromising the fabric’s integrity. This method is particularly useful for producing high-performance textiles used in military and aerospace industries, where both durability and flame resistance are critical.
Copolymerization: Copolymerizing flame-retardant monomers with Nylon 6 during fiber production creates fibers with integrated flame-retardant properties. This approach is commonly used in specialized textiles for automotive interiors, aircraft upholstery, and high-risk industrial environments. The copolymerized flame-retardant Nylon 6 fibers offer superior flame resistance and can be engineered to maintain or even improve the mechanical properties of the fabric. However, the complexity of the copolymerization process and higher production costs remain significant considerations.
These applications require high standards for material flame resistance, comfort, and durability. It has been reported that the interaction of flame retardants with the environment is the second most impactful of all polymer additives, second only to plasticizers. The data offered highlights alternatives to halogenated flame retardants but, from an industrial perspective, the specific application of halogenated flame retardants is the true replacement for some classic flame retardants that have proven serious environmental and health concerns. The relationship between non-halogenated flame retardants and thermoplastic plastics (especially condensation polymer matrices, which are also more prone to degradation in biological processes) will continue to develop and will call for new solutions that are more environmentally friendly, with less human contact toxicity, and safer under critical conditions (emitting less toxic gases in hazardous situations). At least for the next few years, non-halogenated flame retardants will remain a priority. 95
The aerospace industry demands extremely high performance from materials, including very high flame retardance, excellent mechanical strength, and resistance to chemicals. The application of non-halogenated flame-retardant Nylon 6 in this field includes cable insulation inside aircraft, seating, and other interior materials. Materials that meet aerospace standards also must undergo strict safety testing to ensure reliable performance in case of emergencies.
Conclusions
Summary of challenges and solutions for different halogen-free flame retardancy methods in Nylon 6 fibers.

Key challenges and future opportunities
Despite notable advances in halogen-free flame-retardant modifications of Nylon 6 fibers, several fundamental challenges remain before large-scale industrial implementation can be achieved. One of the most pressing issues is the need to balance flame-retardant performance with mechanical integrity, particularly in blending systems where the incorporation of additives often compromises fiber strength and spinnability. In the case of in situ polymerization, although dispersion uniformity has been significantly improved, the long-term durability of flame-retardant effects remains suboptimal. Copolymerization techniques, while enabling intrinsic flame retardancy via covalent integration, are frequently constrained by complex reaction pathways and elevated production costs. Moreover, the synergistic interactions among multiple flame-retardant agents are not yet fully understood, hindering their effective application in practical formulations.
Future research should prioritize the development of multifunctional flame-retardant systems that simultaneously ensure mechanical robustness, high-efficiency flame resistance, and environmental sustainability, while simplifying synthesis procedures and reducing production costs. In parallel, emphasis should be placed on the use of environmentally benign and renewable flame-retardant sources, accompanied by comprehensive life-cycle assessments to guide sustainable material innovation. Importantly, fostering stronger collaboration between academia and industry will be critical to accelerating the translation of laboratory-scale discoveries into commercially viable flame-retardant technologies. Such partnerships can help align fundamental research with real-world performance requirements, regulatory standards, and scalability considerations, thereby expediting the adoption of advanced halogen-free flame-retardant Nylon 6 fibers in safety-critical and environmentally conscious applications.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the National Natural Science Foundation of China (NSFC 52203074), Natural Science Foundation of Zhejiang Province (LTGG24E030001), the China Postdoctoral Science Foundation (2023M731331).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
