Abstract
A novel and practical approach has been employed to decompose and carbonize cotton waste using vapor-phase assisted hydrothermal treatment at low temperatures. This method represents an innovative solution for processing cotton waste while effectively minimizing environmental impact. The reaction involves hydrochloric acid (HCl) with varying doses, reaction times, and temperatures. During the process, the fabric’s color changes from white to dark brown and ultimately to black after depolymerization and carbonization. Compared to the untreated samples, the treated samples exhibited area shrinkage of approximately 4–13%. The mass yield was around 81%, indicating a weight loss of 19%, which reflects the effective conversion of cotton into a carbon-rich product. The CS9 and CS10 samples, treated at 100°C for 2 h with 1 mL and 2 mL of HCl, respectively, demonstrated the best carbonization results. Scanning electron microscopy (SEM) images revealed significant changes in the surface morphology and texture of the fibers, accompanied by a reduction in fiber diameter. Additionally, fourier transform infrared (FTIR) spectra confirmed the presence of C = C bonds, supporting the formation of carbonized structures and the occurrence of cross-linking reactions. Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) measurements illustrated enhanced thermal stability, attributed to the formation of pre-graphitic structures with aromatic compounds at higher temperatures, producing carbonaceous materials.
Introduction
Cotton is widely employed in a variety of applications due to its availability, flexibility, strength, chemical resistance, sustainability, and hydrophilicity. It is the most significant and extensively cultivated crop in the world, mostly utilized to produce textile fibers. 1 Currently, dealing with waste textiles is a major concern for both experts and the general public. Recycling cotton fibers has garnered attention among the various textiles due to its large production volume, accounting for about 85% of natural fibers. Physical methods, such as loosening fabrics to obtain recycled fibers, are used, but they can reduce the value of textiles after weaving or gluing. Unfortunately, the process of loosening and weaving recycled textiles can only is repeated a few times before they have to be discarded. This raises the issue of finding a solution for the recycling of cotton fibers.
Cotton primarily comprises cellulose, a natural polymer with the highest carbon content (44%). This linear polymer consists of β-D-glucopyranosyl residues linked by β-1,4-glycosidic bonds. 2 There is a growing interest in the potential applications of biomass as a material resource. Cellulose-derived carbonized materials have garnered significant attention. Carbonization processes primarily involve slow pyrolysis and hydrothermal carbonization (HTC). 3 Hydrothermal carbonization, a thermochemical conversion process, is conducted in neutral or acidic aqueous solutions at relatively low temperatures (180°C–260°C), typically under autogenic pressure. 4
The HTC process has been utilized to convert a variety of waste biomass, including materials such as glucose, sucrose, fructose, starch, cellulose, rubber wood, olive mill industry waste, loblolly pine, agro-waste, food waste, organic fraction of municipal solid waste, paper mill industry waste, and plastic waste. Despite the differing nature of the treated feedstock, most HTC studies have focused on examining the influence of operational variables such as temperature, residence time, and biomass-to-water ratio on the energy, chemical, and morphological properties of the resulting hydrochars. These hydrochars hold potential for applications as solid biofuels and valuable carbon materials.5–7
Cellulose, hemicellulose, and lignin are the major ingredients of most biomass materials; the amounts of these components vary between materials derived from various sources. 8 Since cellulose is the most prevalent organic molecule on Earth, efficient techniques for hydrolyzing or depolymerizing it is a promising starting point for the synthesis of biofuels and biochemicals. Hydrothermal carbonization (HTC) has garnered significant attention in research circles owing to its favorable reaction conditions, straightforward operational procedures, cost-effectiveness, utilization of water as a solvent instead of organic solvents, high carbon yield, and the prevalence of an increased number of oxygen functional groups on the surface. Notably, hydrothermal carbons have demonstrated pivotal utility in fabricating porous carbon materials.9–11 Recent advancements have expanded the scope and efficiency of HTC by modifying process parameters and integrating supplementary techniques.12–14
Sartova et al. prepared activated carbon (AC) utilizing waste materials from the cotton processing industry. This process involved carbonization within the 500 to 800°C temperature range, followed by activation at 800°C using a water stream. 15 This approach underscores the utilization of cotton biomass as a renewable and cost-effective alternative source for textile and food products. Li et al. carbonized the cotton fibers in polyester/cotton blend fabrics by treating them with dilute phosphoric acid at high temperatures. 16 Several studies have explored the impact of additives such as H2SO4, HCl, and acetic acid on cellulosic biomass’s Hydrothermal Carbonization (HTC). These studies have resulted in an enhanced hydrolysis efficiency of cotton fibers. This enhancement can be attributed to the susceptibility of cotton fibers to hydrolyze into polysaccharides in concentrated acids, given the sensitivity of the glycosidic bond to high doses of H+.17–20 The long-standing method of homogeneous acid hydrolysis of cellulose has some drawbacks, including reactor corrosion, high temperature, low catalyst recyclability, challenging product separation, and waste effluent treatment issues. Enzymatic depolymerization of cellulose shows good selectivity for the hydrolysis of cellulose to glucose; however, enzyme recovery from the reaction mixture is laborious, costly, and slow.21,22 Dry gel conversion (DGC), which uses the sol-gel chemistry of inorganic materials and keeps the solid species isolated from the aqueous phase, has become a significant substitute method for zeolite crystallization based on silicate polymerization reactions. It has advantages over traditional hydrothermal treatment, such as low H2O levels and rapid crystallization.23,24
Conventional hydrothermal decomposition processes primarily depend on liquid-phase reactions; however, recent advancements have introduced vapor-phase-assisted methods that enhance material breakdown and transformation. In these vapor-phase-assisted hydrothermal systems, volatile intermediates or reactants in the vapor phase can significantly affect reaction kinetics, leading to more efficient depolymerization and improved product selectivity. For instance, vapor-assisted hydrothermal carbonization has demonstrated the ability to convert biomass into valuable carbonaceous materials under relatively mild conditions. 25 A novel vapor-phase hydrothermal method for sucrose carbonization at 200°C produced porous carbon with a high surface area (52 m2/g) and pore volume (0.18 cm3/g), outperforming conventional HTC. 23 In a previous study, Amino, carboxyl, and thiol-functionalized carbons were successfully prepared using a one-step vapor-phase assisted hydrothermal carbonization method. These functionalized groups significantly improved Cr(VI) adsorption performance compared to unmodified hydrothermal carbons. 26 This simple vapor-phase-assisted hydrothermal carbonization method could be extended to prepare diversely functionalized carbons. These findings suggest that utilizing vapor-phase dynamics in the decomposition of cellulose waste could reveal new pathways for sustainable material valorization. Previous studies have investigated a variety of hydrothermal carbonization methods for cellulose. In contrast, our approach presents a novel vapor-phase-assisted process that enhances the reaction’s efficiency and scalability. Traditional methods typically require substantial volumes of water, elevated temperatures, and prolonged reaction times. However, our method minimizes water usage while operating at lower temperatures and reducing processing time, offering a more sustainable and cost-effective alternative. This significant advancement paves the way for large-scale applications in carbon-based fabrics, water filtration, composites, and the production of other high-value materials. Recent advancements have highlighted vapor-phase-assisted hydrothermal decomposition and carbonization as promising methods for biomass valorization. The present study introduces a novel vapor-phase-assisted hydrothermal approach specifically targeting cellulose waste. The goal is to develop sustainable and efficient methods for producing high-value materials.
Currently, there are no reported studies on using vapor-phase-assisted hydrothermal decomposition and carbonization of cellulose to produce solid carbon in the presence of acids. This study proposes a new vapor-phase-assisted hydrothermal method for carbonizing cellulose at low temperatures and dose of HCl acids. The study examines the effects of temperature, treatment time, and HCl dose on the decomposition and carbonization of cotton fabric. The carbonized cotton samples were analyzed using scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS) analysis, fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), and differential scanning calorimetry (DSC). The results show that this processing method significantly improves the conversion of cotton waste into carbonaceous materials. The research introduces a method for recycling cotton waste through decomposition and carbonization, with potential applications in carbon-based textiles. It offers a sustainable solution for textile waste, suggesting uses in energy storage, filtration, and advanced textile applications.
Materials and methods
Experimental design and parameters for carbonization of the cotton fabric.

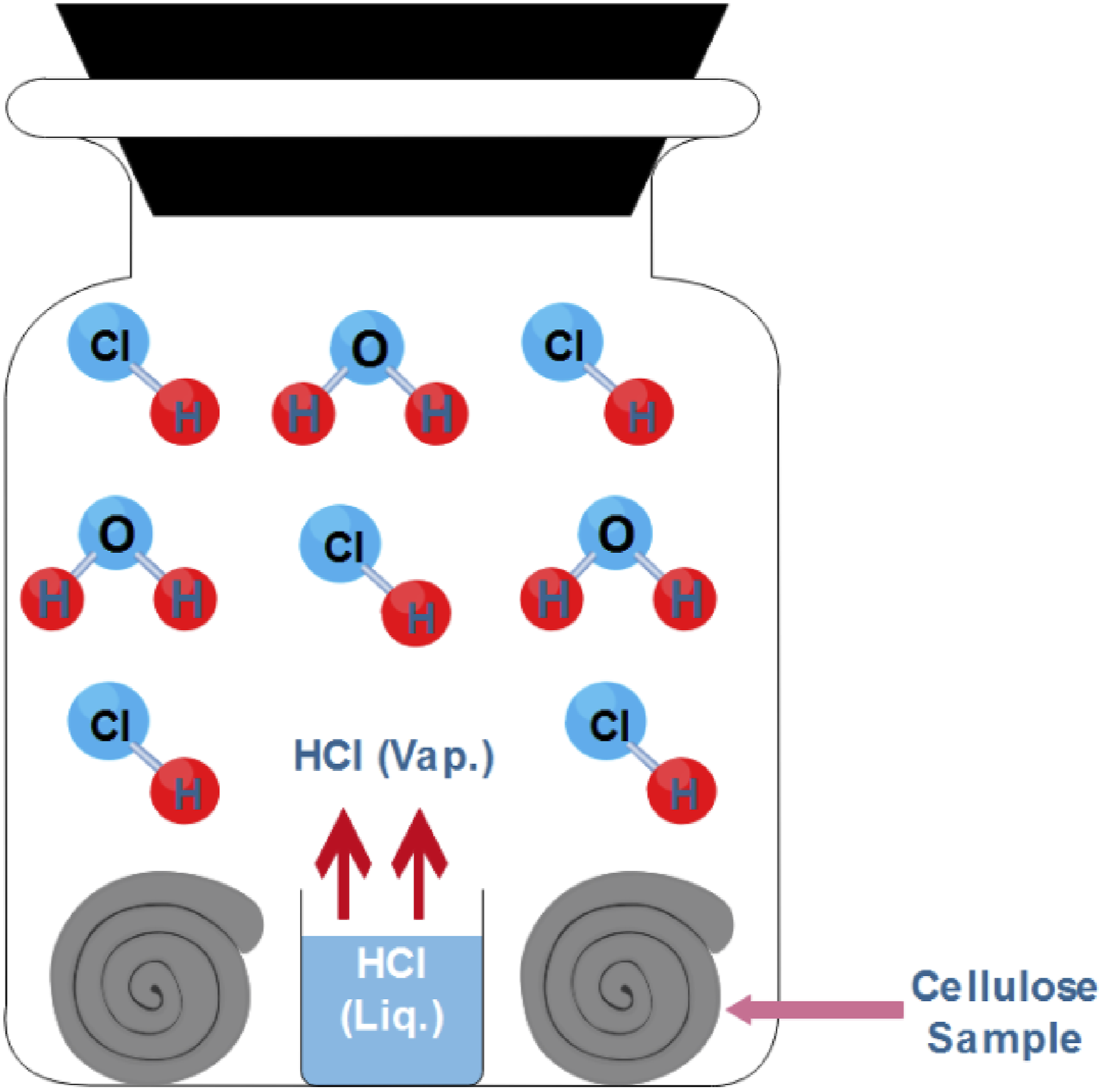
Schematic diagram for vapor-phase assisted hydrothermal carbonization of cotton.
In this study, 10 different cotton samples underwent hydrothermal treatment under various conditions, with each sample assigned a unique code (CS1 to CS10). The coding was based on specific combinations of temperature, reaction time, and hydrochloric acid (HCl) dose. Sample CS1 was treated at 90°C for 1 h using 1 mL of HCl. Samples CS2 and CS3 were processed under the same temperature and duration but with increased HCl volumes of 2 mL and 3 mL, respectively. Samples CS4 to CS6 were treated at 90°C for a longer duration of 2 h, utilizing HCl volumes of 1 mL, 2 mL, and 3 mL. Samples CS7 and CS8 underwent treatment at a higher temperature of 100°C for 1 h, with 1 mL and 2 mL of HCl, respectively. Finally, samples CS9 and CS10 were treated at 100°C for 2 h, with HCl volumes of 1 mL and 2 mL, respectively.
SEM analysis
A Zeiss Ultra Plus scanning electron microscope (SEM) from Germany equipped with an Oxford X-max 20 Energy Dispersive X-ray spectrometer was used to analyze the morphology of the carbonized samples. Platinum sputtering was used to apply a conductive coating on the specimen’s surface before SEM analysis. An accelerating voltage of 2 kV was used to view the samples.
Color and physical analysis
The fabric dimensional and color change after treatment were calculated using scanned images and Image J software. The average dimensions were used to determine the shrinkage and color change. The carbonized fabrics were scanned at 600 dpi, and the resulting images were analyzed using ImageJ software to calculate the whiteness index. The carbonization activity of the treated fabrics was quantitatively estimated by measuring the gray scale values of the samples. The gray scale measurements were utilized to determine the whiteness index, where higher values indicate greater whiteness and lower values correspond to darker colors. This method provided an objective way to compare the color changes from the carbonization process. To assess the material loss during carbonization, we measured the initial and final weights of the fabric samples using an analytical balance. The mass yield (%) was calculated by taking the weight of the carbonized sample, dividing it by the original sample weight, and multiplying it by 100. Weight reduction (%) was determined by subtracting the mass yield from 100%, which indicates the percentage of material lost during the carbonization process.
FT-IR analysis
Analyzing a material’s chemical structure is essential to assessing its performance and quality. The carbonized cotton samples were analyzed using Fourier transform infrared (FT-IR) spectroscopy using Thermo Fisher Scientific’s IZ10 model.
Thermogravimetric analysis (TGA)
The thermogravimetric analyzer (TGA), Mettler Toledo (TGA/SDTA851e) produced by TA Instruments (USA), was employed to evaluate the thermal characteristics of the materials. The samples were heated at a controlled rate of 10°C/min from 30°C to 600°C in a synthetic air environment. In the textile industry, TGA is a well-established and reliable technique for assessing the thermal properties of textiles.
Differential scanning calorimetry (DSC)
All samples were analyzed using a Mettler Toledo brand (DSC 3+ Star System) differential scanning calorimeter (DSC) made in the USA. Each sample weighed between 5.00 and 10.00 mg for the DSC measurement. The samples were initially heated from 25°C to 400°C to observe the structural evolution and thermal process. The DSC measurement involved heating and cooling at 10 K/min rates, with a flow of nitrogen (N) gas at 20 mL/min.
Results and discussion
Morphological analysis of carbonized cotton fabric
Preliminary experiments were conducted to assess the effects of temperature and reaction time on cellulose waste’s hydrothermal decomposition and carbonization. Trials were run at 20°C, 50°C, and 80°C with reaction times from 30 min to 5 h. Higher temperatures and longer durations significantly improved decomposition and carbonization efficiency, while hydrochloric acid (HCl) dose had minimal impact at lower temperatures. Consequently, 90°C and 100°C were chosen for the main experiments, with 1 h and 2 h reaction times for effective conversion. A range of HCl volumes (1–3 mL) was tested to evaluate its effect on fiber shrinkage and morphology, optimizing for adequate reaction activity without excessive cellulose degradation. In Figure 2(a), it is evident that the fabric changed from white to dark brown, and then to black after undergoing depolymerization and carbonization processes. Figure 2(b) displays the untreated cotton fabric, while Figure 2(c) shows the carbonized fabric. The change in color was assessed by analyzing the gray scale values of the carbonized cotton samples using ImageJ software. As the fabric darkened, the gray scale values decreased, whereas it increased when the fabric became lighter or white.
27
The CS10 sample showed the lowest value of 25.7 compared to the other carbonized samples. Following the treatment, all the cotton samples exhibited a transformation in color, turning dark brown and black. (a) Gray scale values (Whiteness index) of the carbonized samples, (b) Optical images of the untreated cotton sample, and (c) Treated cotton sample (CS10).
Figure 3 illustrates the changes in the warp dimension, weft dimension, and surface area of the cotton samples before and after the acid treatment, showing a decrease in area. The dimensional reduction of the carbonized samples followed a trend similar to that observed in color change, with the fibers displaying uniform shrinkage patterns, which indicates the influence of treatment conditions on fiber shrinkage. The carbonized samples showed an approximate 4–13% reduction in area compared to the untreated cotton samples, with the sample CS10 exhibiting the highest reduction of 13%. It is evident that the behavior of fiber shrinkage is influenced by energy flux and temperature.
28
Moreover, the fibers became rougher and finer with a decreased diameter due to the pyrolysis of cellulose molecules.29,30 Dimensional change analysis of the carbonized samples, (a) Warp, (b) Weft, and (c) Area.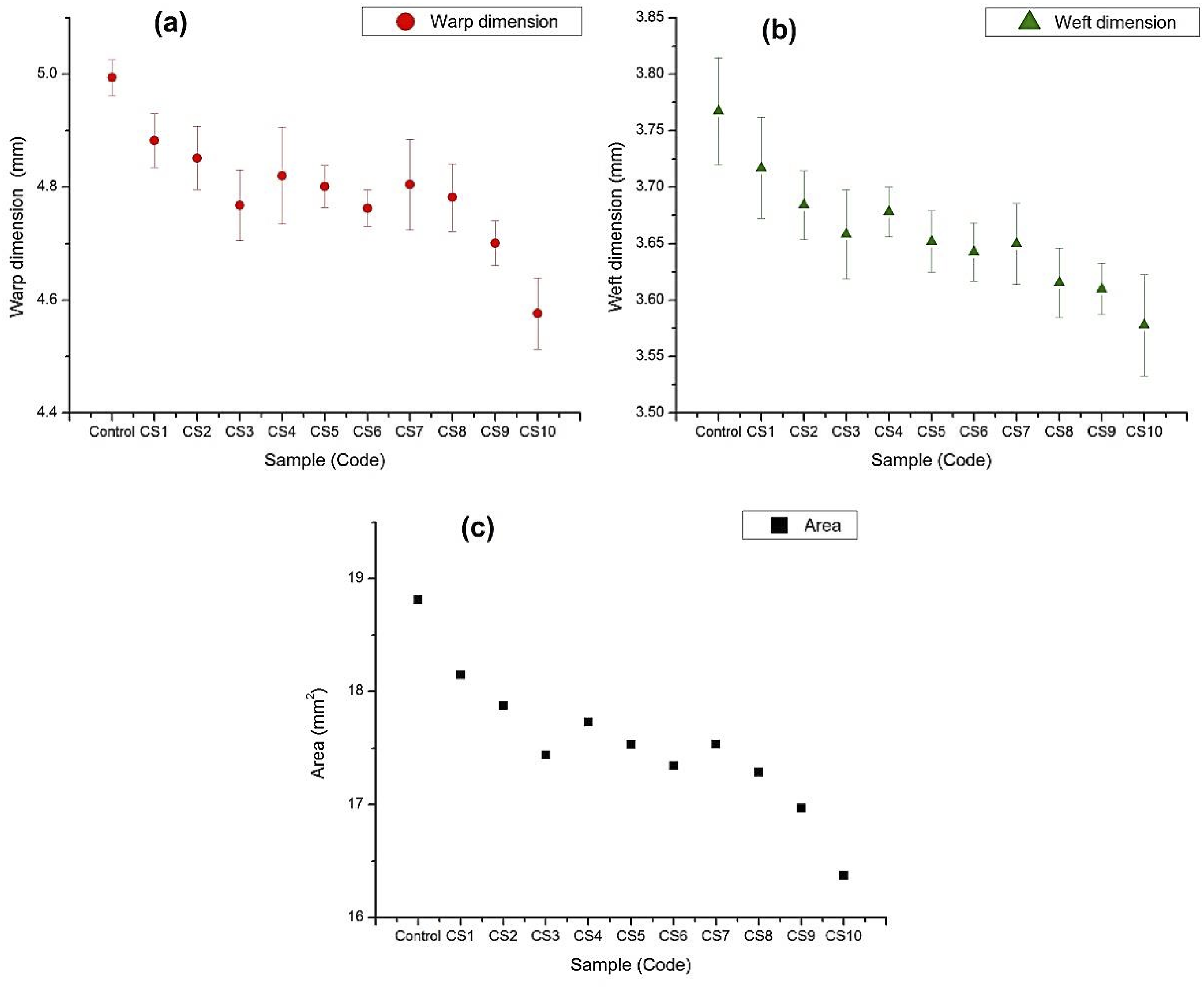
The carbonization process significantly reduced fabric weight due to thermal decomposition and the release of volatile compounds. The original weight of the fabric samples was 1 g, and after carbonization, it decreased to 0.81 g. This corresponds to a mass yield of approximately 81%, indicating a weight loss of 19%. This weight reduction is typical of the carbonization process, during which non-carbon elements such as hydrogen, oxygen, and nitrogen are emitted as gases, leaving behind a carbon-rich structure. The observed mass yield aligns with typical values reported for the thermal degradation of cellulose-based materials, further confirming the successful completion of carbonization. The surface morphology images presented in Figure 4 depict cotton fabrics subjected to various doses of hydrochloric acid for duration of 2 h. Samples CS4 to CS6 were treated at 90°C for a longer duration of 2 h, utilizing HCl volumes of 1 mL, 2 mL, and 3 mL. Samples CS9 and CS10 were treated at 100°C for 2 h, with 1 mL and 2 mL HCl volumes, respectively. The scanning electron microscope (SEM) images illustrate the presence of broken fibers with a wrinkled surface morphology and reduced diameter. The catalytic effect of hydrochloric acid, combined with severe activation conditions, has led to the disintegration of the fibers. Consequently, optimizing the activation and impregnation content conditions is imperative to attain optimal results. Figure 4(a) displays the untreated cotton fibers’ natural curvature and smooth surface. In contrast, Figure 4(b)–(f) present the surface morphology of the treated samples, revealing noticeable changes following the decomposition and carbonization processes. Furthermore, Figure 4(g) and (h) provide high-resolution images of sample CS10, demonstrating a significant increase in surface roughness. Overall, the treated samples exhibit rougher textures than the untreated cotton fabric, indicating successful structural modification during processing. Following the 2 h treatment, the structures of the cotton fibers have been significantly compromised. Additionally, granular substances and residues of the impregnation agent are observable on the decomposed samples.
31
The visual evidence indicates that cotton fibers’ structural integrity is damaged at low hydrothermal temperatures. These findings may offer opportunities for the development of innovative processes and carbon-based materials through the low-temperature vapor-phase hydrothermal carbonization of cellulose. Morphology of cotton samples: (a) untreated cotton, (b) CS4, (c) CS5, (d) CS6, (e) CS9,(f) CS10; and EDS spectra of (g) Untreated cotton, (h) CS10, and (g, h) High resolution image of CS10.
Figure 5 depicts the EDS analysis of the untreated cotton and treated cotton samples. According to the EDS analysis, the treated material primarily comprised oxygen, accounting for 88.4% by weight, followed by carbon, at 11.6% by weight. The untreated cotton sample exhibited weight percentages of 91.7% for oxygen and 8.3% for carbon. The calculated carbon-to-oxygen (C/O) ratio further reinforced the findings, which increased from 0.09 in the untreated sample to 0.13 in the treated sample. The C/O ratio is an essential indicator of the dehydration and carbon enrichment level that occurs during carbonization. A higher C/O ratio suggests a more graphitized or carbon-rich structure, which aligns with the chemical dehydration and decarboxylation typically resulting from acid treatment. Similar studies have noted this trend, where higher C/O ratios result from losing oxygen-containing functional groups during carbonization.
32
These findings indicate that the hydrochloric acid facilitated the dehydration and carbonization of the cotton fibers.
16
EDS spectra of (a) Untreated cotton, and (b) Treated cotton.
Mechanism analysis of hydrothermal carbonization
It is commonly believed that the hydrothermal carbonization of cellulose involves three main processes: gasification, liquefaction, and carbonization. During hydrothermal liquefaction, cellulosic materials can be transformed into various products, including glucose, fructose, organic acids, and more. Subsequently, the resulting soluble oligomers undergo dehydration, polymerization, polycondensation, and aromatization to generate a range of carbonaceous materials, such as carbon spheres and nanofibers.33–35 In the existing literature, two hydrothermal mechanisms have been delineated. One of these, referred to as the soluble pathway, hinges on the initial cellulose hydrolysis process, notably facilitated at elevated temperatures. This process yields glucose and soluble oligomers, subsequently progressing to furan derivatives via dehydration, ultimately culminating in carbon particles’ formation through condensation and polymerization processes.
36
In the solid-state pathway, the complete cellulose molecule undergoes dehydration and condensation processes, forming a modified polymer known as intermediate cellulose. This intermediate cellulose transforms into the final carbon material by incorporating soluble compounds through condensation reactions.37,38 The proposed mechanism for vapor-phase-assisted hydrothermal treatment, utilizing a cellulosic substrate, underwent reactions including aromatization, intramolecular condensation, dehydration, and decarboxylation.39–41 These reactions result in the production of carbonaceous materials (see Figure 6). Additionally, there is likely a minimal degree of hydrolysis, which leads to glucose formation during cellulose’s interaction with water vapor. It’s important to note that this reaction mechanism applies specifically to the solid pathway of the hydrothermal mechanism. In the absence of acid, cellulose pyrolysis primarily occurs through depolymerization reactions that release levoglucosan, while dehydration reactions play a minor role. In contrast, the depolymerization reactions that release levoglucosan are significantly suppressed during acid-catalyzed cellulose pyrolysis. Instead, acid-catalyzed cellulose pyrolysis mainly proceeds with hydrolysis reactions, followed by dehydration at low temperatures.42,43 Proposed mechanism for vapor-phase assisted hydrothermal decomposition and carbonization of cellulose.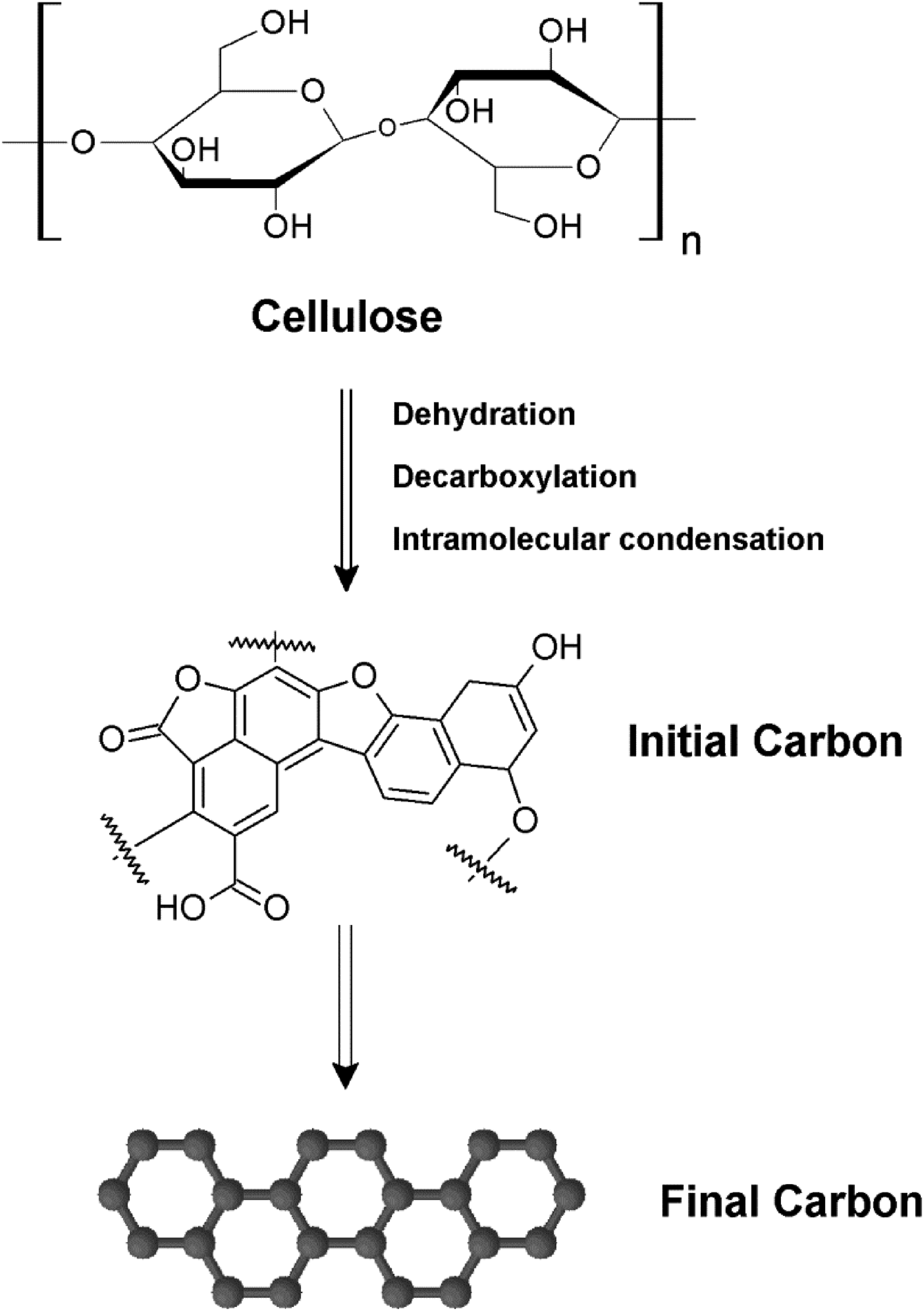
FT-IR analysis
The FTIR spectra of the untreated cotton and the treated cotton samples in the region 400–4000 cm-1 are shown in Figure 7. In the pristine cotton spectra, absorption peaks at 3332 and 2902 cm-1 correspond to the O-H (hydroxyl or carboxyl groups) and aliphatic C-H stretching vibration in the cellulose molecule.18,35 These peaks reflect the presence of functional groups characteristic of cellulose. However, the treated cotton samples exhibit notable changes in the intensities and appearance of specific absorption peaks, indicating the formation of new functional groups. The O-H stretching absorption band peaks exhibited a notable increase in intensity within the spectra of the carbonized cotton samples which suggests a modification in the structure of the hydroxyl groups during treatment. Additionally, a marked increase in the intensity of the C-O stretching peak at 1319 cm-1 and the bending vibration of the C-O groups at 1150 cm-1 further supports the chemical transformations occurring in the cellulose structure.
16
The intensity of the absorption peaks corresponding to the oxygen functional groups has risen. This shows that new linkages between the composite particles and the cotton fibers have taken the place of the oxygen functional groups on the surface of the cellulose molecules. This indicates the formation of new oxygen-containing functional groups, such as ethers or esters, which result from the cross-linking and chemical bonding that takes place during the carbonization process. The C = O (carbonyl, quinone, ester, or carboxyl) and C = C vibrations are represented by the bands at 1708 and 1616 cm-1, respectively,
19
and the bands in the 1000 to 1450 cm-1 region correspond to the C-O (hydroxyl, ester, and ether) stretching vibration and O-H bending vibration. The presence of the C = C peak at 1616 cm-1 further confirms the occurrence of carbon-carbon double bonds, which are indicative of the carbonization structure and cross-linking reactions that take place at elevated temperatures.36,44 The peak at 1425 cm-1 corresponds to alkyl groups such as methyl and methylene groups.
45
FTIR spectra of the untreated cotton and HCl treated cotton samples.
TGA and DSC analysis
The thermal properties of untreated and carbonized cotton fabric were studied using thermogravimetric analysis (TGA). The TGA analysis was conducted on the original cotton and carbonized fabrics under different treatment conditions. The TGA graph of the untreated cotton fabric revealed that the polymer structure decomposed in three stages: volatilization, main degradation, and carbonization as shown in Figure 8(a)
46
The initial decomposition temperature (Ti) and the final decomposition temperature (Tf) are two temperature values that can be used to characterize the thermal behavior of carbonized cotton fabric. Significant weight loss results from depolymerization during the first stage (Ti = 390–430°C), caused by breaking the cellulose structure’s main chain. The evaporation of absorbed water is the cause of weight loss that happens below 100°C. Regarding original cotton, weight loss is virtually nonexistent up to 300°C, rapidly between 310 and 380°C, and slowly up to 600°C. This pattern shows the usual cellulose pyrolysis sequence.
20
The behavior of the samples treated with hydrochloric acid was notably different. The carbonized cotton sample exhibited slow weight loss at 200°C and significant weight loss at around 300°C. The weight curve plateaued at 370°C, with a slight decrease continuing to 600°C. Unlike the one-step reaction of pure cellulose pyrolysis, the HCl-treated cellulose showed a two-step process: a low-temperature phase at 200−300°C and a high-temperature phase at 300−600°C. Similar results were observed for cellulose pyrolysis when inorganic material was added.
34
The degradation process of the original fabric under a nitrogen atmosphere exhibited similarities to the carbonization process. Significant weight loss occurred within the 280−450°C temperature range, primarily attributed to the evaporation of non-carbon elements resulting from high-temperature pyrolysis, accounting for approximately 80% of the total weight loss. Notably, the CS9 sample displayed exceptional thermal stability, retaining over 25% of its mass at 600°C. Experimental findings indicated that the untreated cotton fabric experienced nearly complete degradation below 600°C, resulting in an approximate 88.7% weight loss. Furthermore, the residual weight of HCl-treated cotton samples exhibited higher residue at 600°C, with percentages of 17.6%, 20.7%, 19.6%, 24.6%, and 19.1% for treated samples CS4, CS5, CS6, CS9, and CS10, respectively. TGA and DSC analysis of the untreated and HCl treated cotton samples.
The findings obtained from Differential Scanning Calorimetry (DSC) also revealed a significant structural transformation in the treated samples following thermostabilization after vapor-phase treatment with hydrochloric acid (refer to Figure 8(b)). Both the untreated and treated cotton samples exhibited DSC curves featuring a broad endothermic peak at lower temperatures, ranging from 50 °C to 100°C, which is attributed to the desorption of water molecules adsorbed onto the fiber surface. 34 The DSC thermographs of the untreated and treated cotton fabrics displayed an asymmetrical endothermic peak (indicated by the black dashed box in Figure 8(b)), followed by minor exothermic peaks in the carbonized cotton samples. This suggests the occurrence of crystallization, which was not evident in the thermographs of the untreated cotton (Figure 8(b)). Furthermore, the untreated cotton fabric observed an intensified and wider peak exhibiting an endothermic reaction at temperatures exceeding 300°C (indicated by the red dashed box in Figure 8(b)). This peak is associated with cellulose and hemicellulose decomposition reactions, resulting in the production of levoglucosan and its decomposition by-products. 45 For the carbonized samples, the intensity of this peak decreased while its width increased, except for the original cotton sample. This confirms the decomposition of the majority of fibers, turning them into thermosets.
Comparative analysis
A comparative overview of biomass carbonization techniques.

Conclusion
The study found that the vapor-phase-assisted hydrothermal method significantly improved the conversion of cotton waste into carbonaceous materials. This process’s key parameters included temperature, treatment time, and HCl dose. The use of HCl enhanced hydrolysis and deoxygenation, resulting in a color change of the fabric from white to dark brown to black. The carbonized samples demonstrated a reduction in area of 4% to 13%, with the CS10 sample showing the most significant decrease of 13%. The mass yield was approximately 81%, indicating a weight loss of 19%, reflecting effective cotton conversion into a carbon-rich product. The CS9 and CS10 samples, treated at 100°C for 2 h with 1 mL and 2 mL of HCl, respectively, showed the best carbonization results. SEM images revealed fiber surface, texture changes, and decreased diameter. FTIR spectra confirmed the presence of C = C bonds, indicating carbonized structure, cross-linking reactions, and a reduction in cellulose crystallinity. Measurements using differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA) demonstrated improved thermal stability due to forming pre-graphitic structures with aromatic components, producing carbonaceous materials. The research outlines an effective method for decomposing and carbonizing cotton waste for recycling, highlighting its potential for large-scale application in carbon-based textiles. It presents a sustainable approach to valorizing textile waste, suggesting energy storage, filtration, and advanced textile applications. The study contributes to the circular economy and sustainable material development by converting cotton waste into valuable carbon materials. However, further work is needed to scale the process for industrial use, evaluate the environmental impact of hydrochloric acid (HCl), and optimize the treatment parameters. Additionally, an economic evaluation of the process is necessary. Future research should focus on these aspects to assess this method’s practical and sustainable application.
Footnotes
Acknowledgement
This work was supported by the Project Eco-clothing collection, reg. no. TQ01000450, which is co-financed from the state budget by the Technology Agency of the Czech Republic under the Programme for Support of Applied Research and Innovation SIGMA.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project Eco-clothing collection, reg. no. TQ01000450, which is co-financed from the state budget by the Technology Agency of the Czech Republic under the Programme for Support of Applied Research and Innovation SIGMA.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
