Abstract
The textile industry is striving versatile range of coatings, which imparts multifunctional properties to them. There are many fields where coated textiles perform effectively, where standalone nanoparticles or finishes fail to perform adequately. However, the chemicals, finishes, and synthetically produced nanomaterials currently in use have negative ecological impacts. Hence the review focused on current developments in ecologically friendly biosynthesis techniques for the magnesium oxide nanoparticles, their coating over the textile structures and outlining the potential and novel applications of textiles coated with MgO-NPs in different areas. Due to the rising need for green chemistry and the perspective of nanoparticles in a variety of uses, magnesium oxide nanoparticle production using sustainable and environmentally clean processes has gained popularity. Current advancements in the ecologically pleasant creation of MgO-NPs are those mediated by plants, microbes, and other natural processes. The objective of the current work was to gather all potential methods for the environmentally friendly synthesis of MgO-NPs. Secondly, elaborates on all possible novel applications that green synthetic MgO-NPs coated textiles could have in several industries. The various MgO-NP attributes including size, shape, surface area and their functional characteristics like flammability and antipathogenicity, biocompatibility and redox potential are discussed. The numerous uses of the MgO-NPs coated textiles are also highlighted, including their use as wastewater treatment agents, energy systems, drug delivery, catalysts, and biomedical fields. There is also discussion of these particles in some possible applications in other fields, like nanomedicine and food packaging. The current challenges and future directions in the environmentally friendly MgO-NP production along with applications are also addressed.
Keywords
Introduction
In recent years, the textile industry is actively pursuing the use of sustainable and multifunctional nano-materials. Their aim is to produce the novel multifunctional textile substrate for innovative applications, where standalone nanoparticles do not perform effectively. Nanoparticles are defined as particulate structures that range in size category of 1-1000 nm and subgroups of components with a size category of 1–100 nm. 1 Environmentally friendly product and process development has become more prominent in current years. In the interest of enhancing the manufacturing of metal and also metallic oxide nanoparticles using environmentally friendly resources, researchers have been working in a number of different ways. Magnesium oxide (MgO), has remarkable physical and chemical properties including an excellent refractive index, exceptional corrosion barrier properties, 2 a high degree of thermal conductivity, fairly little electrical conductivity, 3 shielding resistance, predictability athletic ability, good structural properties, combustion, and high light visibility, is an important chemical nanoparticle that is environmentally sustainable, economical, and effective. Magnesium oxide has a general formula of MgO and a crystalline structure made up of Mg2+ ions and O2 ions coupled together by ionic bonds. 4 Magnesium oxide nanoparticles have great characteristics, but their practical applications are restrained by their huge crystalline size and poor surface area. 5
With regard to the synthesis of particles on a nanometer scale and with surface tension, planning techniques for magnesium oxide nanoparticles have been developed. These procedures include drying of the Mg(OH)2, warm decay of a number of precursors, sol-gel methodology, synthetic fume affirmation, 6 aqueous approach, and surfactant methods. Moreover, the production of MgO-NPs from microwave-assisted sol-gel, atmospheric non-thermal plasma jet system and through co-precipitation approach along with green source applications have also been reported in previous studies. 7 In plasma jet systems, several tools are used such as power supply (continuous and alternating voltage, jetting needle, gas control unit etc. to carry the synthesis procedure. K. alvarezii extract was employed as a stabilizer by the researchers in a one-step co-precipitation method. These artificially produced MgO-NPs have a cubic crystal shape. 8 The green amalgamation of MgO-NPs from different sources is very important. In essence, it is an economical, safe, and environmentally acceptable method for creating highly stable metal oxides. Nanoparticles that are produced by green methods (by bacteria, algae, plant extracts and fungi) have many applications in antimicrobial activity, bioactivity, drug delivery, tumor targeting, as an anti-inflammatory agent and in various devices. Certainly, it is a fact that MgO nanoparticles possess certain characteristics but when these nanoparticles are applied on fabrics, various fabrics turn into efficient and multipurpose materials suitable for various industries. Whether in the fields of healthcare, environmental engineering, energy, and food industries, MgO-coated textiles show promising possibilities to redefine various industries.
After extensive review and investigation, current study was focused on compiling the all possible ways have been adopted by researchers for green amalgamation of MgO-NPs. Second, every potential unique application of green synthesised MgO-NPs in various sectors was detailed in this work all possible ways to impart the MgO-NPs over the textile structures. Our specific goal was to give a thorough understanding of the areas in which MgO-NPs coated textiles may be effectively used and offer services. These areas include their effective use in medical applications, wastewater treatment technologies, product manufacture and in devices, agriculture (fertilizers, pesticides, insecticides, fungicides), biosensors, pollutant removal processes etc. All above subsections of review also entail the novel methodologies for green amalgamation of MgO-NPs and their prospective applications in new areas. However, more research work is required in green synthesis techniques to further unfold more applications and advances in nanotechnology.
Importance of MgO nanoparticles
Magnesium oxide nanoparticles, that is metallic oxide nanoparticles, must stayed extensively used in medicine because of their biocompatibility and outstanding persistence under challenging conditions. Due to its distinctive composition, nano-magnesium oxide exhibits a variety of thermal, optical, mechanical, electrical, chemical, and magnetic properties. 9 By inducing a leak in the membrane of their cells and eventually causing their death, MgO-NPs exhibit good antibacterial activity especially for food-borne pathogens including Salmonella enteritidis and Escherichia coli. 10 Magnesium oxide has been used in bioscience to treat sensitive stomachs, heartburn and to mend broken bones. Besides their remarkable antimicrobial effect, magnesium oxide nanoparticles were recently been utilized in cancer therapy. 11 Green synthesis is alternative methods using natural or biological based sources including plants, bacteria and algae. Green synthesis and physiological synthesis are multiple alternatives for this environmentally benign and economically advantageous form of synthesis. Since then, more study has been done on the eco-friendly synthesis of MgO-NPs. 12 This review is aimed at describing the present development of magnesium oxide nanoparticles by green synthesis. The bio-based or naturally synthesized nanoparticles possess many different applications such as in DNA analysis, cancer treatment, optimized reaction rates, gene therapy, magnetic resonance (MRI) and separation science. A modern generation for safe nano-biotechnology has begun with new advances in the green synthesis of nanomaterial because of their productivity, affordability, and eco-friendliness.
Synthesis methods of MgO-NPs
Typically, MgO nanoparticles are formed by physical, chemical, and biological processes. Hydrothermal, co-precipitation, sol-gel, vapor deposition method,
13
micro-emulsion, ultrasonic irradiation
14
and plasma irradiation and chemical reduction techniques can also be utilized in order to produce MgO nanoparticles.
15
In comparison to other methods of chemical synthesis, the sol-gel technique is commonly used for synthesizing MgO nanostructures because it produces more material, is convenient to use, and requires low temperatures.
16
This strategy generally requires metal forerunner I-e for example nitrates, acetyl acetonato, chlorides, acetic acid derivations moreover oxalates of magnesium, sulfates and a compound diminishing expert/body like polymers and citrates for preclusion of hydroxide and for setting pH of the arrangement.
17
Once the required oxide form of nanoparticles has been obtained, the resulting mixture can be heated up to 1000°C. These methods often demand powerful machinery and significant energy to produce magnesium oxide nanoparticles. Therefore, green synthesis is the greatest replacement for the previous methods due to its easy, affordable, and environmentally friendly processing technique. In order to reduce the destructive character of the product, natural substrates including algae, bacteria, plant or fungi are generally utilized in place of chemical anchoring and dissolving agents. Figure 1 is showing the different synthesis approaches of nanoparticles. A schematic layout for different synthesis approaches of nanoparticles. Reprint with permission from [Gaurav Pal], [Green synthesis of nanoparticles: A greener approach for a cleaner future]; published by [Science Direct], [2019].
18

Green synthesis of magnesium oxide nanoparticle
Synthesis of MgO nanoparticles from bacteria
Cell wall of bacteria is crucial in the production of nanoparticles because necessary metals should move through the wall into the cytoplasm and ultimately come back through the meshwork of the wall for the purpose of extracellular release. The content in peptidoglycan such as amino acid of cell wall provides with poly-anions for the stoichiometric interaction between chemical reactive groups of the wall and metals which leads to inorganic deposition of metal. The physiochemical growth parameters such as pH, particle size, culture duration, growth media composition, temperature and light or darkness related growth conditions may influence the resistance of bacteria to metallic ions and the synthesis of metallic nanoparticles. The technique of extraction and production of metal nanoparticles from metal exhibiting bacteria delivers potentially good optical and electrical characteristics along with several uses in the future applications.
19
Bacterial nanomaterials can be fully extracted and have numerous uses in chemical analysis, metal ion recuperation, the medical field especially in arthritis, and medication administration. As a subsequent step in the bio-sorption process used to recover metals from the environment, the metals are bio-reduced into NPs. Therefore, combination strategy of biosorption along with bio-reduction for conversion of heavy metal waste into NPs is becoming more and more intriguing in the context of industrial nanoparticle fabrication. Because of their innate capacity to adjust to drastic changes in their environment, bacteria that are particularly thermally sensitive, represent a better candidate for the green amalgamation of MgO nanoparticles. For the green amalgamation of MgO nanoparticles, Lactobacillus plantarum has been chosen.
20
The bacterial societies barely made a move against the elements with their sterile, un-vaccinated MRS stock. After being weakened, the way of life was assigned the numeral 0. Two substances were used during the experiment titration; 1M magnesium nitrate and after that; 0. To halt this change interaction 2 M NaOH was slowly added into the flask. After approximately 15–20 min, the behavioral pattern was fixed in a water shower heated to 40°C to allow the white incentive to settle. Subsequently, the way of life was, in the end, finished 10 hours post-hatching with the chickens reared at room temperature.
21
Then at that point the way of life was centrifuged for 15 min at 5000 r/min subsequently. Finally, particles were taking out from the supernatant and two washes with milli-Q water were done with much care. The nanoparticles were then gently dried to the formation of powder it was done under great care. The delivered nanoparticles were in the hydroxide structure when they were scorched at 300°C for 4 h in order to convert into the oxide structure.
22
Both external and internal nanoparticle synthesis is possible in bacteria. However, there are some restrictions on bacterial nanoparticle manufacturing. Due to a lack of a full comprehension of the mechanics, the two primary problems are the difficulty in controlling the geometry and second the purification of nanoparticles.
23
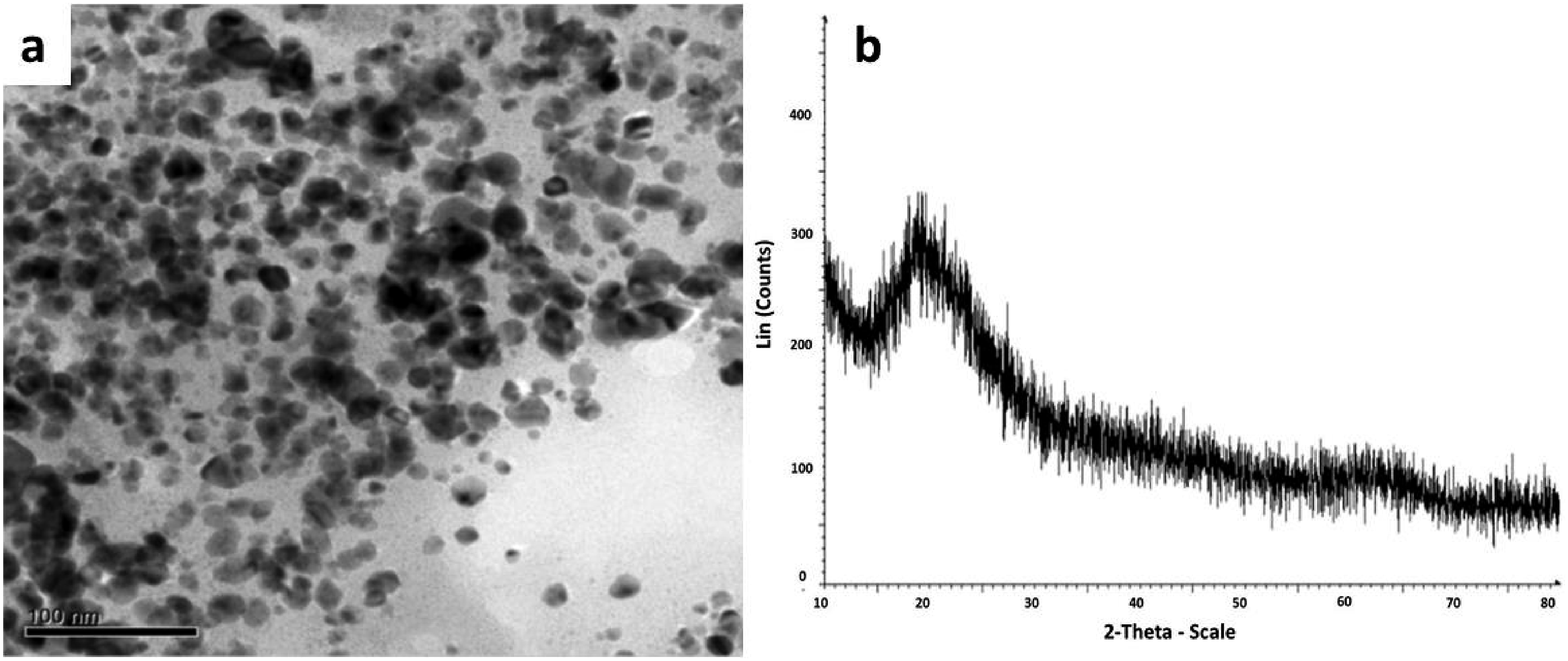
Controlling particle size, shape, and ability to manufacture monodispersed particles on a large scale is difficult. Therefore, before these technologies can be taken into consideration for industrial usage, more research is required to better understand the whole process. In a recent study, Mohanasrinivasan et al. described a microbe-mediated low-cost but reproducible and recyclable technique for the green amalgamation of MgO-NPs. Their work was based on a microbe specie i-e Lactobacillus plantarum (acting as a reductant) and the obtained NPs indicated great property to inhibit or stop the cancerous cells by almost 60%. The TEM images of MgO-NPs with various shapes, as presented in the Figure 2(a). The acquired MgO-NPs were almost spherical and the distribution was symmetrical to a very high level. Therefore, through TEM microscopy, it was identified that about 80%
24
of the nanoparticles had size distribution practically less than 30 nm in size. Similar findings were supported by the past investigations and reports as well. (a) TEM micrograph of MgO-NPs (b) XRD design for L. plantarum intervened nanoparticles. A flowchart shows the biosynthesis of MgO-NPs. Reprint with permission from [Mohanasrinivasan], [Biosynthesis of MgO Nanoparticles Using Lactobacillus Sp. and its Activity Against Human Leukemia Cell Lines HL-60]; published by [Springer], [2017].
24
Synthesis of MgO nanoparticles from fungi
The green synthesis of MgO-NPs using fungi is an innovative approach. The method reinforced the ecofriendly and sustainability by effectively utilizing natural resources. In a recent study, the biomass filtrate of Rhizopus oryaze strain E3 was carried out with the purpose to incorporate it as a biocatalyst for the climate facilitating preparation of MgO-NPs. Three circles of parasitic culture that could be estimated to be around 0. Approximately 8 cm in breadth, the explants were vaccinated into 100 mL of malt extricate stock (MAB) media. These stock media plates were incubated for nearly 5 days at 30 ± 2°C accompanied by shaking in states of 150 r/min. At the time when the incubation period was over, the immunized MAB was centrifuged and tried to get the parasitic biomass. The contagious biomass was mixed in with a forerunner I-e Mg (NO3)2. Using the balanced equation 6H2O the manufacture was done with a conspicuous sign of variety change of parasitic biomass from dull to foggy white. In addition, to support this result, the variety change was moreover characterized utilizing UV-Vis spectroscopy to identify the center SPR. Similarly, Jeevanandam et al.,
25
elucidated that periodic size of created MgO-NPs will be small as the advertised SPR is less than 300 nm, however could be more anisotropic at a lot higher advertised SPR like 300 nm. The further morphological properties of myco-blended MgO-NPs in terms of, shape agglomeration, size and qualitative and quality compound pieces were analyzed using SEM-EDX and tem analysis. As seen from the Figure 3(a) and (b) that are SEM pictures; the NPs were round in shape and almost dispersed and had very little agglomeration. Characterization of myco-synthesized MgO-NPs. (a) TEM image, (b) SEM image. Reprint with permission from [Saad El-Din Hassan], [Rhizopus oryzae-Mediated Green Synthesis of Magnesium Oxide Nanoparticles (MgO-NPs): A Promising Tool for Antimicrobial, Mosquitocidal Action, and Tanning Effluent Treatment]; published by [MDPI], [2021].
25

Synthesis of MgO nanoparticles from Algae
Microalgae are microscopic, multicellular, oxygenic photoautotrophs that make antioxidants that are homologous to plant extracts.
26
The algal extract acts as a suitable capping agent and absorbent because it contains functional groups like hydroxyl and carboxyl.
27
It is obvious that the mechanisms indicated by the algal extract and the plant extract are analogous. Furthermore, polyphenols, peroxidases, carbohydrates, alpha-tocopherol, and beta-carotene are abundant in algal extracts. The discovery of clean, environmentally friendly methods for synthesizing various metallic and metallic oxide nanoparticles, such as silver, zinc, and platinum, and one of them, MgO, has shown great promise in the photo reduction of algae.
28
Both activities occur instantly in algae and can either be intracellular or extracellular based on their surroundings. As a result, many scientists have a keen interest in studying the biological process by which nanoparticles are synthesized and produced by algae. Algal cells can now produce intricate inorganic internal or extracellular structures as a result of evolution. By raising the pH of the medium to 11, Vandamme worked on the emulsification of microalgae like C. vulgaris with magnesium.
29
The brown algae Cystoseira crinita metabolites were used by Fouda et al. to study MgO NP production. The analysis of produced NPs’ antibacterial properties showed their significant effect on gram-negative and gram-positive bacteria. It was revealed that MgO-NPs’ most notable antibacterial effect came from their reaction and creation of volatile oxygen class. While the production of nanoparticles from algae is safe and good for the environment, it is more time-consuming, requires more steps, and is less effective than NPs from plants. The use of the marine brown alga S. wightii in the production of MgO is also documented.
30
Though algae can be used to produce nanoparticles in a green manner, there are still some drawbacks that must be resolved before this process can be regarded as long-term sustainable. Comparing the extract preparation procedure to chemical and physical approaches, the yield of NPs is lower and takes more time.
31
To increase the prospects of algal synthesis in the future, several control factors involved in the synthesis need to be carefully investigated. According to Amr Fouda et al., brown algae, Cystoseira crinita, secretes metabolites that are employed as a bio-catalyst for the green amalgamation of magnesium oxide nanoparticles (MgO-NPs). The biomass of C. crinita is first cleaned, dried, and crushed into a powder. An algal extract was produced by heating, mixing this powder with water, and centrifuging the mixture. MgO-NPs saw a colour shift since light yellowish to yellow-brown as a result of the extract’s reduction and stabilisation. Figure 4 illustrates the flowchart about the biosynthesis process of magnesium oxide nanoparticles (MgO-NPs) derived from algae. The obtained NPs were thusly calcined for 4 h at 400°C. Various technique were used in characterizing the created MgO-NPs namely transmission electron microscopy (TEM), X-ray diffraction (XRD), fourier transform infrared spectroscopy (FT-IR), scanning electron microscopy coupled to energy-dispersive X-ray analysis (SEM-EDX), and X-ray photoelectron spectroscopy (XPS). The information revealed that when the maximal surface plasmon reverberation was 320 nm, nanosphere and crystallography MgO-NPs of 3-18 nm size were made. A flowchart shows the biosynthesis of MgO-NPs. Reprint with permission from [Fauda], [Enhanced antimicrobial, cytotoxicity, larvicidal, and repellence activities of brown algae, Cystoseira crinita-mediated green synthesis of magnesium oxide nanoparticles. Frontiers in Bioengineering and Biotechnology; published by [Frontiers], [2022].
32
Steps involved for the synthesis of MgO nanoparticles by using plant extract. Reprint with permission from [S. Abinaya], [Green synthesis of magnesium oxide nanoparticles and its applications: A review]; published by [Science Direct], [2021].
5


Synthesis of MgO nanoparticles from plant extracts
Due to being more readily available, less expensive, ecologically responsible, simple to prepare and handle, and safe for microorganisms, plants have become a popular natural medium for magnesium oxide nanomaterials. Moreover, there are fewer health implications compared with bacteria-assisted MgO nanoparticle synthesis because the majority of plant extract processing solvents are distilled water and ethanol. To create MgO nanoparticles, plant removes from an extensive variety of plant parts, for example, the root, bark, leaves, blossoms, organic product mash, and strips, are utilized. Plant extracts have been reported to contain significant levels of antioxidants, which are vigorous composites including phenolic acids methylxanthines, saponins, and flavonoids. These antioxidants aid in the removal of free radicals chelated metal complexes, and reactive oxygen species. Plant extracts can therefore operate as bio-reductors and regulators. 33 Plant biological molecules possess significant potential in converting metal salts into nanoparticles. The process of making MgO-NPs from plant extract generally involves three stages. In the main stage, metallic ions are reduced to metal atoms, and then the reduced metal atoms nucleate. During the next stage, the thermodynamic stability increases and small nearby nanoparticles merge into larger particles. In the final step, the process comes to an end and the NPs are given their final shape. 34 Various dynamic biomolecules present in the plant separate guide in the decrease and security of metal particles in the arrangement. Figure 5 is illustrating the steps involved for the synthesis of MgO nanoparticles by using plant extract.
Trigonella foenum-graecum was used in the Green synthesis of MgO nanoparticles, as described by Mary Vergheese et al.
35
It is a simple technique that works well for a series of natural uses. This method of building nanoparticles makes them suitable for use in antimicrobial research. The following are the magnesium oxide nanoparticles made from fenugreek leaf extract. 30 mL of the plant extract were placed in a 500 mL beaker, and 150 mL of the recently made 5 mL of magnesium nitrate solution were added drop by drop with a burette. Additionally, 1 mL of NaOH was added drop by drop while stirring continuously for 2 h at an 80⁰C temperature. A dramatic change in colour from pale green to brown was seen upon the addition of magnesium nitrate solution, indicating the creation of Mg (OH)2 nanoparticles. SEM image demonstrates that the biosynthesised MgO-NPs were made up of a variety of tiny, spherical shapes as shown in Figure 6. Its measurements were discovered to be spherical and within the range of 36.7 and 69.6 nm). Table 1 is showing the different methodologies use to synthesized MgO-NPs by various kinds of plants, bacteria, and fungi. (a) Collected and washed leaves of Trigonella foenum-graceum, (b) SEM image of synthesized MgO-NPs. Reprint with permission from [Mary Vergheese], [Green synthesis of magnesium oxide nanoparticles using Trigonella foenum-graecum leaf extract and its antibacterial activity]; published by [Journal of Pharmacognosy and Phytochemistry], [2018].
35
Magnesium oxide nanoparticles made using a green process with different plants, bacteria and fungi.

Application methods of MgO nanoparticles over the textile fabrics
MgO nanoparticles can be processed onto textile fabrics through different techniques and padding technique is one of the most common techniques used in lab as well as industrial level. This process involves treating the fabric with a solution containing MgO nanoparticles then rolling the fabric to ensure that it has a uniform coating. The applied chemicals in the fabric are then allowed to dry and cure to make the application process compete. It has been reported recently that using Mechanism of coating of nanoparticles on the surface of fabric.
54

Another recently adopted method to impart nanoparticles over the textile surface is the
Applications of green synthesized MgO nanoparticles and their coated textiles
The application of green-synthesized MgO nanoparticles spans a wide range of industries and biomedical fields for osteogenesis, as pills for fungal and bacterial suppression, in cryoinjury, as well as in the ligand binding of uranium ionic species, photo-catalysts, battery storage, and raw sewage monitoring among many other uses. Textile fabrics coated with magnesium oxide nanoparticles (MgO-NPs) have emerged as a promising technology in agricultural systems due to their antimicrobial, UV-protective, and water-repellent properties.
Applications in agricultural system
Green synthesised Magnesium oxide nanoparticles, In the last few decades, have been applied to agriculture primarily in two ways: either as synthetic fertilizers that boost the production of agricultural goods, or as small-molecule pesticide to get rid of pests, diseases, and weeds that are impeding cultivation plant growth.
As a plant protection against pathogens
It is amazing how magnesium oxide nanoparticles have so many benefits for agriculture. They can help with plant protection and boost agricultural production. Nanomaterials have definitely made a big impact in this field. Lin Cai et al. 56 utilize the magnesium oxide nanoparticles to fight against combat Ralstonia solanacearum (a phytopathogen that affects tobacco production), represents a significant breakthrough. These nanoparticles showed innovative and effective solution to control harmful effect of pathogens and reduced biofilm formation. The greenhouse tests with tobacco plants also showed positive results, with increased height and weight after treatment with MgO-NPs.
In agricultural sector, the cotton fibers that is treated with MgO NPs has been applied as a possible interest in seed germination, and in nano- fertilizers, and nano-pesticides, phyto remediation and in treating plant diseases. Because of their inherent anti-microbial qualities the coated textiles could be drawn upon when handling plants or crops or during the harvesting process in order to reduce contamination plant diseases. Due to their inherent antipathogenic properties the coated textiles can be used as a protection against harmful microbes during plant handling or harvest, minimizing cross-contamination.
57
In a laboratory setting, MgO-NPs at concentrations of 50 and 100 mg per liter were tested against the fungus A. dauci which causes leaf blight. As Figure 8 illustrates, the fungus was considerably impeded in its growth by the MgO-NPs at these concentrations. The decrease in fungal growth was significantly greater at higher NP concentrations. After 10 days, it was discovered that at 50 and 100 mg per liter, respectively, A. dauci’s growth had decreased by 38.60% and 56.96%. The number of MgO-NPs employed determined how detrimental they were to the fungus; 100 mg per liter showed the highest level of inhibition. The impact of MgO-NPs on Aspergillus flavus. Figure shows the contagious state development and % of development hindrance in-vitro condition against test separates. T1 = control; T2 = A. T1 = control; T2 = dauci; T3 = MgO-NPs (50 mg L-1); T4 = MgO-NPs (50 mg L-1)+A. T1 = control; T2 = dauci; T5 = MgO-NPs (100 mg L-1); T6 = MgO NPs (100 mg L-1)+A. dauci. Information are mean ± SE (n = 5). Reprint with permission from [Lukman AHAMAD], [Exploring the nano-fungicidal efficacy of green synthesized magnesium oxide nanoparticles (MgO NPs) on the development, physiology, and infection of carrot (Daucus carota L.) with Alternaria leaf blight (ALB): Molecular docking ]; published by [ScienceDirect], [2023].
58

As a plant protection against sun light
MgO-NPs coated fabrics showed UPF values of greater than 50, that is excellent in blocking UV raysThus, the MgO-NPs have UV-blocking properties, and as such can be good for shade nets, plant/net protectors; mulching nets, insect nets, bird nets etc., horticulture, floriculture etc. Additionally, the texiles coated with MgO-NPs even shield the plants from over-exposure to ultra violet radiation that is crippling crops leading to low yields. The second potential application of MgO-NPs coated textile in referred agriculture sector may be in the compaction of the soil.net, insect net, bird net, etc.), horticulture, and floriculture. Moreover, the texiles coated with MgO-NPs protect plants from excessive UV radiation, which can damage crops and reduce yield. 52
In a recent reserch, ppy–MgO and ppy–MgO–CNT composites were synthesized with different weight ratios using in situ chemical polymerization method. In that specific UPF application, the ppy–MgO–CNT composite was indicated to have a UPF of 30. In the antibacterial study, the zone of inhibition was measured in all the test samples and the MRSA and PAO1 bacteria. The zone of inhibition was observed to be 4.0, 3.0 mm for the ppy – MgO – CNT composite. Therefore, the ppy – MgO – CNT composite was reported to demonstrate effectiveness. 59
As a plant protection against soil
The second potential application of MgO NPs coated textile in agriculture sector is the consolidation of soil. Cohesive soil stabilization by using MgO-NPs coated cotton fiber was also examined, where; the fabric with particles used showed the ability to decrease the water absorption capability of the plant to about 170%. Metal particles coating was to protect coir fiber from moisture content that otherwise shortened its life. Following studies by Brahmachary et al. gave the illustration of soil improvement whereby natural bamboo fibers modified with MgO-NPs to increase the strength and stiffness of soil. The percentage of the quantity of bamboo fiber used had a direct proportion with the California bearing ratio value of the soil which was greatly enhanced at 1.2% bamboo fiber dosage. 60
MgO nanoparticles as nano fertilizers
The formation of nanomaterial structures is mostly accomplished using metal oxide, such as magnesium oxide (MgO). When plants are initially subjected to nano-fertilizer, MgO nanoparticles act as a nano-fertilizer for sound defense of tomatoes against wilt invasion; slightly effective if the plants are first subjected to enhanced parasite growth, improved photosynthesis, and crop yield in cowpea.
61
Deficient magnesium keeps plants from growing and actually slows down production. Additionally, MgO-NPs perform crucial structural and regulatory roles in plants, especially when it interacts with nucleophilic ligands. Where, MgO-NPs play a role of micronutrient and acts as a catalyst to initiate more chemical reactions (in comparison to other nutrients). A recent study represents the comparison of the impacts of incorporated NPs compared to the control plants and their representative composts, including ZnSO4. 7H2O (Zn- The results indicated an increase in plant height due to Mg-based fertilizers, showing growth of approximately 4%, 18%, 32%, and 33% at concentrations of 10, 20, 30, and 40 ppm, respectively. These values reached optimal enhancement points, with increases of 8%, 33%, 50%, and 50% compared to the control plants when treated with MgO nanoparticles (Figure 9).
62
The appearance of the new bit on account of nano-composts is due to the fact that they have considerably larger capability in the providing of the plants supplements than the typical compost. (a) the application of Mg-bulk and MgO-NPs to plants resulted in a higher height than control (b) the difference between the control and Mg-bulk and MgO-NPs applications in terms of the growth in fresh biomass of plants. The average values of three replicates are shown, along with standard deviations. The letters (a–d’) on the bars stand for the various treatment variations. Reprinted by permission of Ushna Khalid from [ScienceDirect], [2021] [Comparative effects of conventional and nano-enabled fertilizers on morphological and physiological features of Caesalpinia bonducella plants].
62

A study by Sharma et al. details how black chickpea is affected by MgO-NPs nano-priming. When exposed to 10–150 μg/mL NPs for an prolonged era of time, black chickpeas displayed changed shape along with variations in antioxidant, protein, and carbohydrate content. After priming with MgO-NPs, the shoot and root lengths rose by 171–142 and 217%–328%, respectively. Chickpea biomass and glucose levels were enhanced by 112 and 15%, respectively, through the usage of MgO-NPs to raise the amount of chlorophyll. 63 To make such a decision, Faizan et al. had to direct a review to assess how the beneficial application of 500 mgL−1 MgO-NPs either with or without the addition of 150 µM ascorbic acid, could reduce arsenic toxicity in soybean plants. Another research work aims at analyzing the short term behavior of nanoparticle coated natural fiber impregnated soil, and its application in sustainable pavement subgrade. Tensile strength and moisture absorption test was also conducted on all untreated and treated natural fibers. The nanoparticles were deposited on the fiber surface and caused increase in surface roughness of the fiber and in turn increases the tensile strength on the composite material. The nanoparticles shown to be impregnated on the fiber surface prove the applicability of chemically treated natural fiber for developing improved subgrade strength of embankment material as a sustainable infrastructure solution. 64
MgO nanoparticles as nano-pesticides
Natural and antimicrobial magnesium oxide nanoparticles (MgO-NPs) are widely preferred and established because of their excellent natural characteristics. Drawing from past research work, approaches have been demonstrated concerning the approach that MgO-NPs might be utilised as an acceptable antibacterial substance, individually or in composition with other antimicrobial substances. Imani et al.
65
demonstrated that green-synthesized MgO nanoparticles effectively inhibited the growth of Staphylococcus aureus and Escherichia coli. Notably, when exposed to 20 mg/mL of MgO nanoparticles, there was a significant reduction in bacterial colonies. Additionally, Cai et al.
56
observed that MgO nanoparticles significantly inhibited the growth of the pest pathogen Ralstonia solanacearum. The effectiveness of nanoparticles against expected mosquitos has been previously studied. The activities of MgO-NPs as mosquitocidal specialists can be summed up into two places: how they can furnish responsive oxygen species (ROS), and how cosmopolitan they are to sharp the cell wall. MgO-NPs react with A. stephensi and disperse into the environment giving Mg2+ and O2− ions. Higher levels of O2− ion formation cause formation of reactive oxygen species, which cause oxidative injury and lipid peroxidation. Further, the particles of cell parts are also emitted due to the disturbance in the cell balance that occur due to an increase in Mg2+ concentration and causes death of the mosquito cell as shown in Figure 10. Some researchers observed that nanoparticles address the thiol group of amino acids or the phosphate group of nucleic acid thus altering their functionality.
66
Potential processes for biosynthesized MgO-NPs as a mosquitocidal agent. Adapted with permission from [Amr Fouda], [An Eco-Friendly Alternative to the Control of Pathogenic Microbes and Anopheles stephensi Malarial Vector Employing Magnesium Oxide Nanoparticles (Mg-NPs) Manufactured by Penicillium chrysogenum]; presented by [MDPI], [2021].
66

Polypropylene enhances the chemical stability of the magnesium oxide nanoparticles by increasing the amount of methyl parathion an organophosphate pesticide, which the reactive sites can degrade. Aluminium oxide and magnesium oxide nanoparticles have been shown to decrease the damaging impacts of diazinon, a chemical that the EPA (Environmental Protection Agency) banned due to its high toxicity to humans as well as plants. Abhijit et al.
67
did a study on the quantity of pesticides in surface and drinking water, which has become a serious danger to health. In this work, MgO-NPs were made from purple-colored rice leaves (Crossa) and used to eliminate three pesticides from water simultaneously: thiamethoxam, chlorpyriphos, and fenpropathrin. As the pH rose after 2 h, more thiamethoxam, chlorpyriphos, and fenpropathrin adhered to the surface, as Figure 11(a) illustrates. This increase in adsorption capability at higher pH levels is that MgO-NPs had a relatively high pHZPC value of 10.8. The enhanced electrostatic repulsion forces at higher pH values may facilitate adsorption. The adsorption capability increases from 10 to 50 mg/L of pesticides in the water, as Figure 11(b) illustrates. The best amount of time needed to reach the adsorption balance is shown in Figure 11(c). This experiment aimed to maximize the adsorption capacity while using the least amount of adsorbent. Figure 11(d) shows that the percentage of pesticides removed went up when the dose of MgO-NPs increased from 10 to 100 mg while keeping other factors like the initial concentration of the adsorbate (30 mg/L), pH (7), and volume of the adsorbate (30 mL) constant. It was observed that the percentage of pesticide removal increased as the dose of MgO-NPs increased for all three insecticides. Factors influencing pesticide removal by MgO NPs: (a) pH, (b) initial concentration, (c) contact time, and (d) adsorbent dosage. Conditions: pH 40 mg adsorbent, 30 mg/L adsorbate, 30 mL volume, 2-h contact; concentration 40 mg adsorbent, pH 7, 30 mL volume, 2-h contact; time 40 mg adsorbent, pH 7, 30 mg/L adsorbate, 30 mL volume; dosage pH 7, 30 mg/L adsorbate, 30 mL volume, 2-h contact. This information was taken from a publication by Abhijit Kar, titled “Facile synthesis of novel magnesium oxide nanoparticles for pesticide sorption from water,” published by Springer in [2023].
67

For the identification chemical toxins in the environment
Some of the metal oxide based nanoparticles for which adsorption characteristics have been very high and active are MgO, CaO, TiO2, ZnO, Al2O3 and Fe2O3 containing toxic chemicals including air pollutants; and chemical warfare agents (CWAs) acidic gases and pesticides. Destructive adsorption, takes place on the outer surface of the nanoparticles, and in the process the adsorbate is chemically decomposed, and hence rendered non-hazardous.
68
The MgO-NPs coated textile used in Housed in the large surface area showed an imense photocatalytic activity that can break down toxic chemicals and organic pollutants in the air like odour molecules, bacteria and viruses.
69
In addition to their photocatalytic properties, MgO-NPs integrated fabrics can be used as sensor-based materials. It also makes the fabrics into sensor-based materials and can transform the mechanical forces exerted to electric signals and be useful for tracking body activity by the heart rhythm and pulse if worn under the skin. A plausible reaction process for the disintegration of chlorpyrifos on the surface of MgO. Reproduced with permission from the Royal Society of Chemistry. Reproduced with permission from [Adrián Leonés], [Potential Applications of Magnesium-Based Polymeric Nanocomposites Obtained by Electrospinning Technique]; published by [MDPI], [2022].
72

MgO nanoparticles ability to act as cleansing mediators is widely exploited in many different contexts, which include the detection and eradication of chemical pollutants. The very dangerous pollutant 2,4,6-trinitrophenyl, which is associated with cancer, hepatic problems, dermatitis, etc., is decomposed by magnesium oxide nanoparticles and zinc oxide nanoparticles.
70
Another study demonstrated, the use of MgO-NPs coated textile as pollutant absorbent in the environment. Thus, as the environment experiences a problem of high amounts of waste, it becomes important to obtain optimum methods of cleaning it. Gao et al.
71
of Chinese University of Hong Kong has established a technique to synthesize MgO-NPs with four specific structures and highlighted that it had an outstanding capacity for the removal of the heavy metal ions as well as the organic pollutants present in water. A lot of research articles have reported on the adsorption mechanisms of Mg based nanoparticles for different pollutants. These processes are largely related with the electrical attraction of dye molecules and the surface coordination of hydroxyl groups on the MgO adsorbent materials. Also, MgO nanoparticles are reported to catalyze oxidative degradation resulting in the disruption of the P-S bond of organophosphates and the decomposition of chlorpyrifos (an organophosphate insecticide) via destructive chemisorption, as illustrated in Figure 12. Thus, the researchers concentrate on the creation of electrospun nanocomposites reinforced with MgO-NPs for the removal of toxic substances. Schematic portrayal of the mathematical design of magnesium oxide (Mg4O4)- Sarin adsorbed framework (adapted from Michalkova et al.). Reprint with permission from [Tuğba Baysal], [Nanofibrous MgO composites: structures, properties, and applications]; published by [Taylor and Francis online], [2020].
74

From the coatings of MgO-NPs on cotton fabric, the element revealed that the self-cleaning/stain removal performance of the fabrics was improved to Grade 5 and Grade 4 categories respectively. With regard to UV protection coils of ZnO or MgO-NP coated fabrics had UPF values of more than 50, implying that they were very effective in blocking UV rays. MgO-NPs showed 20% efficacy in cleaning up the effluents contaminated with reactive dye whereas ZnO-NPs offered 4% cleaning efficacy and thus improve this kind of treatments at low cost. Ren 65 was involved in the synthesis of the MgO coated textile fibers and the use of the produced material as an adsorbent medium for the radioactive metal ions. They also found that MgO coated textile fibers were biosorption material that could effectively remove uranium (VI) ion from water and biosorption increase at pH 6. It an indication that the system has attained an equilibrium adsorption capacity of 90 mg/g in 120 min contact time. From the studies it is clear that through MgO coated textile fibers we can have a positive answer of MOX like uranium (VI) from the water system which will be more helpful in water treatment and environmental protection. Almasian et al. 73 have considered the preparation of mesoporous MgO textured with polypropylene glycol coated textile fibers for remediation of heavy metal ions from aqueous streams. In this case, mesoporous fibers with BET surface surface area of 185 m2/g were synthesized. In the study the enhancement of using nanofibers for the removal of Pb, Cu and Cd ions was discussed and it was evaluated that the maximum absorption capacity of the nanofiber reach to its peak point at pH 7 undefined. This implies that these nanofibers could selectively adsorb these heavy metal ions in solution, which is a step towards the improvement in the use of nanofibers for environmental purposes. Figure 13 is showing the schematic portrayal of the mathematical design of magnesium oxide (Mg4O4)- Sarin adsorbed framework.
Smoking poses a great danger to human health. It is crucial to discover how to reduce the effects that cigarette smoking has on health. There are two strategies to lower the rate at which pollutants are released from cigarettes: Two of the pledges made are (a) reducing cigarette consumption and (b) using new generation filters to reduce emissions from cigarettes. The purpose of cigarette filters is to lessen the quantity of tar, nicotine, smoke, and tiny particles that are breathed when smoking cigarettes. In addition to absorbing some of the toxins in smoke, cigarette filters can dilute smoke by adding more air. Using filters reduced the amount of tar and nicotine in US cigarettes by 68%, according to a report. 75 Diminish reconsideration has been made on MgO nanoparticles and discharge feeble of CO, NOx, absolute hydrocarbons and CO disclosed small by cigarettes. Quantitative determination of the degrees of nitrogen oxides, carbon dioxide, total hydrocarbons, and mono carbons in tobacco smoke when able to channel upgrade, activated the scientific intelligence of the company to consider possibilities of the better channels. It is even proposed that while dealing with the superior channel only, complete carbon dioxide emissions could be cut down to 56% of the total possible emissions together with 30% of the nitrogen oxides and 44% and 43% of complete hydrocarbon and carbon monoxide respectively. Figure 14 is showing the components of the illustrated enhanced cigarette filter containing the MgO nanoparticles.
Environmental pollution is a significant problem that impacts countries at all levels of development on a global scale. Although here numerous approaches to address environmental pollution, one drawback is that certain cleaning products have unintended consequences that turn them into toxins in and of themselves. Nanoparticles have become an excellent substitute for a number of conventional environmental cleaning techniques. Because of their solid retention abilities, huge surface region, and phenomenal reaction limit, specialists are concentrating on metal oxide nanoparticles as expected sponges for hazardous gases like sulfur dioxide and nitrogen dioxide. Researchers have established that magnesium oxide NPs could be used to reduce air pollution, particularly when used in automobile emission controls. Nitrogen oxide and carbon monoxide emissions from diesel engines can be greatly reduced by magnesium oxide nanoparticles. These are already considered possible means of extracting harmful chemicals like Sulphur dioxide and nitrogen dioxide because they have excellent absorbing qualities, a huge external area, and a high reaction capacity.77–79
Applications of MgO-NPs in organic transformation
Using this MgO-nanoparticle as a catalyst, we may engage in various important heterocyclic reactions as low as oxidation, as high as reduction, epoxidation between oxygen and hydrogen atoms or elements, condensation into one point upon itself and so forth. Besides, with its help we can form C - C bonds which consist of two carbon atoms joined together in series, C - N which contains one carbon and then nitrogen atom next to it on their periodic tables and when two different elements join as partners for life they are called C-S or O-H complexes and that is just the third category you are dealing with this kind of fertilizer from beginning till end. Applications of MgO-NPs in organic reactions is showing below in Figure 15. Other components of the illustrated enhanced cigarette filter containing the MgO nanoparticles are: (1) first segment of the filter; (2) an intermediate filter; (3) the vest of the cigarette; (4) the tobacco; (5) a thin layer of MgO nanoparticles; and (6) the second segment of the filter. Reprint with permission from [Amirreza Talaiekhozani], [Enhancement of cigarette filter using MgO nanoparticles to reduce carbon monoxide, total hydrocarbons, carbon dioxide and nitrogen oxides of cigarette]; published by [Science Direct], [2019].
76
Applications of MgO-NPs in organic reactions. Reprint with permission from [H Dabhane], [MgO nanoparticles: Synthesis, characterization, and applications as a catalyst for organic transformations]; published by [EurjChem], [2021].
80


Use of MgO-NPs against water treatment
The aquatic ecosystem has become poisoned as a result of increased wastewater discharge into the environment, industrialization, and excessive chemical use. Since the water obtained from the organic resources has biological bacteria dyes, pesticide, detergents, inorganic fluoride, arsenic, copper, mercury and radiological impurities such as cesium, plutonium and uranium the water is not fit for human consumption. A research work described the use of MgO-NPs coated textile fibers against heavy metal detection, including nickel, cadmium, and copper, that are present in drinking water, aquifer steam, and surface water. 81 The most prevalent method for removing contaminants from water is adsorption because it is reasonably priced, simple to use, and does not really generate any secondary pollutants. 82 MgO and numerous other nanomaterials have indeed been successfully employed as adsorbent materials to remove pollutants from water, such as heavy metals, azo dyes, and other substances.83,84 As the high adsorption area in the novel synthetic sorbent and the comfort with which heavy metal decision can be managed to accomplish from extremely minute real sample amounts, the purpose of heavy metal in various samples was carried out using graphene oxide nano-sheets that had been modified with magnesium oxide. The final measurements were carried out using the technique of flame atomic absorption spectrometry. The dyes and textile as well as other chemical industries generate a large amount of wastewater containing dangerous transitional ions, raw components (aromatics) and intermediate products as well as the color. Sectors such as these, which produce toxic waste, are always researching for better, less hazardous ways of purifying wastewater contaminants. Ni, Pb, Cd, Cr, Zn, and Cu are identified to be the most toxic minerals in the wastewater. These toxic discharged metals are fatal to animals, plants, and components of environment like air water, and soil and thus the solution to these problems, has developed a significant discussion for several researchers. In the last few years, scientific organizations have welcomed the removal of chemically reactive ions or elements present in sewerage by using semiconductor, photocatalytic, and bio degradation processes. Heavy materials and toxic substances have been segregated employing various methods such as physicochemical, biological, and electrochemical systems, and application of correct sorts of nano-catalysts. Thus, depending on the ICP-OES analysis results, the Cu, Cr, Ni, Cd, and Zn, together with the Pb, have been selectively removed from industrialized wastewater. 69
Magnesium oxide nanoparticles improve ferrate VI ability to oxidize blue dye, a water-polluting dye used in the textile and pharmaceutical sectors.85,86 Most organizations in the textile and apparels have adopted different dyes to be used in the production of their products with indigo carmine, a dye extracted from water waste being amongst the most popular ones. Exhaustion and decolorization are two methods used in the textile process; the main reagents used in expulsion are magnesium oxide nanoparticles that have the ability to change indigo from blue to blackish color. This process entails breaking down of a dangerous chemical called 2,4-Dichlorophenol (2,4-DCP) into a less hazardous one. Nonetheless, 2,4-DCP is generated after construction works and is released into different water systems, thus, it contributes immensely to pollution in freshwater ecosystems. Magnesium oxide can also be utilized as a catalyst in the ozonation process in order to remove 2,4-DCP from aquatic environments under laboratory conditions and is also used for the degradation of this dye.87,88 It has been known for 2,4-DCP to have negative effects on individuals who consume it, including plants, animals, and humans. Organic pigments including reactive black dye, methyl orang, congo red, methylene red, and methyl blue can be successfully removed using MgO nanoparticles.
Consumption of MgO-NPs in biomedical applications
Recently, magnesium oxide (MgO) nanoparticles because of their biomedical compatibility, stability, and multifunctional application as antibacterial, antiviral, anticancer, antidiabetic, antioxidant, drug delivery, tissue engineering, and bioimaging properties. Therefore, the growing use of magnesium oxide nanoparticles in medical applications requires further investigation of these nanoparticles. Furthermore, MgO nanoparticles explains in detail other significant uses in biomedical fields together with the possible ways through which they exert their effects and briefly discuss toxicity data. 89
Antibacterial activity
The use of nanoparticles (NPs) incorporated in textile materials promises self-cleaning, UV-blocking, protective clothing, and usage in medical sector etc. The medical fabrics are particularly good surfaces for microbial growth particularly in humid environment of the hospital and contribute to many hospital acquired diseases. The measures, including antimicrobial fabrics and dressing materials should be taken in order to minimize the transfer of various types of pathogens; it is possible to prevent the infections to a great extent. Textile fabrics particularly those derived from natural fiber like cotton present favorable breeding grounds for microorganisms because of the large surface area together with the characteristic of absorbing and holding moisture. Additionally, moist cotton compound with body fluids break out as a perfect medium for bacteria to multiply hence creating super infection or smell. Lately, NPs have received unprecedented interest to endow the medical textiles with antimicrobial properties. In which, the metal oxide NPs has been claimed to possess antibacterial activity suitable for biomedical and antimicrobial use. Among the metal oxides semiconductors (MOSs) like TiO2, ZnO, MgO, and CaO, there is considerable interest due to their chemical inertness that makes them quite stable under sever process conditions and because they are known to be nontoxic to humans and animals. 90
Bacterial resistance to the antimicrobial agents is one of the major complications that surface in wound healing and can be as follows: Longer healing time or development of inflammations. Piezoelectric materials may be used in creating bioactive electrically charged surface. This paper focuses on producing two groups of bio-composite from polyvinylidene fluoride with addition of Nano-magnesium oxide (3, 5 and 7 %) wt. by two methods: This was followed by electrospinning and spin coating under special condition for the production of bio film as sensor for wound healing. 91
Antiviral activity
There is Gabbay’s patent on an antiviral textile, ionic copper particles that can be employed for condom sheath, surgical gloves, and tubing. The author provided an overview of the different natural (silk, cotton, wool and linen) and synthetic fibers like cellulose and cellulose acetate fibers, regenerated protein fibers, PU fibers, acrylic fibers vinyl fibers polyolefin fibers were proposed by the author for the formation of the textile , the author rejects polyester and nylon fibers. The activated textile will be produced after selecting and immersing the appropriate textile material in a bath that includes one or more metal cationic species with two or more oxidation states. Then a subsequent reduction process has to take place with the textile interacting with the metal to create a metalized textile. 92
The chosen textile material in this invention is polyamide, incorporating water-insoluble ionic copper particles in a powder form supplied by adding 25 g of a mixture CuO/MgO and Cu2O/MgO2 powder forming a 1% mixture. An in vitro antiviral test was carried on the MT-2 cells to determine readiness of CuO-Cu2O /MgO-MgO2 impregnated fibers in preventing HIV-1 infection. These outcomes demonstrated that 1% CuO and CuO/MgO and MgO Fibers were the effects of 26% inhibition while the positives controls of 70%. They then saturated a latex glove with 1%+ Cu2+/Mg2+ ions and in 20 minutes of exposure, more than 95% subsequent virus infectivity of the lymphocytes, which is the major target of HIV-1, was inactivated. 93
Later on, Gabbay demonstrated a practical way of adding copper or magnesium oxide (CuO/MgO) particles whose size ranges from 0.5 to 2 µm with the rayon fiber that is produced from regenerated cellulosic fiber with the main intention of creating antiviral fibers. The effectiveness of the fibers in the current study convenes in the irreversible process of the Cu2 + ions during contact with water and/or water vapor. The method is comprising these steps: first, the rayon viscose continues in the presence of MgO particles in the quantity of 0.25 and 10% of the initial cellulose dry weight, then the rayon fibre is produced using an acid bath containing sulfuric acid by extruding the viscose through a spinnerette into the bath, and a group of particles will be deposited on the surface of the fibers extending from there. The produced fibers will be antiviral. 94
Drug delivery
Pharmaceutical industries invest heavily in developing advanced drug delivery methods for anticancer therapy. Electrospun nanofibers and nanoparticles loaded with drugs offer versatile biomedical applications, including wound healing and cancer treatment. This study aimed to fabricate elastomeric poly(L-lactic acid-co-ε-caprolactone) (PLACL) nanofibers incorporating Aloe Vera (AV), magnesium oxide (MgO) nanoparticles, curcumin (CUR), and β-cyclodextrin (β-CD) to inhibit MCF-7 breast cancer cell proliferation. The fabricated nanofibers PLACL, PLACL/AV, PLACL/AV/MgO, PLACL/AV/MgO/CUR, and PLACL/AV/MgO/β-CD achieved bead-free, uniform fibers with diameters of 786 ± 286, 507 ± 171, 334 ± 95, 360 ± 94, and 326 ± 80 nm, respectively. PLACL/AV/MgO/CUR reduced MCF-7 cell proliferation by 65.92% on day 9 compared to PLACL/AV/MgO, with 1% CUR interacting with MgO nanoparticles showing the highest inhibition, highlighting its potential for breast cancer therapy. Conventional breast cancer treatments like surgery, radiation, and chemotherapy, while effective, cause side effects and fail to prevent recurrence. Electrospinning-based nanofibers address these limitations, offering advantages such as high surface-to-volume ratio, enhanced drug efficiency, bioactivity preservation, excellent encapsulation, high porosity, ease of fabrication, and cost-effectiveness. Applications include multiple drug delivery systems, natural extract-loaded scaffolds, drug-loaded implants, functionalized polymers, and DNA delivery for gene silencing.
MgO nanoparticles, known for antibacterial and antioxidant properties, show cytotoxic effects on various cell lines and hold FDA approval as safe (21CFR184.1431). Their biological effects depend on size, composition, and concentration, making them promising for selective cancer therapy by targeting cancer cells while sparing normal cells. 95
Tissue engineering
Nanocellulose membranes fabricated via coaxial electrospinning demonstrated superior hydrophilicity compared to single-nozzle methods, likely due to the gelatin shell. Magnesium ions (Mg2⁺), crucial in regulating cellular behavior, influence adhesion, proliferation, and osteogenic differentiation via pathways like TRPM7/PI3K and Wnt/MAPK. However, Mg2⁺ effects are dose-dependent, with excess amounts inhibiting cell growth. Coaxial-MgO membranes provided a controlled, sustainable Mg2⁺ release, preventing the burst release observed in Blending-MgO. This core-shell structure enhanced hydrophilicity and cell adhesion, promoting the adhesion, proliferation, and osteogenic differentiation of human periodontal ligament stem cells (hPDLSCs). Coaxial-MgO also upregulated osteogenic markers such as ALP and Runx2, making it ideal for guided tissue regeneration (GTR) membranes. Coaxial-MgO membranes exhibited significant antibacterial activity, reducing bacterial colonies compared to neat nanocellulose membranes, which lack inherent antimicrobial properties. While Blending-MgO showed a stronger antibacterial effect, this variation likely stems from differences in fabrication methods, warranting further study. Coaxial-MgO has properties that highlights its potential for periodontal tissue engineering and antimicrobial applications. 96
Using the Agar Diffusion method, the antibacterial properties of the fabric coated with the synthesized MgO-NPs were assessed against S. Aureus and E. coli in a recent study. The inoculated agar plates were covered with 15 mm by 15 mm patches of the coated fabric. Both MgO-NPs were found to have bacteriostatic properties against E. Coli when coated on fabric, as demonstrated by a clear zone of inhibition. Gram-positive and gram-negative bacteria differ structurally, which explains similar findings that have also been found in other investigations. Additionally, the low migration rate of magnesium oxide nanoparticles from the coating onto the nutrient agar may be the cause of the absence of an inhibition zone in the case of gram-positive bacteria. These materials have anti-bacterial, self-cleaning, anti-corrosive, and antifungal properties. 52 When given the right conditions—nutrients, moisture, oxygen, and temperature—fabrics are an excellent environment for microbes to grow. Cotton and other natural fibers are more vulnerable to microbial attack than synthetic ones. Therefore, the antibacterial activity of cotton fabric coated with magnesium oxide nanoplates against Escherichia coli and Bacillus subtilis was assessed. MgO nanoplates showed a clear zone of lysis against the bacteria, suggesting that they may be preferred over other chemical compounds that are readily available and minimize bacterial activity. 74
Tissue designing is an area of exploration that utilizations residing cells in different techniques to upgrade tissues and organs by developing and shaping three-layered tissues from refined cells that are then put on bioactive, biodegradable platforms.
97
Tissue engineering has recently aided in the treatment of orthopaedic problems since it makes use of engineering principles as well as biology and chemistry, making it more successful than conventional methods.
98
Magnesium oxide nanoparticles are used in muscle therapy because they enhance the connection between osteoblasts, are applied on HA-PLLA nanocomposites, and provide them with mechanical characteristics suitable for applications in cancellous bone. Moreover, since magnesium oxide nanoparticles promote osteoblast growth, they can be used to modify the mechanical characteristics of other biomaterials added to the mixture.
99
Bacterial cells are disrupted by antibacterial surfaces, which results in cell death. The goal is to decrease or even completely eradicate the adherence of microbes to the surface in order to stop biofilms from forming. Nanofibers can be designed to postpone or even stop microbes from adhering for this reason. By adding metal oxide nanoparticles (NPs) like ZnO, TiO2, SiO2, CuO, ZrO2, and MgO to nanofibers, this property is improved and has a favorable impact on antibacterial properties.However, MgO is becoming more and more popular among the available metal oxides due to its many benefits, including its high surface area, abundance of edge and corner sites, cheap cost precursors, and biocompatibility. Additionally, it can be used with other metallic or nonmetallic materials to improve its resilience and biocidal properties on substrates. Singlet oxygen, hydroperoxyl, OH−, and O−2 are favorable radical generation mechanisms of magnesium oxide (MgO) that impact antibacterial activity by inducing oxidative stress (Figure 16). MgO nanoparticles’ surface oxide ions attach to bacteria and degrade their cell walls and cytoplasmic membrane. Surface oxide ion concentrations increase with decreasing MgO nanoparticle size, improving antibacterial action in this procedure. Gram-negative bacteria are more resistant because of their more complex cell walls, which include periplasmic space and a layer of lipopolysaccharides, whereas gram-positive bacteria are often more susceptible to many antimicrobial chemical and herbal components. However, after coating MgO-NPs, materials might develop antibacterial properties against both gram-positive and gram-negative bacteria.
90
Because NF embedded MgO combines the qualities of two effective nanomaterials to produce antibacterial and biocompatible materials, it is becoming more and more popular According to Dhineshbabu et al.74,100 Electrospinning was used to create nylon-6/MgO hybrid NFs, which were subsequently coated onto cotton fabric to test the product’s antibacterial properties. Antibacterial experiments conducted on gram-positive Staphylococcus aureus and gram-negative Escherichia coli indicated that the use of MgO reduced growth by approximately 67% and 63%, respectively. Although MgO-based systems have demonstrated antibacterial activity additionally, reports of nanofibrous outputs with this capability are limited.74,100 Illustration of (a) potential antibacterial properties of magnesium oxide (derived from Navik et al.) MgO nanoplates’ potential defenses against B. subtilis and E. coli (derived from Ponnuvelu et al.).
90

MgO-NPs coated textile fabric as flame retardant
Because of MgO’s high melting point (2850°C) and excellent thermal stability, it can be added to NF to create nonflammable textiles. Venkatram et al. created Ag/MgO/Nylon six mats for this purpose. Two marks were left on the fiber mats that were made according to specific dimensions, separated by 60 mm, and the burn times between these two marks were noted for every sample. MgO (3 wt%)-AgNO3 (0.25 wt%)/Nylon 6 NF produced the slowest combustion rate, 1.56 mm/s. In addition to its thermal characteristics, MgO reacts with Ag to create a barrier that reduces the rate at which heat and air move Dhineshbabu et al.
101
examined how well the as-synthesised MgO NP/nylon-6 hybrid NF resisted flames. Three distinct materials were subjected to burning tests: (1) uncoated cotton (UC) fabrics; (2) cotton that had been six-coated (N6 C); and (3) MgO/Nylon six hybrid (MgON6 C) fabrics. They showed that uncoated cotton textiles (6.3 s) and Nylon 6 NFs (15.4 s) began burning earlier than MgO/Nylon 6 hybrid NFs (18.5 s) (Figure 17).
74
Methylene blue (MB) (a), methyl orange (MO) (b), and rhodamine B (RhB) (c) solutions’ ultraviolet-visible (UV-vis) spectra at initial and residual concentrations using TiO2 and TiO2/MgO nanorod (NR) photocatalytic textiles, as well as the UV-vis spectra of MB solutions at initial and residual concentrations following various cycles using TiO2/MgO NR photocatalytic textiles (d).
102


Applications of MgO NPs in energy system
Magnesium oxide nanoparticles (MgO-NPs) coated on textiles hold potential applications in the energy sector due to their thermal stability, electrical properties, and resistance to extreme conditions. In a research work carried out by Pingfan Du et al. Co-electrospinning, also known as coaxial electro-spinning, was used to create TiO2/MgO core-sheath structured nanofibers (NFs). The long NFs were subsequently treated with ultrasonic waves to create short NRs. Two innovative technological textiles were created based on the TiO2/MgO-NPs: a photocatalytic functional textile that immobilizes photocatalyst and a photovoltaic smart textile that integrates flexible dye-sensitized solar cells. It was explained how the semiconducting TiO2 core was affected by the insulating MgO coating. The TiO2/MgO NR-based photovoltaic and photocatalytic textiles perform better than their pure TiO2 NR counterparts. When TiO2 is coated with MgO, the charge recombinations that take place in both photovoltaic and photocatalytic processes are significantly reduced, which is the common cause of the performance improvements. Lastly, some possible uses for these two kinds of textiles were suggested. By combining traditional textile research with modern energy and environmental technologies, this work provides a fresh perspective on the creation of exceptional fabrics. The most straightforward and practical method for creating photovoltaic textiles is to construct a solar cell as a structural component and then apply it to any target fabric. Practical engineering demands that the solar cells used in photovoltaic textiles be flexible and lightweight. Apparently, this criterion can only be met by flexible solar cells. Cost-effective flexible dye-sensitized solar cells (DSSCs) have attracted a lot of interest among the several varieties of flexible solar cells.The photoanode film, which is the main component of DSSCs, especially flexible ones, is typically composed of anatase TiO2 nanoparticles (NPs). The modification of NPs by covering TiO2 with another oxide thin layer to form a core–shell structure has been thoroughly studied in order to optimize DSSCs. MgO has a broad bandgap and is an insulating oxide. The energy conversion efficiency of DSSCs can be significantly increased by using magnesium oxide (MgO) as the coating material for TiO2. Two primary causes may be responsible for the enhanced efficiency: the coating layer’s reduction of charge recombination and its enhancement of dye adsorption. 102
Thus, depending on the consumption of the non-renewable energy sources, finding a substitute is as simple as basic. Therefore, in the long run, there will be an opportunity for power modules, solar cells, and batteries as petroleum products’ substitutes. Therefore, the ability of magnesium as compare to other metals is relatively higher in the absorption of hydrogen. Magnesium has a benefit over other halides in that it is abundant in the earth crust and possess a much greater capability to hold hydrogen, which makes it more cost-effective and environmentally suitable. 103 They were found to be extremely promising hydrogen-generating materials that can be used to attain hydrogen through the process of hydrolysis in water. However, this reaction provides hydrogen irreversibly, so this technique is only applicable for a single onsite hydrogen supply.104,105 Moreover, as their exceptionally high surface area and reactivity, magnesium oxide nanoparticles can store and absorb hydrogen through a process known as physisorption, in which hydrogen molecules are drawn to the surface of the nanoparticles by weak Vander Waals interactions. By adding metal nanoparticles to the surface, MgO nanoparticles hydrogen absorption and emission kinetics will be enhanced.
One of the best fillers to enhance the efficacy of polymer electrolytes, particularly the thermal stability behavior, is nano MgO filler. For functional Na-ion batteries, hard carbon is considered to be the most reliable alternative electrode materials because it possesses completely regulated characteristics regarding working potential, reversible capacity, duration of cycle, and available resources. 106 One of the methods for producing porous carbon with controllable internal pore size and distribution is known as template synthesis, which uses inorganic materials like MgO, 107 zeolites, 108 and silica 109 as templates. Within these layouts, the MgO-format blend has appealing points, including that MgO can be digested by using a reduced acidic solution. In addition, there are additional ways of obtaining nano-sized MgO in the carbon framework; for example, pyrolysis of natural mixtures containing explicit Mg can provide nano-sized MgO in the carbon structure, and the acidic unloading of MgO particles can lead to the growth of porosity in carbon. In this manner it is possible to control the size and number of MgO-format pores on the basis of that part of the unrefined components containing Mg. In the current review, while using MgO-layout method and the post-heat treatment at 1000 °C–15008°C are distinctly different, another combining of hard carbon powder materials which subsequently yields very high reversible Na storage capacity over 450 mAh g-1 is demonstrated. Stacked spongy nanocomposites based on carbon (C/MgO), electrodes that are negative were produced to manufacture a high-performance hybrid capacitor battery, and MgO nanoparticles acted as a reservoir for OH ions from the electrolyte. In this context, MgO served as a functional component within the layered, permeable carbonaceous composites, C/MgO, due to its reduced planned surface area and sufficient dynamic sites, which contribute to a specific level of capacitance, despite its elevated SSA and desired pore structure. 110
MgO used as a biosensor
Because of its extensive surface area, electrochemical reactions at the nanoscale are essential for developing various biosensors that can detect extremely small amounts of chemicals. These reactions show significant activity, catalytic properties, and also have a high capacity for absorption, which is useful for attaching enzymes to surfaces. 111 Electrodes called MgO-NPs use the catalase enzyme to detect hydrogen peroxide. Catalyzed enzymes are used to break down hydrogen peroxide, and MgO-NPs are utilized as an electrochemical transducer to aid in accelerating the transmission of electrons. 112 Incorporating synthesized MgO-NPs, using the process of thermal evaporation into gold electrode generates a second biosensor that can identify hydrogen peroxide in hydroquinone as a mediator. High sensitivity and quick reaction are two characteristics of this biosensor. 113 Another option is the nonenzymatic sensor, which is designed to address the issues of low repeatability and prolonged operation. Nanoporous MgO which is environmentally safe and has strong electro-oxidation activity against hydrogen peroxide is one of the non-enzymatic sensors. Since there are no potential health dangers, it can also be used to find hydrogen peroxide in food. 114 MgO nano-belts, which are very responsive to vitamin C, are used in another type of biosensor. 115 The biomolecule of MgO-NPs might be used as a superior electrochemical biosensor material for identifying nucleic acid molecules because of its biocompatibility, high sensitivity, and effective surface area. 116 Figure 18 is showing the UV-vis spectra of different dye solutions at initial and residual concentrations.
MgO nanoparticles as catalysts
Magnesium oxide (MgO) nanoparticles are widely recognized as efficient catalysts due to their high surface area, strong basicity, and thermal stability. These nanoscale materials provide enhanced catalytic activity by offering abundant active sites and facilitating faster reaction rates. Their strong basic nature makes them ideal for base-catalyzed reactions, such as transesterification for biodiesel production and aldol condensation in organic synthesis. The textile and leather industries generate substantial wastewater laden with chemicals and pollutants from various processing stages. In their study Amr Fouda et al use of MgO nanoparticle-coated textiles as catalysts for treating wastewater from crude textile and tannery effluents. MgO nanoparticles (NPs), synthesized using a green method with the novel Aspergillus carbonarious strain D-1, exhibit effective catalytic and adsorptive properties. Key parameters for the biosynthesis of these NPs such as incubation period, contact time, pH, temperature, and precursor concentration were optimized. The NPs synthesized under optimal conditions were characterized using UV–visible spectroscopy, TEM, XRD, DLS, and FT-IR Spectroscopy to confirm their structure and efficacy. In this study, MgO-coated textiles demonstrated impressive catalytic activity, especially in decolorizing textile and tannery wastewater. A concentration of 100 mg of MgO-coated textiles achieved optimal decolorization, with an effective treatment time of 4 h. Under these conditions, the chemical oxygen demand (COD), pH, total dissolved solids (TDS), total suspended solids (TSS), and conductivity were significantly reduced, compared to untreated controls. Furthermore, GC–MS analysis showed that major pollutant compounds in the textile wastewater were diminished, transformed, or completely degraded following treatment. MgO-coated textiles proved highly efficient in reducing the COD of tannery wastewater by 97.5%. Additionally, the MgO NPs exhibited substantial capacity for removing heavy metals, reducing chromium (Cr) levels by 87.06%, with notable removal of other contaminants such as lead (Pb), nickel (Ni), and cadmium (Cd) compared to α-Fe₂O₃ NPs, which were also tested. These findings demonstrate that MgO-coated textiles can serve as effective, reusable catalysts in wastewater treatment, particularly in reducing chemical pollutants and heavy metals, thus supporting cleaner production processes in the textile and leather industries. Figure 19 is showing the use of MgO-coated textiles that serve as effective, reusable catalysts in wastewater treatment.. 117
Furthermore, both types of catalysis frequently employ magnesium oxide. Because of their potency in accelerating the transesterification of crude oil into biodiesel, magnesium oxide catalysts are used. The photo catalytic efficiency and calcination of MgO nanoparticles were found to be significantly influenced by thermal treatment. 118 At a reaction temperature, magnesium oxide nanoparticles exhibited impressive catalytic activity with a high conversion rate. Magnesium oxide nanoparticles were easier to work with and showed catalytic activity that was on par with or better than that of KOH and NaOH. MgO is also frequently applied in photo-catalysis for a various catalyst processes such as benzylation of aromatics, production of aromatic molecules and their derivatives, removal of halogen atoms from hydrocarbon molecules, the addition of oxygen to alkenes, and the removal of water from alcohols, 119 dehydrohalogenation of halogenated hydrocarbons, 120 production of pyranopyrazole and its derivatives, 121 and epoxidation of alkenes. 122 Magnesium oxide is also exploited as a precursor for Wittig reactions, 123 Cyanosilylation and Aldol. 124
Applications in food and storage and packaging
Magnesium oxide (MgO)-coated textiles have gained significant attention in active food packaging due to their strong antimicrobial properties. This study focused on enhancing the effectiveness of MgO coatings by increasing surface oxygen vacancies, a technique that amplifies their antimicrobial capabilities. Previous strategies for improving MgO performance in packaging applications have often struggled to outperform commercial options, and questions remain about whether such improvements persist when MgO-coated textiles are incorporated into biodegradable polymers. In a research work, MgO-coated textiles were developed using a urea-based hydrothermal method, resulting in coatings with enriched surface defects and crystal facets. These defect-rich MgO-coated textiles released higher levels of superoxide anions, which boosted their antibacterial effects. Compared to commercial 27 nm MgO NPs, the newly developed textiles demonstrated superior antimicrobial activity against Escherichia coli and Staphylococcus aureus. Among the samples, textiles treated at 650°C in a nitrogen environment showed particularly high antimicrobial performance, especially against S. aureus. This optimal MgO-coated textile was then incorporated into a cellulose acetate (CA) matrix to create an 8% MgO-NPs coated CA composite. This composite displayed increased antibacterial activity against S. aureus compared to the commercial standard while maintaining similar effectiveness against E. coli.
The CA-MgO-coated textile composite also exhibited a 27% improvement in tensile strength and a 29.5% increase in water vapor transmission rate (WVTR) compared to pristine CA. Although it did not surpass the commercial composite in terms of overall tensile strength, thermal stability, or barrier properties, the study suggests that the developed MgO-coated textiles hold promise for sustainable active food packaging. By combining antimicrobial effects, increased durability, and water vapor resistance, these materials could serve as biodegradable packaging solutions designed to extend food freshness and shelf life. 125 In another study, comprehensive review was carried out for the synthesis methods and physicochemical properties of MgO-NPs, along with their effects on critical BFPF properties, such as water solubility, mechanical strength, water vapor transmission rate, UV-blocking capacity, thermal stability, and antibacterial activity. The addition of MgO-coated textiles to BFPFs offers significant advantages, extending the shelf life of fresh foods and preserving sensory quality. Given MgO has excellent biocompatibility with natural polymer matrices; it is extensively used not only in the food industry but also in pharmaceuticals, semiconductors, and electronics. For food packaging applications, MgO serves as a functional additive, interacting with the BFPF matrix to enhance its functional properties. Recent research highlights the efficacy of MgO-coated textiles in a variety of polymer matrices, including chitosan, gelatin, alginate, carboxymethylcellulose, and starch.
The incorporation of MgO in BFPF matrices not only strengthens physical and mechanical properties and antimicrobial capabilities but also acts as a robust antibacterial agent. This feature enhances the bioavailability and controlled release of active ingredients, significantly extending the shelf life and maintaining the sensory quality of fresh foods. MgO-coated textiles for food preservation applications have received increasing attention in recent years due to their remarkable antibacterial effects, which contribute to prolonged shelf life and high-quality food storage. This study outlines how embedding MgO-NPs into BFPFs affects mechanical and barrier properties, with a substrate-dependent impact observed. Additionally, the study identifies an optimal concentration range (1%–5%) for MgO-NPs to maximize these benefits. 126 Methods for food preservation that use antimicrobial packaging give the product superior barrier properties. 127 MgO-NPs have a chance to be used as a natural supplement to increase the shelf life of food goods by preventing growth of the bacteria, fungi, and other bacteria because of their antibacterial qualities. As per the results of Eghbalian et al. 128 fresh salmon steaks can now be preserved in refrigerators for longer periods of time thanks to antibacterial films constructed of sodium caseinate-gelatin nanofiber rugs containing MgO-NPs and Mentha spicata, which render E. coli and anthraquinone inactive, according to a study. 129 In another experiment, MgO was shown to be the most efficient at removing bacteria. 130 Because of their potent antibacterial action, heat tolerance, non-toxicity, electrical insulation, UV shielding capacity, and photocatalytic qualities, 131 ntriguingly, MgO NPs have also been used as an inorganic material in developing of the packaging of the product that is active. Different bacteria from foodborne diseases, mostly Shewanella baltica, E. coli, S. aureus, and L. monocytogenes, 132 have demonstrated MgO-NP inhibition. The el of bactera may die as a result of the Mg2+ ions released from MgO NPs damaging the composition and thickness of the bacterial wall layers. 128 Another study emphasized on the determination of the effects of MgO nisin, and heat on the biocidal activity of the milk medium. 133 By employing the solvent casting method to strengthen MgO-NPs in polylactic acid (PLA) biopolymer, this study seeks to produce biofilms. The main mechanical, antibacterial and thermal properties of MgO nanoparticles (up to 4 weight percent) strengthened in PLA biopolymer were examined in this work for food packaging applications. After a 12-h treatment, the 2 weight percent reinforced PLA films among the manufactured biocomposite films demonstrated increasing damage and the mortality of about 46% of the E. coli bacterial culture. Figure 20 presents various types of biofilms developed using magnesium oxide nanoparticles (MgO NPs).
Lotted stress-strain curves allowed for the determination of important mechanical parameters for the bio-composite films, including elongation at break (EAB), elastic modulus (EM), and tensile strength (TS). Figure 21 shows the mean and range of the property values in addition to the typical stress-strain correlations of the films. Schematic diagram of MgO-coated textiles that serve as effective, reusable catalysts in wastewater treatment.
117
various kinds of biofilms that have been prepared: PLA/MgO biofilms (P4), 1 wt%, 2 wt%, 3 wt%, and neat PLA (P0). [Chetan Swaroop], [Polylactic acid biofilms reinforced with nano-magnesium oxide for use in food packaging], reprinted with permission.]; published by [ScienceDirect], [2018].
134
(a) Typical stress-strain curves for the biocomposite films that were made and (b) Tensile properties of prepared films. Reprint with permission from [Chetan Swaroop], [Nano-Magnesium Oxide Covered Polylactic Acid Biofilms for Food Packaging]; published by [ScienceDirect], [2018].
134
Regression analysis of weight gain (g) against the dietary MgO-NP for M. rosenbergii. These points are the mean of triplicates averaging for each of the treatments. The breakpoint did not appear in this polynomial regression analysis, so the optimal requirement of MgO-NPs in the diet is more than 500 mg kg−1. Reprint with permission from [Veeran Srinivasan], [Dietary Supplementation of Magnesium Oxide (MgO) Nanoparticles for Better Survival and Growth of the Freshwater Prawn Macrobrachium rosenbergii Post-larvae]; published by [NIH], [2016].
136
Different applications of MgO nanoparticles. Reprint with permission from [Heba Mohamed Fahmy], [Review on MgO nanoparticles multifunctional role in the biomedical field: Properties and applications]; published by [Nanomedicine Journal], [2022].
137





The 2 weight percent produced films had the highest EAB and the strongest tensile strength among all of the films, which is consistent with the NPs’ even dispersion inside the PLA matrix. Other investigations also indicated improvements in tensile characteristics along similar lines.
As a flavor enhancer and nutrient supplement
MgO-NPs can improve the flavor of nutrient products. The food products with nano-texturing are said to have enhanced texture, taste, and general acceptability. 135 Moreover, food goods include MgO-NPs as a dietary supplement. Magnesium is a core element for the body, and adding MgO-NPs to food items can help people consume more of it. Vanillin is used as a flavoring and in fragrance manufacturing to mask or cover up disagreeable tastes or odors in pharmaceuticals, animal feed, and cleaning supplies. The efficacy of the dietary MgO-NPs supplementary diet was evaluated in the experimental Macrobrachium rosenbergii post-larvae (PL) using a research study. MgO-NPs were added at 0, 100, 200, 300, 400 and 500 ppm to the basal diet comprising 0.95 g Mg kg⁻1; the total Mg in diets supplemented with MgO-NPs was also enhanced (1.07, 1.15, 1.24, 1.37 and 1.46 g Mg kg⁻1 respectively). The fish, M. rosenbergii PL (initial weight 0. 11 ± 0. 04 g) which was acclimatised to the experimental condition were fed with MgO-NPs supplemented diets for a period of 90 days. Improved outcomes in terms of survival, development, activities of protease, amylase, and lipase, as well as the levels and compositions of total protein, amino acids, carbohydrates, and lipids, including those of proteins, amino acids, fatty acids, and haemocytes, were observed in M. rosenbergii PL as a result of supplementing with 100–500 mg kg−1 of MgO-NPs over the control group. Figure 22 is showing the regression analysis of weight gain against the dietary MgO-NPs. Moreover, Figure 23 illustrates the primary application branches of MgO-NPs coated textiles, highlighting their potential in food packaging and various other multidisciplinary domains.
Challenges and future prospects
Major challenges in green synthesis of MgO-NPs include its reproducibility, toxicity, stability and various characterizations. The natural extracts, which generally vary in composition and efficacy, are often employed in the synthesis of MgO-NPs through green processes. Data comparison could be hardly accurate due to the variability in the size, shape and other properties of the produced nanoparticles. This green synthesis is mostly done in small quantities, which might not be feasible for mass production. It is extremely difficult to develop scale-able green production techniques for them. MgO-NPs made using traditional methods may be less stable and more prone to aggregation and deterioration over time. It is crucial to create methods to increase the stability of nanoparticles. Also, green-synthesized MgO-NPs can have complex and heterogeneous structures, making it difficult to accurately describe their size, shape, and other characteristics. Moreover, it is always necessary to assess the toxicity and biocompatibility of these nanoparticles. Understanding the possible effects of MgO-NPs made through green synthesis will require more study. However, they have encountered a number of constraints on the way, the green production of MgO-NPs has great prospects in the future. There are some advantages of green synthesis of MgO nanoparticles over the conventional methods normally used. MgO nanoparticles have potential applications in numerous fields such as catalytic application, medical and biological application, application in cleaning up pollutants, batteries and energy, and many other. In case of organic synthesis and environmental catalysis, the results of catalytic activity of MgO-NPs in various chemical reactions have been demonstrated. They can be employed in the ability of heterogeneous catalysis of organic synthesis, green chemistry, and environmental remediation. These nanoparticles can be useful for tissue engineering, imaging, cancer therapy and drug delivery. It has been realized that MgO nanoparticles demonstrate high adsorption capacity for both the heavy metals and the organic contaminants. The use of MgO nanoparticles in soil remediation, air purification, and water treatment are potential future applications in environmental remediation. Such nanoparticles may be used in agriculture to increase soil fertility and plant growth, among other things. Their use as fertilizers and insecticides in agriculture has the latent to reduce the use of hazardous chemicals and advance sustainable agricultural practices. Furthermore, the use of MgO-NPs in energy storage devices like batteries and super capacitors has been researched. They have the prospective to be used in the creation of high-performance, ecologically approachable energy storage technologies in the future. MgO nanoparticles have a bright future in a variety of applications, but further study is required to fully realize their promise in these areas.
Conclusion
Magnesium oxide nanoparticles (MgO-NPs) can be produced in an ecologically approachable manner as an alternative to conventional processes that use toxic chemicals and produce damaging byproducts. In this study, green-synthesized MgO-NPs particularly those from bacteria, algae and fungi were investigated along with their novel applications. That is why these particles are considered as more effective than other metal oxide nanoparticles in view of their high possibilities to be degraded, high catalytic activity, antimicrobial characteristics, stability and effectiveness in wastewater treatment. Without doubt, MgO-NPs are definitely be promising candidate for the nanomedicine because of their biocompatibility and tunable properties. Thus, they enhance the quality and safety of foods and can even be employed in the formation of food packaging materials. On the contrary, the green synthesis presents an opportunity to achieve the development of stable non-toxic NPs for several applications. However, there are some disadvantages of green synthesis that may still be a concern including the matter of reproducing the technology and the others are toxicity effects. However, due to the need for more sustainable and environment friendly processes, the concepts of green chemistry and the application of green strategies for the preparation of different NPs are evolving. Also, further research is required along with increased industrial, governmental, and academic cooperation to ensure the responsible synthesis and long-term usage of these particles in different areas.
Footnotes
Acknowledgments
The authors are thankful to the Technical University of Liberec Czech Republic and Riphah international University Faisalabad Campus for providing their guidance to compile this novel review for PhD work.
Authors contribution
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
