Abstract
Due to the shortcomings of traditional medical dressings, the development of new dressings has become a hot topic. However, the mechanical properties of hydrogel fibers are relatively poor, so they are rarely spun and knitted, and tend to be in the state of sheet gels or laminated with fabrics as dressings. In this study, a novel fiber-type medical dressing with excellent biocompatibility, pH-monitoring function, drug slow-release effect, and fluid-absorbing ability was created using sodium alginate (SA), polyvinyl alcohol (PVA), and curcumin (CUR) based on wet spinning technology and weft-knitting technique. The X-ray diffraction and the tensile tests demonstrated that PVA could enhance the crystallinity and the tensile properties of the fiber, facilitating the formation of fabrics. In addition, CUR gave the dressing the ability to monitor wound pH in real-time. Moreover, the tests indicated that the SA/PVA/CUR hydrogel fiber fabrics studied in this paper have good swelling properties, drug retardation function, air permeability, moisture permeability, and bio-tissue compatibility, thus demonstrating the potential of this SA/PVA/CUR hydrogel fiber fabrics as medical dressings.
Introduction
When a wound is formed by skin injury, the medical dressing can replace the skin of the wound during the healing period, preventing the wound from exposure leading to bacterial infections, and it can absorb the wound exudate in time to promote hemostasis, thus providing good conditions for the repair of the skin wound. 1 Traditional dressings such as bandages, gauze, Band-Aids, etc. Are inexpensive and have the above functions, but still have limitations such as poor liquid absorption, insufficient antimicrobial properties, and adherence to the wound, resulting in secondary damage when replacing the dressing. 2 In the face of these limitations, new wound dressings have emerged with the emergence of new materials and technological advances. Hydrogel is a hydrophilic polymer with a 3D network structure, which is soft, elastic, and excellent in water absorption and biocompatibility. 3 Generally, its physical and chemical properties are similar to those of natural extracellular matrixes. 4 Compared with traditional dressings, hydrogel dressings, as a new type of medical dressing, are characterized by high fluid absorption and high moisture retention, 5 so they can rapidly absorb wound exudate and provide a moist environment.
Sodium alginate (SA) is a linear anionic polysaccharide mainly derived from the cells walls of brown seaweeds. It is highly valued in the field of wound dressings 6 and drug delivery 7 due to its biocompatible, biodegradable, and hydrophilic properties. In the presence of divalent cations (e.g., calcium ions), SA undergoes a solution-gel transition to form a stable and elastic gel structure. 8 Due to the presence of hydrophilic groups and network structure, this hydrogel can absorb wound exudate and form a moist environment that temporarily replaces the skin as a barrier to avoid bacterial infection, thus promoting wound re-epithelialization. 6 In addition to the advantages in wound healing, SA hydrogels also show promise in drug delivery. This is because SA is able to encapsulate drugs, biomolecules, or cells during the sol-gel conversion process, resulting in a drug delivery system. 7 For example, Karimi et al. used the properties of SA to prepare hydrogel scaffolds for the delivery of the drug Daphne mucronate (DM), accelerating skin regeneration. 9 However, SA hydrogels have lower mechanical strength and stability and weaker antimicrobial properties, so a large number of studies have been conducted to combine SA with other polymers to optimize their properties. Gholivand et al. developed a sodium alginate composite hydrogel filled with cross-linked chitosan to improve the mechanical properties, and the Cu (II) complex provided antimicrobial activity. 10
Polyvinyl alcohol (PVA), a widely used synthetic water-soluble polymer, can be obtained by polymerization and alcoholysis of vinyl acetate, which has excellent biocompatibility, mechanical properties, and moisture absorption.11,12 Since PVA hydrogel is similar to natural tissue, 13 it is a proper medical material for preparing a wound dressing, too. Zhang et al. developed a hydrogel dressing with self-healing, strong stretching, adhesion, and photothermal antibacterial activity from polyvinyl alcohol, borax, tannic acid (TA), and ferric chloride (III) for joint skin wounds. 14
The pH of the skin wound microenvironment can reflect the condition of the wound. Normal skin tissue is acidic, with a pH of 4-6. Bacterial colonies proliferate and produce alkaline by-products, 15 and wounds that have lost their skin barrier are alkaline, with a pH generally greater than 7. Therefore, monitoring changes in the pH of the microenvironment of wounds can help to identify difficult-to-heal wounds, and can be informative for medical diagnosis and prescription use. 16 Curcumin (CUR) is a natural diketone compound extracted from turmeric. In addition to its antibacterial and anti-inflammatory effects, curcumin is also a pH-sensitive substance. As an interconvertible isomeric compound, curcumin exists mainly in the form of enol in acidic and neutral media in yellow color, while it exists mainly in the form of ketone in alkaline conditions in reddish-brown color, so it can be used as a pH indicator. 17 Using this property of curcumin and loading it into hydrogels, hydrogel dressings with pH monitoring can be prepared. Changes in the pH value of the wound environment will cause the color of the dressing to change, and the colorimetric method is used to determine the pH value of the wound, thereby realizing real-time monitoring of wound pH, which is conducive to determining the status of the wound and avoiding bacterial infections caused by unnecessary dressing changes. In the study of Arafa et al., curcuma longa extract was used as a pH sensitive indicator loaded in a hydrogel with a hydroxyethyl cellulose base grafted with itaconic acid, resulting in the preparation of transparent and soft pH sensitive wound dressings capable of showing significant color changes at different pH. 17
In this study, sodium alginate and polyvinyl alcohol were used as hydrogel substrates, and curcumin was used as a drug and pH monitoring agent. SA/PVA/CUR hydrogel fibers were prepared by the wet spinning technique, and the tensile properties of hydrogel fibers were improved by adjusting the content of PVA. Then the morphology structure (SEM), chemical composition (FTIR), crystallinity (XRD), swelling properties, in vitro drug release properties, and pH sensitivity of the hydrogel fibers were investigated. Finally, the hydrogel fibers were prepared into knitted fabrics using the weft-knitted technique, and their structural parameters, air permeability, moisture permeability, and biocompatibility were investigated and compared with three commercially available medical dressings, which demonstrated the potential of this SA/PVA/CUR hydrogel fiber fabric as a medical dressing.
Materials and methods
Materials
The PVA (formula (-CH2CHOH-)
Preparation of spinning fluid and coagulating bath
Series of concentrations of SA solution (1.5, 2, 2.5, three and 3.5 wt%), SA/PVA solution (SA 2.5 wt%, PVA accounted for 30, 40, 50, 60, or 70 wt% of the mass of SA, respectively) and SA/PVA/CUR solution (SA 2.5 wt%, PVA 1.5 wt%, CUR concentration of 15 mg/100 mL) were configured: weighing a certain amount of the corresponding raw materials into a beaker, adding deionized water, and placing the beaker on a magnetic stirrer (Rapid JB-6, Xiamen Rapid Co. Ltd, China) to stir until complete dissolution.
As mentioned earlier, sodium alginate cross-links with calcium ions to form a network structure; therefore, calcium chloride was chosen as the coagulation bath in this paper. 5 wt% CaCl2 solution to be used as a coagulating bath for sodium alginate was configured: weighing 40 g of CaCl2 powder into a beaker, adding deionized water to a volume of 800 mL, and placing it on a magnetic stirrer and stirring until completely dissolved.
Preparation of hydrogel fibers and fabrics
Wet spinning using fluid dynamics.
18
The spinning fluid prepared as above was left to defoam for 12 h. The coagulation bath was poured into the spinning collection platform. A 5 mL syringe and a spinning head with an inner diameter of 0.84 mm were used. The injection rate was set at 0.1 mL/s, the height of the spinning head from the liquid surface of the coagulation bath was 15 mm, and the rotational speed of the spinning collection platform was 8 r/min. The spinning fluid was transferred to the syringe, and the parameters were set to carry out wet spinning using the smart syringe pump controller (XMSP-2C, HeiMai Nano Technology Co. Ltd, China), as shown in Figure 1. Then, an appropriate amount of dried hydrogel fibers was taken and knitted using the weft-knitted technique to form plain stitch weft-knitted fabrics (Figure 1). Specifically, the hydrogel fibers were placed on the knitting needles in the weft direction in a sequential order to form loops and were strung over each other in the longitudinal direction to form the knitted fabric. Plain stitch fabric is material-saving, thin, soft and porous, thus reducing costs, adapting to the curved surface of the skin and aiding gas exchange at the wound site. Schematic diagram of the preparation process of hydrogel fiber fabrics (By Figdraw).
Characterizations
Morphology observation
The surface morphology of the hydrogel fibers was observed under an optical microscope (ECLIPSE E100, Hitachi High-Tech Corporation, Japan) at four times magnification. The morphological analysis for the hydrogel fibers was performed a field emission scanning electron microscopy (FESEM) (SU8100, Hitachi High-Tech Corporation, Japan). The samples were adhered to the electron microscope stage with conductive adhesive and were coated with gold.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared spectroscopy (FTIR) of the hydrogel fibers was acquired using a spectrometer (Nicolet is10, Thermo Fisher Scientific (China) Co., Ltd, USA) over the range of 500-4000 cm−1, The scanning resolution is 0.05 cm−1. 19
X-ray diffraction (XRD)
The hydrogel fibers were analyzed using an X-ray diffractometer (D2 PHASER A26-X1-A2E0B2A0, Bruker Corporation, Germany) to obtain X-ray diffraction spectra, which were used to analyze the improvement of the crystallinity of the hydrogel fibers by the addition of PVA.
Rheological properties test of spinning fluid
The higher the mass fraction of SA or PVA in the spinning fluid, the higher the viscosity of the spinning fluid and the better the mechanical properties of the fibers will be in wet spinning, 20 but at the same time, the spinning difficulty will increase. Hence it is necessary to test the rheological properties before spinning using a Physica MCR301 rotational rheometer (Anton Paar GmbH, Austria). At a constant temperature of 25°C, the spinning fluid was placed on the test bench, and the shear rate was varied linearly in the range of 1-100 s−1, to detect the variation rules of viscosity (mPa·s) and shear stress (Pa) of different components of the spinning fluid, and to explore the effects of SA and PVA content on the rheological properties of the spinning fluid.
Tensile properties
The hydrogel fibers were tested for tensile properties at room temperature using a yarn strength extensometer (XL-2, Shanghai New Fiber Instrument Co., Ltd, China) with a clamping distance of 250 mm and a tensile rate of 1000 mm/min, and were stretched until the samples broke. Each sample was tested five times and the average value was taken.
Swelling property
In order to investigate the ability of hydrogel fibers to absorb wound exudate, the swelling properties of the hydrogel fibers were tested by gravimetric method
21
at 37°C. Several prepared dried hydrogel fibers were weighed and the mass was recorded as 
In vitro drug release studies
The release of CUR was performed in PBS at pH 7.4 in vitro.
22
50 mL of spinning solution (2.5% SA, 1.5% PVA) was mixed with 7.5 mg, 15 mg, and 30 mg of curcumin, respectively. Wet spinning was carried out as previously described and dried. A number of hydrogel fibers of the same mass (0.1 mg) were taken into a reagent bottle separately and a total of 20 mL of a 1:1 mixture of PBS and 75% ethanol was added. The reagent bottles were placed in a thermostatic oscillator at 37°C and the frequency of oscillation was 100 r/min 400 μL of the released solution was taken out with a pipette and the same volume of solution was replenished at 1, 2, 4, 8, 12, 24, 36 h. The absorbance of taking out the released solution at 420 nm was measured by using a multimode microplate reader (Synery H1, Bioteck, America) each time. The concentration of curcumin was calculated from the standard curcumin concentration curve, and then the cumulative curcumin release rate (
pH-sensitive studies
pH sensitivity of curcumin solution: 0.5 mg/mL of curcumin solution was prepared by mixing curcumin powder with PEG solution. The curcumin solution was mixed with a series of pH buffer solutions (pH 4-8) at a ratio of 1:10 and the color change was observed to compare the color of the curcumin solution at different pH values.
pH sensitivity of SA/PVA/CUR hydrogel fibers: Submerge the CUR-carrying hydrogel fibers in different pH buffer solutions and observe the color change of the fibers to compare the color of the CUR-carrying hydrogel fibers at different pH values.
Air permeability tests of fabrics
Considering the patient’s comfort with use, the dressing needs to have a certain degree of breathability. Hydrogel fiber fabrics and three kinds of commercially available dressings (medical gauze dressing, sterile wound dressing, hydrocolloid dressing) were tested and compared for breathability. Referring to the standard GB/T 5453-1997, the air permeability test of each dressing was carried out under the condition of area 20 cm2 and test pressure difference 100 Pa by using a fully automatu permeability instrument (YG461E-III, Ningbo Textile Instrument Factory, China). Each sample was repeated 10 times with a different position each time, and the average value was taken.
Moisture permeability tests of fabrics
Previous studies have shown that dressings with suitable moisture permeability properties facilitate the proliferation of epidermal cells and fibroblasts.
20
The moisture permeability of the fabrics was tested using the cup method.
23
An appropriate amount of distilled water was taken and filled into the moisture-permeable cup. The sample was placed face down, fitted with a gasket and a compression ring, taped on the side, and then placed into a computerized fabric permeability tester. 1 h later, the mass of the test set was weighed to obtain the mass of the test set at equilibrium, and then the weighing operation was repeated after 1 h to obtain the mass of the test set after evaporation, and the difference between the two masses was 
Cytotoxicity assay
Leachate preparation: SA, SA/PVA, and SA/PVA/CUR hydrogel fiber fabrics were impregnated into a complete medium (ratio of 0.1 g/mL) containing 89% high sugar Dulbecco’s modified Eagle medium (DMEM), 1% penicillin-streptomycin and 10% fetal bovine serum (FBS), respectively. Bacterial filtration was performed after 24 h of maceration to obtain the three leachates.
Cell viability assay 24 : The cryopreserved Calcein-AM solution and PI solution were taken out and placed in a room temperature environment for 30 min. Calcein-AM solution and PI solution were added to PBS buffer (final concentration of Calcein-AM was 4 μmol/L and the final concentration of PI was 6 μmol/L) and mixed thoroughly to make staining solution. L929 mouse fibroblasts were inoculated into 96-well plates, and the cells were allowed to incubate in a culture medium for 24 h until the cells were adherent to the wall. The culture solution in the wells was aspirated and SA, SA/PVA, SA/PVA/CUR leaching solution, and control culture solution were added. Each group was repeated three times, and the incubation was continued at 37°C. After 24 h, the leachate in the wells was aspirated, and the cells were washed with PBS once. After 24 h, the cells were washed with PBS for one time, then the PBS buffer was aspirated and the appropriate volume of Calcein-AM/PI staining solution (100 μL per well of 96-well plate) was added, and the cells were incubated at 37°C for 30 min, protected from light. The staining effect was observed under an inverted microscope (ECLIPSE Ti2, Nikon Instruments Inc, China): firstly, the live cells with yellowish green color were observed by excitation at 490 ± 10 nm; then, the dead cells with red color were observed by excitation at 545 nm. The obtained images were counted on Image J software for live/dead cells counting, quantitatively analyzed to obtain the number of live/dead cells, and calculated to obtain the cells survival rate.
Results and discussion
Surface morphology analysis
The surface morphology of SA, SA/PVA, and SA/PVA/CUR hydrogel fibers under optical microscope is shown in Figure 2(a)–(c). The average diameters of SA, SA/PVA and SA/PVA/CUR hydrogel fibers were 75.8 μm, 126.3 μm and 131.5 μm, respectively, and it can be found that the surface of SA/PVA hydrogel fibers was more saturated and smoother than SA hydrogel fibers, and the diameters were obviously larger than those of SA hydrogel fibers. The smoothness of SA/PVA and SA/PVA/CUR hydrogel fibers had similar smoothness but significant difference in color due to the incorporation of curcumin. (a1) Optical microscope image of the SA hydrogel fibers. (a2) FESEM image of the SA hydrogel fibers. (b1) Optical microscope image of the SA/PVA hydrogel fibers. (b2) FESEM image of the SA/PVA hydrogel fibers. (c1) Optical microscope image of the SA/PVA/CUR hydrogel fibers. (c2) FESEM image of the SA/PVA/CUR hydrogel fibers. (d) FTIR spectra. (e) XRD spectra of SA and SA/PVA.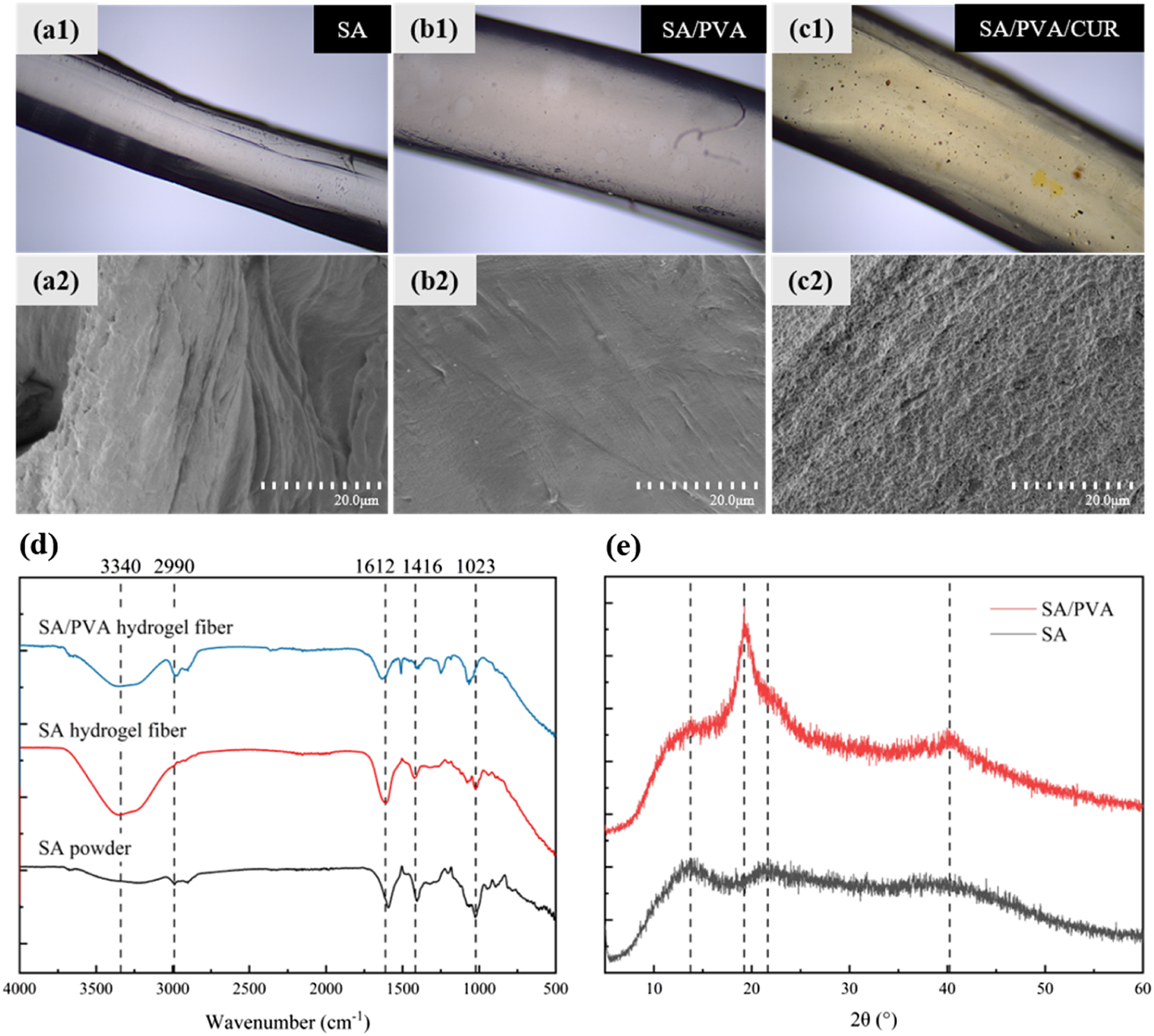
The surface morphology of SA, SA/PVA, and SA/PVA/CUR hydrogel fibers under FESEM is shown in Figure 2(a)–(c). It can be clearly observed that fibers of the SA/PVA hydrogel are less folded than the surface of the SA hydrogel fibers. This is because the holes in the dried SA hydrogel fibers that originally stored water molecules lost their support due to the loss of water and collapsed into folds. In contrast, hydrogen bonds were formed between PVA itself and between PVA and SA in the SA/PVA hydrogel fibers, which could better support the porous structure of the hydrogel fibers. In addition, the addition of PVA increased the hydrophilicity of the hydrogel fibers, and the rate of water loss was not as fast as that of the pure SA hydrogel fibers, so the degree of slumping was lower. The surface of the SA/PVA/CUR hydrogel fibers was rougher than that of the SA/PVA hydrogel fibers, which showed a small and intensive unevenness. Combined with the curcumin particles that can be observed under an optical microscope, the surface of SA/PVA/CUR hydrogel fibers under a scanning electron microscope is rough because it is covered with curcumin particles.
FTIR analysis
The FTIR spectra of SA powder, SA hydrogel fiber, and SA/PVA hydrogel fiber are shown in Figure 2(d). Both SA powder and SA hydrogel fiber had obvious characteristic peaks near 1023 cm−1, 1416 cm−1, and 1612 cm−1, where the vibration peak at 1023 cm−1 was attributed to C-O stretching, and the absorption peaks at 1612 cm−1 and 1416 cm−1 were the symmetric and anti-symmetric stretching vibration peaks of -COO-, respectively. 25 However, there were obvious differences between SA powder and SA hydrogel fibers: SA hydrogel fibers did not have the characteristic peaks of SA powder at 2990 cm−1, which was due to the formation of the mesh structure after cross-linking with Ca2+ so that it made the C-H feature not obvious; compared with SA powder, there was an obvious -OH telescopic vibration peak at 3340 cm−1, which was due to the formation of hydroxyl groups with water molecules in the SA hydrogel fibers. 26 Comparison of the infrared spectra of SA hydrogel and SA/PVA hydrogel revealed that the asymmetric stretching vibration peaks of C-H at 2990 cm−1 of SA/PVA hydrogel fibers 27 were more pronounced compared to those of SA hydrogel fibers and SA powder, which was attributed to the incorporation of PVA that is rich in C-H groups.
XRD analysis
Figure 2(e) exhibits the XRD spectra of SA and SA/PVA. It was analyzed using Jade software and found that SA had no sharp and obvious strong peaks, only diffuse scattering bun peaks at about 12.95°, 21.91°, and 39.92°, showing an amorphous nature 28 ; SA/PVA, on the other hand, showed obvious strengthening peaks at 19.25°and 40.74°, and non-crystalline peaks at 12.36°, 33.40°, and 22.23°, 29 and its crystallinity was calculated to be 29.62%. The degree of crystallinity was calculated to be 29.62%, showing a semi-crystalline state. Therefore, it can be inferred that the addition of PVA is conducive to the improvement of the crystallinity of hydrogel fibers. The higher the crystallinity, the more regular the molecular arrangement in the fiber, and the stronger the intermolecular bonding force, thus improving the mechanical properties of the hydrogel fiber.
Rheological properties analysis of spinning fluid
From Figure 3, it can be found that both SA spinning fluid and SA/PVA spinning fluid exhibit similar patterns under a series of concentration gradients. With the increase of shear rate, the viscosity of the spinning fluid decreases gradually and the shear stress increases gradually, so it can be inferred that both SA and SA/PVA spinning fluids at a series of gradient concentrations belong to the shear-thinning type of fluids. Besides, it can be seen that the viscosity and shear stress of the spinning fluid gradually increase with the increase of the mass fraction of SA and PVA. In general, the viscosity of the spinning fluid increases with the increase of raw material content. It was also found in the wet spinning process that the higher the viscosity of the spinning liquid, the higher the spinning difficulty. Therefore, 2.5 wt% was selected as the mass fraction of SA in the subsequent experiments for comprehensive consideration. (a) Variation of spun fluid viscosity with shear rate for different SA contents. (b) Variation of spun fluid shear stress with the shear rate for different SA contents. (c) Variation of spun fluid viscosity with shear rate for different PVA contents. (d) Variation of spun fluid shear stress with the shear rate for different PVA contents.
Tensile properties analysis
Since the mechanical properties are too poor for the raw material to be knitted, and one of the disadvantages of natural hydrogels is the poor mechanical properties,
30
this study attempted to improve the mechanical properties of hydrogel fibers by adding PVA to SA hydrogel to form a hybrid hydrogel. The tensile properties of SA/PVA hydrogel fibers with different PVA contents are shown in Figure 4. (a) Breaking load-elongation curves of hydrogel fibers with different PVA contents. (b) Breaking load-elongation curves of SA, SA/PVA, and SA/PVA/CUR hydrogel fibers. (c) Breaking load of hydrogel fibers with different PVA contents. (d) Elongation at break of hydrogel fibers with different PVA contents.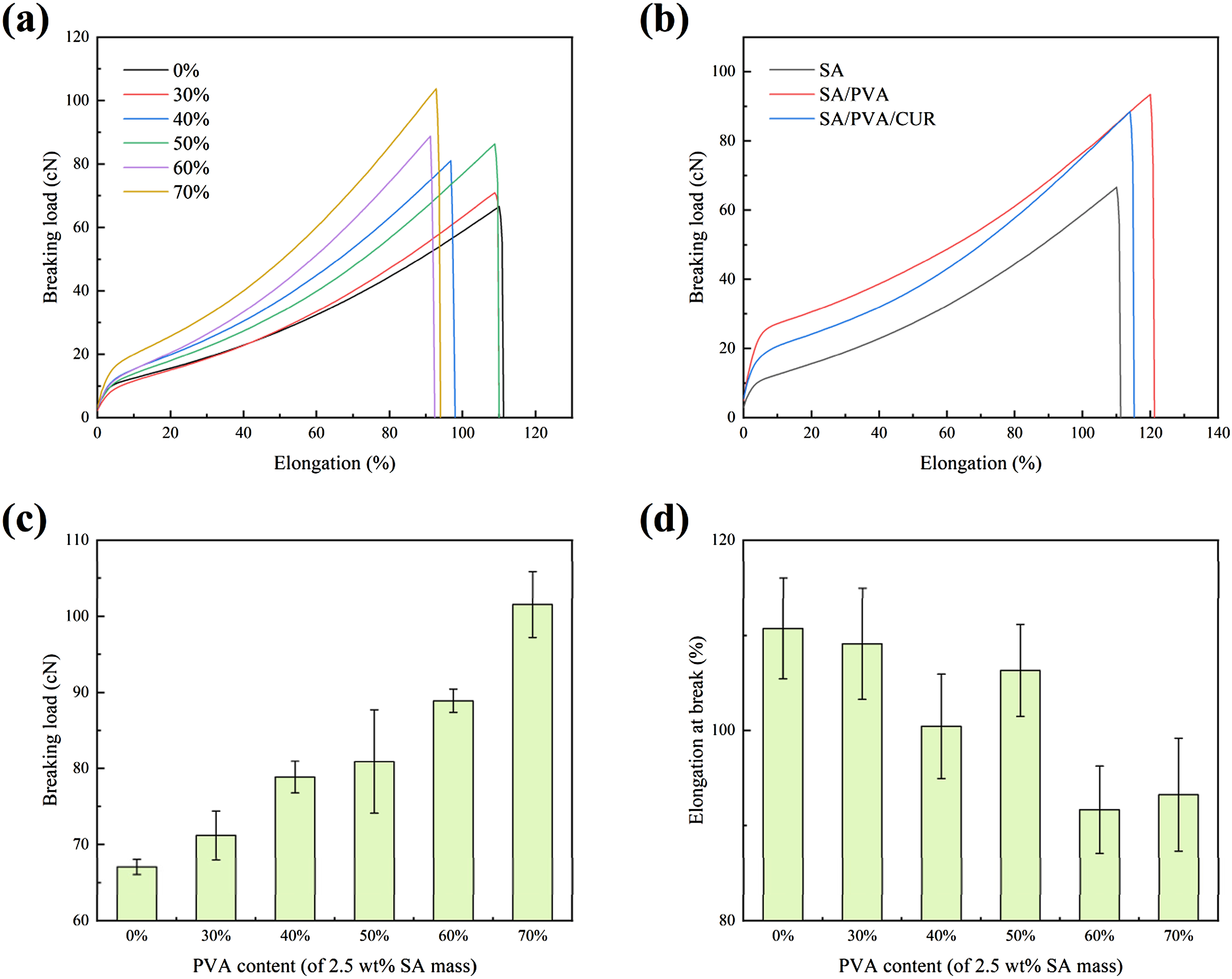
From Figure 4(a), it can be found that regardless of the increase in PVA content, the tensile rupture curve showed a similar pattern: with the increase in elongation, the load first increases rapidly, and then the growth rate of the load becomes slower. This is because at the beginning of stretching, the fiber mainly produces elastic deformation, and as the stretching continues, the plastic deformation of the fiber dominates.
From Figure 4(c), it is obvious that the fracture load of hydrogel fibers increases with the increase of PVA content. That is to say, the addition of PVA is conducive to the increase in the strength of hydrogel fibers and the improvement of their mechanical properties, which is mainly due to the following two reasons: (1) PVA itself has a high degree of polymerization, the molecular chain arrangement is neat, the degree of orientation is high, it is easy to crystallize and it contains a large number of hydroxyl groups that can form hydrogen bonding, so the mechanical properties of fibers added with PVA are improved; (2) PVA and SA both contain hydroxyl groups that can form hydrogen bonding with each other, increasing the intermolecular forces of fibers.
From Figure 4(d), it can be found that the change in PVA content has no obvious effect on the elongation at the break of hydrogel fiber. However, in general, the elongation at break of hydrogel fibers containing a higher content of PVA is lower, because PVA is easy to crystallize, the higher the content of PVA in hydrogel fibers, the higher the crystallinity of the fibers, which makes the breaking strength higher, but also makes the brittleness of the fibers increase and the elongation at break decrease.
Considering that the higher the content of raw materials, the higher the viscosity of spinning liquid and the higher the difficulty of spinning, 2.5 wt% SA and 1.5 wt% PVA (60 wt% of PVA with a mass fraction of 2.5 wt% SA) were subsequently selected for the preparation of hydrogel fibers under the premise of ensuring the feasibility of spinning.
The tensile properties of SA, SA/PVA, and SA/PVA/CUR hydrogel fibers are shown in Figure 4(b). A comparison of the three tensile curves shows that the incorporation of curcumin does not have a significant effect on the tensile properties of the hydrogel fibers compared to the incorporation of PVA. Although the breaking load and elongation at break of the SA/PVA/CUR hydrogel fiber were lower than those of the SA/PVA hydrogel fiber, its tensile properties were still more excellent than those of the SA hydrogel fiber. In the subsequent knitting process, it was also found that the tensile properties of the SA/PVA/CUR hydrogel fibers with this ratio could meet the requirements of the weft-knitted technique.
Swelling property analysis
The swelling property is the main characteristic of hydrogel liquid absorption,
31
the higher the swelling ratio, the more wound exudate can be absorbed by the hydrogel dressing. At the same time, the absorbed water hydrogel dressing can also provide a moist environment for the wound. Figure 5(a) reflected the good swelling performance of SA/PVA/CUR hydrogel fibers, which reached a maximum swelling rate of 266%. From 0 min to 30 min, the hydrogel fiber rapidly absorbed water and the swelling rate increased rapidly, after 120 min, the water absorption rate gradually decreased, and after 1440 min, it was basically unchanged, and the swelling reached equilibrium. The hydrogel fibers had better swelling properties compared to other studies.32–34 (a) The swelling ratio of SA/PVA/CUR hydrogel fiber. (b) Release profile of curcumin-loaded hydrogel fiber. (c) Photographs of curcumin solution at different pH values (4–8). (d) Photographs of hydrogel fibers at different pH values (4–8).
In vitro drug release profile
Figure 5(b) reflects the in vitro drug release profile of SA/PVA/CUR hydrogel fibers containing different concentrations of curcumin. From the figure, it was found that the hydrogel fibers showed a similar drug release pattern irrespective of curcumin concentration. There were two distinct phases of drug release: in the first phase, curcumin was rapidly released in a short period, and in the second phase, the rate of drug release gradually decreased and the release profile leveled off. The duration of drug release could reach 48 h. The cumulative release rate of hydrogel fibers with three curcumin concentrations (15 mg/100 mL, 30 mg/100 mL, 60 mg/100 mL) could reach 83.36%, 88.82%, and 89.56%, respectively. That is to say, it took 2 days to release most of the drug, which proves that the hydrogel fibers have the effect of slow release of the drug. 15 mg/100 mL of the CUR-carrying hydrogel fibers tended to slow down the release curve after 12 h and 30 mg/100 mL and 60 mg/100 mL of the CUR-carrying hydrogel fibers tended to slow down the release curve after 36 h. A comparison of hydrogel fibers with different concentrations of curcumin showed that the higher the drug concentration, the higher the final cumulative release rate and the longer the drug release time. However, the initial burst is higher for lower concentration compared to higher concentration. The change in curcumin content does not affect the change in pore size of hydrogel fibres after water absorption and swelling. Then all three curcumin content hydrogel fibres have similar curcumin release at the same time, so the higher the curcumin content, it has a lower drug release rate instead.
pH-sensitive studies
Figure 5(c) reflected the color of the curcumin solution under different pH conditions. It could be clearly seen that the curcumin solution gradually changed from yellow to orange-red color as the pH increased. The color change was especially evident at pH ≥ 7, which was appropriate for the application of the dressing. During the transformation of the wound to normal skin, the pH changed from alkaline to weakly acidic, and accordingly, the curcumin in the dressing changed from orange-red to yellow. The colorimetric properties of curcumin were thus demonstrated.
Figure 5(d) reflected the color change of SA/PVA/CUR hydrogel fibers under different pH conditions. Similar to the colorimetric properties of curcumin, the hydrogel fibers gradually changed from yellow to brown with the increase of pH, and again the color shift was especially obvious at pH 6-7. The change in color of the dressing can be used by healthcare professionals and patients to determine the status of wound healing. When the dressing is brown, it indicates that the wound is not healing, while a change in the color of the dressing to yellow indicates that the pH of the wound has changed to normal. This allows real-time monitoring of the pH value of the wound, visualizing the wound healing and avoiding unnecessary dressing changes.
Hydrogel fiber fabrics
Figure 6(a) shows photographs of a hydrogel fiber fabric. The hydrogel fiber fabric is a plain weft knitted fabric, which is lightweight and uses less yarn. Table 1 exhibits a comparison of the structural parameters of hydrogel fiber fabrics and other commercially available dressings (medical gauze dressing, sterile wound dressing, hydrocolloid dressing). The hydrogel fiber fabric had the largest gram weight, moderate thickness, and smaller fabric density than woven gauze, and was sparse and porous. (a) Face and back photographs of hydrogel fiber knitted fabrics. (b) Moisture permeability of four dressings. (c) Cell viability of L929 cells in hydrogel fiber fabrics leachate. (d) The fluorescence intensity of L929 cells cultivated with SA、SA/PVA and SA/PVA/CUR hydrogel fiber fabrics. Structural parameters and air permeability of the four dressings.

Air permeability analysis
Table 1 lists a comparison of the air permeability of hydrogel fiber fabrics and other commercially available dressings (medical gauze dressings, nonwoven sterile wound dressings, hydrocolloid dressings). As can be seen, the permeability of hydrogel fiber fabric is closer to that of medical gauze dressing. Because both fabrics have a relatively small density and contain a large number of holes, they have good breathability, which is conducive to vapor exchange between the wound and the surrounding environment. The non-woven sterile wound dressing and hydrogel dressing permeability are much smaller, and poor breathability will affect the patient’s use of comfort.
For knitted fabrics, PT is the number of longitudinal rows of coils on the fabric within 5 cm along the transverse direction, and PW is the number of transverse rows of coils on the fabric within 5 cm along the longitudinal direction. For woven fabrics, PT is the number of warp yarns arranged within 10 cm of the weft direction of the fabric, and PW is the number of weft yarns arranged within 10 cm of the warp direction of the fabric.
Moisture permeability analysis
The moisture permeability of hydrogel fiber fabrics and other commercially available dressings (medical gauze dressings, nonwoven sterile wound dressings, hydrocolloid dressings) is shown in Figure 6(b). Similar to the air permeability rate, the moisture permeability rates of hydrogel fiber fabric and medical gauze dressing are close to each other, and both of them have good moisture permeability properties. On the other hand, nonwoven dressings and hydrocolloid dressings have a tight structure with fewer holes or almost no holes, so their air and moisture permeability is poor, and they are prone to make the human body feel stuffy. Although medical gauze dressing has good air permeability and moisture permeability, it has the limitations of traditional dressings, 35 such as poor absorption of exudate, and the problem of secondary injury caused by replacement. Hydrogel has good water absorption ability and a smooth surface, but its air permeability and moisture permeability are poor. Therefore, it can be said that hydrogel fiber fabric combines the advantages of new dressings and traditional fabrics while having a smooth surface, excellent air permeability, moisture permeability, and exudate absorption ability.
Cell activity
Figure 6(d) reflects the survival status of L929 cells in SA, SA/PVA, and SA/PVA/CUR hydrogel fiber fabric leachate, respectively, where fluorescent green represents live cells and red represents dead cells. It is obvious from the figure that the number of live cells is much larger than that of dead cells in both control, SA, SA/PVA, and SA/PVA/CUR groups. The further quantitative analysis yielded Figure 6(c), which reveals that the mean values of cell survival in the SA group, SA/PVA group, SA/PVA/CUR group, and control group were 94.44%, 94.37%, 96.07% and 96.89%, respectively. According to the current Chinese standard, Biological Evaluation of Medical Devices: GB/T 16886.5-2022, it can be concluded that SA, SA/PVA and SA/PVA/CUR hydrogel fiber fabrics have good biological tissue compatibility and are suitable for use as medical dressings.
Conclusion
In this study, biocompatible SA/PVA/CUR hydrogel fiber fabrics with pH monitoring function were prepared as medical dressings by wet spinning and weft-knitted techniques using sodium alginate and polyvinyl alcohol as raw materials and curcumin as a drug and pH sensitizer. The crystallinity and tensile properties of the hydrogel fibers were improved by adding PVA. The addition of curcumin endowed the dressing with remarkable pH responsiveness, which made real-time monitoring of wound pH possible, and the good hydrophilicity and network structure of SA and PVA enabled the dressing to have excellent liquid absorption ability and slow drug release. Finally, the weft-knit structure facilitates the dressing’s breathability and moisture permeability, which contributes to wound healing. Overall, this study demonstrated the potential of SA/PVACUR hydrogel fiber fabrics as medical dressings. Compared with previous studies, this paper knitted the hydrogel fibres to form a fabric, altering the way the hydrogel was originally moulded to provide more flexibility in controlling the pore size, permeability and thickness of the dressing, and enhancing the designability of the dressing. However, compared with common yarns, the mechanical properties of hydrogel fibers still need to be improved, so there is still a way to go to achieve large-scale commercial production. In the future, the possibility of mass production of hydrogel dressings can be improved by considering more abundant raw materials and more suitable preparation methods.
Footnotes
Acknowledgments
The authors acknowledge the financial support from the National Science Funds of China (52373058), the Key Science Research Project of Wuxi Municipal Bureau of Health (Z202219), the Fundamental Research Funds for the Central Universities (JUSRP62005), and a Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAP).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Funds of China; 52373058, The Key Science Research Project of Wuxi Municipal Bureau of Health; Z202219, the Fundamental Research Funds for the Central Universities; JUSRP62005, Priority Academic Program Development of Jiangsu Higher Education Institutions (PAP).
