Abstract
Lithium ion batteries are one of the most promising electrochemical energy storage systems. They generally consist of four components: anode, cathode, electrolyte, and separator. The separators are crucial for batteries since they prevent physical contact of electrodes and thus short circuit. In this study, reutilization of aramid fabric was highlighted by transforming it into a high value product: battery separator. A waste aramid fabric was used to synthesize aramid aerogels by deprotonation, sol-gel, and freeze-drying processes and then investigated as lithium ion battery separators. Aramid fabric was collected from a scrap plant of an industrial automotive company. Nanoclay or TiO2 nanoparticles were added into this waste-based aramid aerogel matrix in the sol-gel stage to further enhance the performance of the separators. The samples were characterized by scanning electron microscope (SEM), linear sweep voltammetry, electrochemical impedance spectroscopy (EIS) and galvanostatic charge/discharge tests. A uniform and bead-free morphology was observed for all samples with over 60% porosity. Electrolyte uptake and ionic conductivity test results showed that addition of TiO2 nanoparticles increased electrolyte uptake and ionic conductivity up to 365% and 2.2 mS/cm, respectively. The cells prepared by using nanocomposite aramid aerogels with TiO2 exhibited excellent cycling performance with a capacity of around 160 mAh/g in 200 cycles.
Introduction
Lithium-ion batteries are one of the most promising energy storage devices due to their high energy density, large operational voltage, long cycle life and low self-discharge rate.1,2 They are frequently utilized in portable electronics, medical devices, and electric cars. In lithium ion batteries, separators play a significant role by allowing ion passage and eliminating direct contact of electrodes and thus, preventing the short circuit and thermal runaway. 3
Separators should possess certain properties such as mechanical stability, chemical inertness, high porosity, a proper thickness, thermal stability at elevated temperatures, and homogenous pore distribution.4,5 Commercial separators are polyolefin microporous separators, and these separators have drawbacks of poor wettability and low thermal stability. 6 Properties of separators are strongly affected by their production method and there are a lot of ways to produce battery separators. Commercially widespread production methods of separators are dry processes which include extrusion and stretching. Electrospinning, centrifugal spinning, phase separation, melt blowing, and sol-gel methods are other methods used for separator production. 7
An appropriate level of porosity is necessary for a separator to enable ion exchange. It is known that a separator needs to have a minimum of 40% porosity with a uniform distribution. 4 A nano-porous structure and adequate porosity contribute to a high performance where active materials are in contact with electrolyte through porous separator. Commercial separators show 55% porosity in average 8 with unidirectional pores which reduces tensile strength in transverse direction. 9 However, a uniform and spherical morphology would provide a better ionic conductivity, electrochemical performance, and mechanical stability.
Aerogels are lightweight and highly porous materials which have a three-dimensional network with large number of pores. Polymer aerogels are produced by sol-gel method, and they are known for their low density and high adsorption capabilities. 10 Polymer aerogels can be produced by using several polymers such as cellulose, polyvinyl alcohol, aromatic polyamide (aramid), polyimide, poly(para-phenylenediamine), polyaniline, poly (styrene sulfonate), etc. 11 Aerogels have extended application from biomedical engineering to thermal insulation, catalytic materials to absorbers. 12
Aramid is originated from polyamides which include aromatic groups in polymer chains. In aramids, diacid and diamine moieties of polyamide are arranged in the aromatic structure. 13 Aramid is classified as a high-performance polymer due to its distinctive properties such as wear and abrasion resistance, very high tenacity, thermal and chemical resistance. High crystallinity, aromatic structure and hydrogen bonding gives aramid ultimate physical and chemical privilege. Aramid’s unique combination of strength, durability, and heat resistance has positioned it as a critical material in various industries where reliability and safety are paramount. Aramid fibers have 1.44 specific gravity, 62 GPa tensile modulus with a 4% tensile elongation.14,15 Aramid fibers have a high thermal degradation point which is attributed to a mass loss at 579.8°C. 16 Aramid fibers are employed in aerospace, automotive, ballistic protection, construction and industrial sectors, providing reinforcement for composites, cables, and tires. Additionally, their inherent flame resistance makes them ideal for applications requiring fire-resistant properties. Aramid has been used in few studies as a separator.17,18 However, second use of these industrial products is mostly ignored. There are only few examples of recycling techniques in the literature such as pyrolysis, 19 green recycling, 20 and subcritical and supercritical treatment methods. 21 In this study, to recycle the automotive aramid scrap, we utilized deprotonation process as a chemical method and produced nanocomposite aramid aerogels. Aramid aerogels can be produced by sol-gel and freeze-drying methods: firstly, a colloidal suspension of aramid nanofibers (ANF) are produced by deprotonation process in which macroscopic aramid fibers are splitted up by aggressive alkaline solutions 22 ; secondly, water as proton-donor is added to ANF dispersion to restore the broken bonds and form hydrogels by converting dispersed phase into a 3D solid network; thirdly, hydrogels are frozen and finally, the ice phase in the hydrogels is removed by freeze-drying. After the freeze-drying process, highly porous and lightweight aramid aerogels are obtained.
In this study, waste aramid fabric was utilized as an aramid precursor. Aramid fabric was initially used in automotive industry to reinforce the plastic parts. An automotive B-pillar part was produced by over molding process in which polypropylene was injected onto aramid fabric in high pressure. Once the part was scrapped by the operator, aramid fabric was peeled off from plastic part. Herein, the recycle capability of aramid fibers as aramid nanofibers and its transformation to lithium ion cell separator was underlined. As an innovative material, aramid aerogel proposes several advantages for battery separator applications. Those advantages can be elaborated by two perspectives: material and morphology. Combining aramid’s features with porous aerogel structure will emerge as a multifunctional material. A separator made by a highly nano-porous material with a high thermal stability will increase the cell performance and safety. Aramid fibers have competitive ultimate tensile strength, modulus and tribological properties. 23 Moreover, aramid is an excellent thermal insulation material with high degradation temperatures. 24 Thermal stability is necessary in case of a thermal runaway. 25 In separators, high wettability is also required. Polyolefins have hydrophobic surface characteristics, on the other hand, aramid nanofibers have hydrophilic amide groups which increases its wettability. 26 In the literature, few studies have been reported aramid aerogel as cell separator however reutilized aramid fiber- based aerogels and nanocomposite aramid aerogel separators have not been synthesized yet.27,28
Nanoparticle addition into separators can enhance ionic conductivity, electrolyte uptake and electrochemical performance of the cell. The addition of fillers creates Lewis acid-base interactions between the polar surface groups of the inorganic filler and the ionic species in the electrolyte. This interaction generates additional sites for ion migration, thereby improving ion mobility and ionic conductivity. Also, ionic transportation effects ohmic polarization resulting in a gain in discharge capacities at each current density. 29 Prasanth et al. 30 investigated the effect of nanoclay existence in microporous polymer membrane. They demonstrated that adding nanoclay fillers improved ionic conductivity, ion transference number, and stability of the lithium electrode-electrolyte interface. Gao et al. 31 produced TiO2/polyimide composite separator with flame retardancy for a safe lithium ion battery. They showed that addition of TiO2 nanoparticles enhanced the flame retardancy performance and electrochemical performance by improving the interface stability between separator and electrolyte. They concluded that TiO2 addition improved the affinity for liquid electrolyte and thermal stability up to 160°C. Moreover, a distinct morphology with spherical beads was obtained which increased electrolyte uptake (721%) and exceedingly diminished lithium dendrite formation. Xiao et al. 29 produced a composite separator with ZrO2 nanoparticles. They showed that combining ZrO2 nanoparticles with a nonwoven substrate provided improved heat resistance, potentially enhancing battery safety.
This study aims to accelerate reutilization of industrial aramid fabric waste. Aramid is an expensive, high-performance polymer and it has a long lifetime therefore, it must be reused or recycled. Furthermore, recycling materials with high environmental, economic, and technical value is critical for a more sustainable world. The processes studied in literature for the recycling of aramid generally require high energy and they are expensive methods. In the deprotonation method, which is preferred in this study, it is possible to convert waste aramid fabric into aramid nanofiber in a laboratory environment in only a 4-hour process. We also demonstrated nanocomposite aramid aerogel separators for lithium-ion battery with excellent porosity, ionic conductivity, and electrochemical performance for the first time. TiO2 and nanoclay nanoparticles were added into the aramid nanofiber dispersion to obtain nanocomposite aerogels to investigate the effect of TiO2 and nanoclay on morphology, ionic conductivity and electrochemical performance.
Experimental
Materials
Aramid fibers were obtained from Toray Company. It was initially used as an over-molding insert on an automotive thermoplastic B-pillar part. The fabric had a plain weave pattern with 215 dtex weft and warp yarns and the fabric thickness was 0.2 mm. Dimethyl sulfoxide (DMSO, 99,9%), potassium hydroxide (KOH), tert-Butanol (TBA) and n-butanol were obtained from Sigma Aldrich. Nanoclay and titanium dioxide nanoparticles nanoparticles were obtained from Aldrich and Grafen E.
Preparation of aramid nanofiber dispersions
Aramid nanofiber (ANF) dispersion was prepared by proton-donor assisted deprotonation. 32 Firstly, a highly alkaline liquid solution was prepared with dimethyl sulfoxide (DMSO) and potassium hydroxide (KOH). In a large beaker, 1.5 g of KOH was first manually grinded and dissolved in DMSO. Aramid fabric was washed with methanol and dried and cut into small pieces. Afterwards aramid fabric was added into KOH/DMSO solution. Aramid fabric splitted up into aramid nanofibers with the effect of strong alkaline KOH/DMSO solution which is named as deprotonation process. After addition of aramid fabric, 50 mL of deionized water was added into the beaker. Water served as proton donor to facilitate the process by decreasing the viscosity of negatively charged aramid polyanions in DMSO. 32 The liquid phase immediately turned into a yellow color with the addition of deionized water. The temperature was set to 60°C and the dispersion was mixed for almost 4 h until a red transparent liquid was obtained.
Preparation of aramid hydrogels & aerogels
A low viscosity red transparent ANF dispersion was obtained after 4 hour-mixing of aramid/KOH/DMSO/water system at 60°C. TiO2 or nanoclay nanoparticles were added into ANF dispersion to prepare TiO2 or nanoclay aramid aerogels, respectively. The nanoparticle added ANF dispersions vigorously stirred for an hour to spread nanoparticles in the dispersion. For gel preparation, 10 mL water was added dropwise during mixing to allow a slow gelation and hydrogel formation. After an hour of mixing of hydrogels, nanocomposite aramid hydrogels were obtained. In order to get rid of KOH and DMSO in hydrogels, they were washed with water for several times. Finally, nanocomposite aramid hydrogels were washed with tert-butyl alcohol to terminate the solvent exchange. The hydrogels were frozen in the freezer and then put into a freeze-dryer (Christ alpha 1-4LD) under vacuum for 24 h to obtain nanocomposite aramid aerogels. Aramid aerogels without nanoparticles were also synthesized for comparison.
Characterization
The morphology of the aramid aerogels was studied using a field-emission scanning electron microscope (FESEM). Contact angles were measured by a droplet contact angle instrument. X-ray diffraction analysis was performed to characterize the phases and crystallographic microstructures of samples at the voltage of 5 kV and with the current of 40 mA. Raman spectra were obtained by using a 532 nm laser excitation. Fourier transform infrared (FTIR) spectrum was obtained in the range of 500-4000 cm−1. The porosity investigation was conducted by n-butanol uptake tests. In the uptake test, following equation was used:
Electrochemical impedance spectroscopy (EIS) was used to measure the ionic conductivities of the aerogel separators. The aramid aerogels were soaked in liquid electrolyte and placed between two stainless steel electrodes over a frequency range of 1 MHz to 10−2 Hz with AC amplitude of 10 mV at room temperature. Afterwards, the ionic conductivity was calculated by following equation:
Results and discussion
In this study, nanocomposite aramid aerogel separators were produced by a facile approach as shown in Figure 1. Aramid fabric, which was a waste of an automotive company, was recovered using the deprotonation process. Due to its strong alkaline characteristic of KOH/DMSO solution, macroscopic aramid fabric splitted down into nanofibers via electrostatic repulsion which also eliminated any differences from batch to batch.
22
TiO2, or nanoclay nanoparticles were added separately into aramid nanofiber dispersions and stirred to homogenously distribute the nanoparticles and prevent agglomeration. Addition of water served as a proton donor and restored the structure by establishing random bonds between dispersed aramid nanofibers and thus aramid hydrogels were created.
32
Aramid hydrogels were washed with water several times to get rid of residual DMSO and KOH. To arrange the thickness, hydrogels were flattened by a doctor blade. Washed hydrogels were firstly frozen in a freezer for a day and then, hydrogels were finally freeze-dried by sublimating the frozen solid ice phase of hydrogels without any collapse. Freeze-dryer allowed a porous aerogel structure unlike drying in furnace. The produced nanocomposite aramid aerogels have high porosity of above 80% and ultra-lightweight to be carried by a flower as shown in Figure 1. It is notable that the aramid fibers in waste are as same as the original fibers. Furthermore, even after the aramid fiber-based materials have reached the end of life, aramid fiber retained the same structure with only minor damage to their mechanical properties.
19
The scrap fabric was obtained from scrap injection molded part which was subjected to 230°C and 110 bar during process. Even though the fabric was regenerated from injection molding process, due to its thermal and mechanical stability, no significant difference has been detected among different batches of aramid fiber. Furthermore, aramid fabrics were transformed into aramid nanofibers by broking the hydrogen bonds in the chain which means the nanofibers were not affected from the first life of aramid fabric. Schematic illustration of (a) deprotonation and aramid nanofiber formation, (b) nanoparticle addition and nanocomposite hydrogel formation, (c) aramid hydrogels and aramid aerogel on the top of a flower petal.
The innovation of this study can be expressed by preparing a nanocomposite aramid aerogel separator with well dispersed nanoparticles by using waste aramid and facile approach. Few studies reported the aramid aerogel for battery separator application27,28,33 and inorganic nanoparticle addition into the polymer separator matrix is well known and studied.34-36 Herein, we aimed to achieve a robust and high-performance characteristic with the addition of inorganic nanofillers into waste-based aramid aerogels. Introducing nanoparticles into separators can significantly enhance the performance of lithium ion batteries. By introducing inorganic fillers, the nanoparticle surface groups interact to the electrolyte’s ionic moieties, fostering Lewis acid-base interactions. 29 This interaction creates additional pathways for ion migration, thereby improving both ionic conductivity and the absorption of electrolyte in the separator. The positive effects of TiO2 and nanoclay fillers were already studied and an increasing electrochemical performance was reported.37–40 In this paper, it was aimed to show the effect of these particles in the waste-based aramid aerogel structure. Aramid aerogels were produced from waste aramid fabric by deprotonation process and incorporating TiO2 or nanoclay enhanced ionic conductivity and electrochemical performance.
SEM images of aramid aerogel and nanocomposite aramid aerogels are shown in Figure 2. A uniform and tortuous morphology is crucial for separators. The fiber diameter also affects the porosity and electrochemical performance of cell. Aramid aerogel (Figure 2(a)) showed a highly homogenous nanofiber structure with approximately 40 nm fiber diameter. TiO2/aramid aerogel and nanoclay/aramid aerogel (Figure 2(b) and (c) also showed a uniform porosity and fibrous structure with visible pores. All the samples have a homogenous porosity which is beneficial for effective and fast Li ion flux.4,41 SEM images of (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) nanoclay/aramid aerogel.
Figure 3 shows the fiber diameter normal distrubition histograms of aramid aerogel and nanocomposite aramid aerogel separators. All the samples showed similar fiber diameter around 50 nm. Average value of fiber diameters were 51 nm, 51 nm, 50 nm with standard deviation of 5.7, 6.1, and 6 for aramid aerogel, TiO2/aramid aerogel and nanoclay/aramid aerogel, respectively. The contact angles of aramid aerogel and nanocomposite aramid aerogels were measured using a droplet contact angle test, where water droplets were deposited onto the surface and the results are shown in Figure 3(d). Contact angle is corralated with hydrophilicity, surface roughness and porosity.42,43 In our study, aramid aerogel and nanocomposite aramid aerogels showed nearly 0° degree contact angle with water droplet due to the high hydrophilicity of aramid and excellent porosity of aerogel structure. TiO2 and nanoclay addition did not change the hydrophilic behaviour since they were filled and dispersed in the aramid aerogel matrix instead of accumulating on the surface. Similarly, Gan et al. studied contact angle measurements of aramid aerogel membrane and showed that water was immediately speared out and gave a zero contact angle due to the porous structure of aerogel.
44
Fiber diameter normal distrubitions and contact angles. (a) Diameter distrubition histogram of aramid aerogel; (b) Diameter distrubition histogram of TiO2/aramid aerogel; (c) Diameter distrubition histogram of nanoclay/aramid aerogel; (d) Contact angle figure of TiO2/aramid aerogel.
Porosity, electrolyte uptake and ionic conductivity values of aerogel separators.

Electrolyte uptake test was performed to evaluate the ability of the separator to absorb and retain the electrolyte. The compatibility of the separator in an electrolyte influences the ion flow and thus electrochemical performance such as capacity retention.47,48 As can be seen from Table 1, all the aramid aerogels showed much better electrolyte uptake than commercial separator.
Aramid aerogel and nanocomposite aramid aerogels showed over 300% electrolyte uptake. Notably, TiO2/aramid aerogels and nanoclay/aramid aerogels could absorb much more electrolyte than aramid aerogel with 365% and 360% uptake capacity, respectively. An increase in electrolyte uptake can be attributed to high hydrophilicity of ceramic-based materials 49 and sufficient channels for ion transmission. 50 Chen et al. indicated that the multi-porous structure led a better liquid electrolyte uptake in fibrous separators. 51 Sun et al. prepared a TiO2 modified aramid nanofiber separator and it showed improved porosity and electrolyte retention rate after nanoparticle doping. In their study, ANF separator showed higher electrolyte retention rates than commercial PP separator and further TiO2 doping increased the electrolyte retention due to the high hydrophilicity of TiO2. 40 Prasanth et al. investigated the effect of nanoclay addition on ionic conductivity and electrochemical performance of PVDF membrane made by phase inversion method. They indicated that PVDF membrane with 2% nano-clay had 35% electrolyte uptake which was 14% higher than that of the membrane without nano-clay. This increase was attributed to high porosity, membrane morphology and clay content. 30
Ionic conductivity is an important parameter which enables efficient ionic transport and affects the battery performance. 40 Ionic conductivity is closely related to porosity, tortuosity, and electrolyte uptake. 45 In this study, aramid aerogels and nanocomposite aramid aerogels were soaked in the liquid electrolyte and placed between two stainless steel electrodes to measure the ionic conductivities. Table 1 also shows the ionic conductivity values. Aramid aerogel exhibited an ionic conductivity of 1.8 × 10 −3 S/cm which was higher than that of commercial PP separator (0.8 × 10 −3 S/cm). TiO2/aramid aerogels and nanoclay/aramid aerogels showed the highest ionic conductivity of 2.2 × 10 −3 S/cm. The increase in ionic conductivity of nanocomposite aramid aerogel separators could be ascribed to the improved electrolyte uptake and hydrophilicity of nanoparticles since ionic conductivity is correlated with the electrolyte uptake.40,50 Zhu et al. produced TiO2 grafted PE separator which had higher ionic conductivity than that of PE separator. 49 Balkanloo et al investigated clay mineral/polymer composite characteristics and showed that highest electrolyte uptake value belonged to the polymer with 8 wt% of clay due to its high hydrophilicity and surface area. 38 Sheng et al. studied aramid nanofibrous aerogel separators and they showed that the aramid nanofibrous aerogel had much higher ionic conductivity than that of PP separator. 27
XRD analysis was performed to identify crystalline phases in aramid aerogels. Figure 4 shows the XRD patterns of aramid aerogel and nanocomposite aramid aerogel separators. XRD patterns of aramid aerogel separators: (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) nanoclay/aramid aerogel.
Aramid aerogel had two main peaks at around 20° and 23° attributed to (110) and (200) planes. 52 XRD results of aramid aerogel, TiO2/aramid aerogel and nanoclay/aramid aerogel showed the characteristic patterns of aramid aerogels32,53 which proved that aramid aerogel and nanocomposite aramid aerogels still have the crystalline sites of aramid and structural regularity of aramid aerogels. TiO2/aramid aerogel had an intense peak at 25.3° which corresponds to crystal phases of TiO2. 40 XRD pattern of nanoclay/aramid aerogel showed a peak at around 61° which could be attributed to the dominant phase of nanoclay. 54
Raman spectroscopy was used to investigate the effect of addition of nanoclay and TiO2 nanoparticles on the molecular structure. Figure 5 shows the Raman spectra of aramid aerogel and nanocomposite aramid aerogel separators. Raman spectra of aramid aerogel separators: (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) Nanoclay/aramid aerogel.
Raman spectra of all samples showed the identical peaks of aramid at approximately 1280 cm−, 1332 cm− and 1615 cm-. 55 In the Raman spectrum of TiO2/aramid aerogel, typical peaks of rutile phase of TiO2 crystallites appeared at 446 cm− and 609 cm− which indicated the introduction of nanoparticles. 56 In the Raman spectra of nanoclay/aramid aerogels, a broad peak between 450 cm- and 770 cm- could be attributed to the peaks of nanoclay. Raman spectrum identified nanoclays by their unique spectral pattern, revealing specific details about their mineral structure. This included the presence of hydroxyl groups, tetrahedral silicate/aluminate anions, octahedral metal cations, and interlayer cations within the nanoclay mineral family. Generally, these minerals exhibit characteristic peaks around ∼95, 200, 290, ∼430, and 710 nm, particularly noticeable in montmorillonite minerals, where a broad peak is typically observed in the central region. 57
FTIR was used to characterize the chemical interactions of nanoparticles within aramid aerogel microstructure. Figure 6 shows the FTIR spectra of aramid aerogel and nanocomposite aramid aerogel separators. FTIR spectra of aramid aerogel separators: (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) Nanoclay/aramid aerogel.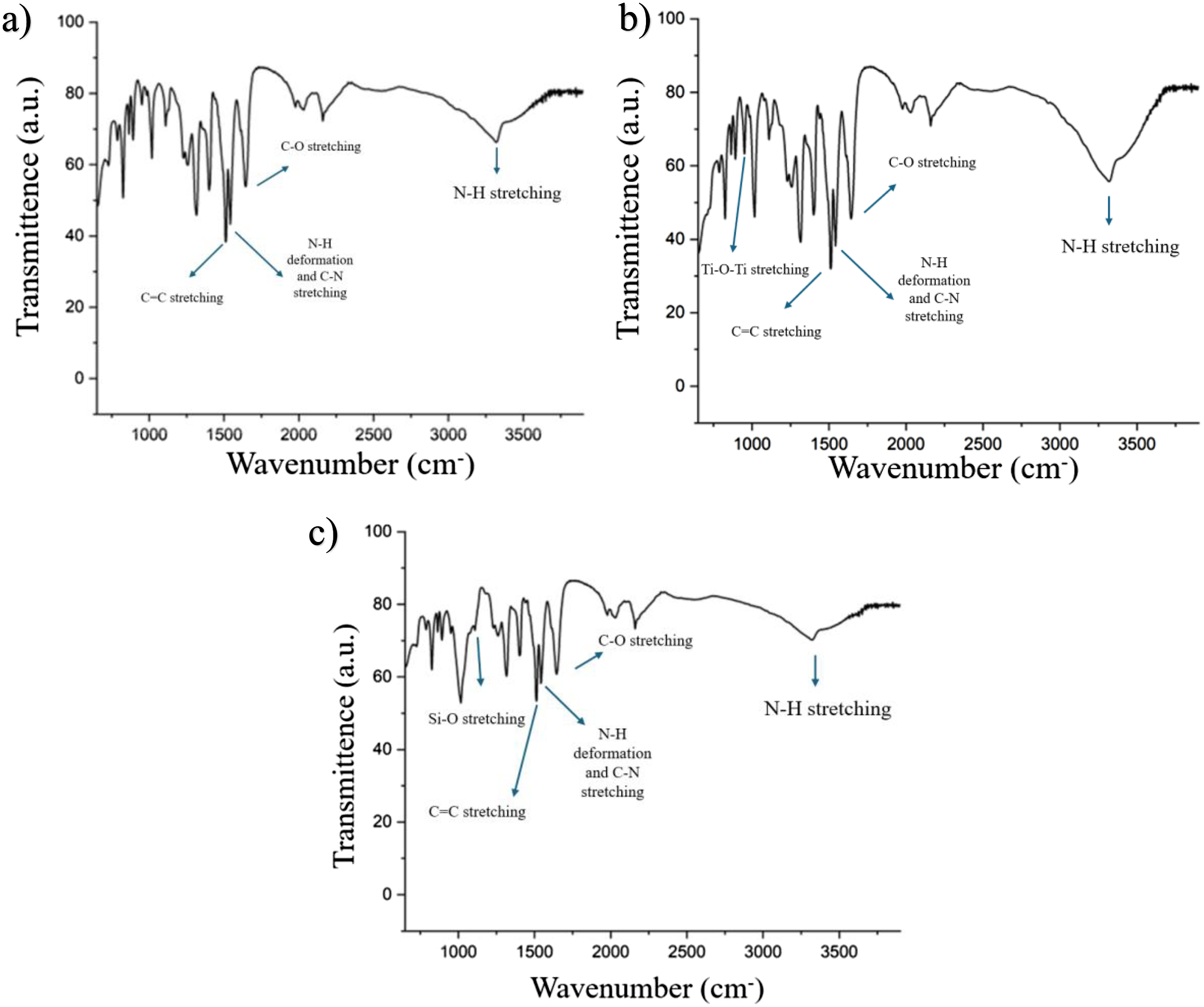
FTIR spectra showed the typical vibrational modes of aramid bonds which are aromatic C = C stretching at about 1515 cm−1, and C = O stretching at 1644 cm−1 and C-N stretching about 1540 cm−1 and a broad peak about 3300 cm−1 region which corresponded to N-H stretching.22,58 The similarity with FTIR spectrum of aramid fiber with ANF confirmed the restoration of structure during gelation stage. The nanocomposite aramid aerogels showed characteristic transmittance of both TiO2, nanoclay and aramid. The band at near 1100 cm− assign to the Si-O stretching that is imparted by nanoclay.58,59 The appearance of this stretching group demonstrated the successful intercalation of nanoclay in the ANF structure. The effect of TiO2 addition into the microstructure was seen at near 970 cm- peak with Ti-O-Ti stretching vibration mode.
Electrochemical oxidation limit is determined by linear sweep voltammetry (LSV) test which investigates electrochemical stability of separators. In this test, the separators were soaked into electrolyte on the Li/separator/SS system and the extended potential is applied.
36
The sharp increase of current in the voltammetry curves represented the electrolyte decomposition and electrochemical oxidation limit. Figure 7 shows the LSV curves of aramid aerogel and nanocomposite aramid aerogel separators. In the curves, current did not show a dramatic increase when the voltage was lower than 4.5 V vs. Li+/Li. It meant that all aramid aerogels could sustain a stable electrolyte below 4.5 V which was sufficient for lithium ion battery operating voltage range.
39
Linear sweep voltammetry curves of aramid aerogel separators: (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) Nanoclay/aramid aerogel.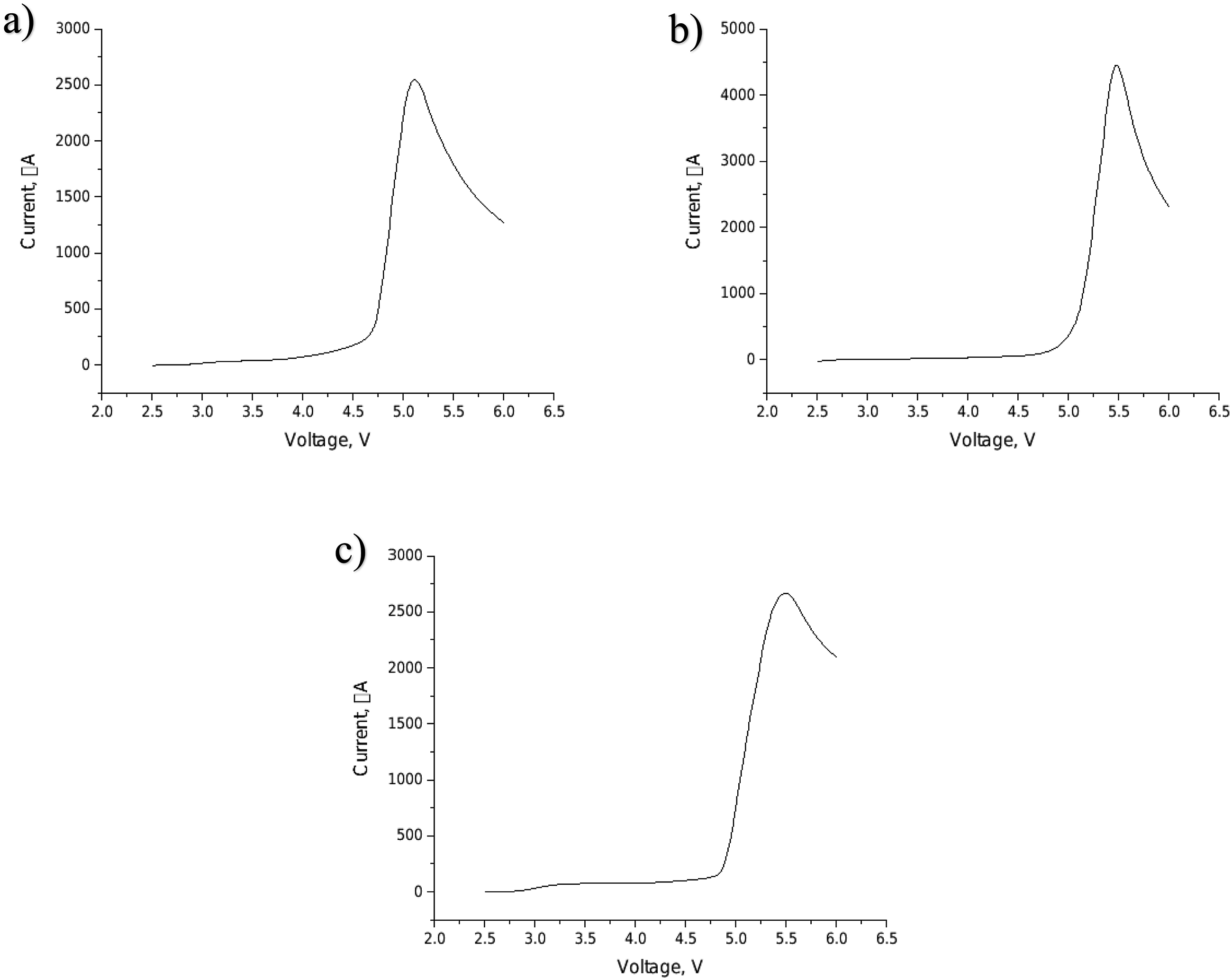
As seen in Figure 7, aramid aerogel, TiO2/aramid aerogels and nanoclay/aramid aerogels showed electrochemical oxidation limit around 4.5 V, 5 V and 4.75 V, respectively (Figure 7(a–c)). TiO2/aramid aerogels and nanoclay/aramid aerogels showed a higher decomposition voltage which meant that nanocomposite aerogels had a better electrochemical stability. This could be attributed to nanoparticle incorporation. Chen et al. indicated that nanoparticle modified separators showed higher decomposition voltage which means a better electrochemical stability. Also, the Lewis acid/base interactions with the ionic species in the electrolyte contribute to minimizing the decomposition of the lithium salt anion, thereby extending the electrochemical potential window, and improving performance. 51 Arya et al. showed the effect of nanoclay addition in polymer nanocomposite separator. The addition of nanoclay enhanced the voltage stability window up to 4.42 V whereas the pure separator showed the stability only up to 3.12 V. The increase in electrochemical stability and maximum voltage limit by nanoclay addition was attributed to cation coordination with a surface charge of clay which provided conducting regions. Moreover, the nanoclay tunnels would help to cations to be hosted in polymer chain which provides faster cation migration. 37
In order to investigate the compatibility of aramid aerogel separators with lithium metal, electrochemical impedance spectroscopy measurements were performed. Aramid and nanoparticle/aramid aerogels were soaked into the electrolyte in symmetric Li/Li cells. In the Nyquist curves, the semicircle at the intermediate frequency region represents the interfacial resistance. The larger diameter means an increased interfacial resistance 40
Figure 8 shows the Nyquist plots for aerogels and interfacial resistances were 225, 75 and 75 Ω for aramid aerogel, TiO2/aramid aerogel, and nanoclay/aramid aerogel, respectively. Moreover, the samples had lower interface resistance values compared to the commercial separator (310 Ω).
60
The difference between aramid aerogel and nanocomposite aramid aerogels could be explained by improved electrolyte absorption and nanoparticle addition which are beneficial for lithium diffusion at the interface between separator and electrode.
60
Sun et al. produced TiO2 modified para-aramid nanofiber composite separator by casting and phase immersion method. They characterized the electrochemical impedance by EIS and reported that the separator with TiO2 showed smaller semicircle diameter and resistance compared to the commercial separator due to the existence of TiO2 nanoparticles. According to their research, the TiO2 particles facilitated charge transfer and enhanced Faraday reaction kinetics.
40
Arya et al. reported that addition of clay reduced the bulk resistance due to the effective changes in the arrangement of polymer chains, facilitating faster ion conduction. The addition of even a minimal quantity of clay resulted in a reduction of the high-frequency arc. The results were ascribed to the improved salt dissociation and the formation of a double layer.
37
Interfacial resistance of aramid aerogel separators: (a) aramid aerogel, (b) TiO2/aramid aerogel, (c) Nanoclay/aramid aerogel.
To further investigate the electrochemical performance, coin cells were assembled with nanocomposite aramid aerogel separators, LiFePO4 cathode, lithium metal anode and electrolyte. Primarily, the capacity of a cell depends on the electrode type and structure. Nonetheless, it is known that the separator’s structure also significantly influences the cell performance due to its ionic transportation ability between electrodes which is a crucial factor in determining cell kinetics.
45
The charge/discharge curves of the first cycle of the cells with aramid aerogel, TiO2 and nanoclay/aramid aerogels are given in Figure 9. The cells were tested between 2.5 V and 4.5 V at 0.2 C. First-cycle charge/discharge curve of Li/LiFePO4 cells with (a) aramid aerogel, (b) TiO2/aramid aerogel and (c) nanoclay/aramid aerogel.
As shown in Figure 9, the cell with aramid aerogel displayed an initial discharge specific capacity of 160 mAh/g while TiO2/aramid aerogel had the specific capacity of 165 mAh/g and nanoclay/aramid aerogel had the specific capacity of 162 mAh/g (Figure 9(a–c), respectively). All the separators showed better results than commercial PP separator in Li/LiFePO4 cell with the first-cycle discharge capacity of 154 mAh/g. 2 The first cycle discharge capacity was increased with TiO2 and nanoclay incorporation. This can be devoted to the higher electrolyte adsorption and higher ionic conductivity of separators. Higher electrolyte adsorption of TiO2/aramid aerogels and nanoclay/aramid aerogels gave rise to higher ionic conductivity which promoted more conduction channels and increased discharge capacity. Chen et al. produced TiO2@PI/PVdF-HFP nanofiber membrane by bicomponent co-electrospinning method and they reported higher initial cycle discharge capacity for their TiO2 nanoparticle incorporated separators. 51
The cycling performance of batteries refers to their ability to maintain capacity and performance over multiple charge and discharge cycles. Cycling properties of Li/LiFePO4 cells with aramid aerogel (Figure 10(a)), TiO2/aramid aerogel (Figure 10(b)) and nanoclay/aramid aerogels (Figure 10(c)) were tested at 0.2 C. Cycling performance of Li/LiFePO4 cells with (a) aramid aerogel, (b) TiO2/aramid aerogel and (c) Nanoclay/aramid aerogel.
All the cells containing aramid and nanocomposite aramid aerogels had a discharge capacity of around 160 mAh/g without an evident capacity lost for 200 cycles at 0.2 C. The cycling performance results validate excellent stability for 200 cycles without a capacity retention. The observed cycling performance corresponds with the efficient liquid electrolyte uptake and enhanced ionic conductivity. These factors facilitate faster ion transport and improved retention of electrolyte during cycling. 51 Parekh et al. produced aramid nanofiber aerogels and compared galvanostatic cycling performance of aramid nanofiber aerogel separator with PP separator. They showed that PP separator was stable over 300 cycles with a 15% fade. On the other hand, aramid nanofiber aerogel separator delivered stability over 300 cycles with only 11% fade. 33
However, Sheng et al. showed that aramid nanofibrous aerogel was stable at 0.2 C in 150 cycles with the specific capacity of only around 150 mAh/g. 27 Liu et al also reported a stable cycling performance with around 150 mAh/g specific capacity for aramid aerogel separators. 28 Zhang et al reported that specific capacity decreased around 140 mAh/g in 100 cycle for aramid separators. 60 Yang et al also reported the specific capacity of only around 150 mAh/g in 100 cycles for aramid/bacterial cellulose separators. 61 Tang et al reported the specific capacity of ANF/PEO separator 128.3 mAh/g in 100 cycles. 62 Patel et al. found the specific capacity of their ANF separator as 123.4 mAh/g at 10th cycle which showed a reduction in capacity with 53% after 50 cycles. 63 Excellent cycling stability with high discharge capacity of the nanocomposite aramid aerogels could be attributed to separator morphology. Aerogel structure successfully provided a porous structure. A uniform distribution and homogenous pore size facilitated faster ionic transport. Also, nanoparticle addition enhanced cycle performance by establishing ion transmission channels and increasing hydrophilicity of separator.
Conclusion
Lithium ion batteries have emerged as a crucial energy storage component, powering a wide range of portable devices and electric vehicles. The performance of these batteries is influenced by various components, and the separator plays a critical role in ensuring safe and efficient operation. In this study, waste aramid fabric was reutilized, and nanocomposite aramid aerogels were synthesized and then used as separators in the lithium ion cells for the first time. The effects of TiO2 and nanoclay nanoparticles in aramid aerogel separators in lithium ion cells were investigated, separately. SEM images illustrated a uniform porous structure, which is beneficial for the efficient electrolyte adsorption and ionic conductivity. Electrolyte uptake and ionic conductivity increased in nanocomposite aramid aerogels with addition of TiO2 or nanoclay. Comprehensive electrochemical tests were conducted to evaluate the performance of lithium ion cells employing aramid aerogel separators and nanocomposite aramid aerogel separators. The results demonstrated that the cells equipped with nanocomposite aramid aerogels exhibited excellent electrochemical performance. Moreover, waste aramid fabric was used as the precursor of ANF dispersion, which were then transformed into a new aerogel. This process effectively reclaimed aramid fabric that had been discarded and would have otherwise ended up in a landfill, contributing to the integration of a sustainability approach in the research. The findings of the study highlight the potential of nanocomposite aramid aerogel separators as a viable option for lithium ion battery. These novel separator materials could lead to improved battery performance, increased energy storage capabilities, and reduced environmental impact in future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Commission Marie Skłodowska-Curie Actions under Grant Agreement ID: 101021759 and the Scientific Research Projects Department of Istanbul Technical University ITU BAP (MGA-2023-43897). The authors are also thankful for the financial support from the National Research Foundation of Korea (Grant No. NRF-2021R1F1A1061200), and the Korea Institute of Industrial Technology (Grant No. PUR24400).