Abstract
Due to its low hydrophilicity, direct sublimation inkjet printing on woven polyester fabric surfaces is challenging. This research experimentally studies the efficacy of dielectric barrier discharge (DBD) roll-to-roll plasma treatment with or without water vapor post-exposure as a novel method to modify polyethylene terephthalate (PET) surfaces to enhance hydrophilicity, color performance, and printing quality. The Response Surface Methodology (RSM) was used to design experiments, and the optimum conditions were analyzed through statistical calculations. Their results were examined using Optical Emission Spectroscopy (OES), Atomic Force Microscopy (AFM), field emission scanning electron microscopy (FESEM), Energy Dispersive X-ray (EDX), X-ray Photoelectron Spectroscopy (XPS), and Attenuated Total Reflectance Fourier Transform Infra-Red spectroscopy (ATR-FTIR) tests. Qualitative and quantitative tests for color performance and wicking test used for hydrophilicity assessment confirms the effectiveness of this Green inline pretreatment for industries to enhance direct sublimation inkjet printing quality on PET by a decrease in absorption time from 12051 to 202 s and an increase in the color depth to 12.49%, 25.89 and 18.07% for Cyan, Magenta and Key (black) respectively. One of the stable optimum conditions in which three printed colors: Cyan, Magenta, and Black (Key), show high-quality color performance is at 15 processing rounds, 2 m/min velocity in the absence or presence of water vapor post-exposure. Water vapor post-exposure exhibits a beneficial, economic effect along with plasma treatment. The results show that the chemical modification aspect of this treatment plays a more significant role in printing quality enhancement than its morphological modification.
Introduction
Direct printing has gained popularity owing to some of its beneficial characteristics such as high speed and quality, less human resources requirements, and environmentally-friendly aspects, to name a few, hence widely applied. Some surfaces, such as raw polyester fabric, are unsuitable for direct printing due to their low surface energy and hydrophobic nature, making them inappropriate for direct printing, thus leading to poor color quality and physical properties. Therefore, the surface must be treated before printing to enhance its surface energy and transform its hydrophobic nature into the hydrophilic one. 1
Plasma processing is considered one of the most effective surface treatment methods compared to other chemical methodologies. It enjoys several advantages, such as its ability to modify the outermost surface layer and prevent changes in the material bulk properties, provide low temperature for temperature-sensitive polymers, ensure high-security levels, not use chemical materials, eliminate the need for water washing, and guarantee the compatibility with the environment in the long run.2–7 In addition, considerable attention has been drawn to the industrial applications of cold plasma owing to its advantageous characteristics: simple production, control, applicability under atmospheric pressure and inline processing, and low costs. 8
Plasma is like a cocktail in which some reactive species, such as ions, electrons, atoms, free radicals, etc., are formed and remain floating, making some physical and chemical changes to the surface due to several phenomena such as etching, ablation, cross-linking, functionalization, and film deposition which occur either separately or simultaneously.9,10
Plasma treatment has emerged since the 1960s, applying various plasma technologies to different polymers for multiple applications. 11 In this regard, extensive studies have been conducted to assess the plasma surface modification of polyesters, considering the possible impacts on some surface characteristics, such as wettability changes caused by morphological and chemical variations. These studies have investigated some properties,6,10,12–17 such as the resultant hydrophilicity18–23 and hydrophobicity levels, variation in the optical17,18 as well as mechanical-tribological6,14–16,20 properties, and depthof penetration.22,23 The mentioned properties were obtained, followed by an examination of the effects of different parameters such as the applied power,12,13,15,20 working gas type,6,13,15,19,21 current frequency , 19 the interelectrode gap, 10 energy deposited by discharge in electrodes,10,12,18 treatment time,10,12–15,17–19 pressure,15,18,19 and polyester type20,22 under optimal conditions. The results from such studies also elaborated the interaction mechanisms between plasma and polyester surfaces to some extent. In this respect, Wrobel et al. 19 discovered that the morphological changes in the woven polyester fabric surface caused by plasma treatment directly depended on the type of gas used for plasma generation and the current frequency. They also found that although plasma treatment of the polyester fabric did not considerably enhance water vapor sorption, it could cause some changes in the kinetic of this process. 19 In 2006, Leroux et al. 20 investigated the effects of atmospheric air plasma treatments on woven and nonwoven PET textile structures. They remarked that plasma treatments depended highly on the polymer crystallinity degree and the textile structural porosity. 20
In recent decades, some other research studies have specifically examined the effects of plasma treatment and determining parameters on the improvement of the polyester printability24–30 and dyeability.5,31,32 The performance of the color deposited on the polyester surface in terms of the color depth,31,32 color strength K/S,5,24–27,29–32 patterns sharpness, and bleeding effect24–27,29,30 was thoroughly assessed in these studies. Using them, researchers could better comprehend the relationships among the plasma treatment, hydrophilicity improvement, and consequent enhancement of printing and dyeing quality on the PET surfaces. For instance, while investigating plasma-treated PET films' adhesion and printability mechanisms, Deshmukh and Bhat reported that plasma treatment could sharply enhance some characteristics, such as adhesion and printability, in short, processing times of up to 2 minutes (which is a desirable result in industries). However, in the processing time range of 2-20 min, this process became relatively slow with slight changes. 28 In another study, Fang and Zhang evaluated the physical-morphological as well as chemical modifications of the surface to improve the color performance of the atmospheric pressure plasma-treated polyester fabrics for inkjet printing. They found that 50% argon+air plasma provided more oxygen-containing groups on the fabric surface than the regular air plasma at the power of 300 W, time of 180 s, and gap distance of 3 mm. They also highlighted the critical role of the chemical modification aspect of plasma treatment in improving inkjet printing quality. 29 In another study, they used four layers of woven polyester fabric, each with a thickness of 0.25 mm, to measure the penetration depth of the atmospheric pressure plasma and the inkjet printing quality on each layer. Then, they confirmed the possibility of the plasma penetration and processing of these multi-layer PET fabrics; however, according to their findings, the modification level decreased upon increasing the fabric depth. Despite that, using up to two layers of fabric intensified the effect of inkjet printing, thus improving color performance. 30
A few studies have investigated the polyester surface when plasma is treated and then post-exposed to water vapor independently or in the plasma phase.9,33–36 For the first time, Jacob et al. evaluated the impact of post-exposure to the humid atmospheric air between the successive helium plasma treatments on the PET foils. They found that such exposure decreased the contact angle and consequent growth of more specific oxygen incorporation. 9 Rodríguez Santiago et al. subjected some polymers, including PET, to the atmospheric processing of dielectric barrier discharge (DBD) water vapor-helium plasma and reported that PET, among the other used polymers, namely ultrahigh molecular weight polyethylene (UHMWPE) and poly-ethylene terephthalate (PTFE), experienced the most significant reduction in the contact angle by 50%. They also reported a considerable alteration in the PET polymer structure, probably resulting from the opening of the aromatic ring by energetic plasma species. They suggested the utilization of water vapor plasma as a cheap and green method for improving the surface activation of polymers. 36
Our previous research recommended direct sublimation inkjet printing with DBD roll-to-roll atmospheric pressure plasma pretreatment with a ceramic dielectric as an environmentally-friendly approach to printing polyester fabrics/textiles. According to our findings, compared to the plasma power, the velocity and round numbers of plasma treatment and the interaction between these two parameters could noticeably affect the color strength and surface energy enhancement. 7 In this research, a new in-line industrial device operating at atmospheric pressure was designed, fabricated, and employed, utilizing air as the working gas and a water-vapor post-exposure stage and DBD Plasma discharge. The advantage of this system is the absence of vacuum and working at room temperature and atmospheric pressure. It also reduces costs. It is worth mentioning that this system can be used for surface modification on a large-scale production at a textile company but most of the other research have used a lab scale setup. On the other hand, the features of this new setup have made it as a non-destructive and noninvasive method which is very important in textile industries. Moreover, this new setup can do a complementary de-sizing and cleaning and attached ozone treatment beside the other features which have been reported. Compared to our previous research, some changes were made to the plasma-generating device, and then polyester was exposed to the water vapor, followed by plasma treatment in order to investigate whether this stage can increase the speed of processing by achieving a higher level of color performance parameters and hydrophilicity levels. The effects of physical and chemical changes of the polyester fabrics printed by inkjet printer were assessed. Finally, the impact of ink overlapping magenta, black, and cyan colors and their color strength was studied. The Response Surface Methodology (RSM) was employed to optimize the experiments. In addition, the capillarity (wicking) test, X-ray Photoelectron Spectroscopy (XPS), Attenuated Total Reflectance Fourier Transform Infra-Red spectroscopy (ATR-FTIR), Energy dispersive X-ray (EDX), Field Emission Scanning Electron Microscopy (FESEM), Atomic Force Microscopy (AFM), Optical Emission Spectroscopy (OES), ozone measurement, and bleeding analyses were employed to confirm the results. Given that no study has been done on the post-exposure to water vapor after plasma treatment on the polyester surface and its effect on printing improvement, further studies are still required to delve into this novel approach.
Materials and methods
Inkjet printing system and materials
100% polyester woven fabric with a surface density of 36 g/m2 was purchased from Yazdbaf textile company (Yazd, Iran), and the black, cyan, and magenta sublimation digital ink was from J-teck company (Guanzate, Italy). The samples were cut in the dimensions of an A4 sheet, i.e., 21 × 29 cm, on which the tests were done. The printer used in this study is Stylus T10 Epson (Japan).
Plasma treatments and water vapor post-exposure procedure
An industrial atmospheric pressure roll-to-roll plasma device (DBD) manufactured by Plasma Fanavar Jam Engineering Company (Tehran, Iran) was used in this experiment. This system consists of a metal roller made of stainless steel with a length of 50 cm and diameter of 13.36 cm, covered with dielectric (silicon) as the ground electrode, and a high voltage (HV) electrode containing a head with 10 electrodes with a length of 31.5 cm. The distance between the two electrodes is 3 mm. An AC power source with the ability to produce sine waves at the frequency of 20 kHz was also utilized in this study. The device's power can be adjusted from 250 to 680 W. Speed of the carrier roller, power, and the number of treatments can be manipulated in this system. This research used a two-L tank with a 2000-W heating element to generate water vapor. While the samples are placed on the roller, they pass through the atmospheric pressure plasma processing place with air as the gas, and then, they will be post-exposed to water vapor. After this stage the treated sample used to be inkjet-printed on and color tests will be done to analyze its color performance. This experiment's plasma power and voltage were set as 550 W and 15 kV, respectively. The velocity and number of processing rounds and vapor application were the variables of this experiment.
Optical emission spectroscopy and ozone measurement
Chemical species present in the plasma have been determined by OES (Avaspec-3648, Avantes-Netherlands), a non-invasive technique for plasma diagnostics. 21 Moreover, the Ozon detector SKY2000-Flygoo (China) has been used to examine the feasibility of Ozone synthesis in this work which can contribute to ozone treatment.
Surface characterization
Atomic force microscope
Atomic force microscope assessed the topography and surface roughness. The microscope used in this study is Nanosurf easyScan 2 AFM (Switzerland). The device is set in contact mode, and the surface is scanned by the probe's contact with the fabric. Root mean square (RMS) and average surface roughness parameters were obtained from images of 5 × 5 micrometers in dimensions.
Field emission scanning electron microscopy (and energy dispersive X-ray spectroscopy
The morphology of the textile surface was scanned using FESEM, Sigma 300HV, produced by ZEISS (Germany). The microscope operates at the accelerating voltage of 3.00 kV. The images of the samples were prepared with 100x, and 1.50 kx, magnification. Before the FESEM test, all samples were coated with gold. In addition, for elemental analysis of the surface, the EDX, the Aztec Oxford system equipped with an Silicon drift detector (SDD) with a resolution of 125 eV, was used that could identify the elements with atomic numbers greater than that of boron.
X-ray photoelectron spectroscopy
X-ray Photoelectron Spectroscopy analysis was employed to investigate the chemical composition of the polyester textile surface using a BESTEC (Germany) (EA10) device that works under the vacuum pressure of 10 mbar with Mg-K (alpha) radiation and hʋ=1256.6 eV. The X-ray anode operated at the power of 250 W, and the high voltage was kept constant at 15.0 kV and a detection angle of 45°. The value of the pass energy of the survey spectra was 97.0 eV, while for the high-resolution spectra, this value was 36.0 eV. The spectra were normalized to the C-C peak located at 285 eV.
ATR-FTIR spectroscopy
An infrared spectrophotometer called SpectrumOne, manufactured by Perkin Elmer (USA) with a ZnSe detector, was used in this experiment. On average, 16 scans were performed for each spectroscopy in the 400-4000 cm-1 range with a resolution of 4 cm−1 at the collision angle of 42°. The crystal used here was made of diamond.
Hydrophilicity measurement
Given the porous structure of the textiles and fabrics, contact angle measurement is strongly affected by the heterogeneous structure of the fabric; hence, the fabric's porosity prevents the correct measure of the contact angle due to the water drops suction effect. For this reason, the capillarity (Wicking) test was done to evaluate the plasma's impact on the PET's hydrophilicity. In this test, the desired sample (in 2width-8 height cm dimension) is placed vertically so that half a centimeter of the sample is placed inside the distilled water, and the time it takes for the water to rise from the fabric strip up to the height of seven cm is measured. Here, the water rises from the fabric due to capillary forces inside the fibers, and the height of the rise is reported per time.13,37 In this regard, the YL-69 moisture detection sensor (hygrometer) equipped with the Arduino Pro Mini board in the Arduino environment was utilized to accurately measure the time it took for the water to reach a height of seven cm. The shorter the absorption time, the higher the hydrophilicity level. 27 The results from the wicking test confirm the efficiency of treatments applied to polyester.
Color performance evaluation
Color strength
Color strength (K/S) and Optical Density (OD) were measured using the Ihara Spectrophotometer Model S900 Portable, made in Japan. The K/S value of the printed samples is calculated from the Kubelka-Munk equation (equation (1)31,32):
The color depth increase percentage, I, can be obtained from equation (2),31,32 as shown below:
Anti-bleeding effect
Digital optical microscope Dino-Lite model AM-314TS from ANMO Electronic company (Taiwan) was used to assess printing quality and ink bleeding.
Design of experiments
Coded levels and range of variable factors used in the design of the experiments.

Results and discussions
Design of experiment and hydrophilicity investigation
Once the experiments were carried out and the relevant hydrophilicity responses were obtained, the effectiveness of the variable factors was evaluated by analyzing five statistical models: the design model (reduced cubic), two-factor interaction (2FI), quadratic, linear, and cubic in DesignExpert software. According to the results, the design model was selected as the best, with an insignificant lack of fit and a maximum R2 value. 38
ANOVA for the reduced cubic model of the wicking response surface.

Std. dev.: 315.04, R2: 0.9931, Adj. R2: 0.9867, Pred. R2 0.9552, C.V. %: 9.56, Adeq. Precision:5 4.9094
It can be deduced from Table 2 that all three parameters of A-Number, B-Velocity, and C-Vapor affect the responses in the proposed model. In addition, the AB, AC, BC, A2, B2, and A2C interactions affect the final response. The p-values less than 0.001 highlight the employed model's statistical significance at the confidence level of 99%.40,41
The adequacy and accuracy of the employed model were confirmed through the diagnostic plots, including the normal probability of the residuals diagram, Figure 1(a), and the Predicted versus Actual diagram, Figure 1(b). The former shows the graph of the difference between the values of the predicted and actual responses. Accordingly, all the residuals are located along a straight line and distributed randomly, indicating the normal distribution of the data set, hence a suitable candidate for statistical optimization.37,38,42,43 The latter graph, Figure 1(b), shows a good agreement between predicted and actual obtained hydrophilicity levels. Design Expert Illustration, Diagnostic plots: (a) Normal plot of Residuals, (b) Predicted versus Actual Wicking (absorption time); Response surfaces of wicking (absorption time) 3D-plotted for the number of processing rounds and velocity (c) without Water vapor post-exposure and (d) with Water vapor post-exposure; ramp plot for (e) without water vapor post exposure and (f) with water vapor post exposure.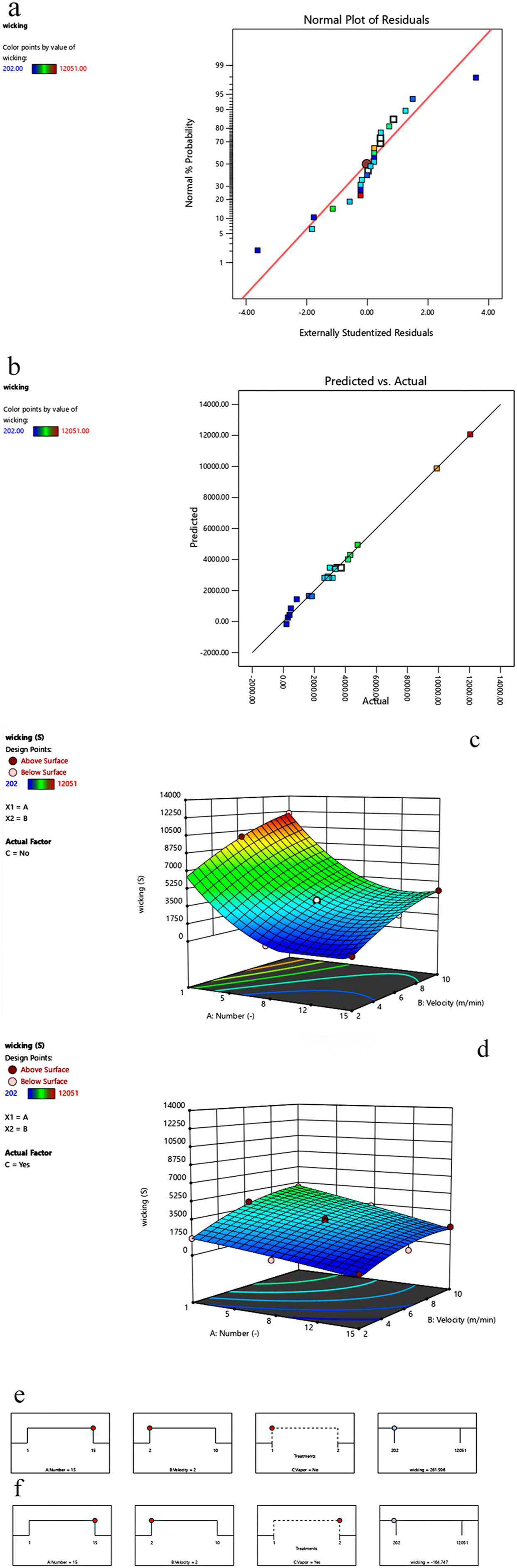
The combination of different input variables' impact on the response is illustrated in a 3D diagram, Figure 1(c) and (d). In the 3D diagram, the more movement towards the blue color, the less the wicking (absorption time) value will be, hence more good hydrophilicity. On the contrary, the more move towards green, yellow, and red colors, the longer the absorption time will be, thus less hydrophilicity. It should be noted that if the number of processing rounds is relatively low, the vapor effect is consequently quite evident. The difference between the wicking values is large and can be economically significant in industries due to less energy consumption. On the contrary, in the case of higher numbers of processing rounds, this difference in the wicking values will decrease for either the absence or presence of post-exposure to water vapor, indicating that the higher number of plasma processing rounds compensates for the noticeable effect of vapor in the lower rounds, thus reducing the significant difference between the hydrophilicity values in two modes of water vapor post-exposure. In addition, lower processing velocity contributes to the hydrophilicity enhancement.
It should be noted that high numbers of processing rounds, post-exposure to water vapor, and low velocity with their synergistic effect contribute to making the surface more hydrophilic. It should be noted that for the optimization process, due to this study's feasibility goal, the goals for variable factors have been considered in range, and Wicking’s goal is to be minimized with the highest importance. According to the experimental and optimization results, the best response with the highest desirability (1) is at the optimal mode of 15 rounds at a velocity of about 2 m/min, in the absence of Figure 1(c) or the presence, Figure 1(d) of the vapor post-processing. For this reason, from this time on, the analyses will be carried out using three samples, one of which is the untreated (control) sample. The other two are subjected to the optimal plasma treatment conditions of 15 rounds at a 2 m/min velocity in either the absence (optimal plasma treatment)OPT or presence of post-exposure to water vapor (optimal plasma treatment + vapor) OPTV.
Wicking test results, PET fabric absorption time for untreated and air plasma-treated with or without water vapor post-exposure samples.

Optical emission spectroscopy
Optical emission spectroscopy analyses are depicted in Figure 2 and Table 4, based on the literature.44–51 As shown in Figure 2, species belonging to the nitrogen family constitute a major part of the spectrum since nitrogen is the principal constituent of air. These peaks mainly belong to the second positive system N2 (C3IIU→B3IIg) (11.1 eV→7.39 eV), which indicates the population of the higher level of N2 (C3IIU), resulting from many excitation and quenching processes. The first negative system of N2+ (B2Σu+→X
2
Σg+) (18.7 eV→15.6 eV), which has a lower intensity than the second positive system of N2 (C3IIU→B3IIg) in the obtained spectra, is the result of inelastic processes of high-energy electron ionization via direct impact. Only a few available electrons appear to have the required transition energy of 18.7 eV.44,46,49,50,52 Despite the use of air, the atomic oxygen intensity is practically invisible; probably due to the faster quenching rate than the radiation rate, making atomic oxygen observation difficult.44,53 Low-intensity OH emission peak(s), attributed to the humidity of the testing environment may be observed due to the electron collisions with H2O around the wavelength of 300 nm. This transition is emitted by low-temperature atmospheric pressure air plasmas containing even a small amount of H2 or H2O (one percent).44–48 Also, low-intensity H2 peaks are observed, which seem to be caused by the recombination of the atomic hydrogens formed due to electron collisions with OH or H2O. Hydrogen-related peaks in the OES spectrum can also be attributed to other plasma degradation products and excited atmospheric air.47,48,54 The transition of CO at 312.7 nm is also worthy of attention as a shoulder that probably results from CO2 dissociation.46,47,55 OES spectra of DBD roll-to-roll atmospheric pressure air plasma at 550 W and 15 kV. Main peaks detected from OES results of atmospheric pressure air plasma at 550 W.

Measurement of the synthesized ozone
The measurement of ozone production from the air, as one of the oldest plasma chemicals processes,
56
shows its amount 35 ppm, 10 s past plasma generation. At the same time, it reaches about 206 ppm, with an increase of almost six times under the optimal condition of 15 rounds. The common mechanism of ozone synthesis in the air
56
is the following:





Ozone is a strong oxidant capable of enhancing surface wettability, hydrophilicity, and adhesion characteristics.57,58 According to the hydrophilicity test findings, followed by OPT/OPTV, the surface's hydrophilicity increased significantly, corresponding to an increase in the concentration of synthesized ozone during the treatment process. Application of this roll-to-roll system can pave the way for further industrial inline applications of ozone, mainly owing to its low costs compared to low-pressure plasma devices with high running costs.
Surface morphology and topography analysis
FESEM analysis of the control, OPT, and OPTV samples shows in Figure 3(a)–(c) the application of this treatment as a non-destructive and gentle treatment. Borcia et al. stated that because of the presence of void spaces at the cross-sections of woven polyester fabric yarns, the created micro-discharge could reach the grounded electrode surface faster than usual, resulting in localized destruction and melting in that area.
59
However, due to the silicon dielectric beneath the woven polyester fabric, direct contact with the grounded electrode is impossible in this system. This localized damage does not occur on the woven polyester surface, which can benefit the textile and printing industries. Figure 3(d) and (f) depicts some spots in the form of strips or clumps with greater magnification, the EDX analysis of which confirms the presence of some elements such as carbon, oxygen, titanium, silicon, sodium, chlorine, potassium, tungsten, calcium, phosphorus, and iron. SEM images of woven PET fabric with different magnifications 100x and 1,5kx,: (a, d) untreated sample, (b, e) air plasma treated sample, (c, f) air plasma treated with water vapor post-exposed sample.
In this semi-quantitative and semi-qualitative microanalysis, the elements with atomic numbers less than Boron cannot be identified.60,61 Therefore, the molecular formula based on the EDX output data cannot be completely determined, hence neglected in this study. These elements are either the remains of the materials used for sizing and the fabric production process in factories or even the remains of the materials used for desizing the size materials from the fabrics.62–70 Due to the effect of plasma on desizing, these contaminations are removed after plasma processing or divided into smaller segments due to etching, thus leaving only a small amount of their remains. In some cases, they are likely to be fermented, transforming their shape. Figure 3(e) shows that the number of these spots decreases, followed by OPTplasma processing. In addition, as observed in Figure 3(f), OPTV causes an even greater decrease. It can be concluded that plasma cleaning would enhance the hydrophilicity and adhesion of the polyester surface.2,10,20,28,38,54 These findings are consistent with the hydrophilicity test results. The color performance section will provide results that confirm the adhesion improvement.
Surface topography analysis
Surface Topography Analysis by AFM, Figure 4(a) and (c), illustrates no noticeable changes in the roughness and available contact surface. This finding agrees with the FESEM introducing this treatment as a non-destructive method. The RMS roughness values for the control, OPT, and OPTV were measured as 20.8216 nm, 21.9147 nm, and 23.9663 nm, respectively. In addition, their average roughness values were obtained as 16.1929 nm, 16.3347 nm, and 20.0409 nm, respectively. A slight increase in roughness parameter values between the control and the plasma-treated samples seems to be attributed to the effect of the mild etching process that led to ablation and cleaning,
57
as discussed in the FESEM+EDX analyses. However, the small difference in value between OPT and OPTV remains unanswered. This slight increase in roughness parameter values can be a reason for their small difference in hydrophilicity. But, it still cannot justify the drastic hydrophilicity difference between the control and them. Busscher et al. remarked that for the average roughness value of 100 nm and above, contact angle variations would be affected by the roughness parameter.
71
Based on this statement, it can be concluded that the obtained values for the roughness parameters cannot adequately justify the surface hydrophilicity enhancement. 3D AFM images of woven PET fabric (a) untreated sample, (b) air plasma treated sample, (c) air plasma treated with water vapor post-exposed sample at 550 W and 15 kV.
Surface chemistry analyses
XPS analysis has a depth of 5-10 nm. As a result, in determining variations in the chemical composition of the outermost layers, this technique is much more sensitive than ATR-FTIR and EDX.72,73 Figure 5(a)–(c)) depicts the overall XPS spectra for the control, OPT, and OPTV samples. The presence of C1s, O1s, and N1s peaks at 285 eV, 533 eV, and 400 eV confirms the presence of carbon, oxygen, and nitrogen elements in the plasma-treated samples. XPS Illustration, overall XPS spectra for (a)the control,(b) air plasma treated in the absence (OPT), or (c) the presence of post-exposure to water vapor (OPTV) woven polyester fabric sample; High-resolution XPS analysis of C 1s peaks of untreated(d), OPT(e), OPTV (f) polyester fabrics; High-resolution XPS analysis of O 1s peaks of untreated (g), OPT (h), OPTV (i) polyester fabrics. High-resolution XPS analysis of N 1s peaks of OPT (j) and OPTV(k) polyester fabrics.
In contrast, only the oxygen- and carbon-related peaks can be observed in the control sample. The intensity of the O1s peak for the control sample is less than that in the two treated samples, and it reaches its maximum for OPT. However, the OPTV air plasma-treated sample exposed to water vapor decreased, yet it was still more than the control one.
Relative Atomic composition and atomic ratios of polyester fabrics determined by XPS.

The atomic percentage of different chemical groups present on the surface of PET woven fabric for deconvolution of C1s, O1s, and N1s high-resolution peaks.

As shown in Figure 5(d)–(f), the C1s peak in the untreated sample can be divided only into three peaks of the C-C/C-H, C-O, and C = O bonds. Then, after plasma treatment, another peak appeared at higher binding energies attributed to the (C = O)-O bond. In the O1s-related spectra, Figure 5(g)–(i), only two bonds of C = O and C-OH can be correspondingly detected in the control sample, while in addition to these two bonds, the O-C = O bond is also observed in the treated samples. This observation confirms the formation of carboxyl groups followed by performing plasma treatment on the surface. In Figure 5(j)–(k), the greater area under the N1s peak in OPT compared to OPTV indicates the decrease in the number of nitrogen groups in OPTV.
Table 6 shows carboxyl groups formed on the surface after OPT. In addition, a comparison between the atomic percentages of the C-OH bonds obtained from C1s and O1s deconvolution revealed that after OPTV, the atomic ratio of the hydroxyl groups on the surface increased significantly. Specifically, the atomic percentage of the C-OH bonds obtained from C1s decomposition increased from about 2.58% for OPT sample to approximately 12.91% for OPTV. In addition, the percentage of the same bond obtained from O1s decomposition increased from about 60.22% for OPT to about 83.46% for OPTV. According to these observations, it can be concluded that the OPT increases the number of carboxyl groups, and in contrast, OPTV raises the hydroxyl groups, improving surface hydrophilicity. Simultaneous surface ablation, cleaning, and functionalization by plasma remove some primary elemental components from the surface by breaking some C-C bonds, removing hydrogen from C-H bonds, and then creating cross-linking and replacing them with oxidation and functionalization products. As a result, an opposite trend in the intensity of both C1s and O1s peaks can be noticeable. Similar observations were reported in the published research papers.9,10,12,14,21,28,29,32,35,48 Since polyester belongs to polymers that contain structurally bonded oxygen in their chain, oxygen can be found in untreated samples. 10 Here, the increase in polar groups justifies the wicking test values.
Regarding the amounts of oxygen and nitrogen received by the surface after plasma treatment, it should be noted that the increase in the amount of nitrogen was much less than oxygen, which is almost surprising mainly because the dominant gas in the plasma environment, i.e., air, is primarily composed of nitrogen. According to the OES results, the intensity of nitrogen species is higher than oxygen ones. The possible reasons for this strange result can be the higher reactivity levels of the reactive oxygen species than those of nitrogen ones, the high dissociation energy of nitrogen molecules, and the lower density of nitrogen atoms than that of oxygen ones.74–76
The intensities in the XPS spectrum provide information on the sample surface's chemical composition and the chemical species' depth distribution. 77 In this regard, the intensity of the oxygen peak observed after OPTV slightly decreased compared to the peak observed after OPT, which is also evident in the reduced area under the diagram. Suppose the intensity decrease is interpreted as a reduction in substance on the sample surface. In that case, it can be attributed to species diffusion, reorientation, and movement towards the bulk interior and near the surface. This phenomenon may indicate the plasticizing effect of water vapor as a universal plasticizer.78–80
In addition to the XPS, analysis of the obtained ATR-FTIR spectra81–83 was used to investigate the functional groups on the surface of the control, OPT, and OPTV samples (Figure 6). Table 7 represents the observed characteristic peaks of polyester used in this article, based on literature. ATR-FTIR spectra for control, air plasma treated and air plasma treated + water vapor post exposed, woven PET fabrics. Wavenumbers and their assigned Characteristic Bands of PET identified by ATR-FTIR.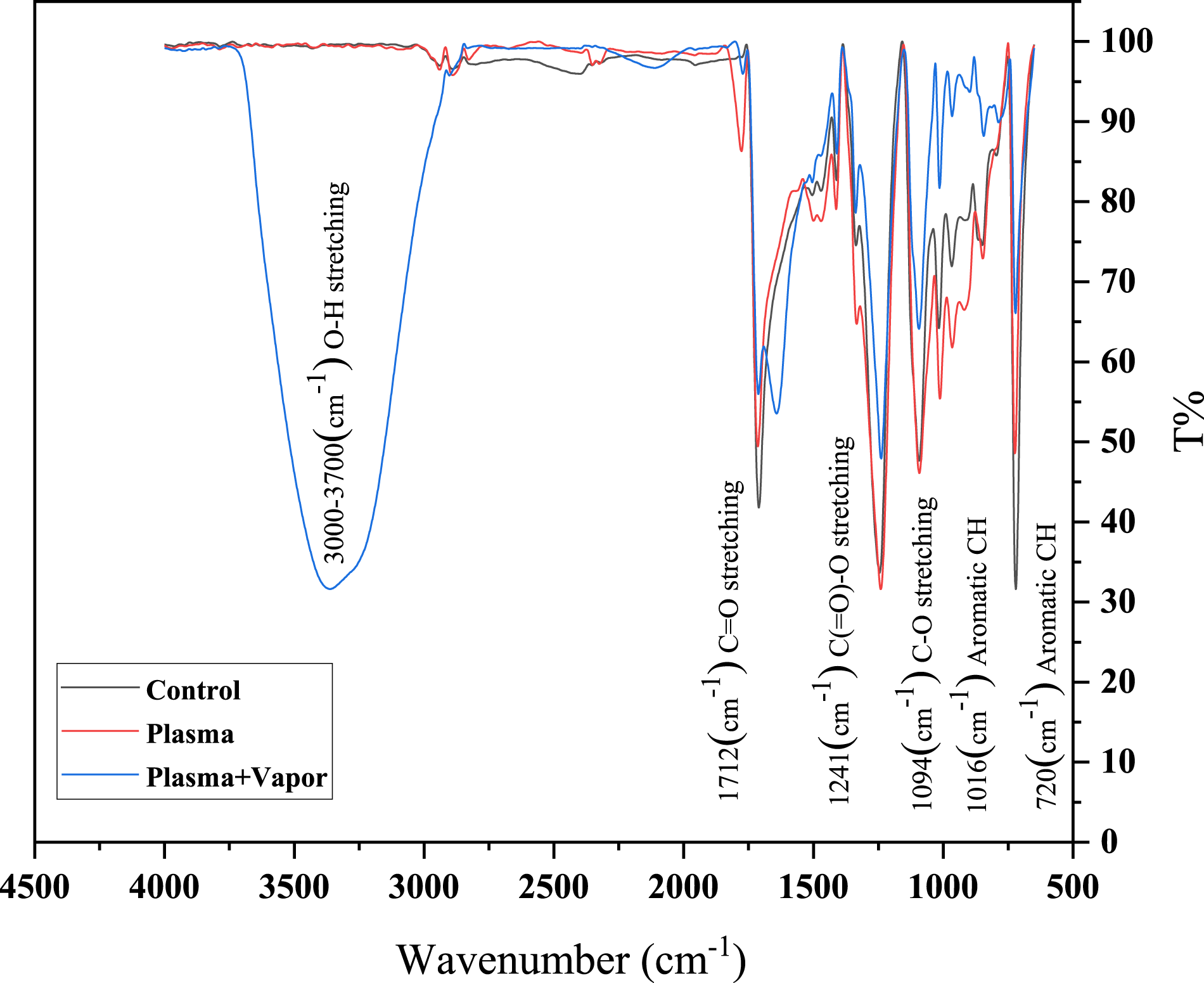

According to Figure 6 and Table 7, the characteristic band of polyester at 720 cm−1, which is attributed to the aromatic CH out-of-plane bending vibrations of benzene rings in the crystalline phase,7,21,84–89 shows the intensity reduction of this crystalline band in the three samples, starting from the control sample, OPT and finally, OPTV which has the least intensity. More specifically, this bending vibration belongs to the wagging type. 89 The absorption region can be attributed to the mentioned band and the C = O out-of-plane bending vibration. 31 The same decreasing trend was observed for the 795 cm-1 and 1712 cm-1 characteristic bands, attributed to C = O rock + - C-O deformation 86 and C = O stretching vibrations, respectively.
Band 795 cm−1 is also attributed to CH ring out-of-plane bending vibrations. 31 The characteristic bands at 847 and 967 cm−1 are representatives of the crystalline phase. 31 The band at 847 cm−1 that results from two neighboring hydrogens in benzene is referred to as the rocking bending vibration for C-H (-CH2-), and that at 967 cm−1 is attributed to 31 trans-C-O stretching + vibrations of ester group and_ in some cases_ to the in-plane bending vibration of the C-H aromatic ring.21,89 The bands at 847, 967, and 1016 cm−1 (with their aromatic ring vibrations) increased their intensity after OPT. However, compared to the control sample, their intensity decreased in OPTV. The band at 1094 cm−1 (with its weak shoulder 1117) is attributed to the C-O stretching vibrations. As observed, it was slightly intensified after OPT but followed by OPTV; it became much less intense than the control band. The intensity of the shoulder at 1117 cm−1, which is representative of the crystalline phase, 31 decreases after OPT and becomes insignificant by OPTV. OPT slightly intensified the bands at 1241 cm−1 (attributed to the C (=O)-O stretching vibrations), 1336 cm−1 (representative of a crystalline phase attributed to the CH2 wagging bending vibration), 1410 cm−1 (with the aromatic ring vibration as a stable group 85 ), 1471 cm−1 (with the crystalline phase 31 that is representative of the CH2 bending vibration), and 1505 cm−1 (attributed to the aromatic ring vibrations); however, their intensity decreased after OPTV, compared to the control sample. The bands at 2940 and 2969 cm−1 belonging to the weak stretching vibrations of the polymer backbone of benzene rings (ν as and ν s −CH2-)88,89 are identified in control and OPT samples; however, they cannot be traced in OPTV mainly because broadband covers them. The formation of C-H radical groups followed by the chain scission process in order of dissociation energy from low to high by plasma on the polyester surface could explain the intensification of some bands after OPT. 88 Also, breaking CH bonds by the active species produced in plasma can decrease the intensity of the characteristic band, for example, the band at 720 cm−1 7. Another weak absorption band can also be detected in the vicinity of 2353 cm−1, which may result from the difference in carbon dioxide concentration in the air inside the infrared cell. 21 In addition, another small peak in all three samples was observed in 1955, which is attributed to the anhydride group. 88 The anhydride formation can be due to the presence of alcohol in the main structure of the polyester, which, due to its ability to react with anhydride and produce ester groups, remains a reactant in polyester. 85 After OPT, the crystalline peaks’ intensities decrease or increase. However, in OPTV, in all cases, they decrease. This may be attributed to a reduction in the crystalline phase of PET's surface atomic layers. Notably, some cases after OPT show a peak shift towards a smaller wave number. This shift may be more observable, followed by OPTV. The peak shift towards a lower wave number indicates an increase in the species' mass and, thus, a lower vibration frequency, which can be attributed to the effect of plasma and its role in the destruction of low molecular weight (LMW) species and/or their transformation into high molecular weight species via cross-linking. This will lead to chain scission and removal of weak boundary layers formed by LMW species, consequently improving surface adhesion. 76 In other words, these shifts are attributed to the interaction of the ester group with the aromatic ring. The crystallinity and molecular orientation of the polymer 90 greatly influence the vibrational spectra of PET. In the present research, no new absorption peaks were detected after OPT and similar behavior was observed after glow discharge plasma treatment of polyester, as reported in. 87 In contrast to the control and OPT samples, the amorphous peak 898 intensity in OPTV, attributed to the Gauche configuration CH2 rock, is visible, confirming the growth of the amorphous structure after OPTV; this growth appears to be beneficial for hydrophilicity.31,86 The band at 1642 cm−1 is another absorption band evident in OPTV due to the H-O-H bending vibration of the absorbed water. 85 In OPTV, there is another broad absorption band in the range of 3700-3000 cm−1 with a peak and shoulder of 3359 and 3288 cm−1, respectively, which belongs to the O-H stretching vibrations,7,31,87,88,90 indicating a strong absorption band from the polar functional group OH, which can be effective in surface hydrophilicity enhancement. For two reasons, ATR-FTIR analysis does not provide us with completely accurate and correct surface chemistry information. First, plasma treatment penetrates at the nanometer scale, whereas FTIR has a micrometer-scale penetration depth. The second reason is the appearance of multiple peaks for a functional group. 48 In this regard, ATR-FTIR can be a complementary method to XPS. According to the XPS results, the oxygenated species peaks for OPTV are less than OPT, which appears to be due to the porosity of the PET fabric and the penetration of water vapor molecules into the depth of the tissue that XPS cannot scan completely but is easily detected in the ATRFTIR spectroscopy. On the other hand, as previously stated, this decrease in intensity can be attributed to species diffusion and reorientation towards the interior parts of the bulk near the surface. The OH band's intensity is vividly reflected in the ATR-FTIR results, whereas that of the other oxygenated species is quite low. Other species' movement appears to have resulted in their placement in points deeper than the ATRFTIR scanning depth or within the depth difference range of ATR-FTIR and XPS. According to the XPS results, the oxygenated species peaks for OPTV are less than OPT, which appears to be due to the porosity of the PET fabric and the penetration of water vapor molecules into the depth of the tissue that XPS cannot scan completely but is easily detected in the ATRFTIR spectroscopy. On the other hand, as previously stated, this decrease in intensity can be attributed to species diffusion and reorientation towards the interior parts of the bulk near the surface. The OH band's intensity is vividly reflected in the ATR-FTIR results, whereas that of the other oxygenated species is quite low. It seems that the movement of other species in the case of vapor post-processing led to their placement in points deeper than the scanning depth of a few micrometers of this analysis or the depth difference range of two ATR-FTIR and XPS scanning.
According to the XPS results, after OPT/OPTV, an increase in the concentration of oxygen-containing species and polar functional groups on polyester surface layers caused an increase in surface hydrophilicity. However, as the findings suggest, one of the reasons for OPTV's slightly higher hydrophilicity than OPT appears to be the crystallinity structure weakening and intensifying the amorphous phases on the polyester surface. This decrease in crystallinity seems to be caused by the plasticizing effect of water vapor. 91 According to one study, cold plasma treatment had little impact on water vapor sorption inside PET fabric, and only minor changes in the kinetics of this process was made. 19 Given the almost dense presence of water vapor to which the sample is post-exposed, it may be possible to propose that water vapor molecules created a polar medium like liquid water molecules, preventing small and fast aging that occurs between the plasma treatment and the hydrophilicity level measurement. 57 However, this idea is valid if we do not consider the oxygen peak intensity criterion in the XPS as an absolute measure of the amount of substance on the surface or do not compare it with another sample; hence, we only rely on the comparison with the species within each sample. Another reason for the difference between the hydrophilicity levels in OPT and OPTV seem to be the presence of OH groups. Specifically, in OPTV, the bands related to hydrogen bonds will intensify due to the absorption of water molecules. The XPS results also justify this increase in the hydroxyl groups on the surface. Apparently, in OPT, the intensity of the carboxyl groups is higher than hydroxyl groups, probably meaning that hydroxyl functional groups have a more significant effect on the surface polarity and wettability. It should be noted that the presence of water molecules appears to strengthen the dipole-dipole interaction caused by hydrogen bonding, resulting in a higher level of hydrophilicity for OPTV.
Color performance
Figure 7(a)-(d) show the amounts of magenta color settling on the polyester surface and anti-bleeding performance in the presence of three colors, magenta, black, and cyan. As observed in Figure 7(a), the amount of ink deposition in the control sample is less than in the rest. OPTV exhibited a better color deposition among the two plasma-treated samples (Figure 7(b) and (c)). The same improvement trend for anti-bleeding can also be detected with clear boundaries. The lowest color performance level of control is mainly due to its hydrophobicity nature. The enhancement trend for treated samples agrees with the hydrophilicity, surface chemistry, and surface morphology tests, increasing the adhesion and speed of ink absorption on the surface and the woven polyester surface's ability to fix the ink particles. Moreover, the used ink is water-based, and its dispersants enhance the hydrophilic properties of the pigments
92
; therefore, as a result of post-processing with water vapor, the interaction of intermolecular hydrogen bonds between the water-based ink and water molecules deposited on the surface of the woven polyester contributes to the intensity of adhesion. In addition, the porosity in the woven polyester used in this experiment increases the color amount and intensity upon increasing their contact in OPT and OPTV compared to the control sample. Images of qualitative evaluation for magenta color settling and the anti-bleeding performance of printed colors: magenta, cyan, and black on the untreated (a, d) air plasma treated (b, e) air plasma treated + post-exposed to water vapor (c, f) PET fabrics at optimum conditions for hydrophilicity (15 rounds and 2 m/min velocity). (Upper row from left to the right: a, b, c, respectively. Lower row left to the right: d, e, and f).
Quantitative evaluation for the color performance of untreated, air plasma treated, and air plasma treated with water vapor post-exposed polyester fabric printed with Cyan, Magenta, and Black (Key) colors.

Conclusion
This study suggests DBD roll-to-roll atmospheric pressure air plasma treatment with or without post-exposure to water vapor as a novel, green, economic pretreatment for woven PET fabrics. This pretreatment can be used in line and improve the hydrophilicity, color performance, and direct sublimation inkjet printing quality. Due to the novelty of this work and the complicated mechanisms of surface chemistry, noticeable results have been concluded, including: Cleaning and mild ablation in this pretreatment not only lead to decontamination and complementary de-sizing of fabric surface but also improve its hydrophilicity. Morphological and topographical changes made by plasma treatment are negligible and suggest this method is a non-destructive treatment. Also, the chemical modification aspect of plasma treatment plays a key role in the enhancement of hydrophilicity and printing quality compared to the physical-morphological aspect of this pretreatment. Besides, changes in surface crystallinity and amorphous phases of woven PET fabrics seem to be effective in mentioned enhancements. Incorporation of oxygenated species, particularly hydroxyl groups, affects hydrophilicity.
These results need more studies to delve into their complicated nature and clarify them. According to this study, the efficacy of DBD roll-to-roll atmospheric pressure plasma treatment for woven PET fabric is unquestionable. Water vapor post-exposure can play a complementary role considering different desirable conditions by a decrease in absorption time from 12051 to 202 s and an increase in the color depth to 12.49%, 25.89 and 18.07% for Cyan, Magenta and Key (black) respectively. This green, non-destructive method can be used inline, facilitating its industrial applications.
Footnotes
Acknowledgements
We gratefully acknowledge the financial support of the Iranian National Science Foundation (INSF) (Grant/Award Number: 99000336). We thank all the Plasma Chemistry Lab members at Laser and Plasma Research Institute.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Iranian National Science Foundation (INSF) (Grant/Award Number: 99000336).


