Abstract
Antibiotics released in the water pose a serious threat to human and ecological health. Therefore, it is of great importance to effectively remove antibiotics from wastewater. In this study, recyclable bacterial cellulose/sodium alginate/zeolitic imidazolate framework-8 (BC/SA/ZIF-8) aerogel composites were prepared by chemical cross-linking and in situ growth in freeze-drying. The successful preparation of the composite aerogel was confirmed by scanning electron microscope (SEM), Fourier Transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) analysis. The results reveal that BC/SA/ZIF-8 aerogel composite has significantly high BET specific surface area of 165.24 m2/g than that of BC/SA at 7.39 m2/g. The BC/SA/ZIF-8 composite aerogel exhibited excellent adsorption performance for tetracycline hydrochloride, with a maximum adsorption capacity of 746.27 mg/g. The adsorption process followed the pseudo-second-order kinetic model and Langmuir adsorption isotherm model. Moreover, the reusability test on BC/SA/ZIF-8 composite aerogel revealed an insignificant decline in adsorption after six cycles. Therefore, BC/SA/ZIF-8 composite aerogel has a high potential as a novel and recyclable adsorbent for efficient removal of tetracycline hydrochloride from water.
Introduction
For many years, antibiotics have been widely used in medical and agricultural activities. 1 Despite the positive applications of antibiotics, they have threatening effects on the environment, which have caused widespread concern among scholars domestic and overseas. 2 In most cases, the antibiotics used will be excreted through the active form and appear in the drinking water, which will not only destroy the balance of the human flora, but also destroy the ecological balance. 3 Such antibiotics include mostly-used Tetracycline hydrochloride (TC) which causes devastating harm when accumulated in the human body. Hence, it is necessary not to underestimate the side effect of tetracycline hydrochloride.4,5 In recent years, various methods for the remediation of wastewater from pollutants have been explored. These include biodegradation and bioadsorption,6,7 physical adsorption, 8 oxidation reduction method, 9 photocatalytic degradation,10–13 ionic resin and membrane filtration.14,15 Adsorption method is noted as one of the most effective methods to remove various organic pollutants in water due to its advantages in operation cost, simplicity, removal efficiency and practicability.8,16,17 Various adsorbents such as activated carbon, carbon nanotubes, 18 graphene oxide, 19 zeolites, 20 porous resins, 21 and metal oxides 22 have been used to adsorb and remove antibiotics.
Metal-organic frameworks (MOFs) is a new kind of crystalline porous material which has found significant use in the removal of organic pollutants. A variety of MOF materials have been used in the field of adsorption such as University of Oslo-66 (UiO-66) MOFs, and Materials of Institute Lavoisier (MIL) MOFs.23,24 Zeolite imidazolate frameworks (ZIFs), possessing the advantages of both ZIFs and MOFs, finds wide application in water treatment because of its excellent crystallinity, porosity and surface area. 25 ZIF-8 is a relatively common kind of MOFs. ZIF-8 can not only be synthesized simply at room temperature, but more importantly, it has the macromolecular rotation gate effect and can adsorb substances larger than its own pore size. 26 It is one of the characteristics of ideal materials for the adsorption of antibiotics in wastewater. Wu et al. reports an extensive improvement in adsorption capacity of ZIF-8 for TC (adsorption kinetic studies was 124.6 mg g−1 at 25°C temperature). 27 Peng et al also synthesized hierarchically porous ZIF-8 adsorbents assisted with sodium dodecyl sulfonate through a facile one-pot self-assembly method. 28 However, the recovery performance of ZIF-8 is poor, and it is easy to agglomerate and cannot be easily reused, which greatly limits its further application in the field of adsorption. Therefore, it is very important to find suitable substrates to enable it to be applied in the adsorption of antibiotics in wastewater and enhance its recovery ability.
Inspired by excellent biocompatibility, non-toxic, fine network structure, high crystallinity, high swelling capacity, bacterial cellulose (BC) synthesized from Acetobacter xylinum has various applications as adsorbent due to its significant advantage over common plant cellulose. 29 BC also shows stability to change in pH value and is widely used in fixed guest materials. 30 Sodium alginate (SA), a natural polysaccharide derived from seaweed, possesses unique hydrogel properties and high biocompatibility, is an ideal filler for polymer nanocomposites. 31 Series of literature reports the effective application of SA based composite for removing pollutant from wastewater via adsorption.32,33 Although composites consisting of alginate and BC have been successfully synthesized, 34 bacterial cellulose/alginate (BC/SA) composite aerogel have rarely been reported as recyclable adsorbents for wastewater treatment. As a result, developing a lightweight and mechanically flexible bacterial cellulose/sodium alginate aerogel as a carrier, with ZIF-8 will not only grant good adsorption but also improve the recyclability of the adsorbent.
In this work, BC/SA aerogel was first synthesized via blending and crosslinking of alginate and calcium ions (Ca2+), followed by freeze-drying to remove water in the system to form an open and interconnected porous structure. 35 Subsequently, ZIF-8 was grown in situ on BC/SA sponge, resulting in BC/SA/ZIF-8 complex sponge. The practicability of composite aerogel as adsorbent was characterized and studied using adsorption of TC. The effects of MOF growth time, adsorbent dosage, pH value, adsorption time, initial concentration and temperature on the experimental results were investigated. By fitting the adsorption kinetics and isotherm experimental data, it was found that the Langmuir isotherm model better describes the isotherm data and the adsorption process of TC occurs mainly as a monolayer adsorption on the homogeneous surface of the composite aerogel.
Experimental
Materials
BC was prepared in-house (Anhui Polytechnic University, China). Zinc nitrate Hexahydrate (Zn(NO3)2·6H2O, ≥99%), 2-Methylimidazole (C4H6N2, >98%), Tetracycline Hydrochloride (C22H24N2O8, ≥96%), absolute methanol, tryptone, D-mannitol and SA were purchased from Aladdin Biochemical Technology Co., Ltd (Shanghai, China). Yeast extract was purchased from Thermo Fisher Scientific (Waltham, Massachusetts, USA). All the chemicals used were of analytical grade.
Preparation
BC pellicles
The synthesis method of BC was based on previous reports with some modifications.
36
In a typical procedure, D-mannitol (25 g), trypsin (5 g) and yeast (3 g) extracts were dissolved in 1000 ml of deionized water and the medium was further sterilized in an autoclave at 121°C for 30 min. Subsequently,
BC/SA aerogel
SA solution (0.33%, w/v) was prepared by dissolving SA in deionized water. At the same time, BC was prepared into uniform suspension (1.5% w/v) by high shear emulsifier, and the prepared SA solution and BC suspension were mixed according to the mass ratio of 3:7 (30% addition of sodium alginate/bacterial cellulose composite aerogel has more advantages in biocompatibility, water absorption, and structural stability). 31 The mixture was poured into silicone molds and freeze-dried to prepare uncross-linked BC/SA aerogel. Afterwards, BC/SA aerogel was fully cross-linked in 1.4% w/v calcium chloride solution (optimal concentration for maximum mechanical strength of calcium alginate gel) 36 for 24 h. BC/SA aerogel was obtained by repeatedly rinsing the residual calcium chloride with deionized water and vacuum freeze-drying.
In situ growth of ZIF-8 on BC/SA aerogel
The prepared BC/SA aerogel (60 mg) was added to the mixture containing 30 ml methanol and 1.5 g Zn(NO3)2·6H2O until completely swollen, and methanol (70 mL) and 2-Methylimidazole (3.3 g) were added to the mixture under continuous magnetic stirring. The mixture was allowed to react for 6 h under magnetic stirring, and then the gel was washed with methanol several times to remove the remaining chemical substances. Finally, the gel was freeze-dried for 12 h to obtain white composite aerogel. The fabrication process of BC/SA/ZIF-8 is shown in Figure 1: the yellowish-gold conical flask represents the culture flask of acetobacter xylinum; the BC represents the obtained bacterial cellulose fiber membrane; the BC/SA and BC/SA/ZIF-8 represent BC/SA aerogel and BC/SA/ZIF-8 composite aerogel, respectively. (a) Schematic fabrication process of BC/SA/ZIF-8 aerogels. (b) Pure bacterial cellulose membrane. (c) BC/SA/ZIF-8 composite aerogel.
Characterization
The morphology of the samples was studied by scanning electron microscope (Hitachi S-4800, Japan, Current 10 μA, voltage 5 kv). The crystal structure and chemical composition of samples were analyzed by X-ray diffraction (Shimazu XDR-6100, Japan, 2θ range 5–40°) and Fourier transform infrared spectrometer (Shimadzu IRPrestige-21, Japan, at the range of 4000–800 cm−1), respectively. In addition, the specific surface area was measured using BET analyzer (Micromeritics ASAP 2460, USA). Prior to surface area analysis, the samples were degassed at 120°C for 3 h. Thermal gravimetric analyzer (TGA550, TA Instruments Corporation, USA) was used to analyze the thermal stability of samples at a heating rate of 10°C/min, temperature range from 50 to 800°C under N2 atmosphere. UV–visible spectrophotometer (UVmini-1285, SHIMADZU, Japan) were utilized to analyze the content of TC.
Adsorption measurements
The kinetic model and isotherm parameters were obtained by batch adsorption experiments. BC/SA/ZIF-8 aerogel (0.05 g) was dispersed into 80 mL TC solution with a concentration of 60 mg/L. The subjects were placed in a shaker at 30°C at 120 rpm. The effects of reaction time, pH, temperature and the amount of adsorbent on the adsorption experiments were studied in detail. Experimental assessment of each sample was performed three times. Subsequently, the supernatant was taken and the antibiotic residues were analyzed by UV-Vis spectrophotometer (SHIMADZU UVmini-1285). The adsorption capacity qe of TC was measured using the following equations.
37


The recyclability of the aerogel composite was examined. After every adsorption cycle, the aerogel composite was added to 75% ethanol to oscillate and desorb TC, and freeze dried overnight (the hydrogen bond between the hydroxyl group of ethanol and TC molecule is stronger than the force between the adsorbent surface and TC, 38 enabling quick separation of TC molecule and aerogel composite).
Results and discussion
Morphological characteristics
The SEM images of pure BC, BC/SA and BC/SA/ZIF-8 aerogels are presented in Figure 2. The observations from Figure 2(a) indicate that the nanofibers of BC exhibit a three-dimensional porous structure, which arises from the sublimation of ice crystals after irregular growth during the freeze-drying process. However, as shown in Figure 2(b), the surface pores of BC/SA aerogel are partially clogged with lamellae, due to the penetration of SA gel between BC fiber bundles, leading to partly occlusion of its pores. Notably, Figure 2(c) and 2(d) reveal that nanoparticles grow uniformly on the surface of BC/SA/ZIF-8 aerogel, and these nanoparticles possess a regular polyhedral structure with 0.3 µm average particle size, consistent with the previously reported morphology of ZIF-8.
39
SEM observations confirm the successful synthesis of BC/SA/ZIF-8 aerogel. SEM images of (a) pure BC aerogel; (b) BC/SA aerogel; (c) BC/SA/ZIF-8 aerogel; (d) enlarged view of BC/SA/ZIF-8 aerogel (the inset is the particle size distribution of ZIF-8 particles).
FTIR analysis
As shown in Figure 3(a), FTIR spectra of BC, BC/SA, BC/SA/ZIF-8, and ZIF-8 were recorded in the wavenumber range of 4000–800 cm−1 to buttress the successful synthesis of aerogel composites. Notably, unlike to pure BC, the BC/SA band at 1647.6 cm−1 is attributed to the carboxyl group on the alginate. Furthermore, the FTIR spectrum of BC/SA aerogel is similar to that of BC/alginate nanocomposite beads reported in the literature.
40
The broad band centered around 3390.3 cm−1 is attributed to the stretching vibration of -OH, which may be due to the adsorbed water. The band at 2932.3 cm−1 is assigned to stretching of C-H.
41
The absorption band at 3127.8 cm−1 is attributed to C-N stretching on the ZIF-8 imidazole ring and the band at 1589.3 cm−1 corresponds to the C = N stretching vibration.
42
The adsorption peaks in 600–1500 cm−1 were assigned to the bending vibration mode of the imidazole ring.
28
The results indicate that the BC/SA/ZIF-8 composite aerogel retains the characteristic peaks of ZIF-8 nanoparticles and BC/SA, in agreement with the XRD results. Therefore, these results indicate that the synthesis of BC/SA/ZIF-8 composite aerogels was successful. (a) IR spectra of BC, SA, and BC/SA aerogel composites; (b) XRD patterns of BC, BC/SA, and BC/SA/ZIF-8 aerogels.
XRD analysis
Figure 3(b) shows the XRD patterns of BC, BC/SA, ZIF-8, and BC/SA/ZIF-8. Three main peaks at 14.5°, 16.6°, and 22.5° appeared in BC aerogel, representing the (1–10), (110), and (200) crystal planes of cellulose I in BC, respectively.
43
However, crystallinity of BC/SA aerogel was significantly lower than that of pure BC. This implies that the original long nanofibrils of BC were cut into shorter nanofibers by the high-shear emulsifier, so the linear polymer of
TGA analysis
The thermal properties of the aerogels and the loading rate of ZIF-8 were examined and presented in Figure 4. Apparently, significant heat loss was observed at 250°C on BC/SA/ZIF-8 and BC/SA and 600°C on ZIF-8, corresponding to the thermal decomposition of BC and ZIF-8, respectively. BC/SA and BC/SA/ZIF-8 have similar thermal decomposition curves; however, the thermal stability of BC/SA/ZIF-8 is significantly higher than that of BC/SA (as seen in the region of 400–800°C), and it had a higher residual char content. The good thermal properties of ZIF-8 in the composite aerogel is related to the production of ZnO, which promote the formation of residual char and thus improved the thermal stability.
46
The incorporation of ZIF-8 in BC/SA/ZIF-8 enhanced its thermal stability compared to BC/SA aerogel. The loading rate of ZIF-8 is calculated by the following equation: TGA curves of pure ZIF-8 powders, BC/SA, BC/SA/ZIF-8 aerogel.

N2 adsorption-desorption isotherms
The Brunauer–Emmett–Teller (BET) specific surface area and porous structure of the aerogel composites were assessed since it is an influential factor that affects adsorption performance. As shown in Figure 5(a), BC and BC/SA aerogels show obvious adsorption hysteresis under high pressure, indicating that the adsorption mechanism on their surfaces is capillary condensation of mesopores or macropores.
38
The specific surface area of BC/SA aerogel gel is 7.3905 m2/g, slightly higher than that of pure BC aerogel (6.2415 m2/g). This is attributed to the open and interconnected macroporous structure of BC/SA aerogel.
48
Nevertheless, the specific surface area of BC/SA/ZIF-8 composite aerogel is up to 165.2471 m2/g, which are almost 22 times that of BC/SA aerogels. Obviously, ZIF-8 crystal (1061.0 m2/g)
45
has a decisive influence on the specific surface area of BC/SA/ZIF-8 aerogel composite. Furthermore, according to the pore size distribution curve (Figure 5(b)), it can be found that the main pore size of BC/SA/ZIF-8 is about 4 nm, indicating the presence of mesopores.
49
(a) Nitrogen adsorption-destruction isotherms of BC, BC/SA and BC/SA/ZIF-8. (b) Pore diameter distribution curve.
Adsorption properties of composite aerogel
Influence of ZIF-8 loading time on adsorption effect
The time of in situ growth of ZIF-8 on fibers has an important effect on the adsorption effect of composite aerogel. According to published literature, ZIF-8 nucleates after 1 h of growth and the relative crystallinity of ZIF-8 increases with time within 24 h of nucleation. As shown in the Figure 6(a), the influence of different in situ growth times on the adsorption effect of TC was studied, BSZ1, BSZ2, BSZ6, BSZ17 and BSZ24 represent the in situ growth time of 1 h, 2 h, 6 h, 17 h and 24 h of aerogel composite, respectively. Obviously, with the increase in synthesis time, the removal rate of TC gradually increased. However, after loading ZIF-8 on the BC/SA aerogel for 17- and 24-h (BSZ17 and BSZ24), the adsorption rate reduced. This could be due to the aggregation of ZIF-8 on the surface of the aerogel. Therefore, BSZ6 is relatively considered the best performing adsorbent. Vividly, increase in adsorption rate was observed as the synthesis time increases, but reached a plateau on BSZ6 around 120 min. In other words, no obvious increase in adsorption was observed after 120 min. This could be due to the saturation of active sites of the adsorbent with TC pollutant. Considering the adsorption efficiency of the adsorbent, the synthesis time of 6 h (BSZ6), was selected as the best reaction time in the subsequent experiments. Effect of ZIF-8 loading time on TC adsorption (a), adsorption capacity at different solution pH (b), adsorbent dose on adsorption (c), and temperature on adsorption capacity (d): The concentration of contaminants was 60 mg/L, contact time 360 min at 150 rpm, and temperature of 30°C.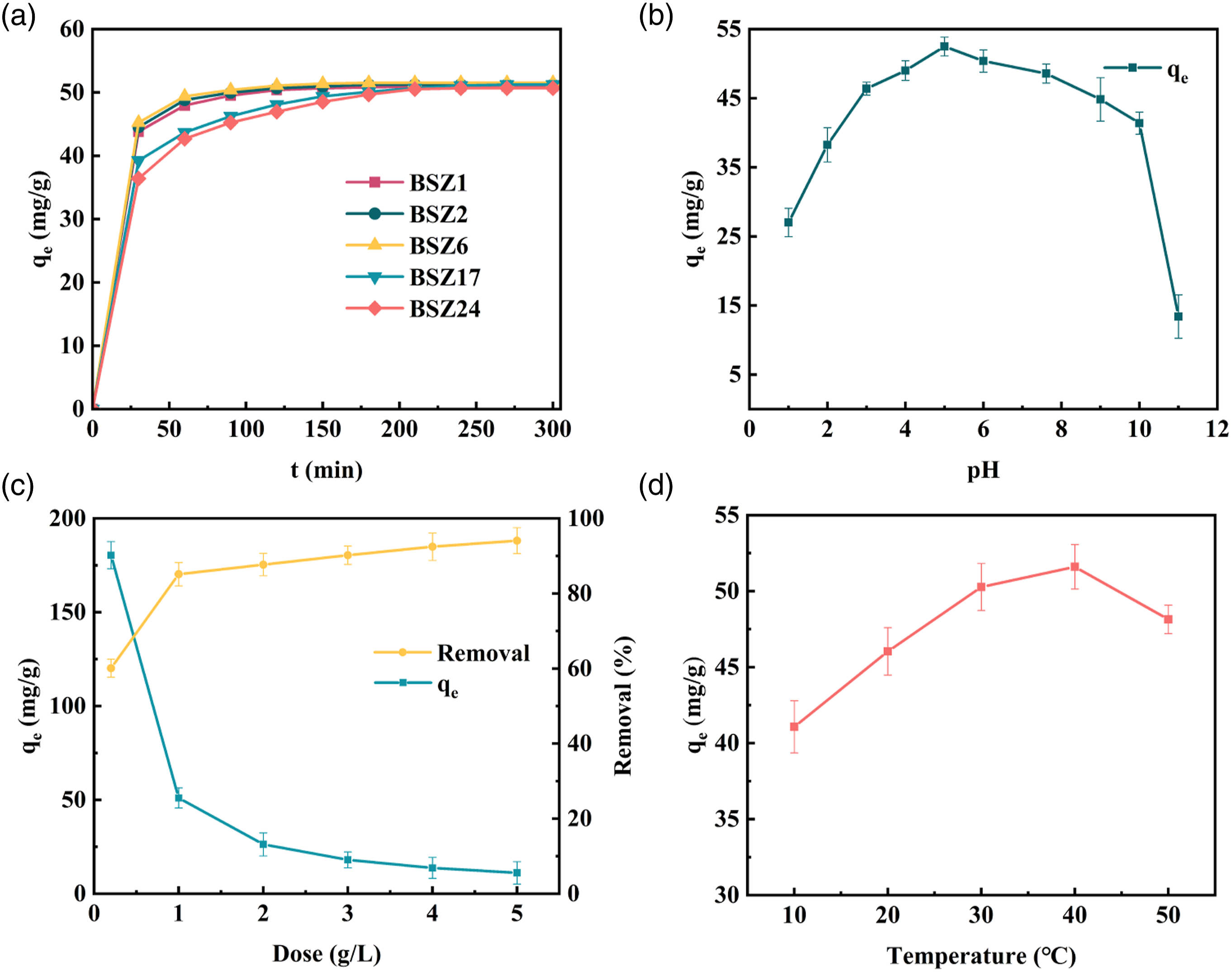
Effect of pH
The pH value has a strong effect on the adsorption process, because pH affects the surface charges of the adsorbent and the adsorbed substance. Therefore, the effect of pH on the adsorption process was studied. As shown in Figure 6(b), the adsorption experiment of TC was carried out in the pH range of 1–11, having 150 rpm at 30°C. Obviously, the adsorption capacity of aerogel composite increases with the increase in pH within the range of 1–5. However, the adsorption capacity gradually decreased when the pH increased above 5.0. This phenomenon can be explained by the fact that ZIF-8 has a point of zero charge (pHpzc) around pH 3.9. 45 Usually at pH <3.34, 3.34 <pH <7.69, 7.69 <pH <9.69, pH >9.69, TC exists as TCH+3, TCH20, TCH− or TC2−, TC2−. 50 When pH is less than 3.9, the surface of ZIF-8 is positively charged, resulting in electrostatic repulsion of TC. However, when pH >3.9 is used, TC mainly exists in the form of TC20, generating electrostatic attraction with negatively charged surface adsorbent, which is conducive to adsorption. In addition, when pH >10, TC exists in the form of TC2- and produces electrostatic repulsion with negatively charged adsorbent, resulting in decreased adsorption capacity. Based on the above experiments, pH = 5 is the optimal pH value for this experiment.
Impact of adsorbent dose
The effects of adsorbent dose on adsorption capacity and removal efficiency were studied in the range of 0.2 ∼ 5 g·L−1. It can be observed from Figure 6(c) that with the increase in the amount of adsorbent, the removal rate of TC increases from 60.1 ± 2.4 to 94.1 ± 0.78, and the adsorption amount qe decreases from 180.44 ± 7.205 to 11.13 ± 0.208 mg·g−1. This implies that the high adsorbent dose provides more active binding sites, making TC easier to combine with functional groups, thus improving the removal rate. However, with the increase in adsorbent dose, the adsorption capacity of aerogel composite decreases. On one hand, the number of unoccupied active sites increases. On the other hand, the active sites on the adsorbent are easy to aggregate or overlap and the length of diffusion path increases, leading to the decrease in the surface area of the adsorbent, which is associated with the decrease in adsorption capacity. 51 Further increase in adsorbent metering had no significant effect on the removal of pollutants. To sum up, considering the adsorption effect and economic benefits, 1 g·L−1 was selected as the best dose of adsorbent in subsequent experiments.
Impact of temperature
Temperature is a crucial factor that influences TC adsorption on adsorbent. The adsorption capacity of composite aerogel was investigated under different temperatures, as shown in the Figure 6(d). The findings illustrate that the adsorption capacity of TC rises with increasing temperature within the range of 10 to 40°C, with the highest adsorption at 40°C, which is considered as optimal temperature for TC adsorption. Nonetheless, when the temperature goes beyond 40°C, the adsorption capacity for TC declines. This can be attributed to weakened interaction between TC and adsorbent due to the continuous enhancement of molecular thermal motion. Consequently, 40°C is the optimal solution temperature for good adsorption of TC by BC/SA/ZIF-8.
Impact of contact time and kinetic study
The effect of contact time on the adsorption capacity of TC was investigated under the conditions of 30°C, 150 rpm and 60 mg/L TC concentration. As shown in Figure 7(a), the reaction reached 87% of the total adsorption capacity in 60 min. This implies that the abundant pore structure on the surface of the composite aerogel provided a large number of adsorption sites for TC at the initial stage of the reaction. However, with the progress of the reaction, the blank active sites available for binding were greatly reduced, resulting in the slowing down of the adsorption rate. Obviously, the adsorption reaction is basically in equilibrium after 300 min. The adsorption kinetics of the reaction was studied to propose the adsorption mechanism. Therefore, the experimental data were fitted by two dynamic models, Pseudo-first-order (PFO) and Pseudo-second-order (PSO). The fitting model was expressed in Figure 7(b) and 7(c), and the calculation formula was represented by equations (4) and (5), respectively.
52
The effect of contact time on the equilibrium adsorption capacity of TC on BC/SA/ZIF-8 composite aerogel (a), Pseudo first order kinetics (b) and pseudo second order kinetics (c) of TC adsorption by BC/SA/ZIF-8 aerogel. Effect of TC initial concentration on equilibrium adsorption capacity of BC/SA/ZIF-8 aerogel composite (d). Langmuir isotherm (e) and freundlich isotherm (f) of TC adsorption by BC/SA/ZIF-8 aerogel composite. Kinetic fitting parameters of TC adsorption by BC/SA/ZIF-8 composite aerogel.



TC Adsorption isotherms
The concentration of adsorbed substrate is an important experimental parameter and one of the main driving forces in the adsorption process. This experiment explored the effect of TC initial concentration of 220 mg/L, 400 mg/L, 600 mg/L, 800 mg/L, 1000 mg/L, 1200 mg/L on adsorption capacity at 30°C (Figure 7(d)). Comparatively, the adsorption capacity of aerogel composite increases dramatically at the initial stage in the same temperature condition with the increase in TC concentration. This phenomenon can be explained by the increase in the driving force of concentration gradient to promote the transfer of pollutants from solution to the active site on the surface of the adsorbent. 53 However, as TC concentration increases to 800 mg/L, the adsorption capacity increases slowly due to the saturation of the active site of the adsorbent, implying the adsorption reaction nearing equilibrium state.
The analysis of adsorption isotherms is essential for understanding the adsorption process. The results of adsorption isotherms can provide information about the interaction between the adsorbent and adsorbate. The Langmuir isotherm model is suitable for surfaces with uniform adsorption capacity and monolayer adsorption, while the Freundlich isotherm model is suitable for multilayer adsorption with non-uniform surface distribution of adsorption heat and affinity. Therefore, the experimental data were fitted into Langmuir and Freundlich isothermal models. The linearized equations for the Langmuir isotherm and the linearized form of the Freundlich isotherm equation were expressed as equations (6) and (7), respectively.
54


Adsorption isotherm parameters of TC by BC/SA/ZIF-8 aerogel composite.

Comparison of TC adsorption capacity of BC/SA/ZIF-8 with other adsorbents reported in the literature.

NB: UiO-66/PDA/BC - UiO-66/polydopamine/BC composite; SA/CMC/CuMOF – sodium alginate/carboxy-methyl cellulose/cu-based metal-organic framework composite.
Reusability of BC/SA/ZIF-8 aerogel composite
It is well known that the reusable performance of adsorbent is one of the important indicators for evaluating the comprehensive performance of adsorbent. The cyclic experimental results of TC removal rate and adsorption capacity of composite aerogels are shown in Figure 8. Obviously, after six cycles, the removal rate of adsorbent decreased from 85.1% to 76.8%, mainly due to the reduction of adsorption active sites on the surface of composite aerogels after repeated elution. The experimental results demonstrate that the adsorbent used in this experiment has excellent reusable performance. The good recyclability of the BC/SA/ZIF-8 aerogel is related to stability of the BC/SA carrier, and the strong interaction/cross-linked of ZIF-8 to the BC/SA carrier. Cyclic stability of TC adsorption by BC/SA/ZIF-8 aerogel composite.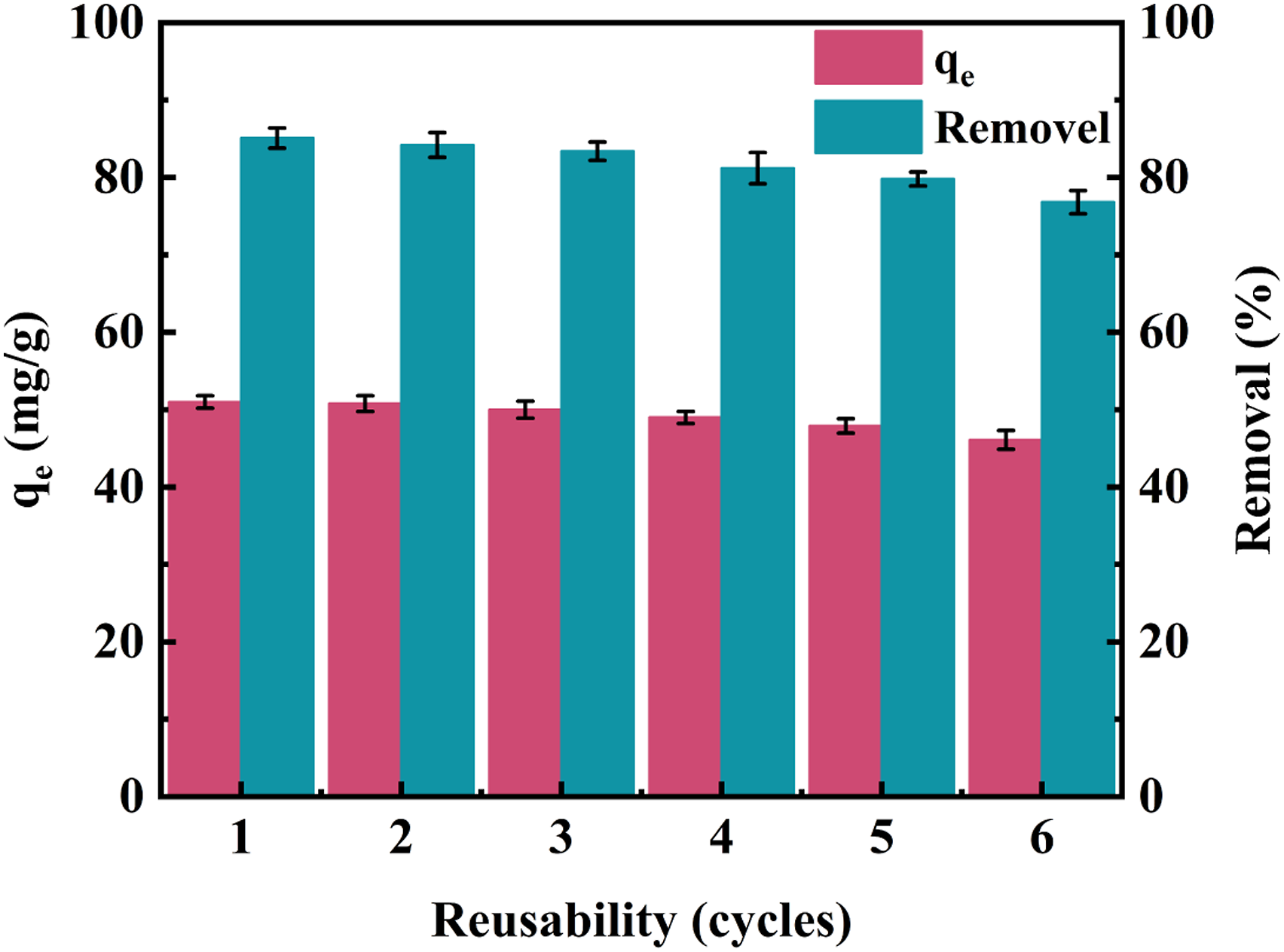
Conclusion
In this study, a flexible and lightweight aerogel carrier was prepared by incorporating BC into sodium alginate, and ZIF-8 was loaded onto the aerogel via in situ growth to achieve a recyclable adsorbent. The experimental results revealed that the optimal pH and temperature for TC adsorption by the aerogel composite were 5 and 40°C, respectively, with a maximum adsorption capacity of 746.27 mg/g. Notably, 87% of the total adsorption capacity was achieved within 60 min. This is attributed to the rich porous structure of the BC/SA surface and the excellent adsorption ability of ZIF-8. 47 The adsorption kinetics and adsorption isotherm model studies showed that the adsorption process of TC by aerogel composites follows a pseudo-second-order kinetic model, and the Langmuir adsorption isotherm model better describes the isotherm data, indicating that the adsorption process of TC is mainly monolayer adsorption on the homogeneous surface of the composite aerogel. 45 Moreover, the aerogel composite can be easily desorbed using ethanol and reused, with only an 8.3% decrease in the removal rate of TC after six cycles. Therefore, the BC/SA/ZIF-8 aerogel composites have great potential for efficient adsorption of tetracycline hydrochloride in aqueous environment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Funding from Anhui Polytechnic University; 2019YQQ008, Xjky03201901, Anhui Province Key Research and Development Plan Project; 2022107020006, Financially Supported by Scientific Research Project of Anhui Provincial Education Department; 2022AH050990, Advanced Fiber Materials Engineering Research Center of Anhui Province; 2023AFMC06 and 2023AFMC16, Natural Science Foundationof Anhui Province; 2008085ME139, the joint Fund Project of Anhui Provincial Textile Engineering Research Center and “Textile Fabric” Anhui Provincial key laboratory; 2021AETKL07.
