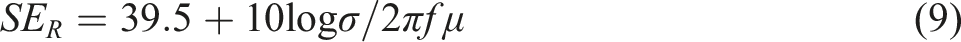Abstract
Nowadays, the research on wearable electronics have received tremendous attraction because of their potential applications in personalized health monitoring and treatment, energy conversion and storage, and human-machine interface system. Herein, we report a facile route for the fabrication of electrically conductive cotton fabric with excellent joule heating and high electromagnetic shielding performances using graphene oxide (GO) and silver nitrate (AgNO3). The GO used in this study is exclusively synthesized from spent batteries in order to minimize the environmental pollution. The surface morphology, elemental analysis, electrical conductivity, thermo-heating behavior and electromagnetic shielding performance have been studied systematically. Due to the high electrical conductivity, the GO-Ag coated cotton with 5 wt% of GO reached high surface temperature of 117.8°C within 35 s, and also it exhibits high electromagnetic interference shielding efficiency value of 79.08 dB. The high flexibility, excellent conductivity, electromagnetic shielding efficiency and joule heating performance of GO-Ag coated cotton fabric suggesting that the GO synthesized from spent batteries will be a potential and valuable resource for the new generation of wearable electronics.
Keywords
Introduction
Over the past few decades, energy storage technology has become a need for day-to-day existence. An energy storage system is a key component of harvesting energy from various sources and converting it into electrical energy. 1 In contrast to the plethora of available energy storage technologies, batteries are incredibly versatile, require little maintenance, and are considered to be one of the least expensive methods.2,3 Among the various types of batteries, one of the oldest and most widely used batteries are zinc-carbon batteries. 4 These batteries also are referred to as house-hold batteries because they are used as energy source for operating TV remotes, flashlights, remote controlled toys (RC toys), radio receivers and so on. Despite their applications, these batteries are limited to their single use.5,6 The spent zinc-carbon contains 20% of manganese, 20% of Zinc, 20–30% of graphite and small amount of plastics, electrolyte and other separator materials. 7 According the reports, approximately 13,000 tons of spent batteries have been disposed as a garbage in South Korea. 8 Among them, 558 tons are zinc-carbon batteries and secondary alkaline manganese batteries. 9 Moreover, the disposed battery wastes do not undergo natural degradation and cause serious environmental issues due to the presence of metallic contents, which are hazardous to environment as well as human health.10–12 Currently these spent batteries are considered as general waste, and the cost for safe disposal of these kind of batteries are quite high. Thus, most of the battery wastes are dumped in landfills or incinerated. The metal ions present in spent batteries will pollute the groundwater if disposed of in landfills, and the incineration of spent batteries will emit harmful gases. 6 Thus it is necessary to develop alternative method to minimize the environmental issues and recycle the metal sources such as Zn, MnO2 and other carbonaceous materials.
Many methods are available for the recycling of valuable materials of spent batteries. 13 The metal/metal oxides which recovered from spent batteries are mainly used for industrial applications, and they can also be used as effective catalyst for the removal of organic pollutant. Other than these metal/metal oxides, the graphite recovery from spent batteries has been received considerable attention due to their immense applications in various fields. 14 Graphite is main source materials for the synthesis of graphene oxide, carbon dots, graphene sheets and so on. Among the various type of carbon materials, graphene is mainly composed of sp2-hybridized carbon atoms which are hexagonally arranged in two dimensional layer, and results in high surface in both planar axes. 15 Graphene materials has significant applications in biomedical field, sensor, composites, etc., due to its unique chemical and physical properties such as high specific surface area (2600 m2 g−1), high electrical conductivity (5000Wm−1 K−1), thermal conductivity and excellent mechanical strength. 16
The graphene based materials, especially graphene oxide has extensively used in variety of wearable and flexible electronics due to its unique properties such as high carrier mobility, environmental stability, and mechanical robustness. 17 Moreover, graphene oxide contains larger number of functional groups that can easily bind with any fiber surfaces. The surface modification of textiles with graphene oxide not only increase its mechanical strength but also imparts some value added properties which includes thermal conductivity, hydrophobicity, fire retardancy and UV blocking, 18 and also the graphene oxide modified textiles exhibits high electrical conductivity, and they are high demand in various fields such as flexible sensors, 19 joule heating materials for thermal regulation,20–23 medical electrode in health care units, 24 flexible energy storage devices 25 and microwave absorption and electromagnetic interference (EMI) shielding materials. 26
Herein, cotton fabric is chosen as a basic substrate in this work to fabricate highly electrically conductive textile. Because cotton is most commonly used natural cellulosic fibers, and it is inexpensive, biodegradable, which not only used for clothing but also used as bandages, dressings, facemask and variety other of applications. Most importantly, cotton fabric available in different configurations, size and shapes, and they are highly hydrophilic, thus it can be easily modifiable based on our specific needs.27,28 Nevertheless, cotton, especially its surface, has a high surface roughness, so simply coating it with carbon material can cause discontinuities in its electrical resistance. To tackle this challenge, herein we use the graphene oxide prepared from spent battery, as primary conductive network/filler over cotton surface, and then the metal particles were deposited over GO-cotton to achieve excellent electro-conductive fabric material.
However, the direct deposition of any metal nanoparticles over textile surface can sometimes causes some discontinuity issues, low effectiveness, and sometimes make the textile materials as rigid one, which causing the high conductivity loss as well as flexibility loss. Moreover, the deposition of hybrid mixture of graphene oxide with any metal particles also results I high conductivity loss. 29 Therefore, this work is planned to use GO, synthesized from spent batteries as effective conductive fillers over cotton surface. Silver nanoparticles are one of the most used electroactive materials and also exhibits excellent mechanical stability and thermal stability. Thus, the electrical and heating performance of GO-coated cotton is further enhanced by deposition of silver particles over GO.
The aim of present work is mainly focused to fabricate thermally stable electrically conductive material using battery waste-derived GO and silver nanoparticles for EMI shielding and joule heating applications.
Materials and methods
Chemicals and materials
Plain weave cotton fabric with 126 EPI (warp), 96 PPI (weft), 150 GSM and 0.4 mm thickness were obtained from local market and spent zinc-carbon batteries are collected from the laboratory. The chemicals used in this work are analytical grade (AR) and directly used without purification. Potassium permanganate (KMnO4, 99.3%), Silver Nitrate (AgNO3, 99.8%), Hydrogen Peroxide (H2O2, 34.5%), Sodium nitrate (NaNO3) from Samchun Pure Chemicals, South Korea. Sodium Hydroxide (NaOH) and Hydrazine hydrate from Sigma Aldrich. Before surface modification, cotton fabrics were pre-treated with sodium hydroxide and de-ionized water.
Materials characterization
The morphology and fabric structures were analyzed by field emission scanning electron microscope (FE-SEM) (SIGMA HV–Carl Zeiss with Bruker Quantax 200–Z10) and energy dispersive x-ray analysis (EDX) and its elemental mapping images were observed with FEI Company (Apreo S HiVac). The X-Ray diffraction analysis (XRD) of as-prepared cotton samples and GO were characterized using Bruker AXS GmbH, D8 advance model (Germany) with Cu Kα1 radiation (λ = 0.15,406 nm) in the range of 2θ (10°–80°). The Raman spectroscopic analysis (NT-MDT Co., NTEGRA Spectra) and X-ray photoelectron spectroscopic (XPS) analysis (Thermo Fisher Scientific, K-Alpha+) were used to confirm the formation of GO and the successful deposition of GO and Ag over cotton fabric.
The thermal stability of as-fabricated samples was done by thermogravimetric analysis (TGA) (TA Instruments, TGA Q500). The weight of each sample were about 5–7 mg and air was used as carrier gas with flow rate of 50 mL/min. The experimental range was 10°–600°C with the heating rate of 10°C/min
The electrical performance of GO-Ag coated cotton fabrics was analyzed by digital multimeter (Chekman, TK-202). The thermal performance was studied by digital thermal recorder with constant DC power supply (UNICORN Co).
EMI shielding effectiveness
The Electromagnetic shielding efficiency (EMI SE) of all coated samples were analyzed by vector analyzer (RODHE & SCHWARZ, ZVA24). In general, the total EMI (SET) shielding is mainly composed of three parts such as reflection (SER), absorption (SEA) and multiple reflection (SEM), the SEM can be ignored when SET >10 dB. The scattering parameters (S11 and S21) in the frequency range of 8.2–12.4 GHz were collected in order to assess the EMI SE of coated cotton samples. EMI SE can be calculated as follows:
30
Synthesis f graphene oxide from spent batteries
The GO was synthesized from carbon rods of dry zinc-carbon batteries by modified Hummer’s method. 31 3 g of graphite powder, obtained from carbon rods of used battery, and 3 g of sodium nitrate are mixed with 75 mL of 95% H2SO4. The mixture is stirred for 2 h at below 50°C. Then 9 g of KMnO4 was slowly added into the mixture, stirring is further continued for 60 min at 20°C, then the temperature was raised to 85°C for 30 min, and 200 mL of distilled water was added into the mixture to abort the oxidation process. Afterwards, 6 mL of H2O2 (30%) was added into the mixture in order to reduce the residual KMnO4. The solution is centrifuged for 25 min to remove non-oxidized graphite. The residue is further washed with 5% HCl and again with copious amount of distilled water till the filtrate reaches the neutral pH. At final, the obtained residual mass is dried for 24 h at 60°C.
Fabrication of GO-coated cotton fabric
The cotton fabrics were cut into 2 × 2 cm2 and washed with NaOH, ethanol and distilled water. The GO suspension is prepared by mixing the GO powder with ethanol and sonicated for 4h. Then, the GO-coated cotton samples were taken out and washed with ethanol-water mixture. The washed GO-coated samples were dried in the hot air oven for 5 h.
The conductive filler ability of battery waste-derived GO was tested with five different samples were prepared by varying the wt% of GO from 1.0 wt% to 5 wt%, and they were named as GO1, GO2, GO3, GO4 and GO5.
Preparation of GO-Ag coated cotton fabric
The electrical conductivity of GO coated cotton fabrics was further increased by deposition of silver particles over it. The Ag over GO coated cotton was fabricated by immersing in 2 mM silver nitrate solution for 120 s, and it was reduced by hydrazine hydrate. After that, the GO-Ag coated cotton samples were washed with ethanol-water mixture. The obtained cotton fabrics were air-dried for 12 h. The final finished samples were called as GO1-Ag, GO2-Ag, GO3-Ag, GO4-Ag and GO5-Ag based on their GO wt%.
Results and discussions
Fabrication of conductive cotton fabric using graphene oxide and silver nitrate
Figure 1 is a graphical representation which illustrates the fabrication process of highly flexible, multifunctional cotton fabric using GO-Ag. In order to achieve the better electrical conductivity, five different samples were prepared by varying the graphene oxide concentration only. The silver nitrate concentration was kept as same for all GO-coated cotton samples. Schematic diagram of fabrication of multifunctional GO-Ag coated cotton fabric.
Characterization of prepared graphene oxide
XRD analysis
The successful formation of graphene oxide from spent batteries are confirmed by XRD analysis. In Figure 2, the diffraction peak at 26.7° attributed to (002) plane, additionally, two peaks around 44.1° and 54.52° corresponding to (100) and (004) reflection planes were observed. The obtained peak values were well matched with standard graphite materials. After oxidation, the characteristic peak corresponds to graphite (26.7°) disappeared and a new peak observed at 11.6° which attributed to the plane (001) of graphene oxide. This results confirmed successful the oxidation of graphite to graphene oxide.
14
X-ray diffraction pattern of (a) graphite (b) synthesized GO.
Raman analysis
Raman spectroscopic analysis is fast, non-destructive and most important tool for the characterization of carbon materials. The Raman spectra for all-graphene based materials mainly consists of several different peaks, namely G peak, D peak which are main characteristic peaks for any carbon materials. The G band is called as graphitic peak while the D band is termed as disordered vibration peak of graphene materials.14,32
Figure 3 illustrates the Raman Spectra for the as-prepared graphene oxide from the recovered graphite powder. The Raman spectrum shows three peaks: G band, D band and 2D band. The first band D is located at 1332 cm−1 is caused due to the breathing mode of κ-point phonon, with A1g symmetry. This D band represents the structural imperfections of GO which is produced by the introduction of hydroxyl and epoxide groups in the carbon structure during the oxidation process. The G band is appeared at 1582 cm−1 which related to the tangential stretching mode of the E2g phonon in sp2 hybrid carbon atoms. The ratio of intensities of D-Band (ID) and G-Band (IG) provide the useful information such as extent of chemical oxidation and proportion of defects in the graphene oxide. The calculated ID and IG of as-synthesized GO is 1.09. It represents the high number of defects in GO that confirms the presence of oxygenated functional groups in graphite powder. In general, 2D band strongly depends on the number of layers of present in the graphene structure. The less intense 2D band occurs at 2658 cm−1 may represent the monolayer of graphene oxide.
33
Raman spectra for synthesized GO.
Characterization of GO-Ag coated cotton fabric
Surface morphological analysis
The surface topographic changes of uncoated and coated cotton fabrics after coating were examined by scanning electron microscopic (SEM) analysis. Figure 4(a) illustrates the surface morphology of uncoated cotton samples, due to ultrasonic cleaning of alkali and alcohol treatment, the surface looks neat, clean and smooth. The pristine cotton exhibits bundle of cellulose fibers with crisscross pattern. The surface topographic images of GO1 and GO5 were given in the Figure 4(c)–(g), in order to get better understand about the surface morphological changes with respect to increase of GO from 1% to 5 wt%. The low magnification image of GO1 coated showed similar morphology of uncoated cotton surface. But the high magnification image showed the cotton surface becomes slightly rougher when deposited the of 1 wt%, it becomes even more rough for GO with 5 wt%. The GO coatings were entirely wrapped around the cellulose fibers rather than the pores between the fibers, which creates the conductive path for the movement of electrons. After in-situ reduction of Ag deposition over GO-modified surfaces, lot of silver particles were successfully attached over the finished fabrics and results on further increase of surface roughness, which creates complete conductive path for the movement of electrons and the results were shown in Figure 4(h)–(m). Moreover, EDX elemental analysis were carried out to analyze the chemical composition of depositions over finished surface and the results showed that elemental composition of C and O was found to be 81.19% and 17.39% respectively in GO5-coated cotton, whereas the GO5-Ag coated cotton showed Ag of 91.33%, the C and O decreased to 4.90% and 3.77% respectively, which also confirms the successful deposition of silver of GO-coated cotton (Figure 5). FE-SEM image of (a, b) pristine cotton (c, d) GO1-cotton (e–g) GO5-cotton (h–j) GO1-Ag cotton (k–m) GO5-Ag cotton. EDX images of (a) GO5 cotton and (b) GO5-Ag cotton.

EDAX mapping images are used to identify the successful deposition of coating over cotton surface. Figure 6 showed the EDX mapping images of GO5-Ag coated cotton. The color images indicated the distribution of carbon, oxygen and silver all over cotton fabric. Elemental mapping images of GO5-Ag cotton.
XRD analysis
The crystallinity of uncoated pristine cotton, GO-coated cotton and GO-Ag coated cotton was investigated by XRD analysis (Figure 7). Pristine cotton exhibits XRD peaks at 15.4° and 16.8° and 22.9° which corresponds to the crystalline lattices of cellulose fibers. After surface modification of cotton fabric with GO and GO-Ag, there is observable change in the sharp crystalline peak of cellulose at 22.9°. This clearly indicates that modification of cotton fabrics by GO and silver particles alter the crystalline nature of cellulose and also ensures the mechanical strength of cotton fabric even after deposition. In Figure 7(b) the peak corresponds to GO at 11.6° was found to be very weak in the GO coated and it was very hard to analyze. This could be due to the fact that the concentration of GO becomes low when we deposit it over the cotton surface. However, in GO-Ag coated cotton, the GO peak was completely disappeared and new peaks at 38.3°, 44.5°, 64.6° and 77.5° attributed to the 111, 200, 220 and 311 crystallographic planes of silver particles which also confirms the successful deposition of silver over GO coated cotton surface.
34
XRD pattern of (a) pristine cotton (b) GO5-cotton and (c) GO5-Ag cotton.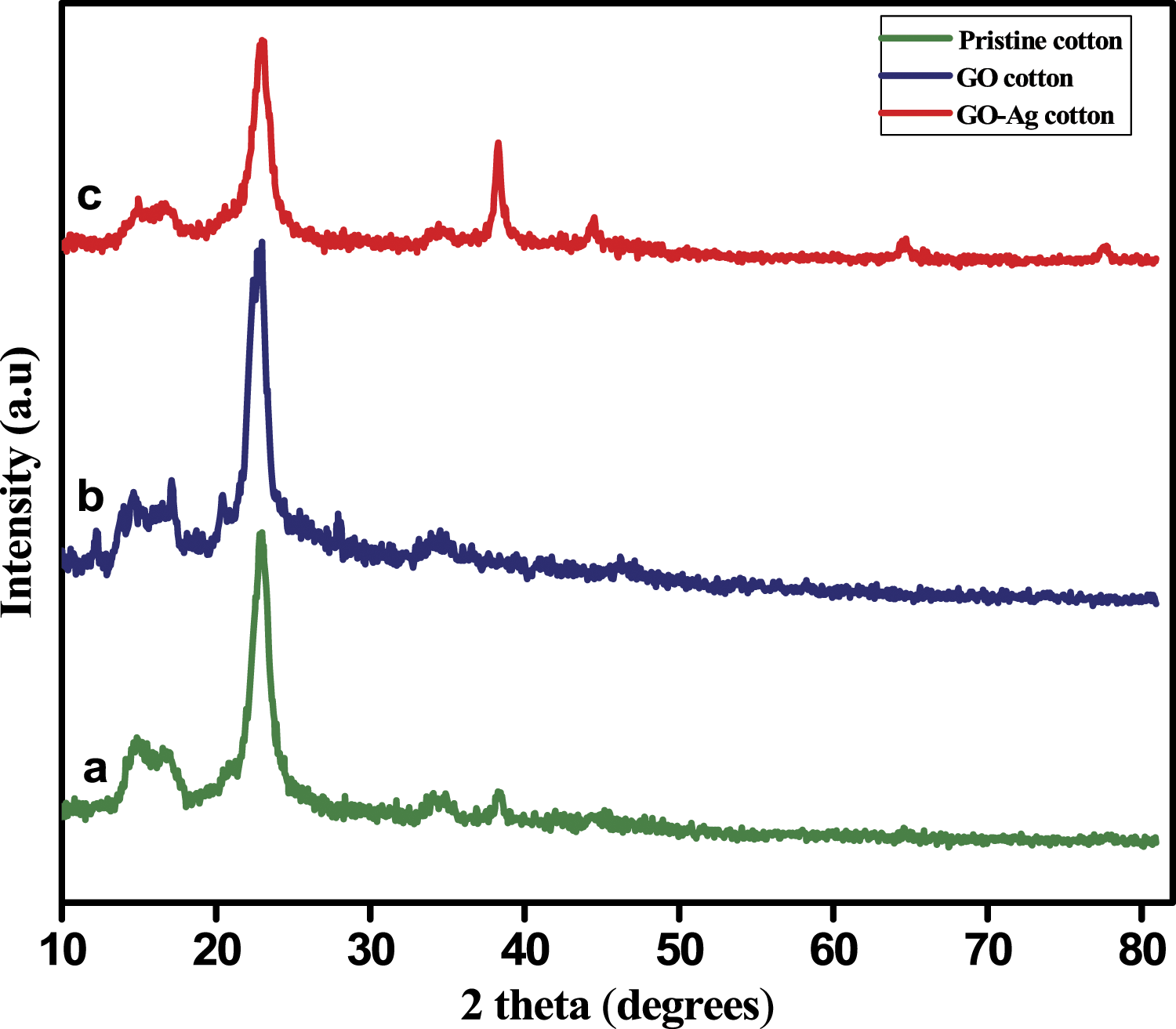
Raman analysis and XPS analysis
As described earlier, Raman spectra is mainly used to characterize the surface modifications of graphitic materials after functionalization with any other materials by detecting the changes in the graphitic structure of carbonaceous materials. Figure 8(a) showed the Raman spectra of GO-coated (inset graph) and GO-Ag coated cotton surface, which helps to analyze the ID/IG ratio before and after deposition of Ag over cotton surface. The D band at 1357 cm−1 which attributed to the oxidizing defects and edge effect on the non-functionalized domains, while the G-band at 1582 cm−1 corresponds to the C-C stretching in graphitic layer, and the ID/IG ratio was observed to be 1.02. This ID/IG ratio was found to be increased to 1.71 after the successful deposition of silver over GO-modified surface. This could be due to the fact that deposition of silver over GO creates more defective structure on graphitic structure and also shift of band occurs on the D and G band from 1357 cm−1 to 1365 cm−1 and 1582 cm−1 to 1588 cm−1 due to the SERS effect of silver particles.
35
(a) Raman spectral analysis of GO5-Ag cotton and (inset) GO5-cotton and (b) XPS survey spectra of GO5 and GO5-Ag cotton (c) C1s spectra of GO5-cotton (d) C1s spectra of GO5-Ag cotton and (e) Ag spectra of GO5-Ag cotton.
XPS analysis were carried out to investigate the chemical state of GO and GO-Ag coated cotton fabrics. In Figure 8(b), the survey spectra clearly demonstrate the existence of C and O in GO-cotton and C, O and Ag in GO-Ag-cotton. Figure 8(c) represents the C1s core level spectrum of GO, which has three peaks at 284.4 eV (C–C in aromatic ring), 286.9 eV (C–O) and 289.7 eV (O = C–OH). These indicates the presence of oxygen containing functional groups such as (carboxyl, hydroxyl, carbonyl, and carboxyl) in GO-coated cotton surface. However, the surface modification of GO-coated cotton surface with silver particles showed the intensity which corresponds to the C–O signal was decreased, whereas the intensity of the C–C signal was found to increased (Figure 8(d)), which reveals that the epoxy and carboxyl groups aid in anchoring of Ag onto graphene network. The binding energies of silver particles were shown in Figure 8(e), Ag 3d5/2 and Ag 3d3/2 peaks were observed at 368.5 eV and 374.5 eV, which clearly the successful deposition of Ag over GO-surface. 36
Electrical conductive studies
In order to evaluate the electrical conductivity properties of treated cotton samples, the electrical resistivity of uncoated and modified cotton samples was examined by two probe multimeter. For each sample, the electrical resistance was measured by three times and average values were given in Figure 9. The uncoated cotton was inherently insulating material. When the GO wt% increased from 1 to 5, GO1 cotton showed very high electrical resistance value almost similar to insulating material which is due to the presence of multiple oxygenated functional groups of GO. But the electrical resistance value decreases with increase of GO wt%. This decrease in resistance is because of formation of percolation network which creates the conductive pathway for the movement of electrons. Once the graphene sheets reached their percolation threshold, any further increase on graphene content does not cause any noticeable change in electrical resistance.
37
In this present study, after 5 wt%, there was no significant change in the resistance were observed. Electrical conductivity of all (a) GO-coated (b) GO-Ag coated cotton samples.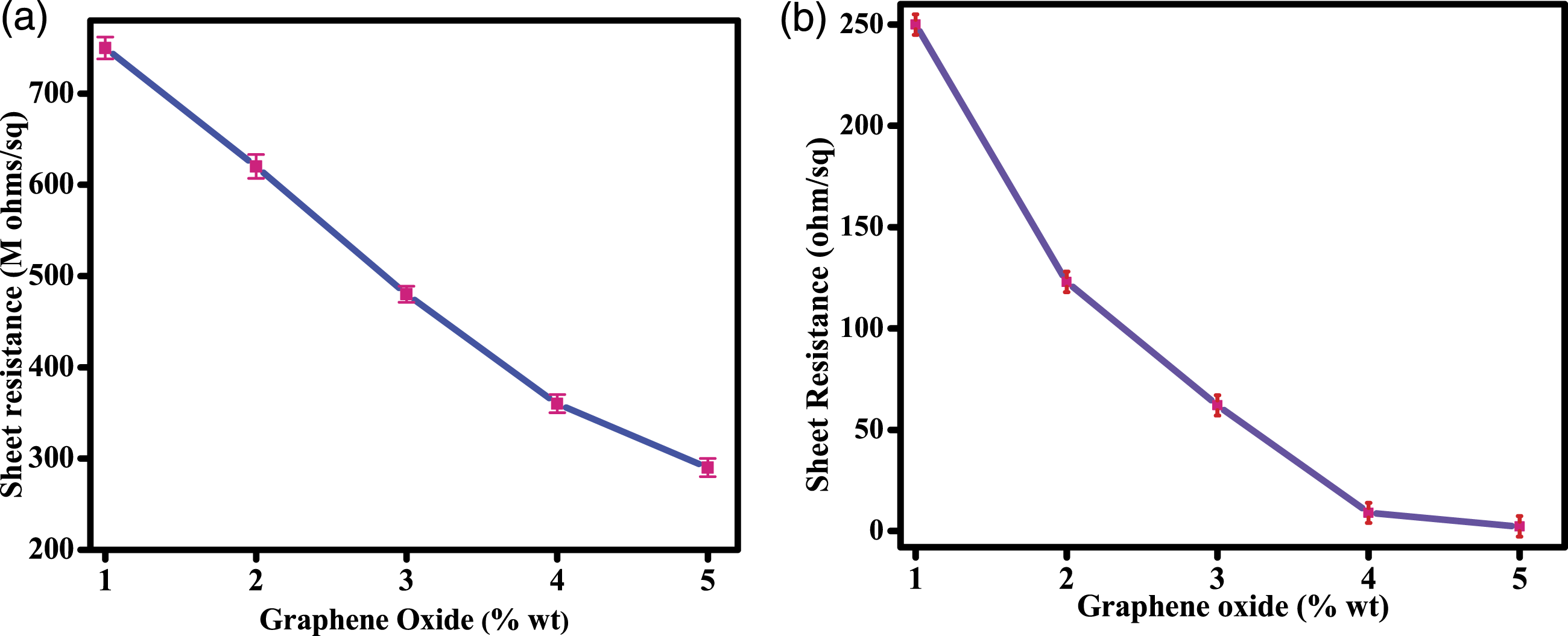
However, there was a significant change of electrical resistance were observed when the silver particles deposited over the GO-coated cotton surface. The electrical measurement values of all GO-Ag coated cotton samples were given in Figure 9(b). The improvement in the electrical conductivity of GO-modified samples by silver particles can be explained by two possible ways: 1) intercalation of Ag within graphitic layer reduces the sheet resistance by weakening the pi bonds of GO. And 2) When bias voltage is applied, the electric field generated around Ag enhances the charge transfer between Ag and GO.36,38,39 The GO5-Ag cotton reached low surface resistance of 2.3 Ω/sq, when compared to all other coated cotton samples. Moreover, all GO-Ag coated cotton samples maintained their surface resistance even after 1000 bending cycles, which shows the electrical stability of as-coated cotton samples (Figure 10(a)). (a) Electrical stability of GO5-Ag cotton and (b) Digital photographic image of electronic circuit to power LED.
Furthermore, the electrical conductivity nature of as-modified cotton was analyzed by creating electrical circuit by connecting the 9 V with the GO5-Ag modified cotton sample. When the circuit was complete, the LED light illuminates brightly which also confirms the electrical conductivity of the as-modified sample. The digital image of the powering LED was given in Figure 10(b).
The fabrication of electrically conductive textile with high electrical conductivity can found potential applications in the field of wearable flexible thermal heater. The as-fabricated GO-Ag coated cotton fabric exhibits good electrical conductivity and flexibility; thus they can be used as wearable heaters for personal thermal management devices. When external electric field applied to conductive material, the inelastic collision occurs between the electrons and photons which results in generation of heat. 23
The electro-thermal behavior of GO5-Ag coated cotton fabric (sized 2.5 × 2.5) was examined by connecting it with external DC power supply. The surface temperature change and sample response were recorded and monitored by digital thermometer and infrared camera. The time dependent-temperature profile of GO5-Ag cotton under different voltage supplies were depicted in Figure 11(a). The graph revealed that the GO5-Ag coated cotton fabric immediately responds and generates heat for the applied voltage and the temperature was gradually increased with increase of input voltage and time. Within 60 s, the GO5-Ag coated cotton fabric reaches maximum temperature in all input voltage conditions. As observed from graph, the GO5-Ag cotton fabric heater at 1 V remains same at the room temperature. When the applied voltage increased from 1 V to 2 V, the temperature elevation starts from 30°C to 60°C within 15–25 s, and reached responsive high temperature within 50 s and eventually it reaches the steady state condition. The further increase in the input voltage of 3 V and 4 V the temperature, the fabric exhibits faster upward trend of heating temperature up to 117.9°C. When the voltage supply stops, all samples were back to room temperature within 30 s. From the results, it evident that GO5-Ag coated cotton exhibited high heating performance because of its low surface resistance and excellent surface morphology. (a) Temperature-time profile of GO5-Ag cotton (b) Thermal stability of GO5-Ag cotton and (c) Average temperature at different voltages.
To evaluate the practical merits of fabricated joule heater, the long-term stability of GO5-Ag was assessed by the driving voltage of 4 V. The thermo-electrical heater with low driving voltage and low sheet resistance are urgent need for these days. 40 Therefore, the heater performance was evaluated at 4 V, because it reaches maximum temperature within 30 s, and it maintained its steady-state condition until the voltage stops. When we considered 4 V is optimum driving voltage, the GO5-Ag wearable heater achieves reasonable thermal stability and it could be maintained for 2100 s (i.e >30 min) (Figure 11(b)). The average surface temperature at different applied were depicted in Figure 11(c).
Besides these study results, thermal camera was also used to analyze the surface temperature morphology of GO5-Ag coated cotton with respect to applied voltage and time. In Figure 12 the electro heating performance of GO5-Ag at applied voltage of 4 V were pictured. It helps to understand the uniform thermal distribution of as-fabricated thermal heater, which is most important character of wearable thermal heater. The first four images (0 s–55 s) showed the heating performance of GO5-Ag coated cotton at the applied voltage of 4 V and next four images (65 s–95 s) showed the temperature was decreases when the applied voltage was removed. Thermal camera images of GO5-Ag coated cotton at various time intervals under constant voltage of 4 V.
The thermal behavior of fabricated thermal heater was also analyzed by fixing the heater in index finger and wrist, in order to study the performance of thermal heater on specific body region, and its corresponding images were also shown in the figure. When the input voltage of 4 V applied, the thermal heater attached in the index finger and wrist reaches the temperature of 65.7°C and 59.4°C respectively. These study results showed the as-fabricated thermal heater may be regard as excellent heating element for personal thermal management and also it can be used in other various applications such as defrosting and deicing also.
Thermal stability analysis
The thermal stability of GO5-Ag coated cotton was analyzed by TGA. The TGA analysis is one of direct and effective method to examine the thermal stability of coated and normal cotton samples. In this method, the thermal stability can be studied by measuring the percentage of weight loss as a function of temperature ranges from 0°C to 600°C, and the results were depicted in Figure 13. The results indicated that the uncoated cotton underwent two degradation steps in the presence of air. The first degradation step occurred at 365°C and it represents the degradation of cellulose and the generation of L-glucose. This L-glucose further broke down into several carbon residues such as aldehyde, ketone and furans at the temperature range between 365°C to 420°C and this represents the major weight loss of uncoated cotton fabric. The second degradation occurred at 440°C, which it corresponds to further degradation of residual carbon matters, and weight loss of unmodified cotton reached almost 99.5% at 500° and it remains same till 600°C.41–44 In GO- coated cotton sample, the major weight loss in GO coated cotton at 227°C–350°C could be due to the decomposition of more stable oxygen functional groups with CO and CO2, and reached 88.6% weight loss at 500°C, whereas in GO-Ag modified cotton, the small degradation step was observed at 180–200°C could be due to the enlargement of Ag particles,
45
and only ∼7% weight loss was observed. The further significant weight loss was observed only at ∼306°C–365°C, and reached 61.2% weight loss at 500°C, showing the thermal stability of GO-Ag coated cotton. Thermogravimetric analysis of GO5-Ag cotton.
EMI shielding performance of GO-Ag cotton fabrics
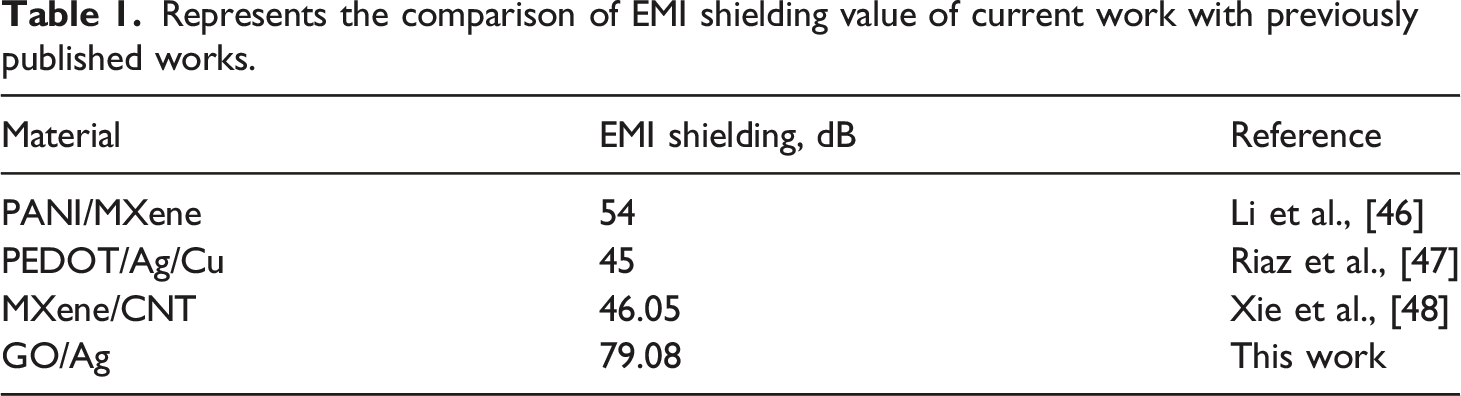
GO-Ag coated cotton fabrics are electrically conductive material but not magnetic material. But the excellent electrical conductivity of GO-Ag plays a major in their electromagnetic shielding performance. The EMI SE of five as-fabricated cotton samples in X-band were tested in the range of 8.2 GHz–12.4 GHz and plotted in Figure 14(a), which shows the EMI SE performance of all coated and uncoated cotton samples. When the GO percentage increases from 1 to 5, the thickness of fabric also increases, thus the conductive network becomes more stronger which results in the increase of the electromagentic shielding performance. When GO wt% increase from 1 to 2, it reaches the SET value of 21.1 dB, and EMI shielding value >20 dB is current demand of dB for commercial materials. The further increase of GO wt% exhibits higher EMI shielding value. Specifically, the GO5-Ag coated cotton sample exhibits maximum EMI value of 79.08 dB at 910 μm thickness. It showed that EMI SE of any shielding materials are thickness-dependent. When thickness increases more conductive network can be formed which interacts with incident electromagnetic waves, resulting in the high EMI SE performance. (a) EMI SET (b) SEA (c) SER and (d) Average EMI SE of GO5-Ag coated cotton.
The EMI shielding mechanism was analysed for the clear understand of shielding performance of GO-Ag coated cotton fabrics, the overall contribution of SEA and SER to SET were depicted in Figure 14(d). From the graph it was noted that SEA was higher than SER throughout frquency range, which clearly showed the our GO-Ag coated cotton sample adopts absorption-dominant shielding mechanism. These high EMI SE and absorption-dominant mechanism always attributed to the low skin depth.
46
The skin depth can be caluculated by the following equation (a) Skin depth analysis (b) Comparison of theoretical and experimental EMI SE and (c) EMI shielding stability of GO5-Ag coated cotton.
In addition to support the absorption-dependant mechanism of GO-Ag coated cotton fabric, theoretical SET, SEA and SER were also calculated by using the following equation.
Represents the comparison of EMI shielding value of current work with previously published works.
Conclusion
In summary, the present work demonstrated the successful fabrication of electrically conductive cotton fabric with high electromagnetic shielding and excellent joule heating performance using GO as conductive filler and silver particles for the enhancement of electrical conductivity. The GO-Ag cotton exhibits very low sheet resistance of 2.3 Ω/sq with the increase of GO wt% from 1 to 5. There is no sigificance change in the electrical resistance even after 1,000 bending cycles, which suggests that the coatings were very stable, when compared the previous work results, EMI performance of GO5-Ag coated exbitis high EMI SE value of 79.08 dB at the thickness range of 910 µm. Moreover, GO5-Ag heater reaches high surface temperature of 117.9°C within the 25–30 s. On view of above study results, GO wt%, which synthesized from waste batteries plays major role on increase of heating performance and EMI SE performance of as-coated cotton fabrics. The recycle and recovery of graphitic materials from spent batteries can prevent the raw material consumption, high cost and also reduce the environmental pollution. Besides fabricating high performance cotton fabrics, the present work also provides an alternative way to utilize battery wastes to create new value-added materials. Overall, the present approach appears to be simple and economical for fabricating electrically conductive cotton fabric with high EMI SE performance and excellent joule heating capabilities. Future study includes further characterization of the effects conductive fillers for understanding and enhancing the material performances and stabilities with the possible consideration of other additives.
Footnotes
Acknowledgements
We would like to thank to the Cooperative Equipment Center at Korea University of Technology and Education for assistance with the sample and device characterization in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2021R1A2C1094233).