Abstract
The VOCs removal from polluted air has been achieved using several different methods but primarily through the use of adsorbent materials or through degradation with photocatalytic oxidation (PCO). Fibres produced by electrospinning have the possibility to easily incorporate additives into the fibres and onto their surface. This can functionalise them for efficient VOCs removal. Cellulose acetate (CA)-based electrospun fibre membranes have been fabricated and doped with activated charcoal (AC) and titanium dioxide (TiO2), either separately or in combination to investigate their toluene removal capacity of the single additive and the synergic effects of adsorption and PCO. Two different methods of functionalisation of the fibres with AC and TiO2 have been used. These methods are air spraying and electro-spraying. Several configurations of the final membranes have been investigated. SEM images indicate that the additives have been successfully distributed on the fibre surface and they affect their morphology by increasing the overall roughness and the thickness of the final membranes. Adsorption with AC achieved 45.5% removal of toluene with a starting concentration of 22.5 ppm. PCO was probably initiated using a blacklight blue UV lamp with a peak wavelength of 365 nm as formation of formaldehyde was recorded. The findings suggest that PCO is affected by the residence time and UV light intensity.
Keywords
Introduction
In modern society, people spend more than 90% of their daily time in an indoor environment 1 and consequently the indoor air quality (IAQ) has a major impact on people’s health, productivity and comfort.2–4 Typical indoor air pollutants are particulates, nitrogen oxides (NOX), carbon oxide (CO), sulphur dioxide (SO2) and volatile organic compounds (VOCs). VOCs are a major group of indoor contaminants, and the main compounds contain alkenes, alcohol, aromatics and carboxylic acids. Formaldehyde, ethanol and toluene are examples of indoor VOCs. 5 The sources of such pollutants are various and are mostly found inside the building, such as cleaning products, building construction materials and furnishings (plywood products and carpets) as well as office equipment like printers and photocopiers, but also human occupancy and related activities.6–9 Owing to the concerns related to IAQ, several different technologies have been investigated and developed to remove VOCs from indoor air, such as non-thermal plasma, 10 adsorption, 11 ozonation 12 and photocatalytic oxidation (PCO). 13
Plasma based methods are related to the use of a high voltage discharge for the destruction of gaseous pollutants. 14 Non-thermal plasma is associated with the production of unwanted by-products like NOX and ozone, poor energy efficiency and unsteady operation.15,16 The use of ozone generators for removal of VOCs was more frequent in the 1990s. Later, it was proven that the efficiency of VOC removal with such technology was rather low 17 and more importantly, ozone has been found to contribute to the formation of harmful secondary organic aerosols 18 and can cause asthma and other health related issues in humans. 19 Adsorption and PCO have been the two methods that attract most interest in the removal and degradation of VOCs.
Adsorption has proven to successfully retain various contaminants on the surface of the adsorbent materials, like zeolites, some polymers, activated alumina and above all, activated charcoal (AC). Carbon materials have demonstrated to be low-cost, highly efficient and stable in removing VOCs and therefore, despite some disadvantages related to their hygroscopicity, have the most potential as adsorbent materials. 20 Activated carbon fibres have demonstrated better performance in adsorbing VOCs compared to zeolites, silica gel and granular activated carbon when investigated under the same conditions. 21 AC adsorption capacity has been investigated for most VOCs including alcohols, aromatic compounds, esters, aldehydes and alkanes and has proven to be effective against a wide range of pollutants. 22
Degradation of VOCs is promoted by PCO, which relies on the presence of a semiconductor as a catalyst and an irradiation source that initiates the reaction. 15 The interesting characteristics of PCOs are related to their activity towards various contaminants, formation of benign final products (CO2 and H2O) and optimal operation at room temperature. 23 Titanium dioxide (TiO2) has been the most used catalyst for this application because of its promising performance as a photocatalyst under UV light irradiation and chemical stability. 24 The main disadvantage of using PCO is the potential formation of harmful by-products such as aldehydes, ketones or organic acids in cases of an incomplete reaction.25–27 Parameters such as relative humidity, residence time, UV source wavelength, intensity and pollutant concentrations are influencing factors in the reaction process of degrading VOCs.28–30 Toluene conversion efficiency has increased from 30% to 90% when the residence time moved from 120 ms to 1920 ms, indicating the importance of this parameter and highlighting the non-complete conversion in any experimental conditions. 29
In order to enhance the efficiency of PCO, the exposure to light irradiance and to the polluted air should be maximised. This can be achieved with combined use with an adsorbent material as a support. The adsorbent would retain the pollutant from the air close to the photocatalyst and this could mitigate the negative influence of a short residence time. Several studies have investigated the use of AC as a support material for TiO2 in removing VOCs from polluted air.31–34 The conversion of toluene using P25 TiO2 was demonstrated to drop down to about 47% when reducing the residence time from 3.7 to 0.6 min, while the reduction was only about 27% when TiO2 was combined with AC. 31 PCO can be used as a regeneration method of AC when located downstream of the adsorbent. 34 Furthermore, the combined use of TiO2/AC has been demonstrated to not only enhance the removal efficiency of the pollutants but also to reduce the amount of intermediate by-products released in the air, as well as delaying the adsorption saturation of AC.33,35
In recent years, electrospinning has attracted a large amount of attention, as it is a technique that allows fast and efficient mass production of fibrous materials. Furthermore, the process allows easy functionalisation with a wide variety of materials, such as TiO2 and AC, to enhance the removal efficiency of VOCs by the fibres.36–39 Polyvinyl alcohol (PVA)-based fibres doped with TiO2 have shown an acetone removal efficiency of 50%, with an initial concentration of 1500 ppm, under a light source of UV-365 nm and a retention time of 10 s. 37 When the retention time was increased from 10 s to 100 s, the removal efficiency of acetone increased to 99%. 37 Polyacrylonitrile (PAN)-based activated carbon electrospun fibres produced by carbonisation and steam activation after electrospinning demonstrated to be an effective adsorbent towards formaldehyde, showing a breakthrough time of 10.5 h at 11 ppm of formaldehyde and in dry conditions. 39 This breakthrough time is remarkably longer compared to conventional activated carbon fibres (FE100), which under the same conditions was 4 h. 39 TiO2-AC complex fibres have been prepared by electrospinning and the fibres were investigated for the removal of Prussian blue dyes by solar dye irradiation, reaching a dye removal of 94–99%. 40
The present study introduces five different electrospun fibre membranes based on cellulose acetate (CA) and loaded with TiO2 and AC either separately or combined to investigate the toluene removal capacity of the single additive and the synergic effect of adsorption and PCO. The membranes were exposed to toluene, which was chosen because it is a common VOC found in indoor air and used as a standard substance for PCO experiments. CA was selected because it is an abundant and biodegradable organic compound, frequently used for applications as filters, artificial skin and protective clothing.41,42 The biodegradability of CA is an advantage compared to the typically employed synthetic polymers, as it means CA does not produce microplastic, both while in use and when it is disposed. 43
The electrospun fibres were doped by using air spraying or electrospraying to include, mix and combine the additives in different configurations. The fibre morphology and characteristic and the toluene removal capacity of TiO2 and AC when used as additives on polymer based electrospun fibres was experimentally investigated and compared.
Experimental
Materials
CA, 39.8 wt% acetyl content, MN 30,000 GPC was purchased from Sigma-Aldrich, together with powder-form AC (MW 12.01 g mol−1). Nanopowder aeroxide TiO2 P25 was given by Evonik Industries. Ethanol, acetone, and 2-Propanol were employed as received from Avantor. All materials were used without further purification.
Synthesis of electrospun fibre membranes
The membranes were prepared using the method described in the previous work. 43 A homogeneous CA-solution was obtained stirring vigorously 31.25 g of CA in 250 mL of acetone. The mixing was performed for 12 h at 20°C (room temperature).
Then, three different dispersions were prepared. The first dispersion consisted of 1.5 g of TiO2 mixed for 1 h in a 100 mL solution of ethanol and 2-propanol (1:4). To ensure uniformity, the solution was kept 30 min in a ultrasonicated bath. The second dispersion contained 2 g of AC mixed in the same solvent mixture (100 mL ethanol and 2-propanol; 1:4). In this case, to ensure a homogenous dispersion of AC, the solution was kept under continuously stirring before being used. 43 These two dispersions were prepared and used in the air spraying process to dope the electrospun fibre membranes. The third solution was prepared to be used in electrospraying and consisted of 2 g TiO2 mixed with 20 mL of acetone.
For the preparation of the CA membranes, electrospinning was performed on a vertical downward configuration, using an electrospinner 2.2.D-500 from Yflow A/S. The electrospinner is equipped with a flat collector and a movable arm that allows for movement of the needle in the X-Y plane via a pre-programmed track. Figure 1(a) shows a schematic representation of the setup. The prepared polymer solution was transferred to a 20 mL glass syringe (Poulten and Graf GmbH). The applied voltage was 14.8 kV, the feed rate was kept constant at 1 mL min−1 through a needle with a 1.5 mm inner diameter, kept at 9 cm from the collector. Continuous electrospinning was performed to use 10 mL of the CA solution for producing a fibre mat.
43
To ensure uniform fibre distribution on the collector surface, the movement of the needle was remotely controlled on a X-Y motion at a constant velocity (50 mm s−1) on the 20 cm × 20 cm collector (Figure 1). During electrospinning, the relative humidity was controlled in the range 27–32% and the temperature was registered in the range 19.8–20.8°C inside the electrospinning box. In order to complete evaporate the solvent, the membrane was kept in a ventilated oven at 70°C for 1 h.
43
Representation of the two setups used for electrospinning in this research project, (a) single needle, (b) co-axial needle.
The pure CA membrane fabricated with electrospinning was the base for three different membrane types: the CA/AC, the CA/TiO2 and the composite membrane. The first two membranes were made air spraying the respective additive solution with an air brush onto the CA-membrane surface. The spraying process was performed by placing the electrospun CA membrane onto a metal grid in a fume hood. The distance between the grid and the air brush was kept at 20 cm. The process consisted of 2 min spraying at a flow rate of 10 L min−1 of nitrogen gas. Each membrane was sprayed twice and the process was interspersed with a 1 h drying phase at 70°C. 43 In the composite membrane, two CA electrospun fibre layers were interspersed with spraying of the two additive dispersions containing AC and TiO2 separately. The four layers of the composite membrane were made up as follows: layer 1, electrospun matrix from 6 mL of CA solution, layer 2, spraying of AC dispersion, layer 3, electrospun matrix from 4 mL of CA solution and layer 4, spraying of TiO2 dispersion. Each step was followed by 1 h drying at 70°C. The air spraying process for the two layers of additives was the same adopted for the single-additive membranes. 43
The fifth membrane was produced differently. The CA fibre mat was electrospun with a coaxial needle and the outer needle provided a sheet flow of TiO2, leading to a more equal distribution of TiO2 on the fibre mat. The co-axial setup is presented in Figure 1(b). The inner needle contained the polymer solution, while in the outer needle the TiO2 dispersion flowed. The feed rate of the polymer solution was set at 1.1 mL min−1 while the dispersion was fed at 0.7 mL min−1. The applied voltage was 23 kV and the collector was kept at a distance of 15 cm from the tip of the needle. The temperature and the relative humidity inside the electrospinning box during the experiment were 22.7°C and 60%, respectively.
Fibre characterisation
Scanning electron microscopy (SEM; Zeiss XB1540) operating at HT of 5 kV was used to investigate the morphology of the fibre surface. Before scanning, a sample of each membrane was coated with a thin layer of gold. DiameterJ plugin for ImageJ (NIH, USA) was employed to assess the average surface porosity of the membranes. A traditional External Micrometer (accuracy ±0.005 mm, Model 965M, Moore and Wright Ltd, Sheffield England) was used to measure the thickness of the membranes. To avoid compression of the materials, the membranes were kept between two metal plates during measuring. 43
Toluene removal
Toluene removal capabilities of the fabricated membranes were investigated with the setup, which is schematically presented in Figure 2. Pure nitrogen at 1 bar pressure was used as a carrier gas throughout the experiment. The total flow was controlled at 3.9 L min−1. The input gas stream was split into wet and polluted streams. A wet stream was used in order to ensure humidity inside the setup, which is a crucial parameter to ensure adsorption and PCO of toluene.27,35 The wet stream was controlled using a compression valve and measured through a mass flow meter (Model 4043, TSI Incorporated, Shoreview, MN USA), whereas the polluted gas stream was controlled through a mass flow controller (5850TR series, Brooks Instrument B.V., Holland). Milli-Q water and toluene were kept separately in two 250 mL gas-washing bottles, which were immersed in a water bath at 21°C to keep the evaporation rate constant. During the experiment, the membranes were kept in a 1-m fused quartz tube, with an inner diameter of 36 mm. The 20 cm × 20 cm membranes were pleated to fit inside the quartz tube. The tube was kept inside a box with a blacklight blue UV lamp with a peak wavelength of 365 nm, employed to activate TiO2 present on the fibres and initiate PCO of toluene. A bypass was built to allow access to the quartz tube while running the experiments and evaluate the toluene concentration before and after passing through the membrane. The flow was directed into a multicomponent FTIR gas analyser (Gasmet DX4000, Gasmet Technologies Oy, Vantaa, Finland) to monitor the amount of water vapour and toluene concentration during the different stages of the experiment and therefore assess the effectiveness of the membranes in removing toluene from the air stream. Schematic representation of the setup to assess the capacity of toluene removal of the membranes.
The toluene removal capacity of each membrane was calculated using the following formula
Results and discussion
Preparation of electrospun fibre filters
Electrospinning and doping conditions used to fabricate the CA-based membranes.

Fibre characterisation
The SEM images of the various membranes are shown in Figures 3 and 4. The pure CA-based electrospun fibres present a flat ribbon shape, as visible in Figure 3(a) and (b). Furthermore, they exhibit a uniform morphology and a randomly oriented distribution over the surface of the membrane. The doped fibres with either AC or TiO2 are visible in Figure 3(c) and (d) respectively. The images show that the additives have been successfully incorporated onto the surface of the membranes. In contrast with the smooth and uniform pure CA fibres, the additives cover the fibre surface of the doped membranes, affecting their smoothness. In Figure 3(d), it is evident how the deposition of TiO2 in form of agglomerates has densely coated the surface of the CA-based fibres, whereas the AC particles are highly SEM images of (a) and (b) pure CA fibres, (c) AC doped fibres, (d) TiO2 doped fibres, (e) composite membranes, second layer, AC doped fibres and (f) composite membranes, fourth layer, TiO2 doped fibres. SEM images of electrospun CA fibres with TiO2 electrosprayed using a co-axial setup.

The SEM images of the composite membrane are shown in Figure 3(e) and (f), where the second and the fourth layer of the membrane are displayed respectively, in order to show the additives on the fibres surface. In this case, the TiO2 seems to be less heavily deposited on the fibres (Figure 3(f)) and morphology is affected in a milder form compared to the CA/TiO2 membrane. However, AC is densely present in forms of agglomerates on the second layer of the membrane (Figure 3(e)).
The fibres from the SEM images of the last membrane, fabricated using electrospraying of TiO2 as part of the electrospinning process, are presented in Figure 4. The flat ribbon shape has remained unchanged compared to the previously described membranes. Again, TiO2 has been successfully integrated onto the fibres surface as visible in Figure 4(b). Figure 4(a) revealed the presence of agglomerates of TiO2 nanoparticles distributed within the fibres and on the membrane surface. These agglomerates could be the result of non-complete evaporation of the solvent during electrospinning and they partly cover the fibres, affecting the surface morphology of the membrane.
Thickness and surface porosity of the electrospun membranes.

The thickness analysis led to the results presented in Table 2. The use of the additives had a significant influence on these results. 43 The pure CA membrane is 0.11 mm thin, followed by the three single-additive membranes with a thickness of 0.14 and 0.15 mm. The composite membrane showed an increase in thickness of 82% compared to the pure CA membrane, due to the use of a layered configuration of fibres and additives.
Toluene removal
The removal capacity of gaseous compounds was investigated by assessing the degradation through PCO and adsorption of toluene from the different membranes. The experimental study was done on each sample and one more investigation was performed combining one CA/AC sample with a CA/TiO2 electrosprayed membrane.
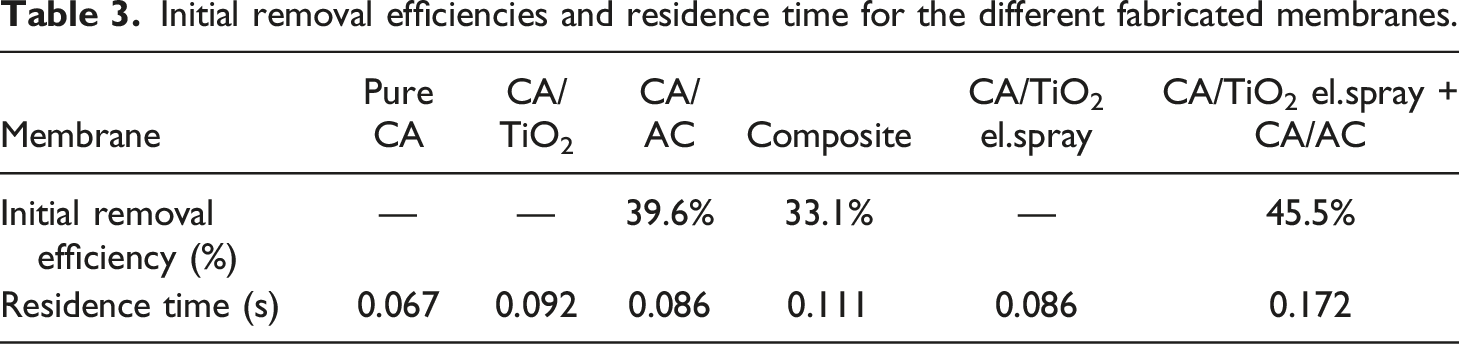
The concentration of toluene inside the setup was stabilised at 22.5 ppm, while the relative humidity was kept at 33% ± 5%. The investigation was run for 40 min on each sample, of which 30 min the UV light was turned on to initiate the PCO of toluene. Figure 5 shows the results of the experiments displaying the changes of the normalised toluene concentration in time for each tested sample, while the initial removal efficiencies and the residence time are presented in Table 3. The pure CA membrane did not adsorb any toluene, as expected. CA has no adsorption capacity in and is used as a negative reference sample. The toluene concentration through the pure CA membrane remained very stable throughout the experiment, with an average deviation from the mean value of 0.15 ppm. The concentration of toluene significantly decreased in a very short time in each sample containing AC, revealing their adsorption capacity. The composite membrane showed an initial removal capacity of 33.1%, while the CA/AC alone and combined with CA/TiO2 electrospray showed an initial removal capacity of 39.6% and 45.5%, respectively. The registered removal is due to the adsorbance capacity of AC. As can be seen in Figure 5, the concentration of toluene started to increase again with a similar development for each of the three samples containing AC. After 40 min of exposure to the pollutant, the removal efficiencies of the membranes have stabilised around 20%. Therefore, they haven’t reached the breakthrough after 40 min of exposure. The enhanced performance of the combined CA/AC and CA/TiO2 electrospray might be due to the increased residence time for the reaction that allowed a higher amount of toluene being adsorbed from the AC. Changes of normalised toluene concentration throughout the experimental study (the yellow dotted lines show the point when the UV light is turned on and off). Initial removal efficiencies and residence time for the different fabricated membranes.
The two membranes that contained TiO2 did not show a clear reduction of toluene concentration. It is noticeable that the concentration of toluene did not present the same stability around the average value as it was before placing the filter in the setup (the average deviation from the mean value was 0.17 ppm) or while testing the pure CA membrane. The CA/TiO2 and CA/TiO2 electrospray showed an average deviation from the mean value of 0.34 ppm and 0.56 ppm, respectively. Furthermore, while the two membranes were irradiated with the UV light, formaldehyde was detected in the air flow, with peaks of 260 and 360 ppb for CA/TiO2 and CA/TiO2 electrospray, respectively (Figure 6(a) and (b)). Formaldehyde is recognised to be one typical by-product of PCO of toluene.24,48 Therefore, it can be concluded that the photocatalytic reaction has been initiated in the set up. Details on the experimental study of the CA/TiO2 (a) and CA/TiO2 (b) el.spray membrane. The yellow dotted lines represent the point in time when the UV lamp was switched on and off.
The reasons behind the inefficient toluene conversion from the TiO2 might be related to different factors. First, the UV lamp shines a rather diffusive and low-intensity light rather than directional and intense. This means that the light was placed along the quartz tube where the membranes were kept and the intensity reaching the TiO2 might have not been enough to degrade the toluene inside the reactor.30,33 The second reason might be related to the contact time, or residence time, which is the time that the gas molecules spend in contact or near the photocatalyst. The residence time involved in this experimental study ranges between 0.067 and 0.172 s, as shown in Table 3. Compared to others, the contact time is kept in the range of seconds to minutes. In a typical air purification system, the residence time ranges from tens to hundreds of milliseconds. 49 This is one main reason behind the difference of performance of photocatalysts, which exhibit acceptable removal efficiencies in the laboratory and demonstrate worse performance under real conditions. 49 Furthermore, the removal of BTEX through PCO with TiO2 has been reported to decrease quickly as the residence time decreases from 3.7 to 0.6 min. 32 Therefore, as the residence time in this experiment was in the range of hundreds of milliseconds, the PCO might not have been initiated efficiently enough. Lastly, when the additives were combined onto the surface of the membranes, it seems that the AC prevents the degradation of toluene from the catalyst. No formaldehyde production was recorded, therefore the TiO2 might have been deactivated from AC, or the formaldehyde had been retained in the charcoal and could not be observed during the experiment.
Typical indoor gaseous air pollutants are found in ranges of concentrations at the ppb and sub-ppb level.50–53 As already mentioned, the concentration of toluene in the setup was around 22.5 ppm, which represents a relative high concentration compared to the typical indoor and outdoor levels of gaseous compounds. Therefore, the amount of AC deposited on the membrane surface might maintain a higher removal efficiency for a longer period of time when exposed to real conditions.
Conclusion
In summary, five CA based electrospun fibre membranes doped with AC and TiO2 were fabricated to investigate the adsorption and degradation capacity and the possible synergistic effect of these two additives when combined. A rougher fibre morphology was concluded to be a direct consequence of the use of TiO2 and AC and the membranes doped with the additives had an increased thickness compared to the pure CA membrane. The AC membranes have shown removal efficiencies for toluene up to 45.5% with a residence time of 0.172 s and a toluene concentration of 22.5 ppm. The PCO was initiated as formaldehyde was detected in the N2 stream, although the conversion rate of toluene was not large enough to be detected. When the two additives were combined to investigate the synergistic effect on the removal of toluene, AC adsorbed the pollutant preventing the initiation of PCO or it has also re-adsorbed the degradation products.
Footnotes
Acknowledgements
The authors thank research technician Peter Kjær Kristensen from the Department of Materials and Production at Aalborg University for his support in performing the SEM images.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Grundejernes Investeringsfond, the Department of the Built Environment, and the Department of Materials and Production at Aalborg University.

