Abstract
The separation of dye/salt through the separation membranes is a great significance for the treatment of dye wastewater. In this study, a novel PMIA hybrid membrane was fabricated via nonsolvent induce phase separation (NIPS) method, in which porous zinc imidazolate framework-8 (ZIF-8) was utilized as a filler. The effect of ZIF-8 content on the structure and performance of PMIA/ZIF-8 membrane was investigated. The results showed that the structure and performance of the PMIA/ZIF-8 membrane could be controlled by changing the content of ZIF-8. The proper content of ZIF-8 was 0.45 wt% in this study. To further enhance the water flux of PMIA/ZIF-8 membrane, the concept of creating nanovoids within the ZIF-8 nanoparticle via wet-chemical etching method was explored in this study. The additional pore of ZIF-8 nanoparticles after wet-chemical etched endows PMIA/ZIF-8 membrane with additional pathways for water molecules. Hence, the PMIA/ZIF-8 membrane after wet-chemical etching showed higher separation performance for dye wastewater. The optimized PMIA/ZIF-8 membrane showed a water permeance of 104.7 L·m−2·h−1 and Congo red rejection of 98% at 0.1 MPa. The water permeance was about 1.5 times than that of the pristine PMIA/ZIF-8 membrane while the Congo red rejection was well maintained. Moreover, the PMIA/ZIF-8 membrane showed good thermal stability during the filtration process. These results indicated that PMIA/ZIF-8 membrane has potential application in dye wastewater treatment.
Introduction
The textile industry plays an important role to develop the global economy and people’s living standards. However, the dye wastewater restricts the development of textile industry as the treatment of dye wastewater is expensive and difficulty.1,2 The dyes in textile wastewater are often toxic, non-degradable and will seriously threaten the environment if it is not handled properly.3–6 Therefore, developing an effective and low-cost method to remove dyes from textile wastewater is important. In order to solve this problem, different technologies are used to treat the dye wastewater, such as adsorption, 7 photocatalytic, 8 filtration, 9 biological treatment, 10 and chemical oxidation.11,12 As a key technology for cleaner production, membrane separation technology which utilizes filtration membranes as a medium to separate solute in the solution has been widely used in recent years due to its high separation efficiency and low energy consumption.13–15 The dyes, inorganic salts, and water molecules in dye wastewater can be separated by membrane separation technology without dye degradation. 16 This signifies that the dyes, inorganic salts, and fresh water can be recycled, thus minimizing wastage. Therefore, membrane separation technology has become one of the hot frontier research topics in dye wastewater treatment. 17
Throughout the dye wastewater treatment, microfiltration (MF) membrane, ultrafiltration (UF) membrane, nanofiltration (NF) membrane, and reverse osmosis (RO) membrane have been widely used. 18 Among the separation membranes, UF membrane and NF membrane have gained more attention due to their high dye rejection, salt passage, and permeability. Non-solvent induced phase separation (NIPS) method is widely used to prepare UF membrane19,20 and NF membrane 21 due to its simple fabrication process and high performance of the prepared membranes. In order to enhance the membrane properties and surmounting the trade-off relationship between permeability and rejection, the nanoparticles such as graphene oxide (GO),22,23 Metal-organic frameworks (MOFs)24,25 and molybdenum disulfide (MoS2)26,27 Karimi et al. are often added to the polymer matrix for the modification of polymer membrane. Among the organic nanoparticles, Metal-organic frameworks (MOFs) have received extensive attention in the field of membrane separation in advantage of its high specific surface area, porous structure, excellent heat resistance and chemical resistance.28,29 The porous structure of MOFs can serve as the additional pathway for selective permeation and increasing the porosity of membrane.30,31 Dai et al. 32 prepared polyphenylsulfone (PPSU)/ZIF-8 resin microspheres (RMs) membranes for solvent-resistant nanofiltration via NIPS method. Liu et al. 33 prepared polyvinylidene fluoride (PVDF)/ZIF-8 mixed matrix membrane with different particle size for dye separation via NIPS method. Sun et al. 34 prepared polysulfone (PSf) UF membrane with hydrophilic hollow ZIF-8 (hZIF-8) as the functional additive. Mahdavi et al. 25 prepared PSf/MIL-53(Al) mixed matrix membranes via vapor-induced phase separation (VIPS)-NIPS method for separation of dye solutions. They all founded that the introduction of MOFs was beneficial to improve the performance of separation membrane.35,36 However, to date, few works can be found in the literature which related to regulating the structure of membrane after the polymer/MOFs matrix mixed membrane was prepared. While the low stability of MOFs under certain conditions 37 made it possible to control the structure of polymer/MOFs matrix mixed membrane.
In recent years, poly (m-phenylene isophthalamide) (PMIA) has gained more and more attentions in the research of nanofiber membrane,38,39 UF membrane 40 and NF membrane22,41–43 due to its superior properties. The large number of rigid benzene ring structures, amide bonds, intermolecular and intramolecular three-dimensional hydrogen bond structures in PMIA macromolecular made it had an excellent rigidity, high temperature resistance and hydrophilicity.44,45 Furthermore, the thermal stability of PMIA makes it a probably promising membrane material for dye wastewater treatment as the textile industry produces wastewater with temperatures that can go up to 90–95°C. 46 Shen’s group47,48 prepared MIL-53(Al) modified membranes with PMIA as the matrix via NIPS method and explored the effect of MIL-53(Al) content on the structure and performance. The PMIA/MIL-53(Al) membrane had a high rejection of dyes while a low rejection of salts, which could realize the efficient separation of dyes and salts in dye wastewater. To the best of our knowledge, there is no works which report the effects of ZIF-8 on the properties of PMIA membrane.
In view of this, the PMIA/ZIF-8 hybrid membrane was prepared via NIPS method in this study. The effects of ZIF-8 content and the wet-chemical etching on the structure and performance of PMIA/ZIF-8 membrane were explored. The ZIF-8 nanoparticles were used as an additive to control the pore structure of the PMIA/ZIF-8 membrane. In addition, the ZIF-8 nanoparticles will be etched via wet-chemical etching method by acid solution. This will further increase the porosity and regulate the pore structure of PMIA/ZIF-8 membrane. The results indicated that the prepared PMIA/ZIF-8 membrane possessed enhanced permeability and improved the separation efficiency of dye wastewater.
Experimental section
Materials
The Molecular weight and structure of dyes.

Synthesis of ZIF-8 nanoparticles
1.098 g zinc acetate dihydrate was dissolved in 50 mL deionized water to form solution A. 8.21 g Hmim was dissolved in 50 mL deionized water to form solution B. Then, two solutions were mixed and stirred for 30 min at 70°C. During this process, white powders were gradually formed in the solution. After that, white powders were separate from the solution via centrifugal separator. The white powders were washed very carefully with methanol and deionized water respectively. Finally, the product was dried at 70°C overnight.
Preparation of PMIA/ZIF-8 membrane
The PMIA/ZIF-8 membrane was prepared by NIPS method (shown in Figure 1). The composition of PMIA dope solution was listed in Table 2. Firstly, LiCl powder, ZIF-8 nanoparticles and DMAc were placed into a three-necked round bottom flask. The mechanical stirring was used to make the LiCl and ZIF-8 dispersed or dissolved in DMAc solution. After that, PMIA staples were added to the above solution at 70°C and kept stirring for about 5 h until PMIA completely dissolved. Then the dope solution was placed in vacuum oven at 70°C for about 1 h to remove the tiny bubbles. The dope solution was cast on a glass plate using a casting knife. Then the glass plate was put into a deionized water bath at room temperature after evaporated 2 min at 70°C. The membrane was kept in deionized water for about 24 h before use. The PMIA/ZIF-8 membrane with different ZIF-8 content was named MB0, MB1, MB2, MB3 and MB4, respectively. The schematic diagram of the preparation of PMIA/ZIF-8 membrane. The composition of PMIA/ZIF-8 dope solution.

Wet-chemical etching of PMIA/ZIF-8 membrane
The PMIA/ZIF-8 membrane was placed in an acid solution with pH = 3 about 48 h after the PMIA/ZIF-8 membrane kept in deionized water 24 h. Then it was taken out and placed in deionized water for later use. According to the content of ZIF-8, the PMIA/ZIF-8 membrane (MBx, x = 0, 1, 2, 3, 4) after wet-chemical etching were named Sx (x = 0, 1, 2, 3, 4), respectively. The ZIF-8 nanoparticles in PMIA/ZIF-8 membrane were etched during wet-chemical etching process as they were unstable in acid solutions. Thereby, the permeability performance of PMIA/ZIF-8 membrane was improved.
Characterization
The morphology of the samples was observed with the field-emission scanning electron microscope (SEM, Sigma 500, Zeiss, Germany). The sample needed to be dried and sprayed with gold before the observation.
The element content on the surface of the sample was tested using the Thermo Fisher Scientific K-Alpha X-ray Photoelectron Spectrometer (XPS). The excitation source used in the test was Al Kα rays, the working voltage was 12 kV, and the filament current was 6 mA. The crystalline structure of the sample was tested using the Ultima IV X-ray Diffractometer (XRD) of Rigaku Co, Ltd The scanning range was 2θ = 5°–40°, the working voltage was 40 kV, and the working current was 40 mA. The static water contact angle of membrane samples was tested by a contact angle meter (WCA, JC-2000, Zhong Chen, Shanghai, China). The mechanical properties of samples were tested using a universal testing machine (SUNS UTM5105, Shenzhen, China) with a tensile speed of 1 mm·min−1. The tensile strength and breaking elongation of membranes were averaged by testing three samples with a size of 5 mm × 3 cm. The surface potential of membrane was determined by streaming potential method using an electrokinetic analyzer (SurPASS, Anton Paar, Austria) with the pH ranging from 10 to 3. The samples were thoroughly washed with deionized water and then freeze-dried for 12 h before test.
The porosity (ε) of PMIA/ZIF-8 membrane was measured by dry-wet gravimetric method and calculated by equation (1)
49

The mean pore radius (r
m
) of PMIA/ZIF-8 membrane was calculated by Guerout-Elford-Ferry equation (equation (2)) on basis of pure water flux and porosity.
50

Membrane permeability
The self-made crossflow filtration system (shown in Figure 2) with an effective testing membrane area of 21.2 cm2 was used to test the permeability of membrane. Each membrane was tested at 0.1 MPa for at least 60 min. The concentration of salt solution and dye solution was 2 g·L−1 and 0.1 g·L−1, respectively. The water flux was calculated by equation (3) The schematic diagram of the self-made cross-flow filtration system.

The solute rejection of the membrane was calculated by equation (4)
Results and discussion
Characterizations of ZIF-8 nanoparticles
The SEM morphology and XRD spectrum of ZIF-8 nanoparticles were shown in Figure 3. As shown in Figure 3(a), the ZIF-8 nanoparticles had a typical rhombic dodecahedron structure. This was the common structural shape of ZIF-8 nanoparticles.
51
Figure 3(b) showed the particle size distribution of ZIF-8 nanoparticles. The particle size of ZIF-8 nanoparticles was almost in 0.6–1.6 μm, while the mean particle size was about 1.09 μm. The crystal structure of the synthesized ZIF-8 nanoparticles was tested with an X-ray diffractometer. The result was shown in Figure 3(c). The position and intensity of the diffraction peaks of ZIF-8 nanoparticles was basically consistent with that of the ZIF-8 simulated crystal structure data. The XRD pattern data was in good agreement with the ZIF-8 simulated crystallization data. It proved that the crystal structure of ZIF-8 nanoparticles in this study was relatively perfect. The SEM morphology (a), particle size distribution (b) and XRD curves (c) of ZIF-8 nanoparticles.
Characterizations of PMIA/ZIF-8 membrane
Figure 4 showed the SEM morphology of PMIA/ZIF-8 membrane top surface with different ZIF-8 content. Figure 4(a1) showed the surface morphology of PMIA membrane without ZIF-8. It can be seen that the surface of the PMIA nanofiltration membrane was relatively dense and smooth. Figure 4(a2-a5) showed the surface morphology of PMIA/ZIF-8 membrane with ZIF-8 nanoparticles. It can be seen that there was some ZIF-8 nanoparticles appeared on the surface of the prepared membrane after adding ZIF-8 nanoparticles to the dope solution. At the same time, the number of ZIF-8 nanoparticles on the surface of PMIA/ZIF-8 membrane increased with the incensement of ZIF-8 content in the dope solution. When the content of ZIF-8 increased to 0.6 wt%, the ZIF-8 nanoparticles on the surface of the MB4 showed an agglomeration phenomenon (Figure 4(a5, b5)). It suggested that the content of ZIF-8 may be excessive, which resulted in the agglomeration of ZIF-8 nanoparticles. The top surface SEM morphologies of PMIA/ZIF-8 membrane with different ZIF-8 content: (1) 0 wt%, (2) 0.15 wt%, (3) 0.3 wt%, (4) 0.45 wt%, (5) 0.6 wt%; (a) before and (b) after wet-chemical etching.
ZIF-8 nanoparticles could exist stably in water, but they were relatively unstable in acid solutions and were easily etched by acid solutions. Therefore, the PMIA/ZIF-8 membrane was placed in an acid solution for the wet-chemical etching. This was helpful to further improve the performance of PMIA/ZIF-8 membrane. The surface morphology of the PMIA/ZIF-8 membrane after wet-chemical etching was shown in Figure 4(b). As shown in Figure 4(b1), the PMIA membrane showed a similar surface morphology compared with that of MB0 which was shown in Figure 4(a1). This indicated that the wet-chemical etching had no obvious effect on the structure of PMIA membrane. While the ZIF-8 nanoparticles in PMIA/ZIF-8 membrane surface appeared a cavity structure (shown in Figure 4(b2-b5)). This indicated that the ZIF-8 nanoparticles on the surface were etched by the acid solution.
Figure 5 showed the SEM morphology of the cross-section of the PMIA/ZIF-8 membrane. As shown in Figure 5(a1), the PMIA/ZIF-8 membrane had a typical asymmetric pore structure. There was a denser skin layer close to the upper surface of the membrane. The separation of the solute was mainly completed by this skin layer. The following were the finger-like pore structure and the sponge-like pore structure, which could provide channels for the passage of water molecules and maintained a certain mechanical property. The formation of the asymmetric cross-sectional structure of PMIA/ZIF-8 membrane was related to the double diffusion process of solvent and non-solvent in the process of NIPS method. When the glass plate was initially immersed in the coagulation bath, a large amount of solvent diffuses rapidly from the surface of the dope solution into the coagulation bath. While the diffusion of non-solvent was relatively slow. Thus, the polymer concentration at the junction of the dope solution and the coagulation bath increases which lead to form a dense skin layer. The part close to the dense skin layer had a faster double diffusion rate which was conducive to the formation of finger-like pores. The bottom of the membrane had a slower double diffusion speed which was conducive to the formation of sponge-like pores. Therefore, the cross section of PMIA/ZIF-8 membrane showed a typical asymmetric structure. The enlarged cross-sectional image was shown in Figure 5(a2). As shown in Figure 5(a2), the ZIF-8 nanoparticles could be clearly seen in the cross section of PMIA/ZIF-8 membrane. This showed that the membrane was successfully modified by ZIF-8 nanoparticles. SEM image of the cross-section of PMIA/ZIF-8 membrane with different magnification: (a) before and (b) after wet-chemical etching.
The cross-sectional morphology of PMIA/ZIF-8 membrane after wet-chemical etching was shown in Figure 5(b). As shown in Figure 5(b1), the cross-sectional structure of the nanofiltration membrane was asymmetrical with the untreated one. It can be seen from Figure 5(b2) that the ZIF-8 nanoparticles in PMIA/ZIF-8 membrane were etched into a hollow structure after wet-chemical etching. Therefore, the wet-chemical etching was helpful to increase the porosity of the membrane which was conducive to the permeation of water molecules. In view of this, the permeability of PMIA/ZIF-8 membrane may be improved after wet-chemical etching.
In order to evaluate the elemental composition of the samples, XPS test was performed on the samples. The result was shown in Figure 6. Figure 6(a) showed the wide scan spectra of the samples. The MB3 and S3 had characteristic peaks of Zn 2p at the binding energy of 1044.7 eV (Zn 2p1) and 1021 eV (Zn 2p3) when compared to MB0. It showed that ZIF-8 nanoparticles were present in MB3 and S3 which indicated that ZIF-8 nanoparticles were successfully added to PMIA/ZIF-8 membrane. Figure 6(b) showed the high-resolution spectrum of the Zn element. The MB0 had no absorption peak of Zn 2p while MB3 and S3 had an obvious absorption peak of Zn 2p. The peak intensity at 1044.7 eV (Zn 2p1) and 1021.6 eV (Zn 2p3) of MB3 was significantly stronger than that of S3. This indicated that MB3 contained more ZIF-8 nanoparticles than S3. The surface elemental composition of various samples was shown in Table 3. The content of Zn contained in the surface of MB3 was 2.45 at% while the content of Zn contained in the surface of S3 was only 0.21 at% which was much lower than MB3. This was because that ZIF-8 nanoparticles were not stable under acidic conditions.
37
The free protons(H+) in acid solution can destroy the coordination bond between Zn and 2-Hmim of ZIF-8 crystals.
52
Therefore, the ZIF-8 nanoparticles in PMIA/ZIF-8 membrane were etched by the acid solution during the wet-chemical etching process which resulting in the decrease of Zn content in the surface of S3. This low stability of ZIF-8 nanoparticles under acidic conditions was used for creating voids inside PMIA/ZIF-8 membrane which was helpful to improve the permeability. The XPS curves of samples: (a) wide scan spectra and (b) high resolution Zn 2p. The surface elemental composition of samples.

To further assess the effect of the addition of ZIF-8 nanoparticles and wet-chemical etching on the properties of PMIA/ZIF-8 membrane, the water contact angle and mechanical strength of PMIA/ZIF-8 membrane was measured. The results were shown in Figure 7(a) and Figure 7(b). As shown in Figure 7(a), the water contact angle of PMIA/ZIF-8 membrane increased with the increase of ZIF-8 content as ZIF-8 nanoparticles were hydrophobic. While the water contact angle of PMIA/ZIF-8 membrane showed a slightly decline after wet-chemical etching due to the decrease of ZIF-8 nanoparticles on membrane surface. The effect of ZIF-8 contents and wet-chemical etching on the hydrophilicity (a) and mechanical strength (b) of PMIA/ZIF-8 membrane; (c) zeta potential of S3.
The stress-strain curves of PMIA/ZIF-8 membrane was shown in Figure 7(b). The elongation of PMIA/ZIF-8 membrane slightly decreased while the tensile strength increased with the addition of ZIF-8 nanoparticles. This was consistent with the previous study.28,53 The proper addition of inorganic component in polymer membranes could modestly increase the mechanical strength of the membranes due to the interaction between particles and polymer chains.28,41 The elongation and tensile strength of S0 barely changed compared to that of MB0 as PMIA is stable in acid solution. 54 Meanwhile, the tensile strength of S1 and S3 slightly increased compared to that of MB1 and MB3. As discussed above, the H+ ions in the wet-chemical solution would lead to the breakage of the Zn-N bonds (Supplemental Figure S1). It is beneficial to form hydrogen bond between ZIF-8 nanoparticles and PMIA chains. 18 Therefore, the tensile strength slightly increased.
The surface charge of nanofiltration membrane has a crucial impact on its separation performance. The surface charge properties of S3 were tested by zeta potential and the result was shown in Figure 7(c). The isoelectric point (IEP) of S3 was 3.24. It means that the S3 exhibited negative zeta potentials when the pH of solution was higher than 3.24. The PMIA/ZIF-8 membrane was negatively charged over a wide pH range. Therefore, S3 had a strong repulsive force to negative solute in feed solution.
Separation performance
In order to explore the effect of ZIF-8 content on the permeability of PMIA/ZIF-8 membrane. The pure water flux of PMIA/ZIF-8 membrane with different ZIF-8 content was tested. The result was shown in Figure 8. It can be seen from Figure 8(a) that the pure water flux of the membrane basically remain unchanged as the test time prolonged. This indicated that PMIA/ZIF-8 membrane had better pressure resistance due to the large number of rigid benzene rings in PMIA macromolecule. This could endow PMIA/ZIF-8 membrane with excellent pressure resistance. It can be seen from Figure 8(b) that the water flux of PMIA/ZIF-8 membrane significantly increased with the increase of ZIF-8 content. This was because the addition of ZIF-8 nanoparticles in the dope solution was beneficial to increase the interface pores between the organic and inorganic phases in the nanofiltration membrane, which was helpful to increase the permeability of PMIA/ZIF-8 membrane. However, the water flux of PMIA/ZIF-8 membrane decreased as the content of ZIF-8 increased to 0.6 wt%. This was because the addition of ZIF-8 will increase the viscosity of the dope solution.
32
The increase in the viscosity of the dope solution reduced the double diffusion rate, which was not conducive to the improvement of the water flux of the membrane.
55
Moreover, the agglomeration of ZIF-8 nanoparticles also lead to the decrease of water flux. Therefore, the water flux of PMIA/ZIF-8 membrane decreased as the content of ZIF-8 increased to 0.6 wt%. Even so, the introduction of ZIF-8 nanoparticles was helpful to increase the permeability of PMIA/ZIF-8 membrane. Effect of ZIF-8 content on the permeability of PMIA/ZIF-8 membrane: (a) time-dependent pure water flux of PMIA/ZIF-8 membrane; (b) the pure water flux of PMIA/ZIF-8 membrane after running for 60 min; (c) the water flux and rejection of PMIA/ZIF-8 membrane with Congo red solution as the feed solution.
In order to further characterize the effect of ZIF-8 content on the permeability of PMIA/ZIF-8 membrane, the Congo red solution was used as the feed solution to test the permeability of the PMIA/ZIF-8 membrane. The result was shown in Figure 8(c). As shown in Figure 8(c), the effect of ZIF-8 content on the water flux of PMIA/ZIF-8 membrane was consistent with that of the pure water flux in Figure 8(b). While the rejection of Congo red decreased slightly with the increase of ZIF-8 content. The result showed that the PMIA/ZIF-8 membrane could realize the efficient separation of Congo red from Congo red solution as the rejection of Congo red was higher than 98%.
In order to verify the effect of wet-chemical etching on the properties of PMIA/ZIF-8 membrane, the permeability of PMIA/ZIF-8 membrane was measured with Congo red as feed solution. The result was shown in Figure 9(a). It can be seen that the water flux of PMIA/ZIF-8 membrane had a significant increase after acid treatment. This was because ZIF-8 was unstable with acid solution and will be etched to a certain extent as shown in Figure 5. It was helpful to increase the porosity of the PMIA/ZIF-8 membrane, which was beneficial to improve the water flux of membrane. Meanwhile, the acid treatment had no obvious effect on the rejection performance of PMIA/ZIF-8 membrane. When the content of ZIF-8 was 0.45 wt%, the PMIA/ZIF-8 membrane had a higher water flux while maintaining stable rejection performance. Therefore, the S3 was used in the following studies. The acid treatment on the permeability of PMIA/ZIF-8 membranes: (a) Congo red solution as the feed solution; (b) the permeability of S3 with different feed solution.
The separation performance of S3 with different feed solution was shown in Figure 9(b). The initial concentration of dye solution and salt solution was 0.1 g·L−1 and 2 g·L−1, respectively. As shown in Figure 9(b), the rejection ratio of S3 for Congo red (CR) and direct red 80 (DR 80) was approximately 100%. While the rejection ratio of S3 for salt molecule was less than 10%. This was because the molecule size of salts was smaller than that of dyes. Hence, the salt molecules were easy to enter the pore and pass through the membrane. Moreover, the water flux of S3 for Congo red solution was higher compared with that of DR 80 solution. Therefore, the Congo red solution was used as the feed solution in following studies.
During the separation process, the operating conditions, such as operating pressure, feed concentration, and operating temperature had great effects on the performance of membranes.
56
Figure 10 showed the effects of operating conditions on permeability of S3 membrane. The feed solution was Congo red aqueous solution. Figure 10(a) showed the effect of operating pressure on permeability of S3 membrane. The membrane was tested under different operating pressure for 60 min, respectively. The operating temperature and Congo red concentration of the feed solution were 25°C and 0.1 g·L−1. As shown in the Figure 10(a), the water flux increased while the rejection of the Congo red slightly decreased as the operating pressure increased. Increasing the operating pressure will accelerate the penetration of water molecules through the membrane, which was conducive to the increase of water flux. At the same time, the higher pressure also allows the dye molecules to overcome the electrostatic repulsive force to pass through the membrane. Therefore, the rejection of Congo red slightly decreased. The influence of dye concentration on permeability of S3 was shown in Figure 10(b). The Congo red concentration ranged from 0.1 g·L−1-0.5 g·L−1. The operating temperature and pressure were 25°C and 0.1 MPa, respectively. The water flux slightly decreased with the increase of dye concentration, while the rejection remained basically unchanged. This was because the high Congo red concentration lead more dye molecules to deposited on membrane surface and block the membrane pores, which caused the decrease of water flux. The influence of operating temperature on permeability of S3 was shown in Figure 10(c). The membrane was tested under different operating temperature for 60 min, respectively. The operating pressure and Congo red concentration of the feed solution were 0.1 MPa and 0.1 g·L−1. The water flux of PMIA/ZIF-8 membrane increased from 104.7 L·m−2·h−1–175.5 L·m−2·h−1 as the operating temperature increased from 25°C to 80°C. Meanwhile, the rejection of the Congo red just slightly decreased from 98.6% to 94.9%. This indicated that increasing the operating temperature could greatly increase the water flux, while had little effect on the retention performance of S3. The viscosity of the feed solution decreased with the increase of operating temperature. This was beneficial to increase the diffusion coefficients of water molecules and enhanced the water flux of S3.
57
Therefore, the water flux of S3 increased with the increase of operating temperature. The slight change in rejection performance indicated that the pore structure of S3 remained undamaged. In order to prove this conclusion, the operating temperature was reduced from 80°C to 25°C. The rejection performance of S3 for Congo red was almost unchanged compared to initial, which further proved that the pore structure of the nanofiltration membrane was not damaged during high-temperature operation conditions. This was because the excellent thermal stability of the PMIA could resist the pore expansion when operating temperature increased. This indicated that the prepared PMIA/ZIF-8 membrane could be applied for the separation process under higher operating temperature. This was consistent with our previous work.44,58 Figure 10(d) showed the photograph of feed solution and permeate solution. It showed that the Congo red in the feed solution was rejected by PMIA/ZIF-8 membrane. The prepared PMIA/ZIF-8 membrane showed a potential application in dye removal from aqueous solution. The effects of operating conditions on the permeability of S3 membranes: (a) Operating pressure; (b) Congo red concentration; (c) Operating temperature; (d) De-colorization of S3 to Congo red.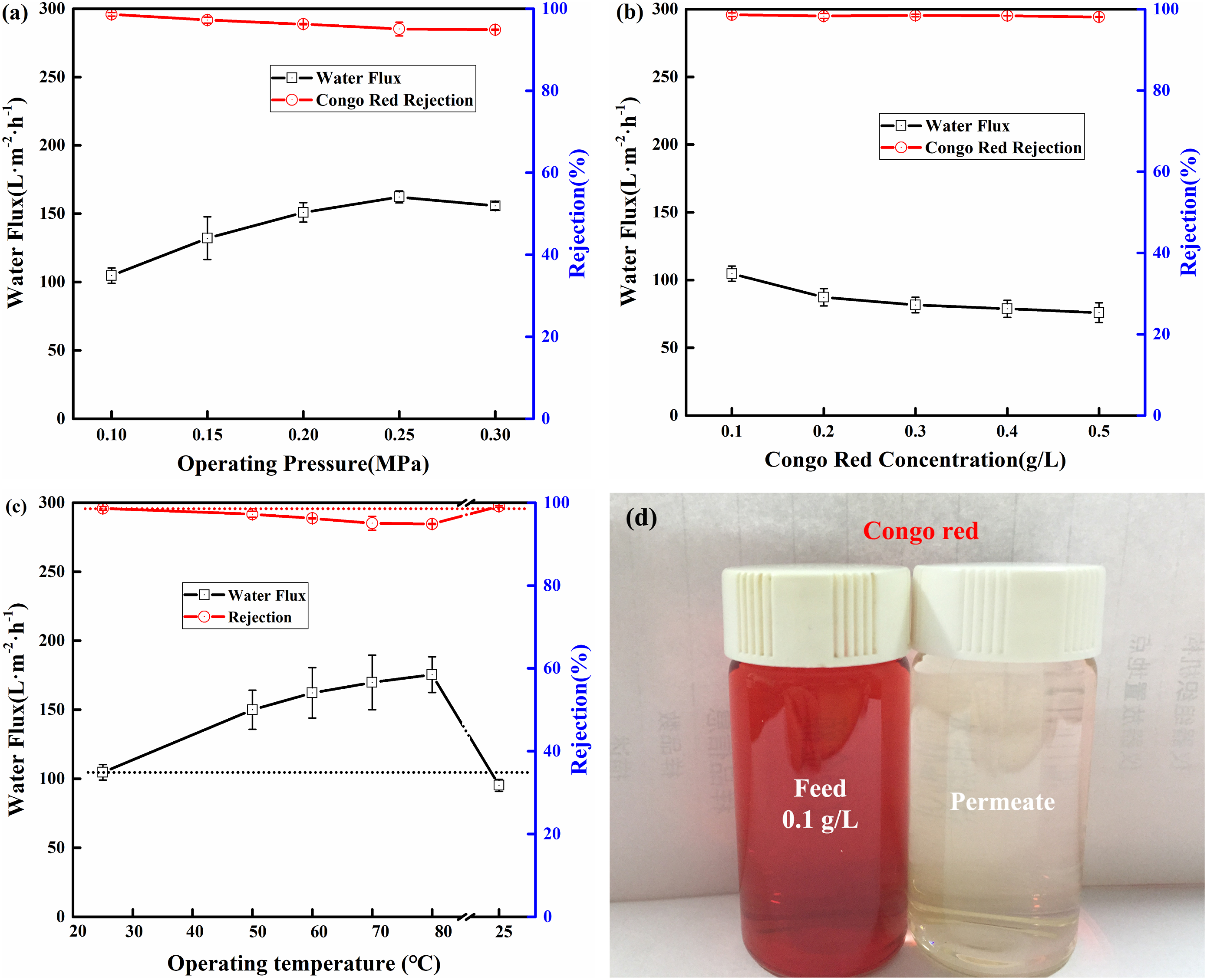
The long-term stability of MB3 and S3 was tested with 100 mg·L−1 Congo red solution as the feed solution. The results were shown in Figure 11. As shown in Figure 11(a), the water flux of MB3 and S3 had a decreasing trend with the extension of the operating time. While the decline of S3 was smaller. The change tendency of the rejection of Congo red dye molecules was similar to that of the water flux. The decrease of water flux and Congo red rejection might be caused by the deposition of dye molecules on membrane surface. The less decrease in water flux and rejection of S3 indicated that S3 had a better anti-fouling performance. While in the filtration tests, both MB3 and S3 showed low salt rejection rate for Na2SO4 (<10%) during the whole process. This indicated that PMIA/ZIF-8 membrane had remarkable fractionation performance for the separation of dye/salt in aqueous solution. The long-term stability of PMIA/ZIF-8 membrane: (a) water flux (b) rejection; (c) flux recovery of PMIA/ZIF-8 membrane during the fouling test.
To further evaluated the anti-fouling performance of PMIA/ZIF-8 membrane, the fouling test via crossflow filtrations was performed. The filtration was firstly operated for 5 h using 300 mg·L−1 Congo red aqueous solution as the feed solution and recorded the water flux every 10 min. After that, the PMIA/ZIF-8 membrane was thoroughly and gently cleaned using polyurethane sponge. 59 Finally, filtration of 300 mg·L−1 Congo red was operated for 1 h by recording water flux every 10 min. The water flux during the fouling test was shown in Figure 11(c). The water flux of MB3 and S3 showed a same decreasing tendence due to the deposition of dye molecules on membrane surface. After the filtration was operated for 5 h, the fouled membrane was cleaned using a polyurethane sponge. The water flux was recovered more than 95% of the original while the recovery ratio of S3 (97.6%) was higher than that of MB3 (96.5%). The result further demonstrated that the properties of PMIA/ZIF-8 membrane were improved via wet-chemical etching process.
Conclusion
In this work, the ZIF-8 nanoparticle incorporated PMIA membrane was prepared via NIPS method. Firstly, the effect of different ZIF-8 loading in dope solution were investigated. The results showed that the addition of ZIF-8 improved the separation performance of PMIA/ZIF-8 membrane. When 0.45 wt% ZIF-8 nanoparticles were added, the water flux of MB3 was 68.9 L·m−2·h−1, which was about 10 times than that of the membrane without ZIF-8, while the Congo red rejection maintained stable. Then, the PMIA/ZIF-8 membrane with 0.45 wt% ZIF-8 loading (MB3) was further treatment via wet-chemical etching process. After wet-chemical etching, the water flux of S3 was about 1.5 times higher than that of MB3 while the Congo red rejection still higher than 98%. This was attributed to that the ZIF-8 nanoparticles in the membrane were etched by the wet-chemical etchant solution. The results showed that the addition of ZIF-8 nanoparticles and wet-chemical etching treatment improved the properties of PMIA/ZIF-8 membrane. The prepared PMIA/ZIF-8 membrane provided a facial method for improving the properties of ZIF-8 modified polymer matrix membrane. The PMIA/ZIF-8 membrane showed certain application advantages in the field of dye wastewater treatment for its thermal stability and high separation efficiency. The improvement in antifouling performance should be a key work on future research before commercializing the product.
Supplemental Material
Supplemental Material - ZIF-8 modified poly (m-phenylene isophthalamide) (PMIA) hybrid membrane for dye wastewater treatment
Supplemental Material for ZIF-8 modified poly (m-phenylene isophthalamide) (PMIA) hybrid membrane for dye wastewater treatment by Kailu Yang, Xibo Liu, Xinya Wang, Tianxu Hao, Wei Zhang, Mingxing Chen in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by Science and Technology Project of Hebei Education Department (QN2022128), the Key Research and Development Project of Hebei Province (20271202D), and the Natural Science Foundation of Hebei Province (E2022208027).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
