Abstract
The main objective of the present review article is to investigate the
Keywords
Introduction
Cardiovascular disease is the leading cause of death in the healthcare system in developing countries.1,2 The replacement of the diseased blood vessel is the most widely used treatment for vascular diseases. 3 The available treatment options include autografts, allografts, and xenografts. 3 Autograft has been presented as the most accepted technique in regenerative medicine.4,5 Saphenous veins, internal mammary and radial arteries have been referred to as the “gold standard” in autograft treatment. 6 But in 30% of patients, the mentioned arteries are not available due to small size, varicosities, anatomic variation, previous removal, or other abnormalities.7,8 Hence, vascular tissue engineering (VTE) and synthetic vascular grafts have been introduced as promising treatment in vascular regenerative medicine.5,9 However, using the synthetic vascular grafts with poor biocompatibility frequently lead to occlusion owing to thrombosis due to platelet adhesion, over-proliferation of smooth muscle cells (SMCs), re-endothelialization, and intimal hyperplasia (IH).10,11
These characteristics can ensure the fabrication of a graft with long term patency in in-vivo conditions. 12
Polycaprolactone (PCL) is a biodegradable, semi-crystalline, and synthetic polyester polymer, and it has shown great potential in being used as a long-term degradable implant in biomedical applications.13–15 PCL, as a strong material with optimum mechanical properties, has been approved for VTE by FDA.16,17
Nevertheless, the cell attachment, proliferation, and endothelialization on the PCL surface have been limited due to its low hydrophilicity, and its biocompatibility is still highly concerned for optimum in-vivo performance in VTE.18–20 Biocompatibility is a significant parameter in success clinical application of synthetic vascular grafts as the blood-contacting structure. 21 Biocompatibility of synthetic vascular grafts involves cell compatibility or cytocompatibility (no cell toxicity and no inflammation) and blood compatibility or hemocompatibility (no thrombosis and no vessel occlusion). 22 The albumin, as the most abundant protein in the blood (50–60%), tends to be absorbed to a hydrophobic surface compared to a hydrophilic surface. 23 Thrombosis formation on the luminal surface of PCL has remained to be an issue in using PCL as SDVGs. 24
Therefore, development and fabrication of the biocompatible synthetic vascular grafts with hybrid structures using other polymers are extremely needed due to their excellent biocompatibility and hemocompatibility.25,26
In many studies, attempts to apply biodegradable polymers supported by PCL to meet the required biomechanical requirements for successful clinical applications have been made. 27
Biodegradable polymers that are currently being investigated for fabrication of the electrospun synthetic vascular grafts polymers can be divided into synthetic and natural polymers. 28 The synthetic polymers have exhibited suitable biodegradable and mechanical properties, but they are limited in VTE due to their hydrophobic surface, low immune response, and poor biocompatibility.29,30
Against natural polymer scaffolds with biocompatibility property and hydrophilic activity can simulate the extracellular matrix (ECM) of native arteries. 31
But their poor mechanical properties are the main drawback to their usage as a material in VTE because they cannot provide sufficient mechanical strength.
32
Therefore, the fabrication of neat electrospun scaffolds with natural polymers as SDVGs in VTE is rarely commercialized. If their mechanical properties are improved, they can be applied in VTE.33,34 Nevertheless, to fabricate of the suitable synthetic graft by blend of two or more synthetic and natural polymers must be used for successful
Collagen, gelatin, and chitosan as natural hydrophilic polymers - attracted much attention in biomedical applications due to their high bioactivity, biodegradability, biocompatibility, renewability, and abundant availability.36,37
According to the best of the authors’ knowledge, numerous studies have been published on the PCL-based scaffolds for VTE.38–41 But the
Accordingly, a significant emphasis and the object of this review is the evaluation of the role of the natural polymers to develop the biocompatible hybrid of PCL-based scaffolds as SDVGs for improvement in growth and monolayer proliferation of ECs based on their
Structure of native arteries and the functional requirements for synthetic vascular grafts:
The wall of arteries and veins (Figure 1) consists of three distinct layers called the intima, media, and adventitia, respectively.42–44 The tunica intima is the thinnest layer consisting of a continuous monolayer of ECs, and sub-endothelial elastic lamina.
45
Structures of a typical Artery and Vein: the arteries and veins consist of three main layers that called: tunica extreme (outer layer), tunica media (middle layer), and tunica intima (inner layer).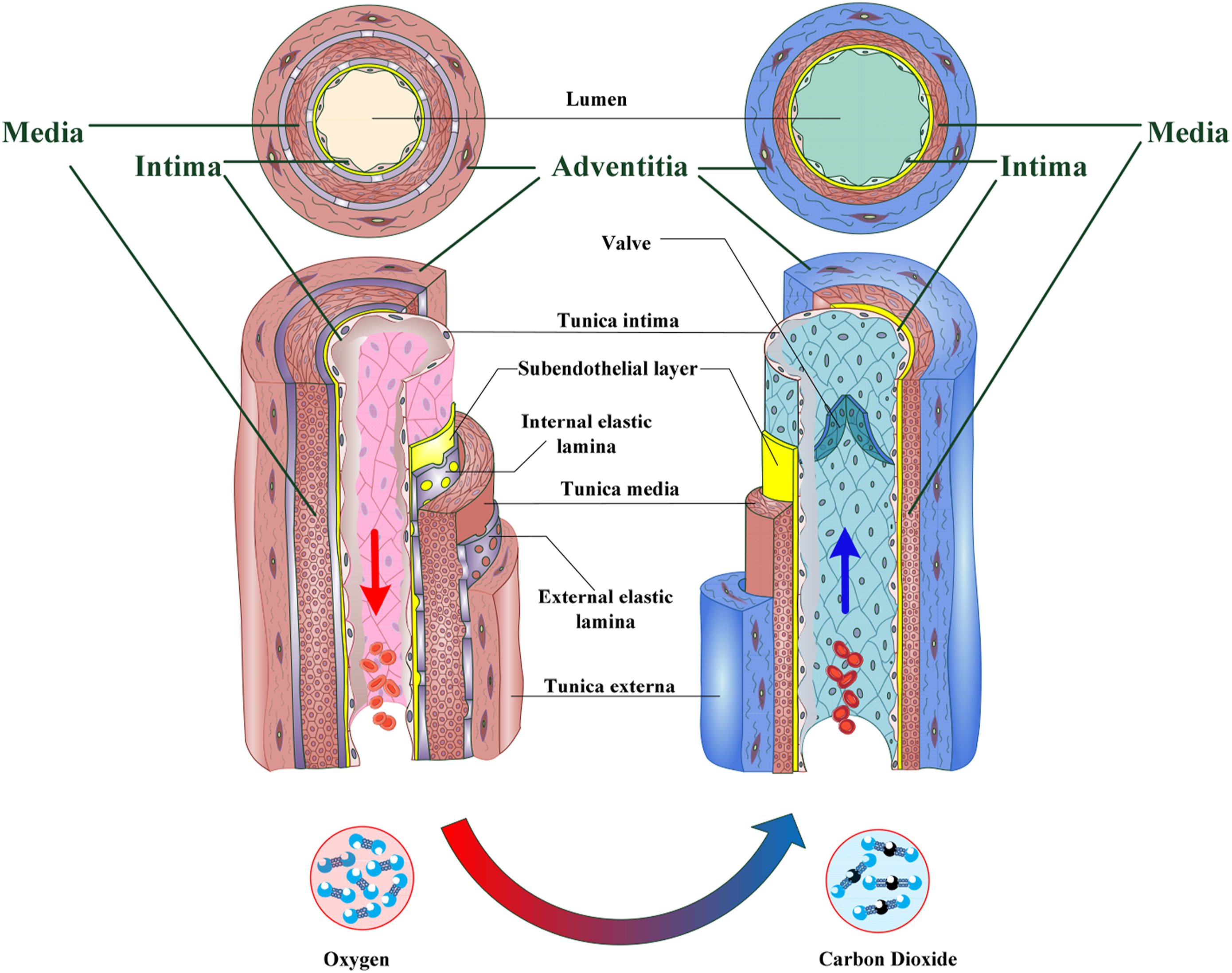
This layer serves as a thrombo-resistant barrier, and covers the luminal surface of the arteries, and plays a key role in monitoring and controlling the endothelialization process. 46 The tunica intima, by recoiling the artery, mediating SMCs proliferation, and ECM depositions-plays a significant role in preventing IH.47,48 The middle layer called the tunica media, contributes to the maintenance of normal blood pressure by controlling the caliber of the arteries.49,50 The outermost layer is called tunica adventitia and consists of connective fibers and is surrounded by an external elastic lamina. 51 This layer is often thicker in veins to prevent the collapse and damage of the blood vessel. 52
In this regard, according to the structure of native arteries, finding a suitable substitute with an anti-thrombogenic structure is an essential challenge in VTE. 53
Vascular tissue engineering: historical perspective, current situation, breakthroughs
The tissue engineering of vascular grafts divide into four main parts that consists of scaffold fabrication, mechanical properties, in-vitro characterization, and in-vivo evaluation (Figure 2).54–58 ‘The primary strategy in this field is based on using bio-stable synthetic materials such as expanded polytetrafluoroethylene (ePTFE, Teflon), polyethylene terephthalate (PET), and polyurethane (PU).59–65 Fabrication methods and evaluation process of small diameter vascular grafts. (1) Scaffold fabrication: The vascular tissue engineering scaffolds can be fabricated by different methods: electrospinning, casting, freeze drying and three-dimensional printing (3D printing), (2) Compliance, burst pressure and axial/radial tensile strength as important mechanical properties of fabricated scaffolds will be evaluate according to the presented standard methods of Association for the Advancement of Medical Instrumentation (ANSI/AAMI/ISO 7198:1998/2001); (3) In-vitro characterization of VTE evaluated using MTT assay, 2D and 3D cell seeding; (4) The fabricated VTE can be implanted directly in-vivo with/without seed cells; the native blood vessel can regenerate in situ.
These structures, as synthetic vascular grafts in large fiber diameter (D > 6 mm), have shown successful clinical application.
66
But they failed success in small diameter vascular grafts (SDVGs, D < 6 mm) due to chronic foreign body reaction with IH, and acute thrombosis.67,68 Fabrication of the suitable scaffold with an anti-thrombogenic luminal surface to permit the integrity of an EC monolayer is considered crucial to ensure the patency and optimum
According to the Figure 2, there are different methods such as electrospinning, freeze drying, and 3D-printing methods for fabrication SDVG.71,72 That among them, the electrospinning introduces as a simple, promising, effective, comprehensive, and novel procedure method. Furthermore, the electrospinning method was developed to generate continuous fibers for fabrication non-woven structures in the sub-micron to nanometer scale range by employing electrostatic forces.73,74 As shown in Figure 2 the compliance, burst pressure and tensile strength introduced as the important mechanical properties for evaluating the clinical applications of fabricated SDVG. 75
Also, their biological properties are analyzed by
An electrospinning setup consists of three major components, as shown in Figure 3: a high voltage power supply, a syringe with a metallic needle, and a collector.78,79 During the electrospinning process, the polymer dissolved into special solvent. When the high voltage is supplied, the polymer solution discharged under the electrostatic forces by a jet at the needle tip in form of a coin shape that called Taylor cone. When the electrostatic field achieves the critical voltage, a charged jet ejects from the tip needle and a fine fiber with micro to nano-scale produce on the surface of collector.76,80 A schematic setup of a standard electrospinning setup.
In-vitro and in-vivo study of hybrid PCL-natural polymer scaffolds as SDVGs in VTE
In this section, the examination of the to-date
Collagen
To date, 28 collagen types have been identified, but collagen I (collagen) has shown excellent biological performance for VTE tissue engineering purposes. 82
Collagen is the key structural protein of native ECM (with a nanometer scale diameter) and it is also the main type that makes up the essential part of collagen in soft and hard tissues such as the bone, cartilage, tendon, skin, and muscle which surround various cells.83,84
Collagen does not show any inflammation and toxicity and it provides a favorable environment for a wide range of cells and consequently has an important role in the improvement of SDVGs
The connective tissue in bone is mainly constructed from type I collagen, forming a framework for organic and mineral phases. 87 Tendon consists of type I collagen fibrils with an average fiber diameter from 30 nm to 300 nm, and it transmits the force of muscular contraction to bone.88,89 Skin is the body’s largest organ and is composed of three layers called primarily, epidermis, and dermis. 80 percent of the dry weight of the dermis layer is mainly collagen.90,91 The cornea consists of three layers called the outer epithelium, central stroma, and inner endothelium. 92 The stroma, as the central layer, is composed of a dense and highly organized ECM of collagen fibrils with the nano-scale diameter. 93
Electrospinning of collagen can produce a fiber with nano-meter scale diameters (similar to the scale size of ECM). 48 But, the lack of elasticity and strength in electrospun collagen nanofibers, is the main problem in using collagen in the form of neat scaffolds in VTE.94,95 Accordingly, collagen/PCL-reinforced scaffolds can be suggested as a promising candidate for VTE.
To improve vascular cell responses, fabrication of the PCL-based scaffolds (D = 3 mm and the fixed composite ratio of 1:1) with collagen (with aligned distribution in the inner layer) and silica (with random distribution in the outer layer) was reported by Park et al. 96 In the mentioned research, the biocompatibility and polymer-cell interaction were evaluated by primarily cell adhesion to the fabricated structures. 97 The Human umbilical vein endothelial cells (HUVECs) and mouse fibroblast cells (L929) were seeded on the inner and outer layers of structures, respectively. They confirmed that adding collagen to PCL in the aligned inner layer has vital role in providing an anti-thrombogenic design. 70 The HUVECs have a positive influence on the release of anticoagulation factors, and that helps to prevent platelet attachment, activation, and complete occlusion and rapid endothelialization. Seeding L929 cells lead to the maintenance of the vascular tone by improving the surface hydrophilicity of PCL.
The structure with the aligned distribution of nanofibers showed better cell adhesion, and proliferation (endothelialization cover) than the other structures. Fabricated structures, due to the favorable biological responses, rapid endothelialization and absence of surface thrombosis and neointimal hyperplasia, showed a suitable healing process and gradual replacement with natural blood vessels. These findings are in agreement by the reported results by other researchers.98–100
Fabrication of the hybrid PCL/collagen scaffolds (D = 5 mm and different composite ratios 95:5, 75:25, 50:50, and 25:75) was reported by Bertram et al. 101 In hybrid scaffolds, the amount of viable adherent T17beEPCs cells significantly increased compared to neat PCL scaffolds. The cell density on the scaffold’s surface is directly related to the composite ratio of collagen. Loss of the confluent monolayer of cells is observed in high concentrations (75%) of collagen. Suitable integration regarding cell viability and functionality (involving adherence, attachment, and spread) were observed in hybrid scaffolds with 25% collagen.
Also, Collagen showed an antigenic switch in ECs and had a higher effect on viable adherent cells and lesser impact on the proliferation of cells than PCL. Electrospun collagen nanofibers have been shown unstable in aqueous environments, and a cross-linking process is essential to stabilize these fibers. 86 Fabrication of the hybrid PCL/collagen scaffolds (D = 5 mm and fixed composite ratio 1:1) using electrospinning (mixed) and co-electrospinning (separated) methods and different cross-linking processes have been done by Chen et al. 18 Collagen is characterized by a fast degradation rate and poor mechanical properties.102,103 Accordingly, Chen and his coworkers used the chemical (glutaraldehyde (GA) and carbodiimides (1-ethyl- 3-(3-dimethylaminopropyl) carbodiimide/N-hydroxysuccinimide (EDC/NHS)) and biological factor (Genipin) in the cross-linking process, for improving the water resistance and mechanical stiffness of collagen. After the cross-linking processes, the fiber of collagen was gathered, and as a result, an increase in the gap between PCL nanofibers was seen. Hydrophilicity and biocompatibility of nanofibers in PCL/collagen scaffolds increased compared to neat PCL scaffolds after the cross-linking process; which is similar to the obtained results by law et al. 102 EDS/NHS and GA considered having cytotoxicity on nanofibers, but, cross-linked scaffolds by genipin (as a natural and biocompatible cross-linking agent and at a specific concentration) were non-cytotoxic. 86
Accordingly, the mixed nanofibers cross-linked with EDC/NHS and the genipin process showed rapid endothelialization, and cell migration, and therefore, fewer adhered platelets were observed on their surfaces in comparison to separated nanofibers.
Tillman et al. developed the hybrid PCL/collagen scaffolds (D = 5 mm and fixed composite ratio 1:1) by the electrospinning method.
7
Fabricated scaffolds support the growth of ECs and SMCs under pulsatile flow conditions (bioreactor) on the inner and outer surfaces, respectively. Fabricated PCL/collagen scaffolds could control the degradation process and facilitate cell growth during remodeling
The long-term patency was observed in the innermost layer of scaffolds (lumen) after ECs cell seeding, and the mentioned structures are a promising candidate to resist platelet adherence. The results of the
Many studies have shown that the electrospun PCL nanofibers, despite having amine groups, don’t lead to a positive impact on cell proliferation and show poor efficacy of endothelialization over the surface.
104
Moreover, the modified process is needed to enhance ECs proliferation behavior on the PCL scaffolds.100,105,106 In this regard, results of
The integrity of the lumen of synthetic vascular grafts plays a key role in preventing thrombosis, and serves to maintain the patency of blood vessels, and endothelium has an essential role in the regulation of vascular hemostasis. Analyzing the properties of fabricated PCL/collagen scaffolds, confirmed the role of collagen in the fabrication of integrated lumen in synthetic vascular grafts.
Also, the strong endothelialization and density of EC growth on the scaffolds indicated significant improvement in EC coverage for PCL/collagen scaffolds compared to neat PCL scaffolds. The present study hypothesized that the efficacy of endothelialization is positively related to the percentage of collagen in hybrid scaffolds, and the 25–50% of the composite ratio of collagen in hybrid PCL/collagen structure resulted in better the endothelialization efficacy in a given period of time.
Gelatin
Gelatin, as a stable and flexible natural polymer, is obtained from the partial hydrolysis of collagen and is the most abundant protein in the ECM.107,108 Gelatin is considered a biocompatible, biodegradable, and edible polymer with poor mechanical properties and minimal immunogenicity. 33 It is an indispensable component for the stability of the cell membrane and adhesiveness for cell attachment and it is present to a great extent in the human epithelium.109,110 Gelatin can be used in contact with the physiological medium of the body without generating undesired reactions and materials. 111 Fabrication of the gelatin nanofibers with an electrospinning process with neutral pH solvents at room temperature highly conserved the activity of the protein reagent embedded in the gelatin nanofibers. 112
Accordingly, gelatin in the form of nanofibers scaffolding received to pay attention in drug delivery systems, skin, vascular, and corneal tissue engineering applications. 114
Gelatin can be combined with PCL to achieve the desired call adhesion and control the degradation properties and biocompatibility. In this regard, Zhang’s group fabricated the hybrid PCL/gelatin scaffolds (D = 2 mm and fixed composite ratio 80 : 20) and modified them with heparin and organoselenium-immobilized polyethyleneimine (SePEI) for VTE.
115
Accordingly, by adding gelatin to PCL scaffolds, the biocompatibility, degradation rate, cell infiltration, and maintained elevated capillary angiogenesis increased in PCL/gelatin scaffolds. The designed scaffolds mimic ECs function and regulate the SMCs layer formation based on scaffold degradation rates as SDVGs applications. Gelatin creates anti-thrombogenic structures by providing optimum conditions for better ECs adhesion and more endothelialization. The Rat abdominal artery implantation was applied to evaluate of the
Nagiah et al. fabricated gelatin-based scaffolds (D = 5 mm and fixed composite ratio 1:5) with PCL, PU, and Poly lactic acid as synthesis and hydrophobic polymers by coaxial electrospinning method. 116 In the following, for fabrication structures the cross-linked process was done by genipin. These structures were fabricated in the form of core (hydrophobic polymer) and sheath (gelatin). All scaffolds showed no cytotoxicity and provided optimum cell growth and biomimetic microenvironments for hECs and hSMCs adhesion on both sides of them. Moreover, PCL/gelatin scaffolds display specific cell-scaffold interactions due to the ability of cell-binding ligands, a denatured form of collagen, good swelling profiles, and fast degradation rates of gelatin.117,118 This scaffold provided better biomimetic environments for adhesion and functions of hECs and hSMCs and prevented the hSMC’s overgrowth compared to other scaffolds.
The SDVGs would endure the force after implantation in the body. Fu et al. described a novel design for the fabrication of the SDVGs of the poly(L-lactide-co-ε-caprolactone) (PLCL)/collagen and PCL/gelatin (D=5 mm, fixed composite ratio 50:50).
119
All structures had good biocompatibility with HUSMCs, and the collagen/PLCL scaffolds showed a significant increase in adherent cells in compared to PCL/gelatin scaffolds. For the
Young’s modulus (strength) of 6 weeks following implantation showed strength reduction in hybrid PCL/gelatin scaffolds because of rapid degradation of gelatin. Also, the Young’s modulus of PLCL/collagen increased due to enhanced neo-artery tissue formation. The hybrid PLCL/collagen scaffolds showed better
In 2018, Joy and his coworkers focused on finding the optimization processing parameters to simulate the tunica adventitia by fabricated PCL/gelatin scaffolds (D = 5 mm and different composite ratios 10:90, 20:80, 30:70, 40:60) using the electrospinning method.
120
They determined the 20:80 ratios as the optimum composite ratio for the fabrication of hybrid PCL/gelatin scaffolds for VTE. The
Moreover, macrophage infiltration, on-going phagocytic degradation, and excessive collagen deposition of PCL/gelatin scaffolds were seen on day seven, and all mentioned parameters were decreased on day 15. The reduction of macrophages and collagen deposition on day 15 indicates a progressive remodeling process in regenerative medicine.
In 2017, the Coimbra group fabricated the PCL/functionalized gelatin (GelMA) scaffolds (25:75, 50:50, and 75:25) in the form of the core-shell pattern by the Co-electrospinning method. 121 The functionalized gelatin reacting with methacrylic anhydride and the obtained results revealed that a more composite ratio of gelatin in meshes’ shell has a positive impact on random distribution and increases biocompatibility in scaffolds. The hemocompatibility of scaffolds was evaluated by hemolysis and thrombogenicity assays by putting the scaffolds in direct contact with rabbit anticoagulated blood. By increasing the composite ratio of GelMA in scaffolds, a decrease in the contact angle was seen, and the hydrophilicity of scaffolds increased. Reducing the platelet adhesion and, consequently, lack of formation of clots resulted by increase in the scaffold’s surface hydrophilicity.
The examined Studies have shown that the hybrid PCL-natural polymer scaffolds were designed to simulate the physiological processes of vascular endothelium, and this aim may explain the enhanced promotion of vascularization, and angiogenesis. Gelatin is obtained by irreversible denaturation of collagen proteins and, in this regard, introduced as a molecular derivative of collagen. 122 Gelatin has a similar structure to collagen. 123 Gelatin creates anti-thrombogenic structures by providing optimum conditions for the establishment of endothelium and more release anticoagulation factors with anticoagulant properties. 124 In hybrid PCL/gelatin scaffolds, due to the ability of cell-binding ligands, a denatured form of collagen, swelling profiles, and degradation rates of gelatin can promote endothelial functions for adhesion and function of hECs and hSMCs and prevent hSMCs overgrowth. It is confirmed that adding gelatin to PCL caused to facility of hostcell infiltration in implantation site which may be attributed to the topographyand strong hydrophobicity of gelatin. Also, gelatin decreased the accumulation of inflammatory cells in in-vivo condition through incorporation of cell-binding motifs (i.e., RGD) and hydrophilic groups. 125 Furthermore, gelatin has achieved more attention in biomedical applications because it is readily available and highly soluble and has important binding moieties for cell attachment.
Chitosan
Chitosan (CS), derivate from chitin, is a linear 1, 4-linked polysaccharide, antimicrobial, and nontoxic polymer with a wide range of applications as SDVGs in VTE.126,127 CS, as an environmentally friendly (‘green’) polymer, is a promising hydrophilic, biocompatible, and biodegradable material that shows sufficient mechanical strength and reactive site for further bio-functionalization.128,129 CS-based scaffolds have inhibited inflammation during implantation, injection, local implementation, and even though ingestion in the
CS has weak mechanical properties, and therefore, in the form of the neat scaffold it has limited application in VTE.133–135 Nevertheless, the usage of PCL with CS in VTE is suggested due to address the poor mechanical properties of CS and slow degradation rate of PCL 136
The Fukunishi’s group fabricated hybrid PCL/CS scaffolds in two diameters (D = 1 and 5 mm and different composite ratios of 5:2 and 5:1) and implanted them in rat and sheep models, respectively.
137
The degradation rate of PCL/CS scaffolds was evaluated by an
The PCL/CS was fabricated by Yang et al. in 2010.
138
The hybrid PCL/CS scaffolds (different composite ratios 1:2, 1:1, 0.5:1.5) demonstrated similar structure to natural ECM in body. The mentioned scaffolds could provide an appropriate surface for endothelial progenitor cells (EPCs) adherence and growth. Better cell growth and proliferation of EPCs were observed in hybrid PCL/CS (1:2) scaffolds compared to neat PCL and CS scaffolds. In another study, the Sultana group focused on the fabrication of synthetic vascular grafts using PCL, CS, and Polyethylene glycol methyl ether (PEGME) (fixed composite ratio 1:1).
139
An i
The synthetic vascular graft by PCL and CS, (D = 1.5 mm, different composite ratios 5:4, 5:2, and 5:1) was fabricated by Yao et al. in 2014. 21 Hybrid PCL/CS scaffolds, showed lower proliferation SMCs than neat PCL scaffolds.
Heparin, by increasing the local concentration of vascular endothelial growth factor, could promote angiogenesis and endothelialization in
In VTE, synthetic vascular grafts are commonly used where cells can form functional regenerated tissue. However, they often become limited due to host cell infiltration and poor remodeling neo-artery. Therefore, the degradation rates of the synthetic vascular grafts need to be controlled. Because, in the rapid degradation rate, the graft failed due to the shape of the graft and in the slow degradation rate, the graft failed due to cell proliferation, and integration. 141 The products of CS degradation are harmless amino sugars that can be entirely absorbed by the human body. 142 Furthermore, the ECM of the blood vessel is constructed of glycosaminoglycans, and the CS consists of glucosamine and N-acetyl glucosamine. Therefore the ECM and CS have a similar structure. Using the CS with optimum degradation rate and similar structure to ECM, it can undergo rapid remodeling neo-artery and can support cell infiltration and cell proliferation in optimal choice for successful clinical application.143,144
Summary, conclusion and future perspective
The idyllic vascular graft must have an anti-thrombotic surface, be resistant to aneurysm, and not have any complications such as calcification. 145 Considering the composite structure of the artery, achieving the necessary requirements for the fabrication of structures with favorable biocompatibility to improve endothelialization on the lumen surface in VTE is challenging. Naturally derived polymers, constituting a temporary scaffold and their other replacement with a complete biological vascular conduit, are promising candidates in VTE. 146 An ideal scaffold for ECM-mimicking blood vessels should have bioactive functional groups to bind with cell ligands and potentially guide cellular behavior towards regeneration patterns. 147
The normal physiologic responses of the blood vessel wall have a key role in controlling thrombotic, inflammation and long-term patency.148,149 Accordingly, the researchers have attempted to design and fabricate the hybrid scaffold as synthetic vascular grafts that more closely mimic the wall of the native artery. 31 According to the above explanations, researchers indicated that the fabrication of hybrid PCL-based scaffolds by electrospinning method and with natural polymers is a suitable approach for improving its biocompatibility for VTE150,151
Bio-polymers support cell growth in two ways. These materials, in the form of the matrix, act as physical attachments for cells and, through molecular signal mechanisms, provide biochemical cues for cells.
117
The biological function of examined natural polymers in the form of hybrid PCL-based scaffolds was analyzed in the last section and their biochemical process was evaluated in the following (Figure 4). According to the mentioned figure, the neat PCL scaffolds due to lack of endothelial coverage, platelet aggregation, and thrombus formation are not particularly suitable for VTE applications.
152
Furthermore, the fast degradation, stopping cell adhesion and migration, losing mechanical properties, and lack of ECs monolayer on the lumen surface introduce as important drawbacks for neat naturally polymeric scaffolds as SDVGs for VTE applications.153,154 Therefore, the PCL-based composite scaffolds can be raised as unique approach for cover these specifications. Biochemical function support cell of hybrid PCL-natural polymers scaffolds with different degradation rate, strong and poor integrin cell adhesion and cell junction and as a results different thrombus formation.
As shown in Figure 4, the collagen has strong integrin cell adhesion and cell junction, fast endotheliazatio and therefore good patency in VTE applications.118,155 Significant structural changes such as protein denaturation might occur during the electrospinning of collagen solution. 156 Collagen denaturation destroys the protein resistance to enzymatic degradation and mechanical loading. 157 But it reported that denaturation might be useful for cellular adhesion if the mechanical properties and degradation rate are properly controlled.
As mentioned earlier, a suitable degradation profile
The researchers have indicated that gelatin may provide a better environment for cell attachment, growth and proliferation in comparison with other biopolymers. 159 Also, it is reported that fabrication the SDVGs in the form of composite structure of PCL/gelatin can be overcome the thrombosis in PCL scaffold due to improve the endothelialization on luminal surface due to surface modification properties of gelatin by to bestow anti-thrombotic properties on to the grafts. 160
Several approaches have been developed to create TEVGs in the last two decades
But collagen has shown an effective collagen-binding domain compared to gelatin (denatured collagen). 161
Also, CS shows slow degradation profiles
In this regard, the integrin-matrix interactions in hybrid scaffolds are very significant for successful clinical applications in terms of attachment, migration, and survival of ECs.
Collagen, as the main integrators with cells, activate the molecular signaling mechanisms and therefore plays the role of adhesion proteins in the biological conditions (Figure 4).
163
In other words, specific interactions between collagen molecules and integrin, enhance optimum cell attachment, proliferation, and good endothelialization
Accordingly, gelatin was introduced as a polymer with fast degradation profile (Figure 4) with poor integrin cell adhesion and cell junction. In this regard, the optimum composite ratio of collagen and gelatin in hybrid PCL-based scaffolds is reported at 25 and 80% and in long time collagen shows higher
CS is an excellent natural polymer for biomedical applications. 165 CS, due to the positive presence charge, can lead to interaction with the negative part of the cell membrane, and in this regard, CS shows hemostatic activity. 21 CS, is difficult to electrospun into a fibrous structure to the many amino groups in its backbone and cationic nature. 166 It is reported that CS is not suitable for blood contacting material despite its biocompatibility. 167 However, CS is not soluble in aqueous solutions, and this is the major disadvantage of CS that limits its widespread application in clinical applications. 168
Also, CS shows poor cell attachment compared to collagen and gelatin (Figure 4) and therefore due to minor thrombus formation in not suitable for VTE applications.169,170 It is shown that the supporting cell attachment of the synthetic tissue as a matrix is related to the host tissue in the body. Collagen is the main structure in the wall of the artery. Accordingly, the hybrid collagen scaffolds show great potential to promote good ECs viability, attachment, and phenotypic morphology and preserve the functions of ECs. In this regard, collagen is a suitable choice for fabricating of PCL-based scaffolds as SDVGs and it is in agreement with the obtained result by Michael et al. 171
Electrospun collagen nanofibers provide the most favorable surfaces for cell adhesion and endothelialization compared to electrospun gelatin and chitosan. Cross-linked or combination of collagen with PCL, improves the weak mechanical properties of collagen without compromising the biological activity. 102 Accordingly, it was hypothesized that the combination of PCL and collagen could provide novel synthetic vascular grafts with controllable properties for VTE, because each blend of components is potentially able to mitigate the other’s drawbacks.
Footnotes
Acknowledgements
I would like to express my deep gratitude to Dr. Hossein Taheri for his comments that greatly improved the quality of the manuscript. Also, the authors also gratefully acknowledge of the Mashhad University of Medical Sciences and National Institute for Medical Research Development (NIMAD), for supporting this research (Grant No. 931488).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mashhad University of Medical Sciences.
