Abstract
This study mainly aimed to evaluate the antibacterial impact of bacterial cellulose (BC) including dendrimer, chitosan, Henna, Green tea and Malva sylvestris in various bacteria such as Staphylococcus aureus and Pseudomonas aeruginosa. The produced bacterial cellulose was evaluated by antimicrobial (AATCC 25923 and AATCC 27853), cytotoxicity, swelling, drug delivery, scanning electron microscopy (SEM) and Fourier Transform Infrared (FTIR) spectroscopy tests. Antibacterial activity raised with increasing concentration of herbals and the most of antimicrobial effect belonged to G. tea. Cytotoxic test demonstrated that all of the samples did not have cytotoxic effect and viability percent for sample of BC with Henna, G. tea, and M. sylvestris extract were respectively 95.87%, 94.69% and 90.64%. Among the samples, the G. tea sample has the most swelling. In addition, delivery of the drug in water and phosphate buffer medium was the controlled release. Additionally, the most of the release related to the herbal drug Henna. SEM results showed BC with a highly porous three dimensional network structure. The considerable role of chitosan and dendrimer was evident in drug uptake and release. FTIR analysis demonstrated the presence of active groups in BC structure.
Introduction
Many microbes are highly pathogenic to humans and cause various chronic diseases. Primarily, modern drugs are used to treat these diseases. However, improper use of these drugs increases the resistance of microbes to drugs and improves public health problems. Currently, the spread of multi-drug resistant (MDR) bacteria does not only occur through nosocomial infections but also in the community. 1 Concerning the infection causing bacteria, alternative antimicrobial agents must be developed and employed to control MDR bacteria. To face this challenge, there has been growing interest in finding antimicrobial compounds from medicinal plant extracts and antibiotics as alternative approaches to discover new antimicrobial compounds.2,3 Skin, as the largest organ of the human body, protects the body from infection, but sometimes the skin itself becomes infected. A wide variety of germs causes skin infections, and their symptoms can vary from mild to serious. Mild infections may be treated with over-the-counter medications and home remedies, whereas other infections may require medical attention.4,5
Today, many different types of dressing are being produced and used for skin diseases. Bacterial cellulose due to its unique structure and characteristics is able to be an appropriate substrate for drug loading, transport and delivery. The use of BC in dressings instead of regular dressings has advantages, including that it reduces the duration of treatment of the disease, which in turn reduces the cost of treatment because of the transparent structure. Due to its nanostructure, it can also stay on the wound for a longer period and prevent microorganisms from passing to the wound surface while oxygen is passing to the wound surface. Therefore, it can be a good alternative to cellulose fiber dressing. 6 The efficiency of bacterial cellulose as a drug delivery material can be improved to provide additional properties. To modify BC, various ways have been proposed to optimize its antimicrobial activity and biocompatibility.7,8
The application of biopolymers such as bacterial polymers like chitosan, dendrimer and bacterial cellulose is one of the emerging adsorption methods for the removal of dyes, heavy metals and drug delivery. The presence of many amine groups on the chitosan and dendrimer chain, increase the adsorption capacity of dendrimer and chitosan. Chitosan, as a nitrogenous polysaccharide mainly composed of poly (b-1–4)-2-amino-2- deoxy-D-glucopyranose is produced through the deacetylation of chitin. Chitosan has several commercial and possible biomedical applications. The chemical structure of the chitosan present in Figure 1. Chemical structure of chitosan.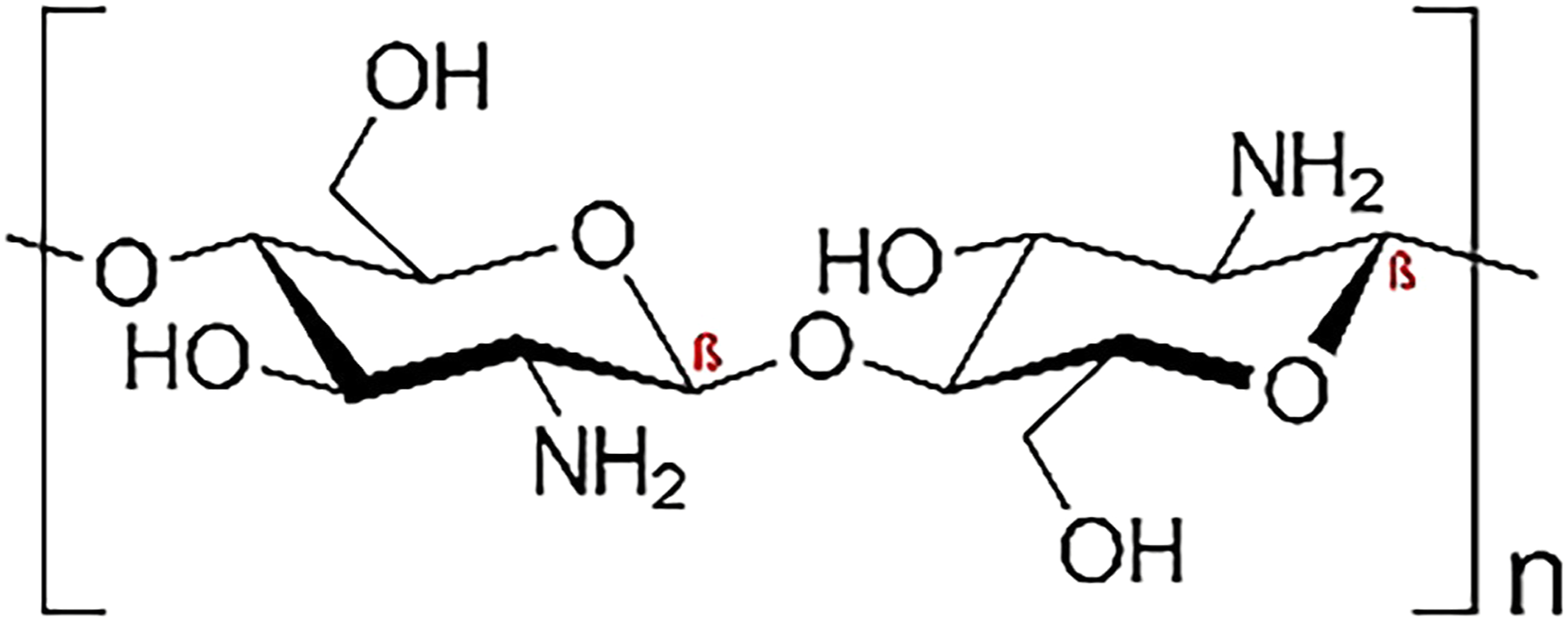
Dendrimers, also known as cascade polymers are tridimensional structures and repetitively branched molecules. Accordingly, the name came from the Greek (Dendron), translated to the tree. A dendrimer is typically symmetric around the core, often adopting a spherical three-dimensional morphology. 9 More recent works by Frechet et al. and Duncan et al. has highlighted the unique drug-loading capabilities and targeting features offered by dendrimers.10,11 The high level of control over dendritic architectures makes dendrimers ideal carrier in biomedical applications. It is appropriate to note that such properties are found in commercially available PAMAM dendrimers. Polyamidoamine dendrimers (PAMAM) are water-soluble, nonimmunogenic, biocompatible compounds 12 and their highly-branched nature could provide enormous surface area that generate great reactivity to microorganisms. Owning to their unique properties, PAMAM have been also evaluated for improvement of drug solubility and drug permeation and as delivery systems for bioactive substances, including antimicrobial agents. 13 Recent work has confirmed earlier reports that PAMAM dendrimers develop predictable nanoscale container and nanoscale surface-scaffolding properties. These unique architectural features offer many new options for the presentation or incarceration of pharmaceuticals, targeting groups. These biomedical functions could be conjugated or incarcerated independently or in combination. 14
One study found that cotton dyeing with reactive dye, electrolyte and alkali increases the pollution load in the textile waste water. In this method, finished cotton with different generations of PAMAM (polyamidoamine) dendrimer was dyed. The dyed samples got good wash, light and rubbing fastness. 15 In other study found that very high temperatures (e.g., ∼98°C) and used hydrogen peroxide (H2O2) bleaching of cotton fabrics could lead to high fabric damages and high energy consumptions in industrial practice. Poly-amidoamine dendrimers were used as a stabilizer in the place of sodium silicate. The low temperature bleaching was optimized using a statistical model. 16 Dendrimers are macromolecules with architecture three dimensional and highly branched. 17 Various dendrimers are used for drug delivery purposes. The most important polyamidoamine dendrimers (PAMAM) are widely used for various biomedical applications, including molecular imaging, drug delivery and gene therapy. 18 Due to the combination of dendrimer and chitosan, more drug delivery occurs because there is a spacing effect of dendrimer structures between chitosan chains. 19 In vivo studies of prepared nano formulation were significant and promising that explored the double concentration of drug due to surface functionality of dendrimer. 20 Moreover, dendrimers are able to favor the interaction of the drug with its target or help with its penetration through membranes. Dendrimeric moieties with various molecular weights can provide different rheological properties of the chitosan matrix mixed with bacterial cellulose. In addition, the dendrimer-bacterial cellulose can allow drug delivery kinetics to improve the amount of drug released. In most recent studies, the interaction of the drug within the dendrimer core structure occurs by a simple physical entrapment. The binding mode of the drug to the dendrimer was found to depend not only on size, the nature of the functional group, and the structure of the drug but also on the dendrimer generation, core structure, and surface charge of dendrimers. It has also been found that dendrimers of higher generation are better capable of encapsulating guests in the interior than a lower generation dendrimer. 21
The chemical structure of the PAMAM dendrimer is presented in Figure 2. In one research, synthesized hydrogel scaffolds were studied in terms of chemical, physical and rheological properties. The ability to release the drug was evaluated using ketoprofen as a model drug, which was included in the scaffold during the gelation process.
22
Plant extract delivery is the method or process of administering a pharmaceutical compound to achieve a therapeutic effect in humans or animals. For the treatment of human diseases, nasal and pulmonary routes of drug delivery are of increasing importance.
23
Plant extract delivery systems are very important since they can deliver a drug directly to a specific organ and thus allow targeting, increase the on-site drug concentration and prolong the drug administration, enhancing its bioavailability and effectiveness.
24
Chemical structure of PAMAM dendrimer.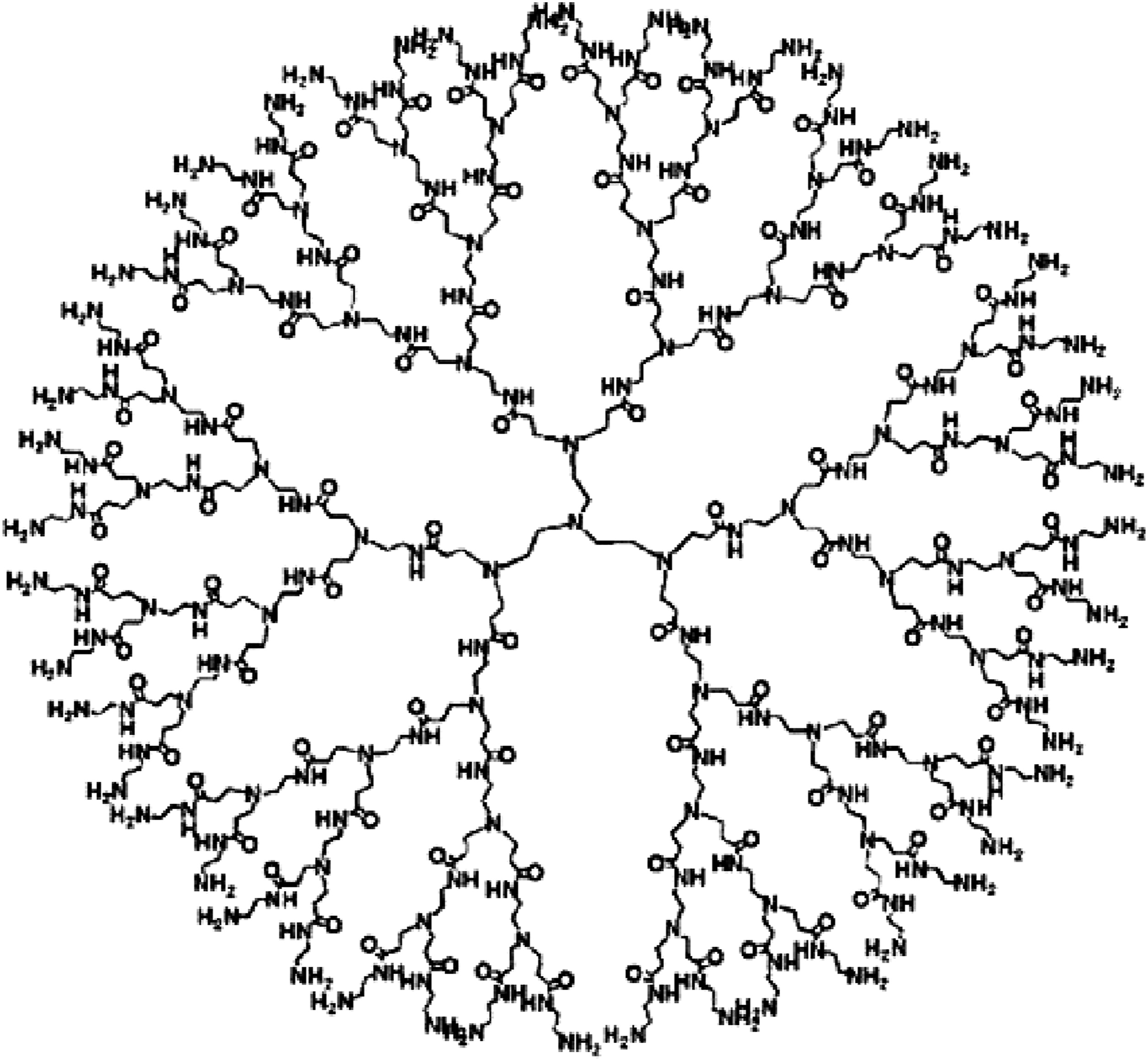
Different herbs have been used as a medicine during different times, but recently, medicinal plants have increased day by day. For the treatment of diseases and primary health care in different countries, medicinal plants are used alone or in synergy with modern medicines and are very effective due to their extensive biological and medicinal activities and higher safety margins. 25 One study found that use of tea industrial wastes can be used for antibacterial finishing of cotton. 26 The researchers used Ordinary mallow as a natural antibacterial agent for dyeing and treatment of woolen fabrics. 27 In other study, the Olive tree leaves for fabric finishing. Bacterial reduction with olive leaf extract was 99.99%. 28 One study found that Viburnum opulus L. fruit and branches in presente cranberry juice had good antibacterial effect on cotton knitted fabric. 29 Lawsonia inermis (Henna) is an antifungal, herbal medicine. Phytochemical investigations of L. inermis leaves showed the presence of some substances, including flavonoids, coumarins, gallic acid, triterpenoids, naphthalene derivatives, lawsone, phenolic glycosides and xanthones. 30 The main compound is Lawsone (2-hydroxynapthoquinone) and about 0.5–1.5% of leaves of Henna is made of Lawsone. 31
Green tea (Camellia sinensis), one of the most popular beverages worldwide, originated in China, and since then, its production and manufacture have spread to other countries in East and Southeast Asia, which is found in abundance in some countries. 32 Green tea displays antioxidant (free radical scavengers) and metal chelating activity and inhibiting lipoperoxidation due to the presence of polyphenols (especially catechin), alkaloids, vitamins and minerals salts with antioxidant, chemoprotective, anti-inflammatory and anticarcinogenic properties. It was reported to have antimicrobial effects. Studies conducted over the past 20 years showed that the G. tea’s (C. Sinensis), polyphenolic catechins, in particular, epicatechine (EC), epigallocatechin -3- gallate (EGCg), and epicatechin -3- gallate (ECg), epigallocatechin (EGC) can inhibit the growth of a wide range of Gram-positive and Gram-negative bacterial species with a moderate potency. Evidence is emerging that these molecules may be useful in controlling common oral infections such as dental caries and periodontal diseases.33,34
Malva sylvestris (M.sylvestris) belongs to the Malvaceae family, and a great deal of (2-methyl-3-methoxy-5,6-dihydroxy-1,4- naphthoquinone) anthocyanins and important natural pigments are extracted from this plant. Anthocyanins and other substances of M. sylvestris have antibacterial properties. In this regard, some reports revealed the presence of malvone A, a naphthoquinone, and different known monoterpenes, aromatic compounds, and tetra-hydroxylated acyclic diterpene. Malva sylvestris showed that the collagen fibers began to form in M. sylvestris, but no collagen formation was reported in the control group. 35 The applications of dendrimer with chitosan is nano-vectors and their detection by tumors is the most important approach to targeting brain tumors. Among the various nanocarriers, dendrimers are synthetic macromolecules with high branching points, mono-dispersity, the three-dimensional globular shape of nano-size (1–100 nm), nanoparticles systems have great potential as diagnostic and therapeutic agents. One study tried to use a nanotechnological polymer therapy approach using a polymer of natural origin such as chitosan to better transfer drugs. Various dendrimers were used for drug delivery purposes.36,37 The most important polyamidoamine (PAMAM) dendrimers. In this study, chitosan and dendrimer were used to finish BC in order to improve drug absorption and delivery due to their active groups and plant extract (Henna, G. tea and M. sylvestris) was used for the treatment of BC. The main feature of this study, in comparison with previous research was the simultaneous finished of BC with dendrimer, chitosan and herbal material) Henna, G. tea and M. sylvestris) were prepared and studied for the first time for drug delivery and antibacterial activity against S. aureus and Pseudomonas aeruginosa. In this project, Henna was used as antimicrobials, G. tea was also used as anti-inflammation and M. sylvestris was utilized as a collagen material.
Material and methods
Materials
Acetobacter xylinum ATCC 23768 and HS agar medium used for BC were supplied from microbiology libratory of Tarbiat Modares University, Tehran, Iran. Chitosan (Medium molecular weight chitosan, 190–310 kDa) and dendrimer (G4-NH2) were purchased from Sigma-Aldrich (Germany) and Alborz Nano Drug Co. Tehran. Acetic acid and other chemical materials, such as sodium hydroxide (NaOH), potassium phosphate monobasic, sodium dodecyl sulfate) SDS) were obtained from Merck Co. (Germany).
Methods
Preparation of nano-bacterial cellulose layers
BC was synthesized by A. xylinum bacteria in Herstrin and Schramm (HS) agar medium containing 2% glucose, 0.5% peptone, 0.5% yeast extract, 0.27% disodium phosphate water, and 0.115% citric acid monohydrate, which were used for long-term storage of the bacteria. Then, the medium was placed in an autoclave in small jars, which were not completely closed. After 17 days of cultivation at 30°C, the cellulose nanolayers were formed on the surface of the culture broth. After cultivation, the nano cellulose layers were removed and rinsed with distilled water. 38
Bacterial cellulose purification
BC was first placed in 2% SDS at 100° C for 3 h, during which additional pigments were removed from the cellulose sheet. Then, BC was washed with distilled water to remove additional materials. Subsequently, it was placed in 4% NaOH at 100°C and then washed with distilled water to remove BC from the culture media and other residues until it was prepared to treat BC with a clear appearance of white color and pH of 7.0. The BC layers in the wet state before and after bleaching are shown in Figure 3(a) and (b).
38
Bacterial cellulose layers in wet state (a) before bleaching (b) after bleaching.
Treatment of samples with dendrimer and chitosan
The dendrimer and chitosan were used on BC as pre-treatment agents by the exhaust method. BC was treated with a mixture of dendrimer and chitosan at 30°C and then at 95°C for 1 h. The liquor ratio was 1:30, and dendrimer and chitosan concentrations were 0.5% and 2%, respectively. All the samples were rinsed after finishing with dendrimer and chitosan. Figure 4 shows the treatment procedure of BC with dendrimer and chitosan.
39
Chitosan and dendrimer treatment bath.
Addition of herbal extract
Bacterial cellulose has poor antimicrobial properties, so an externally herbal product was added to make a good wound healer. Henna leaves, G. tea leaves and M. sylvestris flowers were collected, dried and powdered (99.8% pure and anhydrous). The extract was prepared using the Soxhlet apparatus. The solvent used for this process was ethanol.
Bacterial cellulose treatment
Various concentrations used plant extract on bacterial cellulose with dendrimer and chitosan.

Antimicrobial activity assays
The disk diffusion method was used to test the susceptibility of S. aureus (ATCC25923) and P. aeruginosa (ATCC 27853) to all samples. 1 × 1 cm2 of all samples was used for the test. All experiments were performed in triplicate. The direct colony suspension method was used for the disk diffusion test to prepare a PBS suspension of each microbial strain previously grown in Nutrient Agar plates at 37°C for 18 h. The density of the suspensions was adjusted to a McFarland 0.5 turbidity standard, corresponding to approximately 1–2 × 108 CFU/ml. The test was performed by applying the bacterial inoculums with a sterile cotton swab onto the surface of a Mueller–Hinton agar plate containing 25 mL of medium. The samples and respective controls were applied to the plates within 15 min of inoculation. According to the recommendations, the plates were incubated within 15 min of disk application at 37°C in the air for 18 h for all the strains. The susceptibility assay results were determined by measuring the diameter of the zone of inhibition around each sample using a ruler. 41
Cytotoxic test
The MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (test was seeded using 96 well plates (Tarsons, India). In concentration of about to 100 μL culture medium. After 24 and 48 h of seeding, MTT was dissolved in PBS to remove the insoluble residues. At the indicated time, MTT solution (100 μL medium) was added to all plate wells and incubated at 37°C for 4 h in a CO2 incubator. After that, DMSO was added to all wells and mixed well to dissolve the dark blue crystals and kept at room temperature for a few minutes to ensure the dissolution of all the dark blue crystals. The plates were read on a FLUOstar Omega microplate reader (BMG Labtech, Germany), and the absorbance was recorded at a test wavelength of 570 nm.
OD570c: is the mean value of measured optical density of the negative control after subtracting blank (medium control)
Swelling test
The samples were cut to dimension of 1 × 1 cm2 and packed. The packaged sample was then placed in an oven at 90°C for 20 min. After being taken out of the oven, the sample was weighed. It was then placed on a metal grid. The complex was placed in double distilled water at 37°C. The swollen sample was taken out of the water at regular intervals; its surface was dried and weighed with an accurate scale. Sample weight differences were noted. Water absorption percentage is calculated from the equation (1):
W0: Sample weight before water absorption
Assessment of 8 hour plant extract release
Nanofibers containing three materials were separately placed in 10 mL of distilled water and 10 mL of phosphate buffer, and their release was measured for 8 h. These drugs were detected during release from BC in two solvents, distilled water in pH = 7 and phosphate buffer in pH = 5.5. The method used for determining plant extract release is UV-Vi. This process was carried out for 8 h. Finally, adsorption numbers of Henna, G. tea and M. sylvestris containing samples in distilled water were determined at 256, 348, and 257 nm per hour for the samples respectively. 42
Scanning electron microscopy
The morphology and diameter of the nanofibers’ BC and BC with dendrimer, chitosan, Henna, G. tea and M. sylvestris were observed and determined using a SEM operating at an accelerating voltage of 24 kV (MIRA 3 TESCAN, Czech Republic). 43
Fourier transmission infrared spectroscopy
FTIR spectroscopy was performed to determine the structure of the composite dressing for identifying the presence of the above-mentioned specific functional groups and chemical reactions within a sample. All treated samples with dendrimer and chitosan and treated Henna, G. tea and M. sylvestris, which were analyzed. To prepare the samples for testing, all the samples were first completely powdered and then mixed with potassium bromide powder. The mixture was then pressed under pressure, and the compound was formed into a matrix. The resulting table was placed in a holder in a spectrometer) Bruker Tensor 27 IR, Germany). 44
Results and discussions
Antibacterial test
Inhibition zone measurements against S. aureus and P. aeruginosa.


The zone of inhibition diameters of (a) BC/PAMAM/chitosan/Henna; (b) BC/PAMAM/chitosan/Malvas sylvestris; (c) BC/PAMAM/chitosan/Green tea; d) BC/PAMAM/chitosan against S. aureus.

The zone of inhibition diameters of (a) BC/PAMAM/chitosan/Henna; (b) BC/PAMAM/chitosan/Malvas sylvestris; (c) BC/PAMAM/chitosan/Green tea; d) BC/PAMAM/chitosan against Pseudomonas aeruginosa.
Samples treated with 10, 30, 51.7, 100 mg/mL of Henna, 10, 51.7, 100 mg/mL of M. sylvestris and 10, 90, 100 mg/mL G. tea . All of the samples did not show antibacterial effects and inhibition zones. However, at 90 mg/mL, G. tea showed low antibacterial effect due to high polyphenolic catechins. The catechin components of G. tea are responsible for the observed antibacterial activity and the EGC, EGCg, ECg, EC constitute the most important antibacterial agents. 49 The inhibition zone recorded for BC/dendrimer/chitosan and G. tea against Gram-positive (S. aureus) were higher in comparison to those of BC/dendrimer/chitosan-Henna or dendrimer/chitosan-M. sylvestris. The antibacterial activity of G. tea catechins results from a variety of mechanisms that can be broadly classified into the following groups: (1) inhibition of virulence factors (toxins and extracellular matrix), (2) cell wall and cell membrane disruption, (3) inhibition of intracellular enzymes, (4) oxidative stress, (5) DNA damage and (6) iron chelation. These mechanisms operate simultaneously with relative importance differing among bacterial strains. Studies on antifolate activity in microorganisms showed that G. tea polyphenols could inhibit the enzyme dihydrofolate reductase in bacteria and yeasts, effectively blocking the ability of the microorganisms to synthesize folate. Bioflavonoids (including those from G. tea) inhibit the activity of bacterial ATP synthase, reducing the ability of the microorganisms to produce enough energy. 50 Figure 5 shows zone of inhibition diameters of the samples against S. aureus bacteria. The antibacterial activity of all of the samples on P. aeruginosa bacteria was not good because gram-negative bacteria (such as P. aeruginosa) were less susceptible to gram-positive bacteria (such as S. aureus). Because in gram-positive bacteria, the peptidoglycan layers were porous, allowing antibiotics, chemical compounds, and even herbal drugs to access the cell membrane. The lipopolysaccharide layer and the periplasmic space of gram-negative bacteria were the reason for their relative resistance to antimicrobials. 51 Figure 6 shows zone of inhibition diameters of the samples againts P. aeruginosa bacteria.
Cytotoxic test
Cell viability percent of Green tea.

Cell viability percent of Malva sylvestris.

Cell viability percent of Henna.

Swelling test
By comparing the control sample with the sample finished with plant extract, and adding plant materials to control sample, the thickness of the samples and the porosity increased. This created more spaces in the treated cellulose layers to absorb water and caused an increase in water absorption of the treated samples compared to control bacterial cellulose. Accordingly, the finished bacterial cellulose trap and more water and dried later, so there was more water around the wound.
52
According to the following calculations, G. tea had the highest swelling.



Plant extract delivery
The release of drugs from the surface of bacterial cellulose is shown in Figure 7(a)–(f). The BC multifunction potential and its chemical modifications with dendrimer and chitosan could be useful for binding and releasing different therapeutic agents.53,54 Dendrimers host hydrophilic and hydrophobic drugs, indicating their high versatility. Dendrimers are highly branched polymers with low polydispersity and high performance. They are ideal for constructing effective drug carriers, gene transfer devices and imaging biological systems.
55
The amine-terminated inside PAMAM dendrimers and the amino groups in chitosan are not protonated at physiological pH (7) while all first amines (dendrimer and chitosan) eventually protonated with a further decrease in pH (5.5–6).47,56,57 Pressure and the electrostatic repulsion between the protonated amino groups can release drugs easily, if the pH was under 5, BC with dendrimer and chitosan was eroded. While under alkaline conditions, the swelling degree of the BC decreased markedly and was not changed. This was attributed to the disappeared protonated amino groups.58,59 Also, the results proved that the all of samples had good pH sensitivity under acid conditions. Figures 8 and 9 shows the mechanism of binding Henna, G. tea and M. sylvestris to polymer modified by the dendrimer and chitosan, and the effect of pH on electrical repulsion the structure of bacterial cellulose finished with dendrimer, chitosan and plant extracts, respectivly.
60
The release rate in phosphate buffer was faster than water. A. Drug delivery curve for BC/PAMAM/chitosan/Henna (a) In phosphate buffer; (b) In distilled water B. Drug delivery curve for BC/PAMAM/chitosan/Malva sylvestris; (c) In phosphate buffer; (d) In distilled water C. Drug delivery curve for BC/PAMAM/chitosan/Green tea;s (e) In phosphate buffer; (f) In distilled water. The mechanism of natural extracts plant to polymer modified by the dendrimer and chitosan to antibacterial effect (a) BC/PAMAM/chitosan/Green tea; (b) PAMAM/chitosan/Malvas sylvestris; (c) BC/PAMAM/chitosan/Henna. Effect of pH in protonation of dendrimer and chitosan.


Figure 7(a) and (b) shows drug delivery of BC-dendrimer- chitosan with Henna in phosphate and water, respectively. The results demonstrated that after 8 h of the release, all of the Henna content was delivered from the BC component. Whereas, G. tea and Malva sylvestries release were smaller than Henna delivery. Henna probably had poor communication with the surface of BC and dendrimer and chitosan.
Scanning electron microscopy
BC was treated with dendrimer and chitosan alone and in the presence of herbal materials (Henna, M. sylvestris and G. tea), which was then analyzed by SEM (Figure 10). The sample of BC without treatment (Figure 10(a)) had a smooth surface, and the nano diameter of cellulose fibers was around 17.38 nm. This sample showed a highly porous, three-dimensional network structure, homogeneity surface, and nanoparticles with a spherical pharmaceutics shape. The structure was helpful to absorb a large amount of water and materials, which would be beneficial for sufficient contact between the BC, chitosan, dendrimer and herbal materials in antibacterial activity. However, a large size distribution was evident since particles with diameters of about 231.18, 168.16, 260.01 and 114.92 nm and much smaller nanoparticles between 68.73, 28.21 and 30.46 nm were captured, which increased due to antimicrobial effect using chitosan, dendrimer, and the materials mentioned above.
61
Scanning electron microscopy micrographs of drug entrapment; (a) bacterial cellulose; (b) BC/PAMAM/chitosan; (c) BC/PAMAM/chitosan/Henna (d) BC/PAMAM/chitosan/Malvas sylvestris; (e) BC/PAMAM/chitosan/Green tea; (scale bar 200 μm).
Fourier transform infrared spectroscopy
FTIR spectra of BC treatment with dendrimer and chitosan, dendrimer-chitosan-Henna dendrimer-chitosan-G. tea, and dendrimer-chitosan-M. sylvestris are shown in Figure 11. The peak at 1157.73 confirmed the presence of C-N stretching of amine, and Peak at 2850.70 cm−1 was due to the C-H stretch of CH2. Also, in the spectra of BC treatment with dendrimer and chitosan, a single weak N-H stretching vibration at 3730.94 cm−1 displayed the secondary amine and the peak at around 3330.15 cm −1 might belong to the stretching vibration of the hydroxyl bond (O-H).
62
All the observed peaks in FTIR spectra confirmed the conjugation of PAMAM dendrimer with chitosan and both were also conjugation to bacterial cellulose. Tensile vibration of C = O bonds or tensile vibration of C = C bonds in aromatic rings in Henna, G. tea and M. sylvestris were respectively in 1643.53 and 1644.96, 1646.49. Peaks at 2899.59, 2905.40, 2906.31 cm−1 were due to C-H stretch of CH2 in three materials. In the spectra of these samples, the peak at around 3843.44,3820.58,3852.14 cm −1 could be attributed to the stretching vibration of the (–NH–) bond, suggesting the occurrence reaction between the NH2 group (chitosan) and the hydroxyl group of plant extract with a hydrogen bond. In addition, as shown in Figure 11, by adding plant extract to the dendrimer -chitosan sample, the FT-IR spectrum of dendrimer-chitosan-cellulose had significant changes. According to this spectrum, the tensile vibrations of the OH bonds in this spectrum were transmitted from 3330.15 cm-1 to the wavenumbers of 3405.04, 3448.31, 3419.95, respectively. Therefore, peak of the OH bonds had become considerably wider and more intense, which could be due to hydrogen bonding between groups NH2 chitosan with polar groups (OH) in the chemical structure of compounds in the M. sylvestris, G. tea, Henna. Also, this conclusion indicated that bacterial cellulose was attached with dendrimers or chitosan with OH in bacterial cellulose. These results indicated that the active ingredients (M. sylvestris, G. tea and Henna) were encapsulated in the dendrimer structure.
63
Fourier Transform Infrared spectra of (a) BC/PAMAM/chitosan; (b) BC/PAMAM/chitosan/Green tea; (c) BC/PAMAM/chitosan/Henna; (d) BC/PAMAM/chitosan/Malvas sylvestris.
Conclusion
In this study, BC treatments with dendrimer, chitosan, and herbal extracts of Henna, G. tea and M. sylvestris were prepared and studied for antibacterial, drug delivery, cytotoxic, swelling, FTIR and SEM tests. The advantage of these derivatives was that they enhanced the antibacterial effect due to their functional groups. In herbal extracts like Henna, G. tea and M. sylvestris (free oxygen group), the antibacterial activities increased against both S. aureus and P. aeruginosa with increased herbal concentrations. The results showed that G. tea had the best antibacterial activity. Therefore, the antimicrobial effect increased due to using chitosan, dendrimer, and the herbal materials.
The results of SEM showed that the BC sample had a good porous three-dimensional network structure with a fiber diameter of 17.38 nm. The samples impregnated with chitosan and dendrimer exhibited numerous inclusions on the surface in particles with average sizes of 68.73, 28.21 and 30.46 nm. Some larger particles, approximately 231.18, 168.16, 260.01 and 114.92 nm in diameter, were observed in the sample treated with plant extracts.
The results of FTIR spectra confirmed the conjugation of PAMAM dendrimer with chitosan, and activated groups of M. sylvestris, G. tea, and Henna with PAMAM or chitosan with bacterial cellulose. The cytotoxic test showed that cell viability percent in Henna, G. tea, and M. sylvestris were 95.87%, 94.69%, and 90.64%, respectively. Between these plant materials, the G. tea sample had the most swelling. The release pattern behaviors of all samples were analyzed in phosphate buffer and water, and the former showed the order of ease of release of plant extract.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
