Abstract
Herein, a MnFe2O4/RGO knitted fabric derived from manganese waste was constructed by a simple in-situ assembled coating method, involving the incorporation of Graphene Oxide (GO) and manganese ferrite nanoparticles on polyester fabric, followed reducing by hydrazine hydrate. The reuse of manganese waste from the preparation of GO can reduce chemical waste emissions and endow the absorption performance. The coated particles possess certain magnetism can be attracted and securely collected in seconds, which is convenient for recycling. This fabric gives well microwave absorption with the maximum reflection loss (RL) of −58.6 dB at 9.1 GHz by a thickness of 1.9 mm. In addition, this fabric presents high stable strain sensing under 1000 stretching and bending cycles. Meanwhile, the resistance-deformation-velocity relationship is provided based on the structure, for the analysis of electromechanical behaviors. Moreover, the fabric has the capability for temperature sensing (TCR=−0.738%/°C), and fire alarm. As such, this fabric can be promising alternatives for a wide application on motion and temperature sensing, microwave blocking.
Introduction
Flexible electronic fabric has attracted extensive attention of researchers in various fields such as microwave absorption, strain and temperature sensing.1–3 Sensing usually depends on the conversion of energy, e.g. converting strain into electrical signals based on the piezoresistivity. 4 Functional textiles capable of electromagnetic shielding, 5 radiation protection, 6 far infrared 7 and sensing 8 are widely needed in various fields.
As a two-dimensional carbon nano material, Graphene has become quite popular owing to its excellent mechanical, electrical, thermal and optical properties.9–12 Reduced Graphite Oxide (RGO) composed of multilayer graphene is usually prepared by chemical oxidation intercalation, which is widely considered as excellent wave-absorbing material,9,13 sensing material 14 and green heating material15,16 ascribed to remarkable characteristics of low density, unique conformability and high dielectric loss compared with the traditional materials, 17 as well as low cost and compared with the monolayer graphene. Wang et al. prepared a porous cocoon-like reduced graphene oxide (RGO) by a simple and green reduction reaction using ascorbic acid as the reductant. 18 The RGO exhibits an excellent microwave-absorbing property and its RL is −29.05 dB at 15.96 GHz with a thickness of 2.0 mm. Carbon materials usually have only a single electrical loss mechanism, and the dielectric constant is high, so most electromagnetic waves cannot enter the material, which is not conducive to impedance matching. Ferrite has become one of the most widely used microwave absorbing materials because of its high permeability, low dielectric constant, high resistivity and low cost. Wang et al. obtained magnetic nano MnFe2O4 powder by detonation of specially prepared emulsion explosives. 19 The synthesized nano ferrite has excellent magnetic properties. Ferrite absorbing materials have the disadvantages of narrow absorption band and high density. Loading magnetic nanoparticles on three-dimensional graphene can adjust electromagnetic parameters, improve the attenuation of electromagnetic waves in the material and reduce agglomeration, so as to improve the effect of impedance matching. Therefore, the composite of ferrite and carbon materials can have the physical and chemical properties of different materials, and the defects of the original materials can be effectively improved, so as to obtain good microwave absorption effect.
Meanwhile, graphene manganese ferrite composites have been widely considered. Yang et al. prepared GO/Fe3O4 nanoparticles composite as controlled targeted drug carriers by chemical precipitation method. 20 Nagi M. El-Shafai et al. adopt GO@Fe3O4 to remove pesticides and heavy metal ions from aqueous solutions. 21 S. Sahoo et al. prepared RGO/MnFe2O4/Polyaniline through a facile solvent less thermolysis synthesis procedure for on-site monitoring of nitrite. 22 However, these studies mainly focus on the characteristics and applications of heavy metal adsorption and drug transport. To the best of our knowledge, there are few reports on manganese waste liquid derived MnFe2O4/RGO fabric composite for microwave block, strain and temperature sensing. Zhang et al. prepared MnFe2O4 by a simple hydrothermal, and then ultrasonic treated MnFe2O4 and RGO to obtain MnFe2O4/RGO, and studied its microwave absorption performance. 23 Wei et al. prepared go by Hummers method, and then synthesized nano MnFe2O4 by one-step hydrothermal method, and then obtained MnFe2O4/RGO by ultrasonic treatment, which was used to study the photo-Fenton degradation activity of the MnFe2O4/RGO composites with using the methylene blue under the simulated sunlight. 24 However, the waste emissions such as liquid waste, manganese waste etc. involved in Hummer’s preparation are well known. To produce one RGO, at least 1000 times of the water and 5 times of manganese are discharged, especially during washing of residual acid ion. 25 Therefore, it is of great significance to take advantage of manganese waste in the GO solution to be a green material for microwave block or sensing.
In this study, the residual manganese ions in GO prepared by Hummer’s method are reused in situ, which significantly reduces sewage discharge. Manganese waste derived MnFe2O4/RGO coated fabric is developed, which endows the polyester fabric with conductivity, microwave absorption performance and thermal stability. This waste derived MnFe2O4/RGO fabrics by using a simple in-situ assembled coating method are considered as promising alternatives in a wide range of applications.
Experimental section
Materials
Graphite powder is supplied by American Asbery carbon company. Sodium nitrate is provided by Shanghai Aladdin Co., Ltd. Sulfuric acid and hydrogen chloride are provided by Hangzhou Shuanglin Chemical Reagent Co., Ltd. Potassium permanganate is provided by Zhejiang Sanying Chemical Reagent Co., Ltd. Hydrogen peroxide is provided by Sinopharm Chemical Reagent Co., Ltd. Ferric chloride hexahydrate, ammonia and hydrazine hydrate are provided by Shanghai McLean Biochemical Technology Co., Ltd. The PET fabric is provided by Zhejiang Xinhai Textile.
Preparation of MnFe2O4/RGO
Based on the principle of chemical oxidation intercalation, the acidic GO aqueous solution was prepared by the improved Hummer’s method,
26
as our prior work reported.
25
The process for constructing MnFe2O4/RGO was shown in Figure 1(a). A Illustration showing the structure and preparation process of MnFe2O4/RGO fabric. 
The base PET fabric is plain knitted structure, with a thickness of 0.062 mm, warp density of 200/5cm, weft density of 200/5cm and mass per unit area of 82 g/m2. (i) Cutting several pieces of base PET fabric into a circle with a diameter of 7.2 cm, soaking them in 95% ethanol (CH3CH2OH) for 1 h. Then rinsing with deionized water and drying the treated fabric at 60 °C for 24 h. (ii) Measuring 50 ml of GO acidic aqueous solution prepared in the above process into a washed beaker and weigh 10.03 g of FeCl3·6H2O solid according to the ratio of Fe3+: Mn2+ = 2:1. (iii) Adding ammonia drop by drop to GO solution, and adjusting the pH to 12. Grinding FeCl3·6H2O into powder, adding it to GO solution, and fully react at 95 °C through ultrasonic treatment to prepare MnFe2O4/GO composite solution. (iv) Measuring 30 ml, 20 ml and 10 ml of MnFe2O4/GO composite solution in beaker numbered 1, 2 and 3 respectively, and add 10 ml and 20 ml of GO neutral aqueous solution to 2 and 3 respectively. (v) Placing the polyester base cloth prepared by (i) in beaker 1, beaker 2 and beaker 3 respectively, drying it at 60°C after complete impregnation, and repeating it for 3 times. Meanwhile, weigh and record. (vi) Taking 50 ml deionized water into the beaker, heating it to 90°C through the water bath pot, putting the dried fabrics into the beaker, add enough hydrazine hydrate to reduce the MnFe2O4/GO fabric to MnFe2O4/RGO fabric at 90°C for 2 h, follows by oven drying at 90 °C. At last, MnFe2O4/RGO fabrics with different MnFe2O4: RGO ratios of 3:3, 2:3, and 1:3 are achieved, which are refer to 3M/3G, 2M/3G and 1M/3G.
Characterization
The surface morphology of raw fabric and MnFe2O4/RGO fabric was identified by ultra-5 field emission scanning electron microscope (SEM, ULTRA-55, Japan Electronics Co., Ltd, Japan). The X-ray diffraction (XRD, D8 DISCOVER, Bruker Co., Ltd., Germany) of fabrics is performed with voltage of 40 kV to characterize the crystal structure with the voltage of 40 kV, the X-ray wavelength of the copper target is 1.54056 Å and a scanning range of 5 to 60° at a scanning rate of 2° 2θ/min. The functional group of material was analyzed by a Fourier transform infrared spectroscopy (FT-IR, Nicolet Fourier infrared spectrometer, Thermoelectric Negoly Company) in the spectral range of 500-4000 cm−1 and it adopted ATR attenuated total reflection method. The optical properties of the materials are studied by ultraviolet visible spectrophotometer (UV-Vis, UH4150, Hitachi high tech company).
Microwave absorbing performance test
Microwave network vector analyzer (Agilent, N5222A, USA) was used to measure the parameters of the sample in X-band by waveguide method, including the reflection and transmission of electromagnetic wave.
Motion sensing test
The sample of 3 cm × 1 cm was clamped along the course of the fabric in an fabric universal tension meter (YG065, China), which is connected to the resistance tester (DMM 6500, Tektronix Co., Ltd, China) at the same time to record real-time resistance change. It specifically includes the piezoresistance behaviors of textiles under different strain states of 10%, 15%, 20% and 25% at the same tensile rate, and the relative resistance changes of textiles under different tensile speeds (2, 3 and 4 mm/s) at 10% tensile strain. Then, the sample of 2.5 cm × 1 cm was clamped along the course of the fabric on a self-assembly reciprocating motion test device by a linear stepper motor (KH-01, China) with a clamping distance of 2 cm. 1000 bending cycles at 15 mm/s and 25% strain were performed. In order to evaluate the application of fabric in monitoring and identifying motion signals. The sensor is directly connected to human skin to simulate the activity intensity of body parts during activity. The real-time resistance change was recorded during all the tests.
Temperature sensing and thermal performance test
Temperature Sensing: the sample of 2 cm × 2 cm was put into an oven and connected with DMM6500 digital multimeter outside. The real-time resistance change was recorded during oven heating, which includes the resistance change under the equilibrium temperatures.
In order to verify that the fabric has a certain flame retardant effect, the raw polyester fabric and MnFe2O4/RGO fabric of 2 cm × 2 cm were burned with alcohol lamp under the same conditions, the combustion time and state were recorded. In order to study the thermal stability of the fabric, the raw fabric and MnFe2O4/RGO fabric was analyzed by Thermogravimetric analysis (TG) by TGA/DSC2 thermogravimetric and synchronous thermal analyzer of Swiss Mettler-Toledo company. The test was performed in N2 atmosphere to prevent thermal oxidation degradation, with the nitrogen flow rate of 25 mL/min, the temperature range of 25-800 °C and heating rate of 10 °C/min respectively.
Results and Discussion
Structure of the MnFe2O4/RGO fabric
The morphological characteristics of MnFe2O4/RGO coated fabric was observed by SEM. The features of MnFe2O4/RGO nanoparticle are shown in Figure 2(a). The MnFe2O4/GO was coated on PET fabric, then, in-situ reduced to MnFe2O4/RGO nanosheet by N2H4·H2O. RGO nano sheets and MnFe2O4 nano particles are stacked as sandwich lamination structure, which can evenly be covered on the fiber surface to form a complete conductive film. Generally, there is no attraction between inorganic particles and polymer materials (such as textiles).
27
It can be found that RGO is uniformly distributed on the surface and adheres to the PET fiber through many van der Waals forces or hydrogen bonds, as well as the π-π accumulation between the abundant aromatic rings in PET and RGO.28,29 In addition, graphene which has a long edge and high specific surface area, can be doped and chemically modified, and is easier to accept functional groups. Therefore, graphene can be used as a bridge to make manganese ferrite adhere to the fabric. Nano MnFe2O4 nanoparticles (hundreds of nanometers) are doped between RGO nanosheets (dozens of microns), which are well combined, so that endowing the fabric with certain magnetic loss, magnetism and can be adsorbed by magnets. Characterizations of the MnFe2O4/RGO fabric. 
The crystal structure for fabrics of 3MnFe2O4/3RGO (3M/3G), 2MnFe2O4/3RGO (2M/3G) and 1MnFe2O4/3RGO (1M/3G) were investigated by XRD. As shown in Figure 2(b), there are four significant crystal plane diffraction peaks for the raw PET fabric, which are located at 2θ = 16.5°, 17.14°, 22.6° and 25.8°. 30 The XRD pattern of the coated fabrics shows four identical weakening peaks at the similar position. The intensity of peaks gradually decreases with the introduce of MnFe2O4/RGO coating. At the same time, the intensity of diffraction peak decreases gradually with the increment of RGO ratio. Diffraction peaks at 35.4°, 43.1°, 57.1° and 62.5° are indexed to (311), (400), (511) and (440) crystal planes of cubic phase with inverse spinel ferrite of MnFe2O4 JCPDS no.742403. 22 The intensity of these characteristic peaks gradually decreases with the decrease of MnFe2O4 content.
FT-IR was used to characterize and analyze the functional group of samples. From the FT-IR spectrum of Figure 2(d), it can be seen that the peak near 1704 cm−1 corresponds to -C=O- stretching vibration peak, and the peak near 1236 cm−1 corresponds to -O- stretching vibration peak respectively. 31 The peak at 1704 cm−1 attributed to -C=O- weakens when MnFe2O4/RGO modified coating is carried out as prior work reported. 32 Compared with the raw fabric, the peak intensity of 3M/3G, 2M/3G and 1M/3G decreased at 3417 cm−1, 2210 cm−1, 1704 cm−1 and 1236 cm−1, and the vibration peak disappeared at 2983 cm−1. With the increase of RGO ratio, the strength of each telescopic vibration peak decreases. Based on the above-mentioned analysis, the results prove that the MnFe2O4 particles are linked to the RGO nanosheets.33–35
Based on UV-Vis spectrum of Figure 2(c), the transmittance behavior of samples in visible and ultraviolet bands can be analyzed. The MnFe2O4/RGO composite coating formed by in-situ synthesis on the PET fabric changes the white fabric into black, which significantly reduces the lightness. Color change is contributed to nano MnFe2O4/RGO composite loading on the polymerization surface. The base fabric presents transmission behavior in the UV-vis region. After nano MnFe2O4/RGO composite coated on the surface of the polyester fabric, the transmittance is significantly reduced. This phenomenon is attributed to the effect of the MnFe2O4/RGO composite coating on the visible light UV absorption and reflection behavior. 36 The band gap of RGO is zero, which can be adjusted by doping, so it can be doped with MnFe2O4. Due to a large specific surface area, all MnFe2O4/RGO fabrics shows a low transmittance. For different MnFe2O4/RGO fabrics, the corresponding UV-Vis transmission spectrum shows a decreasing trend with the increment of RGO content. The addition of RGO contributes to the rapid migration of photogenerated electrons and the generation of more active free radicals. The minimum transmittance is in the UV-vis transmission spectrum of 1M/3G, which can be attributed to the reduction of aromatic structures in RGO nano lamellae and the solidification of manganese ferrite.37,38
Microwave absorption performance of MnFe2O4/RGO fabric
The mechanical and electrical properties of MnFe2O4/RGO fabric are very important for its application. The resistance of 3M/3G is 17.37 MΩ. With the increase of RGO content, the resistance of the fabric decreases. The resistance values of 2M/3G and 1M/3G are 7.74 MΩ and 5.93 MΩ respectively. It can be observed from Figure 3(a)-(c) that the reflection loss of the fabric increases with the increase of RGO content. 2M/3G and 1M/3G present better microwave absorption. The maximal RL value of 1M/3G is −17.6 dB at 11.1 GHz with an absorbent layer thickness of 1 mm. MnFe2O4 is a magnetic loss absorbing material, which gives both magnetic loss and dielectric loss mechanisms. The magnetic loss absorbing material absorbs electromagnetic waves mainly by means of hysteresis loss, natural resonance loss, eddy current loss and domain wall resonance.39,40 It has large saturation magnetization and low power loss under high frequency. However, a single MnFe2O4 as an electrode material has the performance defects of easy stacking and agglomeration. Graphene has superior conductivity and large specific surface area, which effectively prevents the agglomeration of MnFe2O4. Besides, the RGO layer can give rise to multiple reflection of electromagnetic wave, which may enhance absorption of electromagnetic waves. (d) RL plots of samples: 3M/3G (
Figure 3(d-i) shows the electromagnetic parameters of samples. The storage capacity of electric and magnetic energy can be expressed by the real part of complex permittivity and complex permeability (ε′ and μ′). The loss of magnetic energy and electric energy is represented by the capacity of the virtual energy part (ε″ and μ″). 41 As RGO gives more effective conductive network and polarization ability. Moreover, RGO is composed of carbon atoms with two-dimensional planar structure, which owns high dielectric loss. 42 Therefore, RGO plays a major role in the complex permittivity (εr = ε′ - jε″) in Figure 3(d)-(e). Overall, the ε′ and ε″ value of 1M/3G > 2M/3G > 3M/3G. The ε′ and ε″ value of 1M/3G reach about 3.8 and 1.7 respectively. This is due to different go contents. With the increase of RGO content, the stronger the dielectric properties of the samples, the greater the dielectric constant. MnFe2O4 is a kind of soft magnetic ferrite material with cubic crystal system, which possesses high permeability and low loss. As can be seen from Figure 3 (g), (h), the value of μ′ is about 1.19-2.84. The imaginary part of 3M/3G is the largest, reaching 1.84, indicating that 3M/3G holds stronger magnetic loss ability. The dielectric and magnetic loss factors (tanδε = ε″/ ε′, tanδμ = μ″/μ′) are also important for the loss capacity of absorber. 32 In general, tanδε(1M/3G) > tanδε(2M/3G) > tanδε(3M/3G), it indicates that 1M/3G has the highest dielectric loss performance. In the frequency range, tanδε < tanδμ for 3M/3G, which indicates that the magnetic loss is dominant in the magnetic field in Figure 3(f), (i). And tanδε > tanδμ for 1M/3G, which indicates that the dielectric loss is the main factor to attenuate the incident electromagnetic wave.43,44 These results show that the difference between complex permittivity and permeability is small, which leads to better impedance matching and better microwave absorption performance. This is attributed to the excellent conductivity and large specific surface area of RGO. It not only solves the problem that MnFe2O4 is easy to reunite, but also enables a better combination of the MnFe2O4/RGO.39,42
Figure 4 depicts 3D RL plots of 3M/3G fabrics with different coating cycles. Where M-2, M-4 and M-8 represent 3M/3G applied for 2, 4 and 8 cycles respectively. It can be observed that the microwave absorption performance is significantly improved with the increase of coating times. The maximum RL value of M-8 is −58.6 dB at 9.1 GHz with an absorbent layer thickness of 1.9 mm. The maximum RL value of M-4 reaches −52.1 dB at 8.5 GHz, and the corresponding absorption layer thickness is 1.8 mm. It can be seen from Table.1 that compared with other materials, M-8 shows superior microwave absorption properties, including strong absorption and thin thickness. This shows that manganese waste liquid derived MnFe2O4/RGO coating is a resultful method to endow with fabric the performance of microwave absorption, and the absorption can be improved by changing the coating cycles and reasonably adjusting the coating thickness.
45
3(d) RL plots of samples coated with 3M/3G times of 2, 4 and 8 respectively: M-2 ( Typical microwave absorbers reported in recent literature.

Sensing performance
The MnFe2O4/RGO fabric not only present well microwave absorption performance, but also possess and sensing performance. It can be used as a sensor with good microwave absorption performance as multi-function fabric. The schematic diagram of strain sensing is shown in Figure 5(a). To further study the reliability of MnFe2O4/RGO sensor, sensing performance under 1000 cycles at 15 mm/s and 25% strain was carried out along the course of the fabric. The sensor’s durability under multiple testing cycles is criteria for the performance and practical applications of a sensor. As can be seen from Figure 5(d), the durability and repeatability of the sample are excellent, with a great overall linear feature apart the initial section, indicating the high stability of this fabric sensor under 1000 cycles. Change of relative resistance is small. Since some RGO nanosheets may slide and separate around during the stretching process, MnFe2O4 particles fill the voids of RGO sheets, even acts like a sliding bead, facilitating slip between sheets, resulting in the high stability of the fabric during multiple stretching.51–53 Sensing characteristics of RGO/MnFe2O4 fabric. 
Figure 5(b) shows the piezoresistance behavior of fabric sensor under different tensile strains of 10%, 15%, 20% and 25% along the course of the fabric. The piezoresistance of the fabric sensor are almost identical among every strain interval, demonstrating good electromechanical stability of sensor. The relative resistance increases with the increase of strain, indicating its potential applications that different strains was required to be identified. Figure 5(c) shows that ΔR/R0 is hardly affected by the tensile rate and the fabric sensor gives fast and stable response to different tensile speeds. The response time in Figure 5(d) is only 0.26s, which means it can sense quickly. The short response time is criterial for excellent sensing performance. 54 These excellent electromechanical performances of the fabric sensor are beneficial for application in wearable intelligent electronic devices.
Schematic illustration of the MnFe2O4/RGO fabric sensor for monitoring human motions by attaching to the body position is shown in Figure 5(e). The MnFe2O4/RGO fabric is attached on a finger and wrist to detect movement. The relative resistance variation ΔR/R0 responses to the bending of finger and wrist are shown in Figure 5(f). As well as the bending angle can be recorded by value of relative resistance variation ΔR/R0 as Figure 5 (g) shown. Then, the relative resistance returns to initial state after the motion recover, demonstrating the repeatability of sensor. Moreover, different body bending behaviors such as the fingers, wrists and arms can be distinguished by the change of relative resistance and the fluctuation of different shapes, indicating its potential applications on body behaviors were required in comparison. In summary, the MnFe2O4/RGO fabric sensor mounted on the body can successfully detect body behaviors of human with well repeatability.
What is noteworthy is that the ΔR/R0 of knitting fabric sensor under course stretching is negative, namely the resistance R decreases during tension; On the contrary, the ΔR/R0 of knitting fabric sensor under course bending is positive, namely the resistance R increases during bending. On the one hand, when the strain be applied, the conductive materials of the conductive network are misaligned or separated, and the contact points between them decrease sharply, resulting in the increment of relative resistance in Figure 5(d), which is corresponding to negative piezo-resistance behavior, as well as prior works reported.4,55 On the other hand, the applied strain usually derived from the stress, and the contact points can increase sharply under stress, resulting in the reduction of relative resistance in Figure 5(b, c, f, g), which is corresponding to positive piezo-resistance behavior, as well as prior works reported. 54
For bending here in Figure 5(d), the misaligned or separated of the contact points is dominant, corresponding to a positive ΔR/R0 and piezo-resistance. And for stretching here in Figure 5(b, c, f, g), the establishment and enhancement of contact is predominant, corresponding to a negative ΔR/R0 and piezo-resistance. This different negative or positive piezo-resistance behavior depends on the textile structure apart from the material. However, the vast majority of published work does not consider this structure-activity, but only the amplitude of resistance change. This issue deserves clarity for related research and application of piezo-resistance sensor.
The mechanism of electromechanical behavior of weft knitted structure of MnFe2O4/RGO fabric is analyzed. Conductivity is mainly related to the contact between adjacent fibers and adjacent yarns. In the initial state, the resistance model of MnFe2O4/RGO fabric is shown in Figure 6(a). Rf represents the resistance of fibers, Rcf represents the contact resistance between fibers, and Rcy represents the contact resistance between yarns. In order to deeply understand the influence of yarn structure on relative resistance, the resistance model is shown in Figure 6b without considering the contact resistance between fibers. A basic unit can be regarded as a parallel circuit. The resistance value (R) can be expressed by the following equation (1): Sensing mechanism of the MnFe2O4/RGO fabric. 

When the applied strain increases, the conductive materials of the conductive network are misaligned or separated, and the contact points between them increase sharply, resulting in the reduction of relative resistance in Figure 6(c), as prior work reported. 54 On the contrary, during the release process, the contact point decreases and the relative resistance increases.
Figure 6(d) shows the relationship between resistance change and deformation during bending. (i) The resistance rises linearly and the bending deformation begins. The misaligned or separated of the contact points is dominant. Which is contributed to the slip of contact points between yarns. The misaligned or separated of the contact points is dominant, corresponding to the resistance rises linearly and the bending deformation begins. (ii) The bending deformation continues and decelerates to the stop stage, and the contact point begins to recover partially. Then, the resistance recovery becomes smaller, corresponding to the first small peak. (iii) The recovery of bending deformation begins and accelerates. The contact point stops to return and begins to slip. The contact points reduced and the resistance increases slightly. (iv)The bending deformation recovers, and the deformation recovers to the flat and tight state. The contact point continues to return. Then the resistance decreases sharply. Summing up the above, the resistance varies from the separation or recover of contact points, which is associated to the structure and deformation of textile, including the contact changes of inter-fibers and inter-fiber tows (highlighted in red in Figure 6(c)). It mainly occurs at the beginning and the last stage of deformation.
Figure 6(e) shows plots of resistance vary with the data acquisition interval, indicating the influence of data acquisition frequency on the ΔR/R0 results. and the corresponding waveforms are completely different when the data acquisition time varies from 0.68s (in green) to 0.02s (in pink). However, the vast majority of published work does not consider this issue of data acquisition frequency, but only adopt the data similar to pink. This issue is of significance to the sensor behavior such as sensitivity, gage factor (GF), response time, etc., which deserves clarity.
Thermal performance
The temperature sensing performance MnFe2O4/RGO fabric is provided as Figure 7(a, b)shown. To further explore the relationship between the resistance and temperature sensitivity of MnFe2O4/RGO composite fabric, the MnFe2O4/RGO fabric was put into the oven to test the resistance varies with temperature as Figure 7(a). The relative resistance has a well linear relationship with temperature, and its resistance temperature coefficient (TCR) is −0.738 %/°C, indicating that the resistance change is caused by temperature change and capable of temperature sensing. In addition, the change of relative resistance is obvious and TCR is superior to our prior works reported about RGO paper, RGO fabric and Silver Nanowire fabric,9,16,56 which is helpful for the use as temperature alarm. There are migrating electrons (em) in the graphite plane and jumping electrons (eh) between the graphite layers. There is a contact between the conductive fibers, which also produces jump conductance. As the temperature increases, the transition between electrons becomes active, resulting in a decrease in resistance.57–59 A Schematic illustration of temperature sensing for RGO/MnFe2O4 fabric. 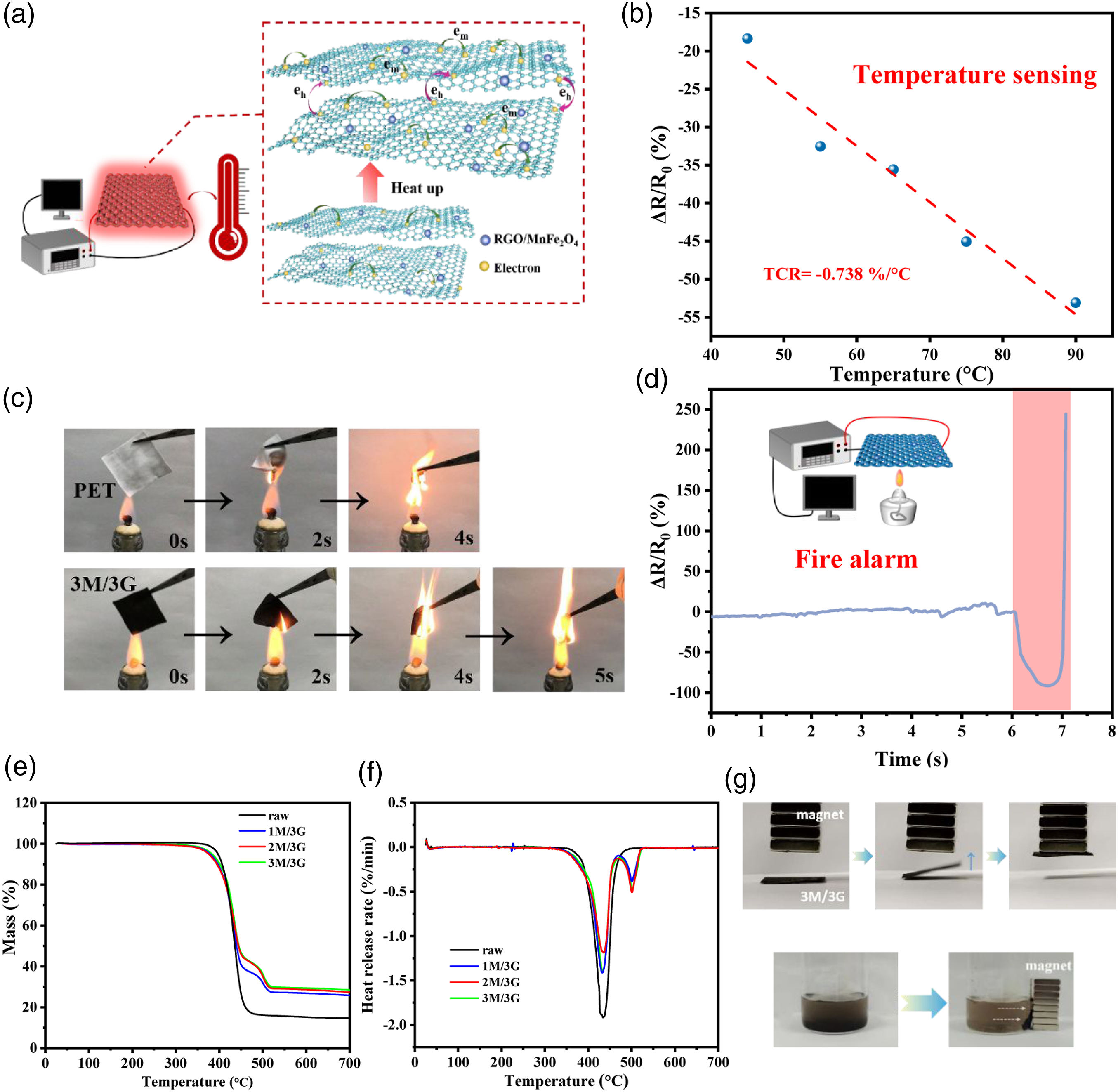
As shown in Figure 7(c), MnFe2O4/RGO fabric has a few flames retardant effect. In detail, the raw polyester fabric is obviously agglomerated together at 2s, while the MnFe2O4/RGO fabric is basically well-shaped. The raw fabric is basically burned out in 4s, while the size of the functional fabric remains about 1/3. Meanwhile, Figure 7(d), measure the resistance change of the fabric in the burning state. From the moment the fabric contacts the flame (about 6 s), the fabric presents an immediate relative resistance change (ΔR/R0) of −92%–91.65%. Then, the ΔR/R0 goes up immediately after burning. The whole process takes only about one second, indicating the capacity for fire alarm.
The MnFe2O4 is densely aggregated in RGO nano sheet, which effectively reduces the surface exposure in the oxidation process, thus improving the thermal stability and the weight of residual carbon 13 according to the analysis of Figure 7(e, f). In this case, MnFe2O4 is densely aggregated in RGO nano sheet, which effectively reduces the surface exposure in the oxidation process, thus improving the thermal stability and the weight of unburned carbon 13 as Figure 7 (e, f) shown. The decomposition process of the original sample and MnFe2O4/RGO fabric in the previous stage is basically the same. In detail, the decomposition process of the original sample and MnFe2O4/RGO fabric below 352°C is basically the same, which is mainly caused by the volatilization of adsorbed water or residual solvent in the sample. Then, between 352 °C - 487 °C, benzoic acid and its derivatives, carbon dioxide, carbon monoxide and other chemicals are cracked. Which provided the maximum heat release efficiency of raw fabric, 3M/3G, 2M/3G and 1M/3G of −1.92%, −1.3%, −1.41% and −1.19%, respectively. The DTG peaks appear around 500 °C is contributed to the introduction of MnFe2O4/RGO. The final residual masses of raw fabric, 3M/3G, 2M/3G and 1M/3G were maintained at 29.7%, 29.3%, 27.4%, and 15.11% respectively, MnFe2O4/RGO fabrics are greater than the final mass of the original fabric, and the residual mass increases with the mass fraction of metal ions. In addition, MnFe2O4/RGO endows fabric with certain magnetism, which is convenient for the recycling collection of materials by magnet after use (Figure 7(g). MnFe2O4/RGO particles in the solution are obviously attracted by the magnet, and the MnFe2O4/RGO particles are completely attracted for about 2.7 seconds, indicating that MnFe2O4/RGO is recyclable and easy to recycle.
Conclusion
In short, a waste derived MnFe2O4/RGO fabric was developed, which is prepared by a simple and environmentally friendly method. This method can effectively reduce a large amount of wastewater from process of preparing GO by reusing waste ions. The MnFe2O4/RGO material possesses certain magnetism and is easy to recycle.
Firstly, MnFe2O4/RGO material shows well microwave absorption performance, with a maximum RL value reaches −58.6 dB at 9.1GHz for M-8. Secondly, it shows high stable stain sensing stability and potential as intelligent wearable electronic devices. At the same time, it is pointed out that the data acquisition frequency has an obvious influence on the data waveform, and resistance-deformation-velocity relationship is provided based on the structure for the analysis of electromechanical behaviors. Moreover, it can respond sensitively to temperature changes and be used as temperature sensor and fire alarm. The firmness of the MnFe2O4/RGO particles coated on knitted fabrics is a common problem to be solved in this kind of research. In the next work, we will focus on improving the adhesion firmness of particles. This material can be used in more aspects, improve the number of recycling and always maintain good results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (NSFC 51803185), Public Welfare Project of Zhejiang Province (LGF21E030005), China Postdoctoral Science Foundation (2020M681917 and 2022T150581), Postdoctoral Foundation of Zhejiang Sci-tech University Tongxiang Research Institute (TYY202013).
