Abstract
In order to enhancement the antimicrobial properties and the thermal stability of the cotton fabrics for outdoor uses, they were coated with formulations of room-temperature vulcanizing silicone rubber (RTVSR)/synthesized zinc oxide nanoparticles (ZnO NPs). The coated fabrics were adjusted to different doses of ultraviolet (UV) irradiation, which act as a crosslinking factor to the silicone rubber, rather than as a photocatalytic agent for enhancing the antimicrobial activity of the ZnO NPs. The synthesized ZnO NPs were characterized by X-ray diffraction (XRD) and transmission electron microscopy (TEM); meanwhile, its impurity was examined using energy dispersive X-ray (EDS) analysis. The thermal stability and surface morphology of the coated fabrics were characterized by thermogravimetric analysis (TGA) and scanning electron microscopy (SEM). The results showed that the mechanical strength (MPa) of the coated fabrics was enhanced by ∼11% after UV irradiation for 120 min, accompanied by decrease in its swelling and solubility by ∼10 and 20%, respectively, as a result of UV crosslinking effect upon RTVSR. Meanwhile, the antimicrobial activity of the coated fabrics was enhanced by ∼50%, as a result of the photocatalytic effect of UV irradiation (120 min), at the constant percentage of 3% (ZnO NPs) in the coating formulations.
Introduction
Cotton fabrics are characterized by their comfortability, ease of handling, and favorable mechanical properties for outdoor or indoor use.1–6 When they adjust to outdoor conditions such as temperature and moisture, which provide a conducive environment for microorganism growth, they lose their tensile strength and impermeability, particularly for sports, military, and work clothing.6,7
Different antimicrobial agents (organic and inorganic) have been used to enhance the microbial resistance of cotton fabrics. Organic antimicrobial agents include chitosan, quaternary ammonium, and N-halamine materials.8,9 Inorganic antibacterial materials are extensively used for their great antibacterial efficacy, particularly in the range of nanoparticles (NPs). Several metal-based NPs exhibit biocidal features, such as Ag, ZnO, Cu-based, and TiO2NPs and other minor metal-based NPs such as iron-based, CeO2, Au, SiO2, Ni, and Pt NPs.7,10–13
ZnO exhibits photocatalytic abilities, conductivity, and photo oxidizing abilities in opposition to self-sterilization. ZnO exhibits major antibacterial activities while the particle range is compact to the nanometer series, and nano ZnO is capable of cooperating with bacterial surfaces or the bacterial central part wherever it enters the cell and consequently exhibits different bactericidal action. 13
Ultraviolet (UV) photocatalysis can induce oxidation progression and create greatly oxidative species (H2O2, O2−, and OH•). 14 Previous studies show that ZnO NPs are highly responsive to UV photocatalysis.15,16 UV radiation highly absorbs and photoexcites ZnO NPs, which function as heterogeneous catalysts, through various reactions, such as hydrogen transfer or dehydrogenation and soft or full oxidation. 17 These reactions cause the removal of oxygen from the surface and create oxygen varieties, which damage bacterial cells. 18 The UV photocatalytic effect is still active in avoiding the UV irradiation resource because of its strong association with the surface electron depletion region to the negative oxygen species adsorbed on the surface. 19
Silicone rubbers are polymers with inorganic backbone comprising alternating oxygen and silicon atoms with organic side groups. This structure exhibits silicone extraordinary properties. Silicone rubbers are elastomeric polymers with high-grade elasticity; they are characterized by hydrophobic nature, resistance to temperature, flame, chemicals, UV, and weathering effects.20,21 They are useful for different applications, particularly as textile coatings in different fields, such as heat-resistant clothing, weather-proof tents, flame-resistant clothing, UV-stable paragliders, and functional clothing for outdoor activities. Room-temperature vulcanizing silicone rubber (RTVSR), a silicone rubber, is a widely used synthetic rubber in many fields because of its unique manufacturing and shaping ease properties. These properties include exceptional thermal stability, decent chemical stability, oxidation resistance, low-temperature hardiness, minimal toxicity, and electrical insulation. Thus, RTVSR has been used to improve the thermal stability and water repellency of cotton fabrics22,23
ZnO NPs have been applied through different methods to treat cotton fabrics against microbial attack, including the coating of cotton fabrics with ZnO/carboxymethyl chitosan (ZnO/CMCS) 24 ;deposition of ZnO nanoparticles on cotton fabrics using the pulsed laser ablation method 25 ; fabrication of multifunctional ZnO/cotton fabrics by surface micro dissolution using sodium hydroxide (NaOH)/urea and zinc chloride (ZnCl2) aqueous solution 26 ; coating with (ZnO)-1,2,3,4-butanetetracarboxylic acid (ZnO-BTCA) with carbon nanotube (CNT) composites 27 ; and treatment with nanocomposites based on ZnO NPs, acyclovir, nanochitosan, and clove oil. 28
This study synthesized ZnO within the nanorange scale in formulations of RTVSR/ZnO NPs. The prepared formulations were applied to cotton fabrics via coating to improve their antimicrobial properties for outdoor use. UV radiation was used to enhance the curing of silicone rubber and antimicrobial activity through photocatalytic phenomena. The prepared ZnO was characterized by transmission electron microscopy (TEM) and X-ray diffraction (XRD) to confirm that the particles were within the nanometer range. The surface morphology and thermal stability of the coated fabrics were characterized by scanning electron microscopy (SEM) and thermogravimetric analysis (TGA), respectively. The effect of the UV irradiation time on the solubility (%), swelling number, mechanical properties, and antimicrobial properties of the coated fabrics was investigated.
Materials and methods
Materials
Cotton textile was providing from El-Nasr Company, El-Mahalla El-Kubra, Egypt. The cotton fabrics were washed and scoured using 2 g/L of nonionic soap (Snadopan), which, supplied from, Sandoz, Switzerland). The fabrics were not displayed to any dyeing processes. Room-temperature-vulcanizing silicone rubber (RTVSR), which consists of H (OSi(CH3)2)n OH with a molar mass of (∼60,000) and (alkoxysilane crosslinker, (C2H5O)4Si) with a ratio of 100:1, and tin dibutyl lauric acid, which used as curing catalyst were kindly provided as ready commercial products for use by Shanghai Rubber Product Institute (China). Zinc acetate [Zn(CH3COO)2·2H2O], with a Molecular weight of 219.50 g/mol, was kindly supplied by Qualikems, India. Toluene (Assay: 99%) and Sodium hydroxide (Assay: 98%) were obtained from El-Gomhoria Company, Egypt. Agar powder (Ash content; 3%, Gel strength ∼600 gm/cm min) was kindly obtained from Nice Chemicals, India. Envoy pathogenic microbes of equally Gram-negative (Escherichia coli) and Gram
Synthesis of ZnO NPs
ZnO NPs were synthesized using the sol–gel method.
29
Two beakers were prepared; the first contained 2 g of zinc acetate dihydrate dissolved in 15 mL of distilled water. The second contained 8 g of NaOH dissolved in 10 mL of distilled water. The contents of both beakers were stirred for approximately 5 min. After mixing, the NaOH solution was poured into the solution containing zinc acetate dihydrate, with continuous stirring using a magnetic stirrer for approximately5 min. Thereafter, a burette containing ethanol was titrated drop wise into the produced solution; a white precipitate was formed by the end of the reaction. The precipitate powder was carefully discarded, washed using distilled water, and dried at 80°C for 24 h in an oven.
Characterization of synthesized ZnO NPs
The particle size of the prepared ZnO was determined by TEM (JEM100CS, JEOL Electron Microscope, Japan), using an acceleration voltage of 80 kV and formfitting through a digital camera (DXM1200, Nikon). The samples were equipped by depositing a dilute solution containing the prepared specimen on a network copper grid (200 mesh) covered with carbon films.
The prepared ZnO NPs were characterized by XRD using the Shimadzu style XD-6000. The diffractogram scan ranged from 2 to 40° at room temperature at a rate of 8°/min. The XRD results were used to determine the mean grain size (D) of the synthesized ZnO particles using the Scherrer equation
The Energy Dispersive X-ray Spectroscopy (EDS) was used to investigate purity and the elemental composition of the synthesized ZnO particles, using the EDX unit (Smart EDX Company, UK).
Coating of cotton fabrics with silicone rubber formulations and UV irradiation
Formulations containing silicone rubber (RTVSR), the catalyst (3% of used SR), and different percentages of the prepared ZnO NPs (0.5, 1, 3, and 5% of the total ingredient volume) were mixed with continuous stirring until homogeneous mixtures were achieved. The prepared formulations were used to surface-coat the cotton fabric substrate using a film applicator, with a thickness of ∼100 μm (Scheme 1). The coated fabrics were exposed to UV irradiation for different durations (5,15, 30,60,120, and 240 min) using a typical UV lamp (category EMITA VP-60, Poland), with a 180-W mercury light, (50 Hz, 220 V) prepared using a monochromatic filter ( Synthesis of ZnO NPs and the coating procedure of cotton fabric.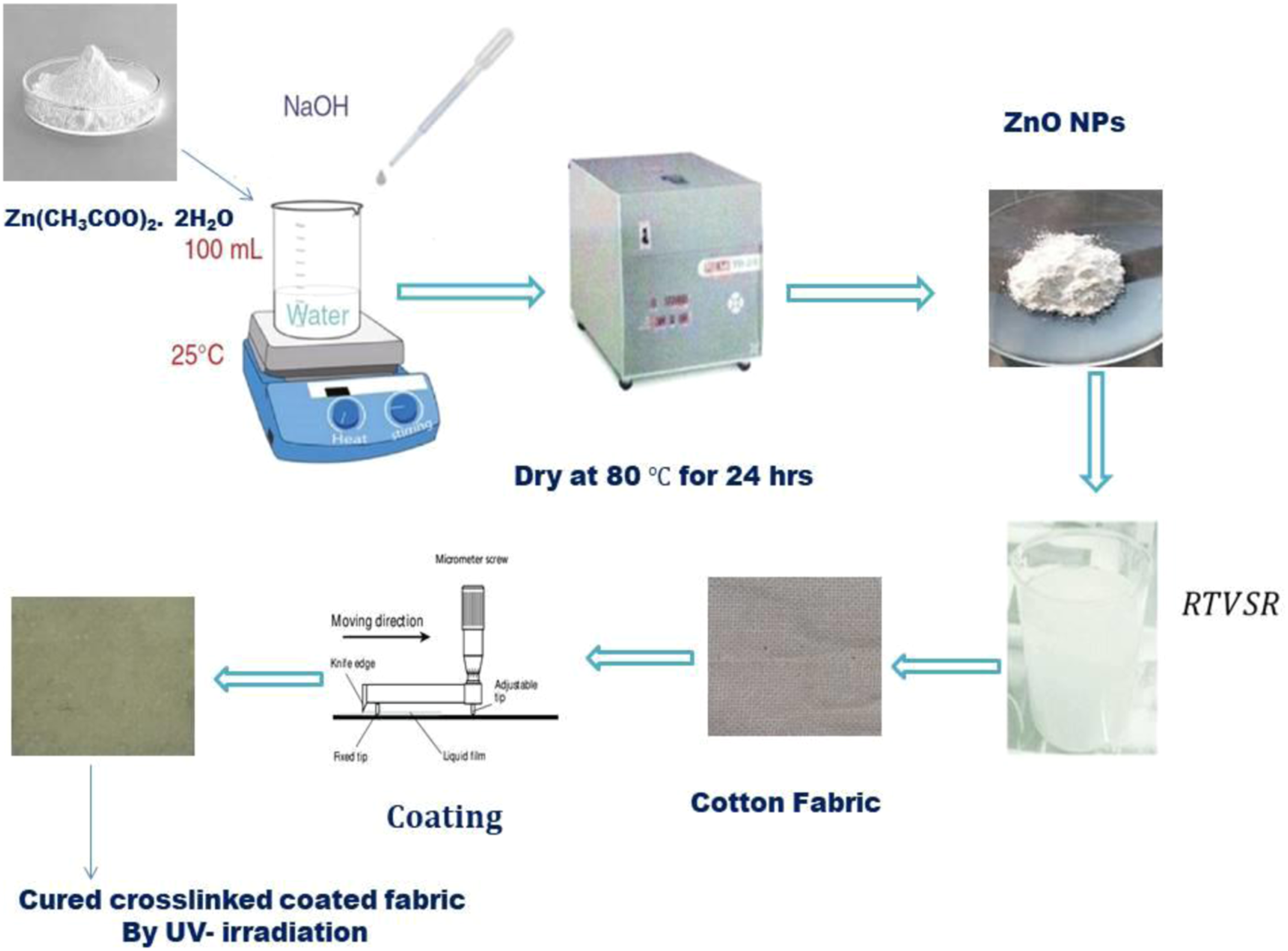

Flowchart for the producing of ZnO NPs and the coating procedure of cotton fabric with a formulation of RTVSR/ZnO NPs.
Characterization of the coated cotton fabrics
Surface morphology (SEM)
The surface morphology of the cotton fabrics prior to and after coating was observed by SEM (JSM–640, JEOL) at 15 kV. The dehydrated samples were sputtered with gold with a microscope sputter coater to view through the microscope.
TGA
TGA was conducted using a TG-50 from Shimadzu (Japan) at a heating system rate of 10–600 °C/min according to ASTM E1641-07. The sample masses ranged between 3 and 5 mg and were encapsulated in platinum pans. The analysis was directed under nitrogen gas at a flow rate of 20 mL/min.
Mechanical properties
The mechanical properties of the cotton fabrics prior to and after the coating process with the SR formulation were studied. The Hung-Ta model HT-9112 (Taiwan) was used to analyze the mechanical properties of the cotton samples, according to ASTM D 412, 1986, at room temperature; the tested samples were cut into rectangles with a width and length of 10 and 70 mm, respectively.
Determining solubility and swelling parameters
The solubility properties of the coating SR formulations on the cotton fabrics were determined by extraction in toluene solvent with heating. The samples were weighed and situated at the top portion of the withdrawal device (Soxhlet). After extraction, the samples were dried in a vacuum oven at ∼50°C until a constant weight was achieved. The solubility ratio (S) was specified by
Evaluation of the antimicrobial properties of the coated fabrics
Antimicrobial agar diffusion system
The diffusion disk experiment is used to determine bacterial activity using the dimensions of the nearby inhibition zone used to treat samples (ASTM E3152-18, 2018). Sterilized nutrient agar intermediate was dispensed into sterilized Petri dishes. Potage culture containing 108 CFU mL−1Bacillussubtilis ATCC 6538 as Gram-positive bacteria or E. coli ATCC 8739 as Gram-negative bacteria was swabbed in excess on the exterior of the nutrient agar dishes. Small samples (1.1 cm2) were gently hard-pressed atop the swab agar plane. The dishes were incubated at 37°C for 48 h. The resistance of the tested microbes was determined by measuring the surrounding region of the inhibition zone.
The burial test method
In this method (ISO 11721-1:2001), the treated samples were adjusted for burying in natural, fertile, and uniform soil (through sieving) after adjusting their moisture content by gradually adding water and placing them in a dry wooden box at a depth of ∼13 cm. The pre-weighted specimens were horizontally buried in the soil for 10 days at room temperature, and the moisture content of the soil was maintained using the test dated between 20 and 30% (built on the dry weight).
Results and discussion
Characterization of synthesized ZnO particles
Diameter measurement (TEM)
The diameters of the synthesized ZnO particles were determined by TEM (Figure 1). The TEM images show that the synthesized ZnO particles were predominantly spherical and within 10–17 nm in size, indicating that the synthesized ZnO particles were in the nanometer range. Transmission electron microscopy (TEM) images of the synthesized ZnO particles.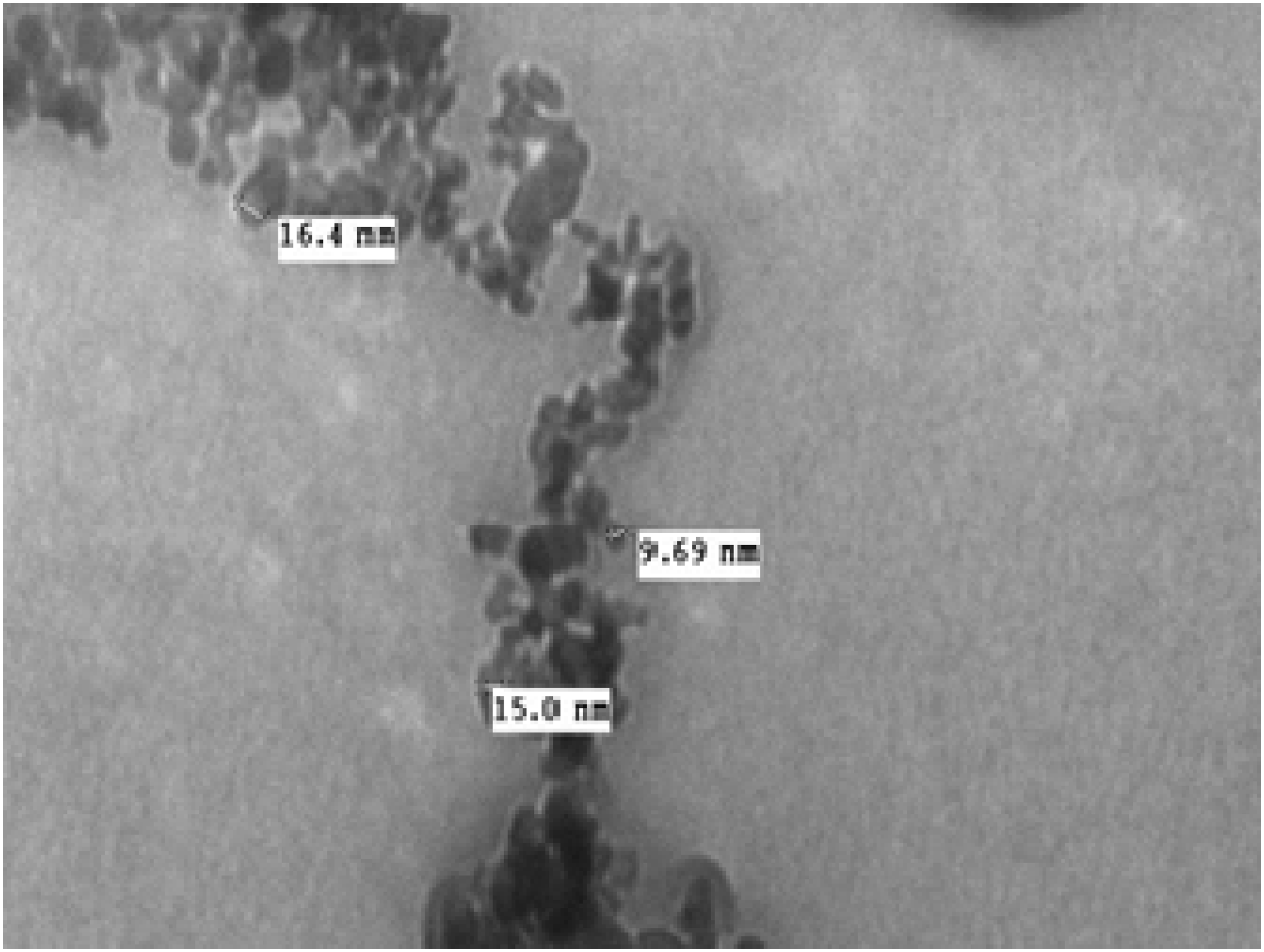
XRD study of the synthesized ZnO particles
Figure 2 shows the XRD patterns of the synthesized ZnO particles. The XRD studies confirmed that the diffraction peaks of the synthesized ZnO correlated with reported JCPDS data, without any strange peaks. Additionally, the mean grain size (D) of the synthesized ZnO particles was obtained using the Scherrer equation. The average calculated diameter of the synthesized ZnO particles using the Scherrer equation was 15.8 nm, indicating that the synthesized ZnO particles were within the nanometer range. X-ray diffraction (XRD) patterns of the synthesized ZnO particles.
EDS study of the synthesized ZnO particles
The elemental composition and the purity of the prepared ZnO nanoparticles were analyzed by EDS. This analysis was performed of the prepared sample to determine the Zn and O ratios. Figure 3 confirmed that only Zn and O elements were present in the prepared sample, thus indicating the high purity of ZnO in the sample. The appearance of sharp diffraction peaks, as seen in the Figure 3, indicated the good crystallinity of the prepared nanoparticles. It observed that stoichiometric percentages of Zn and O are 36.62%, and 63.38%, respectively. Energy dispersive X-ray (EDC) of synthesized ZnO.
Surface morphology of the coated cotton fabrics
The surface morphology of the cotton fabrics, cotton/SR, and cotton/SR/ZnO NPs (3%) was investigated by SEM (Figure 4). For the pure cotton fabric, Figure 4(a) shows the free pull-out fibers of the fabric. After the coating with SR, the SR spread through the cotton fabric, filling the interspaces with good interfacial interaction with the fabric fibers (Figure 4(b)). After adding the ZnO NPs to the SR formulation, homogeneous dispersion of ZnO NPs was observed through the SR matrix (Figure 4(c)). In addition, the smoothness of the coating sheet with SR only slightly decreased after the addition of ZnO NPs. Surface morphology of the (a) cotton fabrics, (b) cotton/SR, and (c) cotton/SR/ZnO NPs.
Thermal stability of the coated cotton fabrics
Figure 5 shows the TGA thermograms and corresponding rate of the thermal decomposition reaction (curved shape) of the coated cotton fabrics with SR and those coated with SR/ZnO NPs (3%). Table 1 summarizes the temperature parameters, where the (T
50
) represents the temperature at 50% weight loss of the substrates, while the (T
max
) represents the temperature at which the maximum rate of the reaction occurred. The thermal decomposition of the coated cotton fabrics with SR and SR/ZnO NPs occurred in two stages. The first stage (200°C–400°C) involved the evaporation of the cellulose water content, whereas the second stage (400°C–560°C) involved the thermal degradation of the coated fabrics. Owing to the two expected possibilities, the overall thermal stability of the coated fabrics noticeably increased when coated with SR, accompanied by a decrease in the thermal decomposition rate. However, noticeable differences existed at the onset, midpoint, endset, and T
max
temperatures, compared with those for the pure cotton fabrics. The thermal stability of the coated cotton was enhanced because of the crosslinking density, and accordingly, the rigidity of the system was enhanced, which in turn improved the thermal stability.
23
Thermogravimetric analysis (TGA) thermograms and thermal decomposition rates of the cotton fabrics, cotton/SR, and cotton/SR/ZnO NPs (3%). Thermal properties of the cotton fabrics, cotton/SR, and cotton/SR/ZnO NPs (3%). aTonset, T10 and T50 are weight loss temperatures at onset, 10% and 50% respectively. bTmax is the temperature corresponding to the maximum rate of weight loss. cThe amount of mass % remaining at 600°C.

Table 1 illustrates the temperature parameters of the thermal decomposition of cotton fabrics, cotton/SR and cotton/SR/ZnO NPs (3%). It can be noticed that the temperature values of the (Tmax) were (349, 353 and 356oC), while the temperature values of the (T50) were (354, 473 and 475oC) for the untreated fabric, coated fabric with SR and coated fabric with SR/ZnO NPs (3%), respectively. Meanwhile, the final char residues were (6, 24 and 27%), for the untreated fabric, coated fabric with SR and coated fabric with SR/ZnO NPs (3%), respectively at 600°C. The final char residue was increased by 15 and 21% more than pure cotton fabrics, after the coating with SR and SR/ZnO NPs, respectively.
On the basis of the temperature parameters values and the final char residues, it can be concluded that, the thermal stabilities of the cotton fabrics were enhanced after the coating process with the different SR formulations. The thermal stabilities can be arranged as follows: Cotton/SR/ZnO NPs (3%) > Cotton/SR > Cotton.
Effect of UV irradiation on the swelling and solubility of the coating formulations
The effect of UV irradiation as a crosslinking factor on the SR/ZnO NPs coating formulation as a function of the soluble fraction and swelling ratio of the formulation was investigated. The solubility fraction and swelling ratio of the coated cotton fabrics with SR and SR/ZnO NPs (3%) after irradiation at different UV doses are shown in Figure 6. The soluble fraction (SF) of the SR on the cotton fabric decreased with an increase in the UV irradiation dose, particularly at a high irradiation dose (>1 h of exposure). This was based on the crosslinking effect of UV irradiation on the rubber composites, resulting in increased hardness and reduced flexibility.
30
Moreover, the addition of ZnO NPs to the SR ultra-decreased the SF of the coating formulation, which was based on the reinforcing effect of the ZnO NPs on SR.
31
However, as shown in Figure 6, the same trend was observed by studying the effect of UV irradiation on the swelling ratio. An increase in the UV irradiation dose led to a decrease in the swelling ratio. The addition of ZnO NPs further decreased the swelling ratio. Effect of UV irradiation time on the solubility (%) and swelling ratio of the cotton/SR and cotton/SR/ZnO NPs (3%).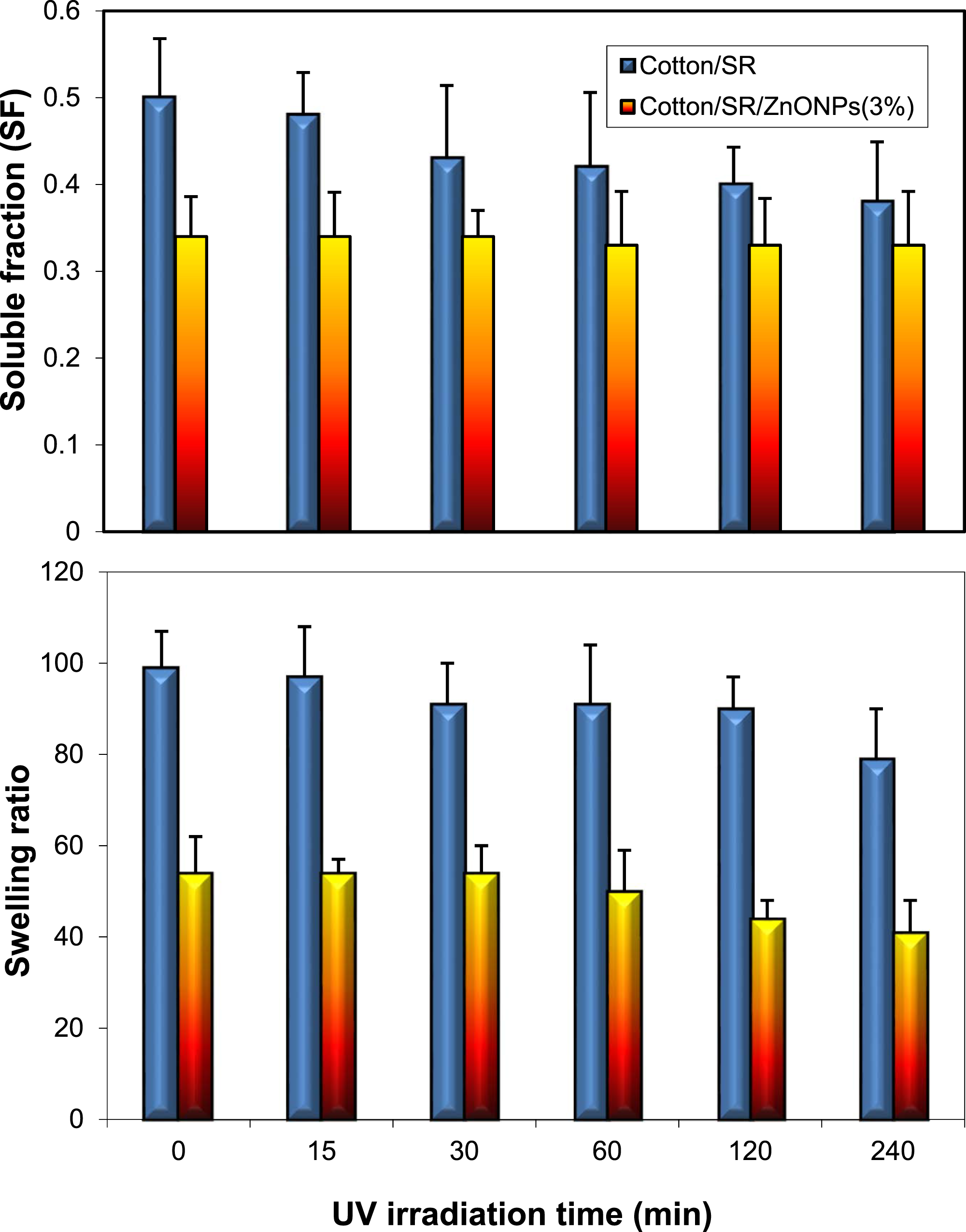
Effect of UV irradiation time on the mechanical properties
The effect of UV irradiation, as a crosslinking factor, on the coated cotton fabrics with SR/ZnO NPs 3% was investigated. Figure 7 illustrates the change in the tensile strength (MPa) and elongation at break (%) for the untreated and coated cotton with the SR/ZnO NP formulation (3%), irradiated at different UV irradiation times (15, 30, 60, 120, and 240 min). For the coated cotton fabrics with only SR, the tensile strength (MPa) increased with an increase in the UV irradiation time, accompanied by slight decrease in the elongation (%) up to 120 min. The further increase in the irradiation time slightly increased the tensile strength (MPa) with a decrease in the elongation at break (%). This could be attributed to the increase in the crosslinking formation of the SR coating formulation with radiation. The addition of the ZnO NPs (3%) to the coating formulation was accompanied by an overall decrease in the tensile strength (MPa) and elongation at break (%). This may be based on the distortion in the SR polymer matrix because of the addition of the ZnO NP powder encapsulation in the coating formulation. Effect of UV irradiation time on the tensile strength (MPa) and elongation at break (%) of the cotton/SR and cotton/SR/ZnO NPs (3%).
Effect of UV irradiation on the antimicrobial activity of the coated cotton fabrics
The ZnO NPs in the coating formulation were highly responsive to UV photocatalysis. Thus, the UV radiation highly absorbed and photoexcited the ZnO NPs functioning as heterogeneous catalysts through various reactions, such as hydrogen transfer or dehydrogenation and soft or full oxidation, which create extremely oxidative species (H2O2, O2−, and OH•). These reactions cause the removal of oxygen from the surface and create oxygen varieties, which damage the bacterial cells.14–18
Antibacterial qualitative agar diffusion method
Figure 8 illustrates the antimicrobial properties of the cotton fabrics coated with RTVSR and RTVSR loaded with varying concentrations of the prepared ZnO NPs (0.5,1, 3, and 5%), as a function of the growth inhibition of the tested microorganisms, indicated by the inhibition zone. The effect of the antimicrobial activity of the coating formulations predominantly depended on the ZnO NP additives, which increased with an increase in encapsulated ZnO NP concentrations, under the conditions of the inhibition zone, toward the used types of pathogenic bacteria (+Ve and −Ve). This effect could be attributed to the antimicrobial effect of ZnO as an inorganic antimicrobial agent, exhibiting considerable antimicrobial action while the particle range was condensed toward the nanometer series. In this range, the ZnO NPs cooperated with the bacterial exterior or center, wherever it enters the cell and consequently, exhibited bactericidal action.7,14 However, at constant concentrations of ZnO NPs, the sensitivity of the Gram-negative (E. coli) bacteria to the coating formulation as a function of the inhibition zone was slightly lower than that of B. subtilis (Gram-positive). This was based on the variation in the cell barrier for both types and the difficulty in infringing the twofold walls of the Gram-negative (E. coli) bacteria. Qualitative antimicrobial activity of cotton fabrics treated with silicone rubber incorporated with different concentrations of ZnO NPs with UV irradiation.
Qualitative antimicrobial activity of cotton fabrics treated with silicone rubber incorporated with different concentrations of ZnO NPs with UV irradiation.

Soil burial test
The efficiency of the coating process for protecting cotton fabrics from degradation as a result of microbial attacks was evaluated using the soil burial test. Figures 9(a)–(c) shows the photographs of the untreated cotton, coated cotton with SR, and that with SR/ZnO NPs (3%) before burying. Figure 9(d)–(f) shows the samples after burying in moist soil, loaded with microorganisms for 10 days Figure 9(d) shows the decay and decomposition of the untreated cotton samples, which gained an intense brownish color because of the microbial attack. However, the coating of cotton fabrics with SR slightly protected the cotton fabrics from microbial attack (Figure 9(e)). The addition of ZnO NPs (3%) to the coating formulation with SR sharply enhanced protection against microbial attack (Figure 9(f)). Photographs of (a) cotton, (b) cotton/SR, and(c) cotton/SR/ZnO NPs (3%) before and (d) cotton, (e) cotton/SR, and (f) cotton/SR/ZnO NPs (3%) after burying in moist soil rich in microorganisms.
Figure 10 illustrates the effect of varying the ZnO NPs concentrations on the coating formulations and exposing the coated cotton samples to UV irradiation at varying times on the antimicrobial attack, before and after soil burial, regarding the change in its tensile strength tests. For the untreated cotton fabrics, the fabrics sharply decayed, accompanied by an extreme decrease in tensile strength. By increasing the ZnO NP concentration, the tensile strength was enhanced because of an increase in the microbial inhibition effect, leading to the protection of the tensile strength of the coated fabrics.
13
Tensile strength (MPa) of the cotton fabrics coated with silicone rubber containing different concentrations of ZnO NPs under the effect of UV irradiation, after burying in moist soil for 10 days.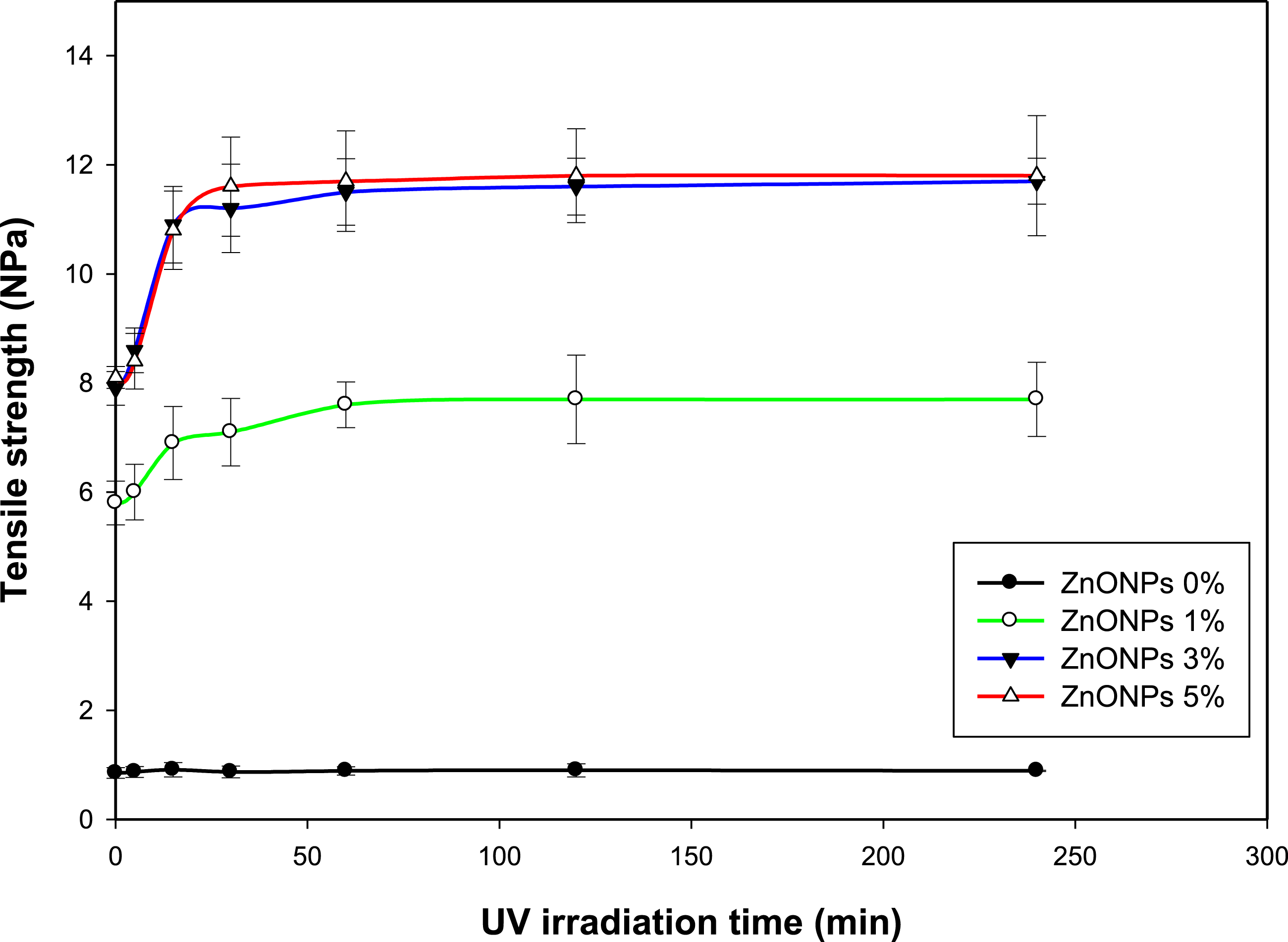
Using various concentrations of ZnO NPs in the coating formulations, the effect of UV radiation as a catalytic factor intended for the antimicrobial effect of ZnO NPs was investigated (Figure 10). At different concentrations of ZnO NPs (1, 3, and 5%), the antimicrobial activity of ZnO NPs increased with an increase in the UV irradiation time, thereby increasing the overall tensile strength of the coated cotton fabrics. Furthermore, the catalytic effect of UV irradiation was sharply enhanced with irradiation for 15–60 min and leveled off with an increase in irradiation time.
Conclusions
This study prepared formulations of RTVSR/ZnO NPs, to improve the antimicrobial properties of the cotton fabrics for outdoor use via coating processes. The coated fabrics were adjusted at different UV doses, which exhibited a dual effect (i) crosslinking factor for the RTVSR and (ii) enhanced the antimicrobial activity of the ZnO NPs via the photocatalytic effect. The results showed that the exposure to UV irradiation up to 120 min enhanced the crosslinking of RTVSR by a ratio of ∼11% of the mechanical strength (MPa), accompanied by a decrease in swelling of ∼10% and solubility ratio of ∼20%. The antimicrobial activity of the coated fabrics was evaluated using qualitative agar diffusion and soil burial test methods, and the results showed an increase of ∼50% in the antimicrobial activity of the coated cotton fabrics because of the photocatalytic effect of UV irradiation (after exposure for 120 min) at the constant percentage of ZnO NPs (3%) in the coating formulations.
Footnotes
Acknowledgements
Authors thankfully acknowledge the substantial monetary care of Egyptian Atomic Energy Authority (EAEA). This study remained reinforced through National Centre for Radiation Research and Technology (NCRRT).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.




