Abstract
In this study, chitosan is obtained from the waste shells of crayfish (Astacus leptodactylus) and characterized. The crayfish chitosan was coated on cotton fabrics using the padding-drying method. The surface properties, flammability and water and air permeability properties of these coatings based on chitosan and a combination of chitosan and nano-TiO2 were determined. The weight of green calico fabric increased to 24.76% after coating with chitosan, while the weight of the blue dyed denim fabric increased to 5.19%. The chitosan coating increased the tear strength of the calico fabric by 20% in the weft direction. Thermogravimetric analysis and vertical burning tests show that chitosan improves the thermal resistance and flame retardancy performance of denim fabrics. While the amount of residue due to thermal degradation of the blue dyed denim fabric was 16.78%, it increased to 34% after coating with chitosan+nano-TiO2. The chitosan coating reduced the mass loss caused by thermal degradation in green calico fabric from 86.3% to 66.0%. Chitosan also improved the post-flame combustion performance of fabrics. While the ember burning time is decreased to 6s in the chitosan-coated dyed calico fabric, this did not occur in the chitosan+nano-TiO2-coated dyed denim fabric.
Introduction
Garments made of cellulosic fibers are flammable when exposed to sources of ignition, such as open fires, stoves and cigarettes, and the flames can spread rapidly, with the user potentially experiencing skin burns of up to 70% of the body surface within seconds.1–3 Since cellulosic fibers exhibit a low limiting oxygen index, high flammability and ease of ignition, 4 fire safety has become particularly important in the textile industry in the last few years and the production of flame retardant protective clothing has become a necessity.5,6 Denim fabrics are widely used by people of all ages in all areas of our lives. Calico fabric, on the other hand, is a type of fabric preferred in clothing and home textiles. Since these fabrics are cotton, they are easily flammable. For this reason, denim and calico fabrics, which are highly demanded by consumers, were preferred to provide flame retardancy.
Fabric flammability is a process that begins at the fabric surface. When the heat transferred to the fabric surface is sufficient, it can cause further degradation and thus encourage a self-sustaining combustion cycle. The chemical and physical properties of a fabric surface significantly affect the ignition and burning processes it can undergo.
7
Flame retardant chemicals act to break the textile’s self-sustaining combustion cycle. For current technology, the greatest problem encountered in the development of flame-retardant chemicals is their harmful impact on the environment and health.
8
In addition, the effects of coating chemicals on global warming and climate change should also be considered. Therefore, significant restrictions have been imposed on the use of potentially harmful chemicals in the textile coating process. Some halogen-based products used as flame retardants are banned in the USA and the EU due to their high toxicity on humans and animals.9–11 For this reason, various additives have been investigated for the design of safe and innovative flame retardant systems. Among them, spherical inorganic nanoparticles (TiO2, ZnO etc.), phosphorus and borate-based compounds, nanotubes and carbon-based graphite are widely used as flame retardant additives in polymer composites.
12
Elsayed et al.
13
developed a new flame retardant and antibacterial textile coating based on inorganic nanotubes and Polyaniline (PANI
Research over the last decade has shown that some biological macromolecules and bio-origin products can be used to design “green” and effective flame retardants 16 Therefore, studies have focused on green biocomposites. 17 Chitosan, a green biopolymer, is preferred for surface modification in textile processes due to its cationic structure, unique biological properties and low cost. 18 Chitosan is a char-forming agent during combustion as a carbon source and acts as a blowing agent by releasing ammonia during decomposition due to its high nitrogen content. 19 These properties of chitosan make it a potential flame retardant. However, previous studies have reported that chitosan cannot provide sufficient flame retardancy when used alone and should be supplemented with various additives. 20 Chen et al. 21 synthesized a composite using chitosan (CS) together with ammonium polyphosphate (APP) to improve the flame retardancy of polylactic acid (PLA). The combination of APP and CS showed synergistic effect in improving the flame retardancy of PLA biocomposites by producing continuous and homogeneous char residue, promoting the crystallization of PLA, and forming a protective layer that swells on the fabric surface. Liu et al. 22 produced phosphorylated chitosan decorated montmorillonite nanoplatelets (PMT) to improve the fire resistance of thermoplastic polyurethane (TPU) composites. PMT increased the char residue, improving thermal stability with the inclusion of aluminum hypophosphite. In addition, the composite coating acted as a thermal insulation layer, which effectively inhibited heat/oxygen conduction in the condensed phase during combustion and prevented organic decomposition volatiles from entering the gas phase. Wang et al. 23 coated a novel intumescent flame retardant combination containing ammonium polyphosphate/p-aminobenzene sulfonic acid modified melamine/chitosan onto cotton fabric surfaces. The authors reported that the composite coating significantly suppressed the heat release and mass loss of fabrics, increased char formation, and the residual content from 8.1% to 24.1% by weight. In the development of flame retardant systems based on chitosan and its derivatives, chitosan cross-linked with bis-(4-formylphenyl)-phenyl-phosphonate, phosphorylated chitosan, melamine salt of chitosan phosphate etc. derivatives have been used. 24 Attia et al. 25 developed the organic compound N-[2,3-dibromo-4-(4-methoxy-3-methylphenyl)-4-oxobutanoyl]anthranilic acid as antibacterial, flame retardant/charcoaling and reinforcing agent and combined it with chitosan. Zhou and Fu 18 obtained chitosan-based composite by doping chitosan with sodium phytate and nano-TiO2-ZnO particles to provide flame retardancy. Kundu et al. 19 synthesized chitosan-metal oxide nanoparticle hybrids in the development of bifunctional polyamide 66 textiles with improved flame retardancy and wettability. TiO2 has been considered a promising flame retardant due to its low toxicity. TiO2 increases the charring during combustion by forming a thermal protective layer on the surfaces of polymeric materials.19,26 For this reason, chitosan and nano-TiO2 particles were preferred to give flame retardancy to denim and calico fabrics.
Chitosan, which is easily soluble in weak acids, is the deacetylated form of chitin. Chitosan is used in many applications, such as biomedicine, cosmetics, wastewater treatment, agriculture, textile, tissue engineering and food industry, as a result of its biocompatibility, biodegradability, non-toxicity and easy processability.27,28 In this study, the crayfish Astacus leptodactylus living in freshwater ecosystems was used as a source of chitosan. This species, which is very common in the fresh waters of Turkey, has a high egg productivity, growth potential and ability to adapt to the environment. 29 Therefore, it has the potential to be cultivated and used as a sustainable chitosan source.
The chemical structure and green nature of chitosan 30 show that it can be successfully used in designing effective flame retardant systems with low environmental impact. 16 In addition to these, chitosan coatings are preferred in terms of providing a better moisture balance between fabric and skin, improving the surface properties of the fabric and increasing its comfort properties. 31 Nano-TiO2 is also used to provide textile fibers with various functions, such as photocatalytic, antimicrobial activity, self-cleaning, electrical conductivity, ultraviolet protection, antistatic properties, thermal stability and flame retardancy. 32 In addition, TiO2 nanoparticles are used in foods as a colorant, and in products such as toothpaste and sunscreen, thanks to their biological properties and low toxicity. 33 Because TiO2 provides good thermal stability, it prevents ignition and propagation of combustion by improving the carbonization process for polymer matrices undergoing combustion.18,26 In this study, the effect of nano-TiO2, together with chitosan, on the comfort properties of denim and calico fabrics was investigated. The effects of the chitosan biomaterial obtained from crayfish waste shells on flame retardancy and water and air permeability are also investigated to develop environmentally-friendly, sustainable and protective textile coatings.
Experimental
Materials
In this study, two fabric types with different structural properties were used: dyed and undyed denim fabrics made of 100% cotton with a classic 3/1 twill weaving type, 368 g/m2 unit area weight (weight), weft density 16 and warp density 27 and green dyed calico fabric produced from 100% cotton, with 105 g/m2 grammage, weft count 12 and warp count 18. In order to reveal the effect of chitosan according to the structural properties of the fabrics and the processes they have undergone, and to determine the most suitable fabric density and weight value for chitosan coatings, calico and denim fabrics were selected. Freshwater crayfish (A. leptodactylus) obtained from fishermen from Altınyazı Dam Lake was used as the chitosan source.Nano-TiO2 (particle size> 21 nm, white powder) were purchased from Merck Company. The nanoparticles were homogenized by vortexing for 10 s before use.
Isolation of chitin from crayfish and obtaining chitosan
The crayfish shells were washed in distilled water, and then dried in an oven at 50°C. Later, the dried crayfish shells were ground. Chitin isolation from the crayfish was carried out in three stages: deproteinization, demineralization and decolorization according to Erdoğan 34 In the demineralization stage, the ground sample was treated with a 2M 250 mL HCl solution (v/v), while in the deproteinization stage, it was treated with a 2M 250 mL NaOH solution (w/v). After treatment with acid and base, it was subjected to depigmentation in a mixture of purified water, methyl alcohol and chloroform in a ratio of 4:2:1. After this process, the isolated chitin was dried in the oven at 50°C, again. Deacetylation of the crayfish chitin was carried out by treating the dry chitin sample in a 70% NaOH solution (w/v) at 140°C for 4 h. After deacetylation, the crayfish chitin turned into chitosan. The obtained chitosan was rinsed with distilled water until the pH was neutral and dried in the oven at 50°C.
Preparation of chitosan coating solutions
Chitosan (10 g) in powder form obtained from freshwater crayfish was dissolved in a 1L solution containing distilled water and 1% acetic acid at 40°C on a magnetic stirrer for 15 min. The prepared chitosan solution was divided into two samples and nano-TiO2 at 0.05% was added to one of them. On this basis, two different chitosan solutions were used in the coatings. The fabric samples prepared were impregnated with coating solutions at a 60% squeezing pressure in a laboratory-type padding machine. The fabrics were then dried (100°C) and fixed at 110°C for 1 min. The amount of TiO2 used in this study was kept low (0.05% by weight) to minimize the health risks.
The TiO2 nanoparticles were homogenized by vortexing for 10 s before use. Then, nanoparticle suspensions were prepared using an ultrasonic homogenizer at 40W for 1 h. In this way, coating suspensions containing homogeneous nano TiO2 were obtained.
Characterization of chitosan
Fourier transform infrared (FTIR) spectroscopy
Chitin and chitosan produced from crayfish shells were scanned 32 times with a spectral resolution of 4 cm−1 in the wavelength range of 4000–625 cm−1 and their FTIR spectras were obtained by using a Perkin Elmer FTIR spectrometer.
Differential scanning calorimetry (DSC)
The thermal properties of the A. leptodactylus chitosan were determined under a nitrogen atmosphere using a TA Instruments DSC Q2000 V24.11 Build differential scanning calorimeter. The chitosan sample (50 mg) was heated in a hermetic aluminum container at a temperature from −50–350°C with an increase of 10°C/min.
Scanning electron microscopy (SEM)
The surface morphology of the crayfish chitosan was visualized at various magnificationsusing an FEI QUANTA FEG 250 scanning electron microscope.
Thermogravimetric analysis (TGA)
The thermal decomposition behaviour of crayfish chitosan was examined with an EXSTARS11 7300 device by heating from 25°C to 800°C in an nitrogen atmosphere.
Examination of structural properties of chitosan-coated fabrics
The TS 250 EN 1049-2 35 standards were used for the determination of the weft and warp densities in the fabrics and the TSE 251 35 test standards were used for grammage determination. The tear strength of chitosan-coated denim and calico fabrics was determined according to the Elmendorf method. This test based on the BS EN ISO 13937-1: 2000 standard.
Surface characterization of chitosan-coated fabrics
The surface morphologies of the coatings made with crayfish chitosan were examined with EVO LS10 and FEI QUANTA FEG 250 scanning electron microscopes. The distribution of nano-TiO2 on the cotton fabric surface was determined by energy-dispersive X-ray spectroscopy (EDX).
Investigation of thermal properties of chitosan-coated fabrics
Thermogravimetric analysis (TGA)
The thermal properties of the fabric samples coated with crayfish chitosan and nano-TiO2 were examined using a TGA analyzer (PerkinElmer Pyris 1) at a temperature range of 25–650°C, with a temperature increase of 10 °C/min. Nitrogen flow rate was 100 mL/min.
Flammability analysis
The flammability performance of the crayfish chitosan and fabric samples coated with crayfish chitosan and nano-TiO2 were determined by the limited flame splash test. The limited flame splash test was performed according to the TS EN ISO 15025:2017 36 standard. This test was chosen because its application closely resembles the actual fabric burn behavior.
Fabric samples prepared in appropriate sizes were conditioned for at least 24 h at 20 ± 2°C and 65 ± 4% RH. They were then placed in a vertical burning test device holder and the experiment was carried out at 23°C and 52% RH.
A flame of 25 mm in height, which was fired with propane gas, was perpendicularly applied to the fabric surface for 10 s. After flame application, changes on the fabric such as flaming, burning (embers), spillage (drip), and hole formation were observed.
Investigation of comfort properties of chitosan-coated fabrics
Air permeability
Air permeability measurements of the denim fabric samples, which were treated with solutions containing chitosan and chitosan and nano-TiO2, were made according to TS 391 EN ISO 9237:1999 37 with a SDL ATLAS M021 A test device. The values were obtained by determining the amount of air (mm) passing from the fabric surface of 20 cm2 at a 100 Pa test pressure in 1 s and the results were given in mm/s.
Water permeability
After the fabrics were conditioned according to the test conditions, water permeability measurements were made according to the hydrostatic pressure method (with a PROWHITE K007 device). Water permeability determination (T) was performed according to TS EN ISO 811:2018. 38 Measurements were taken with a water pressure increase rate of 10 ± 0.5 cm H2O/min or 60 ± 3 cm H2O/min at the 20 cm2 test area and at 100 Pa.
Results and Discussion
Characterization of chitosan
FTIR spectroscopy
The chemical structures of the chitin and chitosan produced from A. leptodactylus were characterized by FTIR spectroscopy. FTIR spectroscopy of α-chitin produces three characteristic absorption peaks at 1550, 1620 and 1650 cm−1, while β-chitin shows a single characterization peak at 1650 cm−1.39,40 These peaks of α-chitin were assigned to N–H bending and C–N stretching (amide II), C = O stretching (amide I) and C = O stretching (amide I), respectively.41,42 In our study, two absorption bands corresponding stretching of amide I, known as the characteristic FTIR bands of α-chitin, were observed at 1655 and 1619 cm−1 for the crayfish chitin, while the single band showing the amide II stretch was seen at 1552 cm−1 (Figure 1(a)). The crayfish chitin was found to be in the αform. These results were also supported by the FTIR results of shrimp chitin reported by.
43
Fourier transform infrared spectra of chitin (a) and chitosan (b) produced from A. leptodactylus.
Carbonyl I and amide II bands, which are distinctive bands for chitosan, were reported to be at 1650 and 1590 cm−1, respectively. 40 In this study, two characteristic bands of the crayfish chitosan were observed at 1654 and 1591 cm−1 (Figure 1(b)). This proves that chitosan was successfully obtained from the crayfish shells.
Differential scanning calorimetry
Thermal decomposition peaks of chitosan from A. leptodactylus.

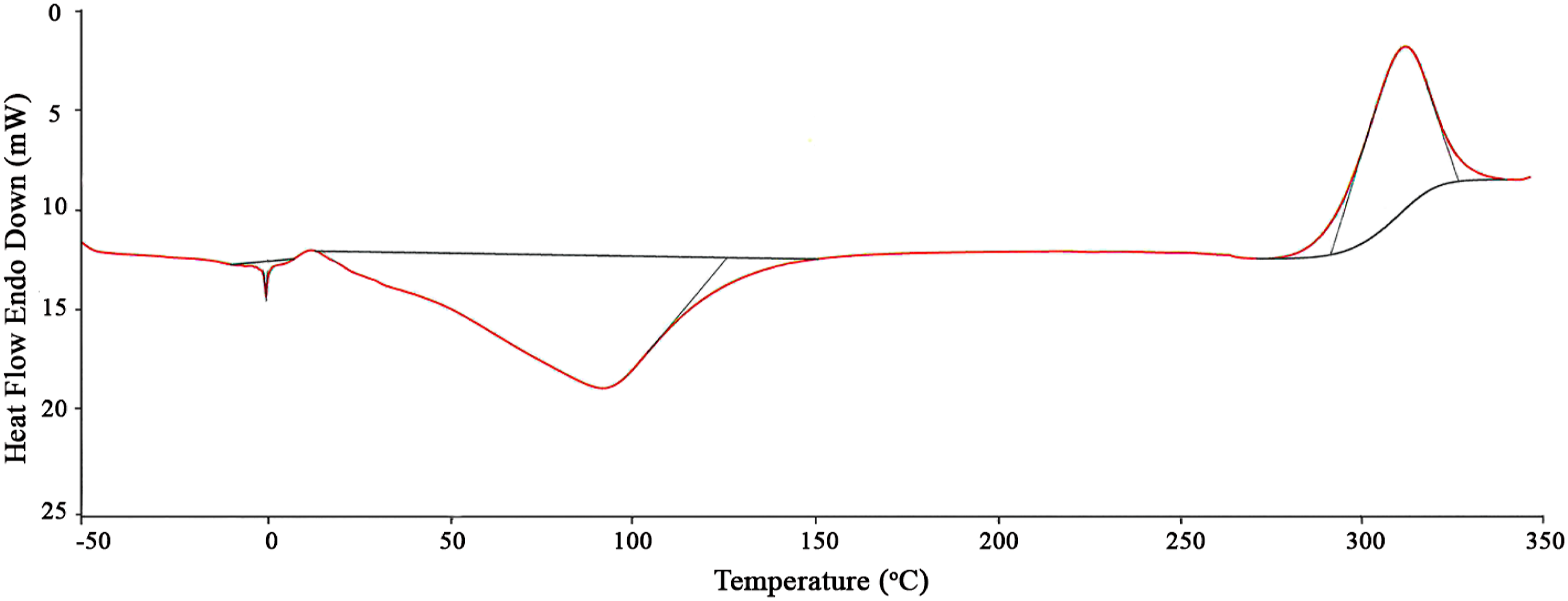
Differential scanning calorimetry thermogram of chitosan from A. leptodactylus.
Scanning electron microscopy
The surface images of crayfish chitosan taken at 40000X and 80000X magnifications are shown in Figures 3(a) and (b), respectively. Crayfish chitosan exhibits a surface morphology composed of nanopores and nanofibers. Pores are homogeneously distributed on the surface and their diameters vary between 95.86 and 186.6 nm. This morphological structure has been earlier reported by Kaya et al.
46
for blue crab chitosan. In recent years, various studies have been published on the use of chitosan, which has a porous structure, in drug release and tissue engineering applications.
47
In this study, the effect of crayfish chitosan with a porous structure on the comfort properties, such as air and water permeability, of denim and calico fabrics was investigated. Scanning electron microscopy images showing the porous structure of crayfish chitosan at a) 40000X magnification and b) 80000X magnification.
Thermogravimetric analysis
The thermal degradation curve of the crayfish chitosan was recorded between 25-800°C under nitrogen atmosphere. The crayfish chitosan lost mass in three stages (Figure 4). According to the TGA curve of chitosan, a weight loss of 11.5% was observed in the first stage, which can be attributed to moisture loss at temperatures below 150°C. In addition, this high degradation ratio in the first stage may be due to the fact that hydrogen bonds between polymer chains increase the initial decomposition temperature [22]. In the second stage, the weight loss between 290–330°C was due to the degradation of chitosan. In the final stage, chitosan continued to degrade at temperatures above 450°C. This may be due to the degradation of the saccharide ring structure in the chitosan molecule. The residual amount remaining undegraded was 4.71%.The maximum decomposition temperature of the crayfish chitosan was determined as 319°C. The thermal degradation curve of crayfish chitosan is similar to the thermal degradation curves presented for chitosan by Chen et al.
21
and Liu et al.
22
The thermal degradation curve of the crayfish chitosan.
Analysis results of chitosan-coated fabrics
Structural properties of crayfish chitosan coated fabrics
Changes in fabric structural properties after coating with chitosan.

Tear strength of chitosan coated denim and calico fabrics.

Surface properties of chitosan-coated fabrics
SEM and EDX analysis
The surface images of crayfish chitosan- and nano-TiO2-coated denim fabrics are shown in Figure 5. The surface image of the fabric coated with crayfish chitosan shows that the chitosan coating wraps the fabric surface in the form of a thin layer (Figure 5(a)). After the coating process with chitosan, no deformation, such as swelling or shrinkage, was observed on the fabric surface. Figures 5(b), (c) and (d) show the presence of nano-TiO2 on and between the fibers. While chitosan wraps the fabric like a film, it also encapsulates nano-TiO2 by surrounding them and enabling them to be coated on the surface of the fabric. The SEM images in this study are similar to the images presented by Dong et al.
50
Scanning electron microscopy images of a) crayfish chitosan-coated dyed fabric, b) crayfish chitosan and nano-TiO2-coated raw denim fabric, and EDX maps of c) crayfish chitosan and nano-TiO2-coated raw denim fabric and d) Titanium particles on the fabric.
According to the EDX analysis, the elemental content of the crayfish chitosan coated dyed denim fabric was determined as 48.77% carbon, 1.37% nitrogen and 49.86% oxygen by weight (Figure 6(a)). The crayfish chitosan and nano-TiO2-coated raw denim fabric was found to contain 44.83% carbon, 1.19% nitrogen, 53.91% oxygen and 0.08% titanium by weight (Figure 6(b)). Titanium also peaked at∼4 keV in the EDX spectrum of the crayfish chitosan and nano-TiO2-coated raw denim fabric, as reported in other studies.
51
The results revealed that the crayfish chitosan and nano-TiO2were successfully coated on the denim fabrics. Energy-dispersive X-ray spectroscopy analysis of a) crayfish chitosan-coated dyed denim and b) crayfish chitosan and nano-TiO2-coated raw denim.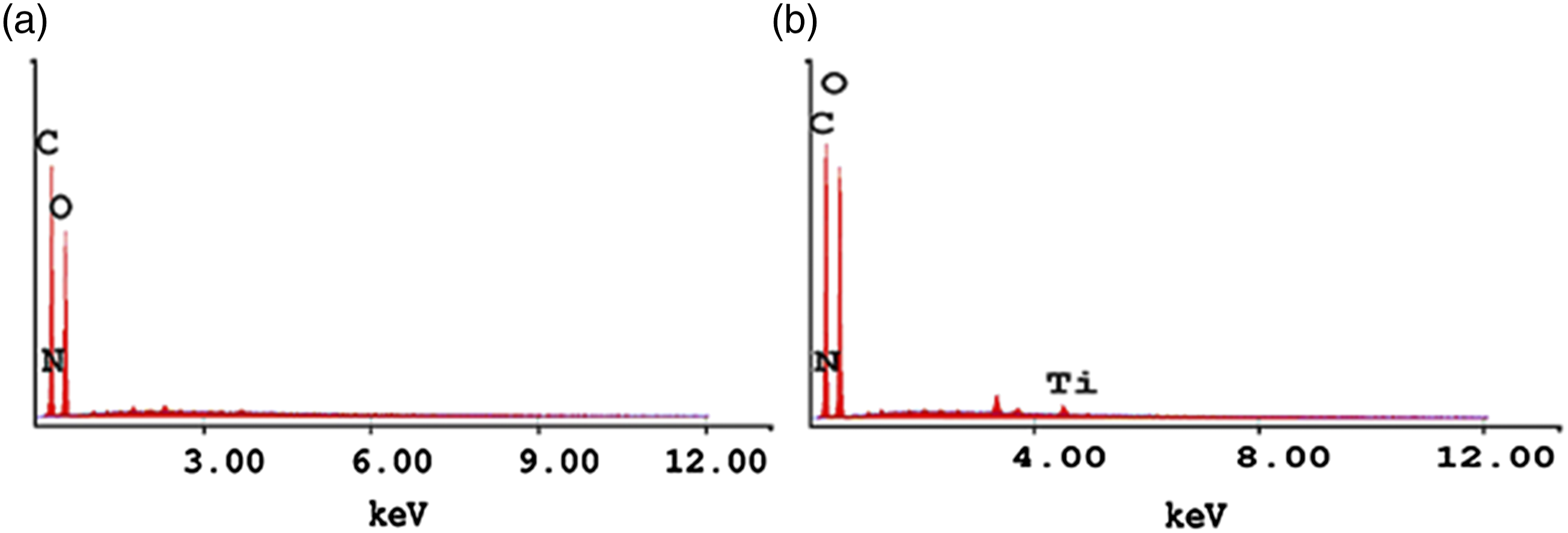
Thermal properties of chitosan-coated fabrics
Thermogravimetric analysis
The TGA of the uncoated fabrics indicated that there was a loss of mass in three stages (Figure 7). In the first stage, all fabric samples lost 4.6–5.0% of their mass between 25 and 100°C. In the second stage, the uncoated blue dyed denim fabric underwent maximum degradation at 360°C and the total mass loss reached 83.3% at 626°C. The maximum decomposition temperature of the uncoated raw denim fabric was 340°C and its total mass loss reached 82.6% when the temperature reached 626°C (Figure 7(b)). The maximum degradation temperature of the uncoated green dyed calico fabric was 380°C and the total mass loss at 626°C reached 86.3%. The highest degradation was observed for the green dyed calico fabric, which has the least residue (13.7%). It has been observed that the degradation rates are similar in dyed and undyed denim fabrics with the same structural properties. Mass losses are due to the loss of water in the first stage, the thermal decomposition of the cellulosic fabric in the second stage and the further decomposition of the degraded products in the third stage.
48
The burning properties of cellulosic fibers vary significantly related to their chemical composition.
52
The results of this study also confirm the previous data. The differences in fabric structure affected the degradation behaviour of the fabrics. In addition, the presence of dye on the fabrics caused a difference in their burning behaviour. Timpa et al.
53
examined the flammability properties of cotton fabrics dyed with indigo and bromine and/or chlorine substituted indigo dyestuffs. The authors observed that TGA curves were continuously shifted towards lower decomposition temperatures as dye addition increased. Compared to undyed controls, fabrics dyed with pure indigo showed little change in thermal degradation. It is also revealed in another study that dyes significantly affected the thermal degradation pathway. Decomposition temperatures in TGA curves decreased at different degrees depending on the dyestuff type.
54
The thermal decomposition temperatures of cotton fabrics dyed with acid dyes varied between 256-303°C. The thermal decomposition temperatures were between 313-326°C for the fabrics dyed with reactive dyes, and between 329-355°C for dyed with fluorescent brightning agents. Thermogravimetric analysis of uncoated fabric samples: a) blue dyed denim fabric, b) undyed raw denim fabric and c) green dyed calico fabric.
Like the uncoated fabrics, the chitosan-coated fabrics also lost mass in three stages. The weight loss in the first stage is attributed to water evaporation, the weight loss in the second stage is ascribed to the formation of gas and charcoal due to the decomposition of chitosan and fabric by the effect of heat and the weight loss in the final stage is assigned to the thermal oxidation decomposition of the char residue produced in the previous stages.
48
The mass losses occurring at 625°C were 88.3% in the blue dyed denim fabric coated with crayfish chitosan, 65.7% in the raw denim fabric coated with crayfish chitosan, 86.4% in the green dyed calico fabric coated with crayfish chitosan and 66.0% in the crayfish chitosan and nano-TiO2-coated blue dyed denim fabric (Figure 8). The highest residue was observed in the undyed raw denim fabric (34.3%) coated with crayfish chitosan and in the blue dyed denim fabric coated with crayfish chitosan and nano-TiO2 (34%). Thermogravimetric analysis of chitosan and nanometal coated fabric samples: a) blue dyed denim fabric coated with crayfish chitosan, b) undyed raw denim fabric coated with crayfish chitosan, c) green dyed calico fabric coated with crayfish chitosan, and d) blue dyed denim fabric coated with crayfish chitosan and nano-TiO2.
While the amount of residue of the uncoated undyed raw denim fabric was 17.4% (Figure 7(b)), caused by thermal decomposition, the residue increased to 34.3% (Figure 8(b)) in the undyed raw denim fabric coated with chitosan. While the maximum decomposition temperature of the uncoated raw denim fabric was 340°C, it decreased to 310°C in the chitosan coated fabric. This effect can be attributed to the ability of chitosan to stabilize the structure of cotton-based denim fabrics, lower the maximum decomposition temperature, and direct the fabrics to form less volatile and non-flammable components. 55 In uncoated blue-dyed denim fabric, the residual after thermal decomposition was 16.7%, while it reduced to 11.7% after coating with chitosan (Figure 7(a) and Figure 8(a)). While the maximum decomposition temperature of the same fabric was 360°C before coating, it increased to 382°C after chitosan coating and the amount of residue decreased. This indicates that the processes that the fabric has undergone before coating greatly affect the thermal decomposition of cotton fabrics and the performance of the chitosan coating. The chemicals used in the dyeing process adversely affected the thermal resistance increasing effect of chitosan on fabrics. However, this disadvantage can be eliminated by adding nano-TiO2 particles to chitosan coatings. The maximum thermal decomposition of the chitosan+nano-TiO2 coated blue dyed denim fabric occurred at 302°C and the residual amount increased to 34.0% (Figure 8(d)). Nano-TiO2 particles added into the chitosan coating cooled the pyrolysis region on the fabric surface with a synergetic effect and caused endothermic degradation.16,19 Chitosan contains nitrogen in its structure. In combustion reactions, nitrogen does not give off heat and reacts by taking heat. The combustion of nitrogen is endothermic. During combustion, chitosan creates a cooling mechanism on the fabric surface thanks to the nitrogen in its structure.20,25 Likewise, TiO2 nanoparticles can catalyze the carbonization process by forming a thermal protective layer that acts as an insulator on the chitosan coating surfaces. 19 Thus, the application of TiO2 nanoparticles together with chitosan reduces the mass loss of the fabric sample. Similar data to the results of this study were obtained in other studies. Hu et al. 56 prepared a renewable nickel chitosan phosphate composite with flame retardant and synergistic effects to improve the thermal properties and flammability of polyvinyl alcohol. Thermogravimetric analysis results showed that this composite led to high char formation and increased thermal stability. In another study, the wood samples coated with chitosan/sodium phytate/TiO2-ZnO composite underwent severe pyrolysis at a lower temperature (250–400°C) than the uncoated one and had the highest residual amount. 25
The chitosan coating did not have a significant effect on the burning performance of the green dyed calico fabric (13.6% residue).Therefore, it is obvious that the structural properties of the fabrics also have an effect on the results (denim fabric 368 g/m2 and calico fabric 105 gr/m2).Furthermore, the structural properties of the fabric were also affected by the dying process applied before coating. The degradation rates of the dyed and undyed denim fabrics with the same structural features are close to each other when they are uncoated, different from when they are coated with chitosan (Figure 8). It has been stated in previous studies that cellulosic textiles show distinctive peak thermal decomposition temperatures and percentage residue related to their chemical composition.32,52 The results of this study also support the literature data.
Nanoparticles are powerful addition agents to improve the thermal properties and morphology of char, due to their strong interaction with polymers and high decomposition temperature.57,58 It has been demonstrated in previous studies that nano-TiO2 provides thermal stability as an effective inorganic barrier.18,59 In this study, we observed that nano-TiO2 increased the thermal resistance of the blue dyed denim fabric and reduced the mass loss.
Flammability
Limited flame spread test results of calico and denim fabrics.


Vertical burning images of fabrics uncoated and coated with chitosan and nano-TiO2:Uncoated blue dyed denim fabric (a1, a2, a3 and a4), crayfish chitosan-coated blue dyed denim fabric (b1, b2 and b3), crayfish chitosan and nano-TiO2 coated blue dyed denim fabric (c1, c2 and c3), crayfish chitosan coated raw denim fabric (d1, d2, d3 and d4), uncoated green dyed calico fabric (e1, e2 and e3) and crayfish chitosan-coated green dyed calico fabric (f1, f2, f3 and f4).
We observed that the physical properties of the fabric affect the performance of the chitosan coating. The amount of post-combustion residue of the uncoated green-dyed calico fabric was 13.7% (Figure 7(c)), and after chitosan coating it was 13.6% (Figure 8(c)).This shows that chitosan coating had no effect on the combustion performance of green dyed calico fabric. In sparse and lightweight calico fabric (before coating: 105 g/m2, after coating: 126 g/m2), chitosan reduced the ember burning time after flame, but could not completely stop it (Figures 9(e) and (f)).The physical parameters of textile materials (fiber type, yarn count, weaving shape, weight, etc.) are important features that determine the burning behaviour. In textile materials, mass per unit area directly affects the flammability and linear combustion rate. The tighter and heavier the fabric, the longer it takes to ignite and burn. In less-thread fabrics, the combustion process takes place easily and quickly, thanks to the easy airflow in the fabric. The high-density, tightly woven fabric made from high-twist yarns provides better flame protection. 61
In denim fabrics, flame propagation is completely prevented as a result of the chitosan coating. Malucelli 16 states that flame retardant additives function by different mechanisms such as char formation, swelling, gas phase reactions, cooling mechanism and dilution. In this study, the flame retardancy mechanism of chitosan changed depending on the structural properties of the fabric and the processes it had undergone before coating. The flame retardancy that chitosan brings to the denim fabric can be explained by the following mechanisms. Chitosan supports the formation of char on the fabric surface during combustion, thanks to its high carbon content and multiple hydroxyl groups (Figure 6). 20 The char formed on the surface of burning fabrics reacts as a heat insulator, restraining the heat transfer from the heat source to the polymer and the mass transfer from the polymer to the flame. 55 The residue after thermal decomposition was 17.4% (Figure 7) in the uncoated undyed raw denim fabric, while it was 34.3% (Figure 8) in the chitosan-coated fabric. The char formation mechanism of chitosan was effective in the undyed raw denim fabric. Chitosan can interfere with the formation of flammable gas involved in flame propagation. The char formation mechanism of chitosan supports this situation. The charcoal formed acts as a barrier to prevent gaseous products from entering the flame. It also acts as a barrier that isolates heat and oxygen exchange. 20 Due to its high nitrogen content, chitosan can act as a blowing agent that releases ammonia during degradation.18,19 In combustion reactions, nitrogen does not give off heat and reacts by taking heat. The combustion of nitrogen is endothermic. During combustion, chitosan creates a cooling mechanism on the fabric surface thanks to the nitrogen in its structure (Figure 6).18,20 This was observed when the maximum decomposition temperature of the blue-dyed denim fabric was reduced to 302°C after coating with chitosan+nano-TiO2 (Figure 8(d)).
Chitosan, combined with other flame retardant addition agents that can directly interact with it, increases the flame retardant performance of fabrics and turns them into stable protective char. 16 Increased char formation during the combustion process is reported to be a good indicator of flame retardancy. 52 Zhang et al. 55 prepared a composite consisting of chitosan, phytic acid and metal ion as a synergist to obtain an efficient flame retardant composite and coated it on cotton fabrics with an easy dip-pad-dry process. This intumescent flame retardant system could accelerate the production of a large number of non-flammable gases through the synergistic effect of metal ions. Also, the phosphate groups in the composite promoted the formation of a charcoal layer that covers the surface of cotton fabrics and inhibits heat transfer. The results of their study showed that this prepared composite effectively improved the thermal stability and flame retardant performance of cotton fabrics, increased the char residues from 3.65% to 29.7%, and reduced the burning rate from 3.3 to 1.5 mm/s. 55 In addition, since the weight gain is less (5.2%), it has less effect on the softness of cotton fabrics. The incorporation of TiO2 nanoparticles into the chitosan coating catalyzes the carbonization process by forming a thermal protective layer on the surface of the polymer substrate. 26 Nanoparticles such as TiO2 provide thermal protection to the underlying material by forming a physical barrier layer that acts as an insulator, thereby reducing the heat release rate and mass loss of fabric substrates. 19 In the uncoated blue-dyed denim fabric, the amount of residue was 16.7% (Figure 7), 11.7% in the chitosan-coated fabric and 34.0% in the chitosan+nanoTiO2 coated fabric (Figure 8). Thus, nano-TiO2 particles prevented ignition and spread of combustion thanks to the cover they formed on the denim fabric surface.
Shafei, ElShemy, and Okeil 62 produced an antibacterial and flame resistant finishing agent for cellulosic fabrics (100% cotton) using nano-TiO2 and chitosan phosphate. According to the authors, in the presence of TiO2, the crosslinking of cellulose chains increases, thereby providing the stability of the cotton structure, increasing the thermal stability of cotton fabrics. In another study, the chitosan/sodium phytate/TiO2-ZnO nanocomposite coated on the cellulosic surface in layers formed a physical barrier and exhibited an effective intumescent flame retardant property. 18 Both TiO2 and ZnO nanoparticles prevented ignition and propagation of combustion by forming a dense cover on the cellulosic surface and provided good thermal stability. It has been observed that the better results are achieved by using nanomaterials together with other flame-retardants and natural fibers due to the significant synergy between the components. 17 However, the results of our study revealed that chitosan alone exhibited a good flame retardancy performance and the addition of nano-TiO2 did not significantly increase the flame retardancy performance of chitosan, probably due to the low amount (Table 4 and Figure 8). The amount of nano-TiO2 used in our study is 0.05% by weight. The ratio of nano-TiO2 used in cotton textile products varied between 0.2%-0.4% 63 and it was used between 3% - 9% by weight in another study. 64 Chitosan alone in providing flame retardancy to the fabric was sufficient. This is an important result in terms of reducing the use of chemicals in textile products. In addition, since chitosan is a recyclable biomaterial, its use provides benefit for the human and environment health.
Comfort properties of chitosan coated fabrics
Air permeability
The air permeability values of the uncoated and coated denim fabrics are shown in Table 4. The highest air permeability value was measured as 1552 mm/s for the uncoated raw cotton fabric, which has a weight of 105 g/m2, a weft density of 12 and a warp density of 18.The air permeability value of the uncoated denim fabric, with a weight of 368 g/m2, weft density of 16 and warp density of 27, was measured as 586 mm/s. This shows that the basic structural properties of the fabrics, such as weft density, warp density and weight are effective on air-permeability values. The increases in the density and weight cause a decrease in the gaps between the yarns, and therefore, a decrease in the air permeability value of this fabrics. This is similar to the results presented by. 65 Therefore, when a lower permeability value is desired, fabric density values as high as the conditions of use allow should be selected.
Air permeability analysis results for coated and uncoated fabrics.

Literature data also support the results of this study. Şahan and Demir 66 observed that chitosan molecules accumulate on the surface instead of penetrating into the fiber and block the pores through which air can pass. For this reason, the air permeability of the fabric gradually decreased depending on the thickness of the coating layer. 67 Another study revealed that the air permeability of fabrics decreased gradually depending on the amount of binder and other chemicals applied during coating and the increase in the concentration of nano-TiO2. 31 Nowadays, manufacturers give air permeability values between 0 and 51 mm/s for commercial products that provide a barrier effect against wind and weather conditions. 68 After coating the denim fabrics with crayfish chitosan and nano-TiO2, the air permeability of the fabrics decreased by 90% and reached the capacity to compete with wind proof commercial products. The results show that the degree of air permeability can be adjusted to the desired level in denim fabrics only using crayfish chitosan or crayfish chitosan and nano-TiO2 coatings, depending on the place of use. This study also revealed that coatings made with crayfish chitosan with a porous structure (Figure 3) promoted the breathability of denim fabrics.
Water permeability
Water permeability measurement results of fabrics.

In this study, the addition of nano-TiO2 to chitosan coatings decreased the water permeability of the calico fabric. If moisture vapour does not freely pass through the fabric, it causes we at accumulation and discomfort by preventing the body sheat dissipation. 31 For this reason, it is important to develop coatings that will provide moisture balance between fabric and skin. As seen in Table 6, chitosan and chitosan+nano-TiO2 coatings reduced the water permeability of the fabric but did not completely prevent it. In coatings made with chitosan, a porous hydrophilic layer is formed on the fabric surface thanks to the porous structure of chitosan (Figure 3).For this reason, we believe that chitosan coatings can provide both waterproof and breathable properties to calico and denim fabrics. In addition, we believe that the moisture balance between the clothing and the skin will be preserved thanks to these features of the chitosan coating.
The clothes that are desired to be reduced their water and air permeability, are generally formed from multi-layered or thick coatings. This not only increases the weight, but also thickens the composite structure and threatens its water vapour permeability. Therefore, some industrial protective clothing is heavy, bulky, movement restrictive and uncomfortable. In this study, while the comfort requirements are met at the minimum level in semi-permeable fabrics with chitosan coating, advantages such as ease of applied to ready-made clothing with a thin coating layer and lightness with a single layer coating can be achieved. Due to its advantages, such as lightness and thin coating, chitosan coatings can be used in the production of garments used in activities that require high performance.
Conclusion
Fourier transform infrared, SEM, and DSC demonstrated successful isolation of fibrous and porous crayfish chitosan. The crayfish chitosan coating slightly increased the weight of the green calico fabric (24.76%) and the blue dyed denim fabric (5.19%), and it also increased the tear strength of the green calico fabric by 20% in the weft direction.The elemental composition of crayfish chitosan and nano-TiO2-coated raw denim fabric was 44.83% carbon, 1.19% nitrogen, 53.91% oxygen and 0.08% titanium by weight. The maximum decomposition temperature of the undyed raw denim fabric (340°C) decreased to 310°C after coated with chitosan due to the cooling effect of the chitosan coating. The maximum thermal decomposition temperature of the chitosan+nanoTiO2 coated fabric was 302°C and the residual amount was 34.0%, showing the synergic effect of chitosan and chitosan+nanoTiO2. The burning time with the ember of the dyed calico fabric (8.66s) decreased to 5.6s after being coated with chitosan. The burning time with the ember was 49 s for the uncoated blue dyed denim. There was no burning with ember after coating. The vertical burning test showed that the chitosan coating increased the charcoal forming characteristic of cotton fabric with combustion and significantly improved the self-extinguishing property of denim fabrics. Chitosan coating provided semi-permeable textile properties by decreasing the water and air permeability, of denim and calico fabrics. We believe that fabrics with improved non-flammability and comfort properties using crayfish chitosan coatings can be used for the production of special-purpose innovative smart clothes ranging from fabric shoes, socks and even gloves.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Trakya University Scientific Research Projects Unit (Project no: TÜBAP-2018/198).
