Abstract
This work aims to obtain reinforced composites of natural fibers that obtained from their agricultural wastes of and conductive polymers to develop an innovation and alternative materials. By the use of natural fibers contributes to the recycle of agricultural wastes, sustainability and further the resulting composite becomes alternative to the metals. Here, flexible conductive composites were obtained from artichoke(A), banana(B) and luffa(L) stem waste fibers(F) by the in-situ polymerization of 3,4-ethylene dioxythiophene (EDOT), pyrrole, and carbazole in the presence of cerium ammonium nitrate, iron nitrate, and iron chloride. Fibers were coated with the conductive polymers mentioned above by the in-situ chemical(C) polymerization and optimum coating conditions were investigated. Effect of EDOT concentration, oxidant concentration was performed to determine the optimum conditions for AF/PEDOT(C). FT-IR, SEM, thermal analysis supported the formation of composite and from the mechanical measurements, modulus of AF/PEDOT(C) was obtained. The highest conductivity of 12.8 S/cm was obtained from AF/PEDOT(C) composite using FeCl3 as an oxidant. Further polymerization of EDOT by electrochemical(E) method was continued on the AF/PEDOT(C) and the electroactivity of resulting electrochemical composite, AF/PEDOT(C)/PEDOT(E) was characterized accordingly. Detailed characterization showed that to use of this composite as a capacitor, one should use 0.03 M EDOT and 0.9 M FeCl3 for chemical polymerization and then continued by electropolymerization by applying 10 cycles in 0.03 M EDOT. All results showed that AF waste could be converted to the valuable AF/PEDOT(C)/PEDOT(E) conductive composites which is potentially suitable material for several electronic applications as charge storage, biosensor, electronic devices.
Introduction
Nowadays, due to the increasing of the environmental concerns and decreasing resources, the use of fiber reinforced composites is important that they impart recyclability, biodegradability to the material and also CO2 neutrality. Conductive polymers (CPs) have occupied very large area in our daily life such as artificial muscles, anticorrosive dyes, biosensors, intelligent textiles, microelectronic devices, rechargeable cells, photovoltaic cells, etc. 1 CPs permit excellent control of the electrical actuator, have great electrical, a high conductivity related to weight ratio, optical properties, and considered biocompatible and eco-friendly. 2 There are many attractive conducting polymers that have been developed since the late 1980s, those based on poly (3,4-ethylene dioxythiophene) (PEDOT), 3 polyaniline (PANI), 4 polypyrrole (PPy), polyindole, 5 polythiophene, 6 polycarbazole (PCz) 7 and their derivatives have attracted a great attention. 8 Of these, the multifunctional PEDOT stands out due to its intrinsic stability, and its capability to obtain processable conductive structures. The expanding significance of PEDOT, can be easily figured out by the various applications in capacitors, natural light transmitting diodes, batteries, sun-based cells photovoltaics, etc.9,10
Since the beginning of the 21st century, there is an increased effort to recycle lignocellulosic natural fibers (NFs). On a smaller level, natural fibers are typically composed of macromolecules or polymers, which are able to pack closely to each other resulting in regions of crystallinity. The crystalline regions provide strength and rigidity to the fibers, while the flexibility and reactivity are related to amorphous regions. The crystalline/amorphous ratio material has an important influence on the properties of the fibers. Traditional NFs, such as wool, cotton and silk, have tenacities. Nevertheless, fibers such as hemp, jute, flax and ramie may have higher strength and stiffness. With the exemption of silk, these are all short fibers, and this prevents the conversion efficiency of the fiber’s strength into yarns and textures. NFs are used in numerous structural engineering applications including bridges, constructions, long-span roof, and thermal insulators. 11
Less investigated NFs can also be assumed as a source of cellulose aimed to enhance the green property of the final composites, thus forcing the industry to incorporate them in such eco-friendly materials.
Artichoke (Cynara scolymus L.) is a consumable vegetable which is a member of Asteraceae family. Artichoke stem is waste product and its disposal is difficult. For this reason, the extraction of fibers from the stem of the native artichoke plant represents an evident environmental advantage. Artichoke fiber (AF) shows a lignin content (4.3%) similar to other fibers such as hemp and flax, whereas good mechanical properties with the help of its high cellulose content (75.3%), thus confirming its feasibility as reinforcement of polymer-based composites.12–15
In the past, NFs were used in constructing applications. Nowadays, NF have become valuable alternative elements to reinforce materials in the composite industry and have taken great interest in recent years because they allow the possibility of obtaining a new hybrid material for different applications.16–21
Wide range of NFs has been used to reinforce different polymer matrices. Such fibers contain wood, bamboo, cotton, coir, wheat straw, flax, jute, pineapple leaf, ramie, oil palm, sisal, kenaf, hemp etc. A lot of factors might influence the resulted NFs composites and can determine their electrical, mechanical, biological characteristics. 22 In spite reinforcing composites with NFs might result in lower mechanical properties in comparison to their traditional counterparts (e.g. carbon fibers and/or glass fiber reinforced composites) NFs reinforced composites achieve significant value because they decrease feed stock material cost and increase recyclability, biodegradability, thermal insulation, and CO2 neutrality. 23 Numerous NFs such as luffa fiber (LF), silk, kenaf, jute, is being used to reinforce thermoset polymers (Polyester, Polyamide, Polyurethane, and thermoplastics (polyethylene, polypropylene, polycarbonate, polyvinyl chloride, acrylonitrile-butadiene-styrene) polymers and elastomers. 24
On the other hand, combining NFs with CPs for lateral use in polymeric matrices to produce NF/CP composites expands the applicability of NFs reinforced composites even more. These interesting features allow such composites to be used in terminals, switches, printed circuit boards, connectors, insulators, industrial and house hold plugs, etc. 25 CP coated NFs have been generally prepared using in situ oxidative procedures or electrochemical methods. The need to develop flexible and wearable electronics in many areas has promoted the development of highly flexible energy storage devices with high performance. The use of NFs as templates in the development of conducting composites is important for all the previously mentioned reason since the presence of the hydroxyl groups in their structure can induce chemical bonding with the functional groups of CPs. Conductivity values of the silk/PEDOT-PSS, 26 CP/silk fiber (SF), PPy/SF, PANI/SF and PEDOT/SF, 27 PP/luffa (LF), 28 PPy/banana fiber (BF), 29 PANI/Coconut, 30 PPy/peach palm fibers, 31 PPy/cotton fabric 32 are relatively similar to pure CPs which supply NFs with useful characteristic without sacrificing the conductivity gained from CPs. The highest conductivity was reported with the PEDOT/silk fiber composite as 0.38 S/cm. 27 However, relatively less research has been reported on the utilization of agricultural wastes of AF as reinforcement in these polymer composites.
Although being an excellent property as mentioned above, however, NF cannot used as an electrode without modification with conductive materials. CPs were one of the best materials for this purpose. CPs and natural fibers can be produced at low costs and are environment friendly and biodegradable. The increasing demand for electronic materials which efficient and having a potential to be more sustainable in various industries can be met by the development of polymer composites with NFs and CPs. As devices get smaller in size, components can be able to store large amounts of energy while occupying the least amount of space in the circuit. To our knowledge, very little work has been done so far on the utility of CP for modifying the NF fiber.
This work aims to obtain reinforced composites of CP and NF by polymerization of carbazole (Cz), EDOT and pyrrole (Py) on natural AF, LF and BF obtained from their agricultural wastes. CP (PEDOT, PPy, and PCz) used to obtain different flexible conductive composites and those NF/CP composites were referred to as AF/PEDOT(C), AF/PPy(C), AF/PCz(C), LF/PEDOT(C), LF/PPy(C), LF/PCz(C), BF/PEDOT(C), BF/PPy(C) and BF/PCz(C) for chemical polymerization where the letter “C” represents “chemical preparation”. To obtain AF/PEDOT(C), cerium ammonium nitrate (CAN), iron nitrate (Fe(NO3)2) and iron chloride (FeCl3) were used as oxidants and FeCl3 was found the suitable one. The conductivities of NF/CP composites were measured and compared. Effect of EDOT concentration, oxidant concentration and type were performed to determine the optimum conditions for AF/PEDOT(C). Further polymerization of EDOT by electrochemical method was continued on the AF/PEDOT(C) and the electroactivity of resulting electrochemical composite, AF/PEDOT(C)/PEDOT(E) was investigated, here the letter “E” represents “electrochemical preparation”.
Experimental
Materials
Acetonitrile (ACN, HPLC grade with water content <0.005%), propylene carbonate (PC), sodium dodecylbenzene sulfonate (SDBS), iron (III) chloride (FeCl3), cerium ammonium nitrate (CAN), iron nitrate nanohydrate Fe(NO3)3.9H2O, lithium perchlorate LiClO4, 3,4-ethylenedioxythiophene (EDOT), carbazole (Cz), pyrrole (Py). All chemicals were received from Sigma-Aldrich reagent grade chemicals of the highest purity and used without further purification.
Cotton fiber (CF), LF, AF, BF fibers were obtained from their wastes, separated and cut according to their diameter33,34 which the values of ∼14.5 μm, 22 ∼200 μm, 35 ∼202.1 μm 13 and ∼182.1 μm 16 respectively and pre-treatments were carried out as described in the following section.
Experimental details
AF and BF were obtained from their wastes from Aegean and Mediterranean Regions of Turkey respectively by separating from plant extracts with the aid of a machine (Registration No 2010 08,487 by Turkish Patent Institute) designed to obtain fiber from lignocellulosic plants. It was waited 20 days in the water-filled container to remove residue and adhering dirt. Appearance of conditioned fibers were shown in Figure 1(a) to (d) respectively. The fibers were shaken under running water until cleaned and they were dried in an oven 70°C for 6 h. After drying, they were conditioned 48 h prior to testing under ±20°C and 65 ± 2 RH% condition as suggested in literature.36–38 Appearance of conditioned fibers of (a) cotton (b) artichoke (c) luffa (d) banana.
LF is a tropical creeping annual plant and it grows well in the areas of the Mediterranean climate, in Turkey. The lengths of fiber were between 400 mm and 600 mm. The LFs were left in water to remove the adhering dirt. (200 C distilled water for 0.5 h) They were dried in an oven 70°C for 6 h. After drying, they were conditioned 48 h prior to testing under ±20°C and 65 ± 2 RH% condition.36–38 CF is a plant derived natural seed and it was obtained from cotton waste 24 with similar to other fibers.
Morphology of the bare and the coated fibers were analyzed by scanning electron microscopy (SEM) model JEOL 7500F with working distance of 10 mm and high voltage of 15 kV.
Chemical groups of the bare and coated NFs were detected by using ATR-FTIR spectroscopy (JASCO 5300) between 500 cm−1 and 4000 cm−1 wavenumbers.
Thermal properties were measured by differential scanning calorimetry (DSC) (7020 SEIKO), the temperature was ramped between 25°C and 350°C under N2 gas with a scanning rate of 10 K/minute. Thermal stability was examined by thermal gravity analysis (TGA) by Netzsch STA 409 PC Luxx.
Mechanical properties of the NF’s before and after coating with CP were tested under tensile stress. Due to the difficulty of controlling the NF geometry, more than 20 samples were tested; at least four samples of AF/PEDOT(C) fiber composites were tested according to the ASTM D3822 standard 39 using Zwick Roell Z010 with 50 kN load cells and average values of four identical specimens were calculated. The highest and lowest values are discarded and the average of the remaining results are taken as the tensile modulus. The cross-sectional area was evaluated from the diameter measured using a digital caliper at five different locations along each sample length, (Supplemental Figure S1) and supported by using Projectina CH-9495 microscope.
Preparation of natural fiber/CP composites
Chemical coating of natural fibers
Since the NFs (BF, LF, AF) are insulator, the electrochemical method cannot be used for coating NF surface with CP and conductivity on the NF surface should be increased to a value enough to conduct electricity. For this reason, first, the chemical polymerization was performed.
There are several methods for preparation of chemical coating and some of them coated the fiber by first dipping it in the monomer solution and then in the oxidant solution. 40 Other method was reported by the slow addition of oxidant to monomer solution that contained fiber. 29 The purpose of all methods is slow down the polymerization reaction in solution, instead, to obtain completely CP coated fiber surface. Here, in this study the former method was applied by some modification since after several try it seemed more effective for coating the NF. Several concentrations were used for both monomer and oxidant and optimum concentrations were determined by using multiple successive dipping of the fiber into monomer and oxidant solutions. When the conductivity of resulting NF/CP coating compared with literature,29,41 it seems similar or higher conductivities can be obtained by the aid of multiple successive dipping method that ensure a complete coating.
During pre-treatment of fibers, SDBS was used as a surfactant to increase surface area and it was added in solution I (0.02 M SDBS +15 mL water) and II (0.02 M SDBS +15 mL ACN). ACN used as a solvent and FeCl3 as oxidizing agent. After this pre-treatment by immersing fibers in solutions I and II for 30 min each, chemical polymerization was performed in separate solutions namely, solution III (0.01 M Monomer +15 mL ACN) and IV (0.6 M FeCl3 + 15 mL ACN). Since the polymerization reaction is fast, in order to increase the yield of CP obtained on NF surface instead of solution, the polymerization was carried out by immersing the fibers in solutions III and IV several times successively. During this process, NFs were left in solution III for 1 h, lastly it was put in solution IV for 10 min and then again in solution III for 10 min. The sequence of immersing in III and IV is repeated for 6 times. A stable continuous stirring at 200 rpm at room temperature conditions were maintained during the whole mentioned steps. Following the procedure, NF was coated with CP and AF/PEDOT(C), AF/PPy(C), AF/PCz(C), LF/PEDOT(C), LF/PPy(C), LF/PCz(C), BF/PEDOT(C), BF/PPy(C) and BF/PCz(C) composites were obtained. They were washed with ACN, dried at room temperature, were weighted as required amount to obtain their pellets and conductivities were measured. Schematic illustration of the NF/PEDOT(C)s preparation, their pellets and NF/PEDOT composite images were shown in Scheme 1. Schematic illustration of the preparation of AF/PEDOT(C) by chemical polymerization, its pellet and images of (a) AF/PEDOT, (b) BF/PEDOT, (c) CF/PEDOT, (d) LF/PEDOT.
For the conductivity measurements, pellets with the diameter of 13 mm were prepared by using a manual laboratory compact pellet press (Scheme 1), with built in hydraulic pump each pellet was pressed up to 10 tonnes pressure for 5 min and the conductivity measurements were taken by Keithley 617 electrometer connected to a four-probe head with gold tip. The current of the sample as pellet form at certain potential was measured and conductivity was calculated using the equation below
Electrochemical coating of NFs
Images of the composite preparation by chemical polymerization were given in Supplemental Figure S2 and according to the conductivity results of chemically prepared composites, the best one was determined as AF/PEDOT (C, 0.03 M EDOT). In order to increase the conductivity and improve the redox behaviour of this composite, it was coated with PEDOT in the presence of 0.01 M and 0.03 M EDOT, in the solution of ACN: PC (9.5: 0.5) containing 0.1 M LiClO4 by potentiodynamic method (Cyclic voltammetry, CV) at a scan rate of 50 mV.s−1. The quasi-reference Ag wire was calibrated using a ferrocene/ferrocenium (Fc/Fc+) couple and the potentials are reported versus an Ag/AgCl reference electrode. Pt wire was used as a counter electrode.
The electrochemical behavior of the NF/CP(C) composite fibers was investigated by CV in a three electrodes cell and they were coated with CPs to obtain NF/CP(C)/CP(E).
To protect the NF electrodes (bare and coated) from getting fully wet upon immersing in the electrolyte solution; each electrode was connected to a copper plate and then to the clamp/crocodile of the potentiostat. Details for preparation of electrodes was given in Supplemental Figure S3 and S4.
Results and discussion
The effect of monomer and oxidant types and concentrations on the properties of resulting composites were investigated. Conductivity, FT-IR, SEM, thermal analysis, and mechanical measurements performed for the characterizations of composites. Results were given below.
The effect of monomer and NF types, on the conductivities of composites
The conductivities of NF/CP obtained by using 0.03 M monomer and 0.6 M FeCl3 were measured and results were given in Figure 2. As it can be seen, AF/PEDOT(C) has the highest conductivity among them. It is expected that the conductivity of PEDOT is higher than the conductivities of PCz and PPy.
42
Further investigations were continued with AF/PEDOT(C). The conductivities of AF/PEDOT(C), AF/PPy(C), AF/PCz(C), LF/PEDOT(C), LF/PPy(C), LF/PCz(C), BF/PEDOT(C), BF/PPy (C) and BF/PCz(C) obtained by using 0.03 M monomer and 0.6 M FeCl3.
The effect of the oxidant type on the conductivities of composites
Figure 3 shows the conductivities of the three different AF/PEDOT(C) prepared by using 0.9 M Fe(NO3)3.9H2O, CAN and FeCl3 as oxidants. The highest conductivity was obtained in the presence of FeCl3. Since the oxidation potential of FeCl3 is lower than that of CAN, the polymerization reaction is slower and for this reason, the amount of PEDOT coated on to the fiber surface is probably more than the amount of polymer in the solution.43–45 On the other hand, -Cl- behaves as a better dopant than-NO3-, and resulted in higher conductivities in the case of FeCl3 as suggested in literature
46
and it was selected as an oxidant for further experiments. Conductivities of AF/PEDOT(C) obtained with 0.9 M of Fe(NO3)3.9H2O, CAN and FeCl3.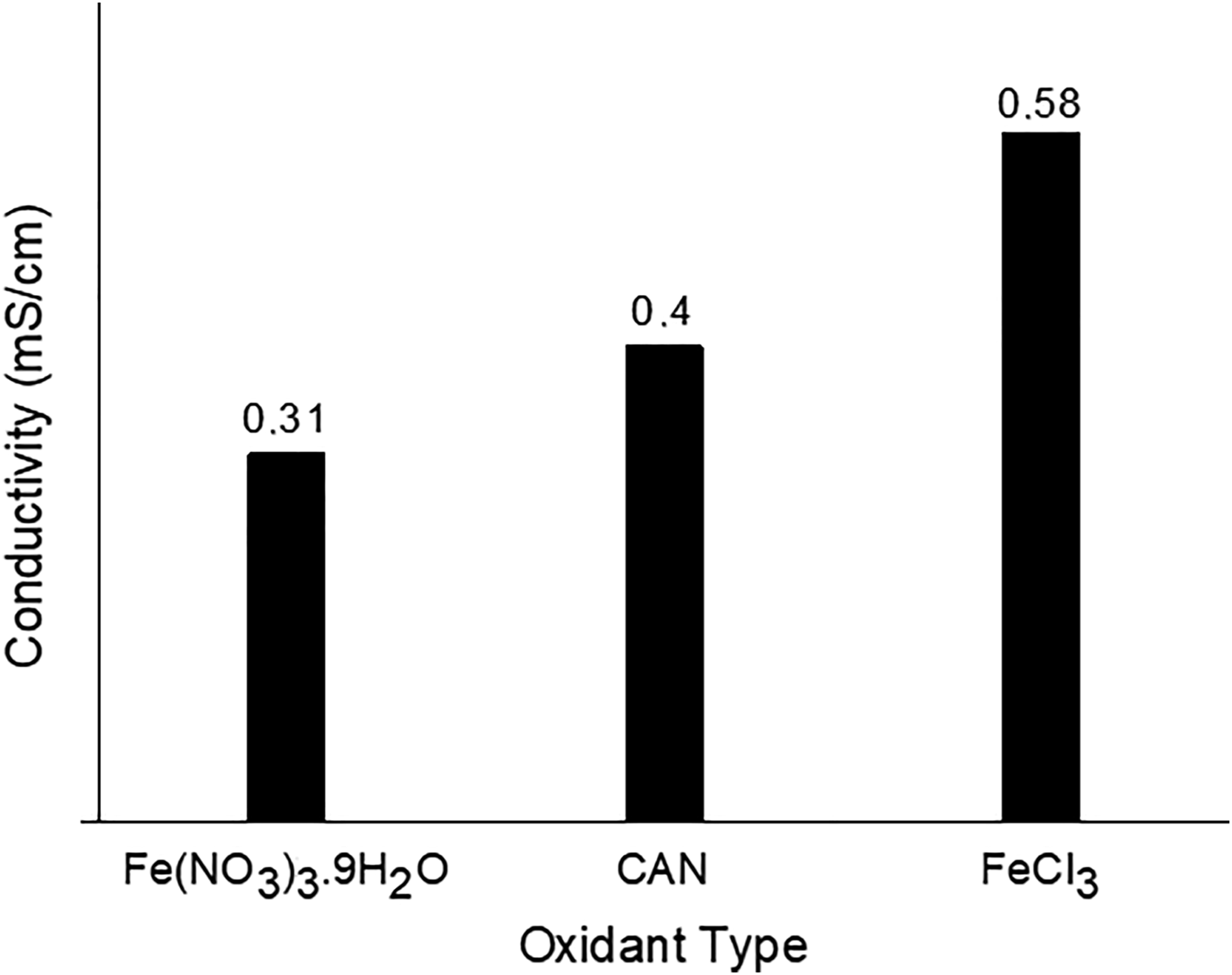
The effect of the oxidant concentration
After finding the best oxidant type, it is important to specify the most efficient concentration of FeCl3, in order to do the polymerization process in shorter time with the best results. Conductivities of five different AF/PEDOT(C) composites were compared in Figure 4. The conductivity was the highest when 0.6 M FeCl3 was used and the film was stable to provide enough electron flow to light the LED lamp continuously (Supplemental Figure S5). Conductivities of AF/PEDOT(C) prepared with different concentrations of FeCl3.
At the higher concentrations of oxidant cause side reactions and/or excessive oxidation as reported in literature 47 that makes the chains crosslink. In addition, the polymerization possibility of EDOT in solution instead of on the AF surface taken place that resulted in the decrease of conductivity. On the other hand, although the conductivity of AF/PEDOT(C) composites increased with concentration up to 0.6 M FeCl3, 0.3 M was determined as optimum concentration since higher FeCl3 concentrations caused thicker and unstable deposition of PEDOT on the AF surface.
The effect of the monomer concentration
To determine the efficient monomer concentration, AF/PEDOT(C) composites were prepared by using different concentrations of EDOT and results were given in Figure 5. The best conductivity was obtained in the case of 0.03 M EDOT, at higher concentrations, the resulting PEDOT film delaminated from the surface as suggested in literature
48
and 0.03 M was selected as the concentration to obtain stable film with high conductivity. Conductivities of AF/PEDOT(C) obtained at different concentrations of EDOT.
FT-IR Results of AF/PEDOT(C)
ATR-FTIR results of AF, PEDOT and AF/PEDOT(C) were represented in Supplemental Figure S6. In the spectrum of AF, the characteristic bands were located at 3300 cm−1, 2900 cm−1, 1737 cm−1, 1422 cm−1 and 1000 cm−1 which are attributed to -O-H stretching vibration, -C-H stretching vibration, -C=O stretching vibration, -CH2 symmetric bending and -C–O stretching vibration respectively and they are in agreement with literature.49,50
The disappearance of the -O-H and -C-H bands in the NF structure in the FTIR spectrum of resulting product and the observation of characteristic -C-S-C vibrations of thiophene (Th) ring in PEDOT at 676 cm-1 and 823 cm-1 supports the formation of the composite.
As it can be seen, the peak at 2347 cm−1 in the spectrum of AF that correspond the -C-H stretching vibrations was not observed in the spectrum of PEDOT and has disappeared in the spectrum of AF/PEDOT(C) indicated that there was a chemical bonding between the AF and PEDOT.
The broad peak in the range of 1600–2300 cm−1 in the spectrum of AF, becomes a sharp peak at 1600 cm−1 in the spectrum of AF/PEDOT (C), similar to the spectrum of PEDOT and proves the formation of the composite.
In addition, the peak of the dopant anion (Cl−) observed in the spectrum of PEDOT at 1032 cm−1 increases as the amount of oxidant increases in the spectrum of the AF/PEDOT(C). This result indicated the increase of the PEDOT content in the composite.
When the EDOT concentration used during polymerization increases from 0.01 M to 0.05 M during the chemical polymerization, resulted the systematic increase in the intensity of all peaks in the spectrum of AF/PEDOT(C) as expected. In addition, when the concentration was further increased to 0.06 M, the intensity of the peaks of -OH at 2972 cm−1, -C-H at 2926 cm−1 and -C = O at 1603 cm−1 of AF decreased, while peak that correspond the dopant anion of PEDOT at 1032 cm−1 increased. These results were supported the formation of AF/PEDOT(C).
Morphology of AF/PEDOT(C)
Properties of the coated films depends on the morphology and in order to gain information from the surface properties of the film, SEM images were taken. SEM images of AF, PEDOT powder and AF/PEDOT(C) surfaces were examined comparatively and given in Figure 6 and Supplemental Figure S7. Smooth surface of AF (Figure 6(a)) as compared to porous structure of PEDOT powder (Figure 6(b)) having similar morphology with the literature51,52 was clearly seen. Observation of similar porous structure on AF/PEDOT(C) surface (Figure 6(c)) confirmed that a good coverage of PEDOT on AF surface. SEM images of the (a) bare AF 500x magnification, (b) PEDOT(C) powder 4000x magnification and (c) AF/PEDOT(C), 500x magnification.
Thermal Stability of AF/PEDOT (C)
TGA curves were analyzed in order to understand the effect of two different EDOT concentrations on the thermal properties of PEDOT/AF(C) (Supplemental Figure S8). The degradation profiles of AF and PEDOT/AF(C) showed four weight loss steps. In the first step below 100°C weight loss occurs due to moisture evaporation from the fiber structure. In the range of 250–360°C the following thermal decomposition step, that corresponds to the weight loss mainly caused by the decomposition of hemicellulose and cellulose was observed as reported in previous studies on hemp fibers. 53 The third step ranging from 360 to 520°C belongs to decomposition of lignin. At higher temperatures than 520°C, the fibers are thoroughly degraded so that the residual mass remained unchanged with increasing temperature.
Comparison of the thermal analysis curves of AF and AF/PEDOT(C) showed that decomposition temperatures shifted to lower values in the case of composites due to effect of thermal properties of PEDOT. 54 As suggested in this literature the major thermal decomposition of PEDOT occurred from 140–330°C and there was around 40 wt% carbon left after heating to 500°C which supported the observation of lower decomposition temperature of AF/PEDOT(C). It was also observed that EDOT concentration did not significantly affect the thermal properties.
DSC measurements was performed to record the glass transition temperature of PEDOT and the AF/PEDOT(C). Results suggested that Tg value (64°C) of PEDOT as reported in literature 41 increased to the value of 76.2°C in the case of AF/PEDOT(C). The incorporation of PEDOT, having higher stiffness than AF caused an increase the Tg.
Mechanical analysis
Samples for mechanical measurements were prepared according to ASTM D3822 Standard Test Method for Tensile Properties of Single Textile Fibers (Supplemental Figure S9 and S10).
There are many properties affecting mechanical behaviour of composites. Although the same coating process applied during the deposition of polymer film on fiber surface, some deviations might observe for different samples. The stress-strength test was carried out for four identical specimens and the average values were taken and the stress-strain curves for bare AF and AF/PEDOT(C) were given in Figure 7. Tensile strength was calculated from the load-elongation data and cross-sectional area of fibers. Young’s modulus data were given in Supplemental Table S1. The cross-sectional area, assuming that the fibers are cylindrical in shape, was evaluated from the diameter measured using a caliper at five different locations along each sample length. Stress-strain curves for bare AF (…) and AF/PEDOT(C) (─).
Results for four different samples were summarized in Supplemental Table S1. Differences might cause random deposition of PEDOT on fiber surface since it can be significantly change place to place. Although results were suggested that composite formation has not significantly effect on the mechanical properties of natural fiber, AF/PEDOT(C) showed more stiff-plastic characteristic than elastomer like untreated fiber 55 as expected due to inclusion of the rigid properties of PEDOT. 56 A similar effect, in literature, was reported for the tensile properties of PANI coated polyethyleneterephlatate (PET) that is better than uncoated PET 57 and was explained by filling the blanks on PET with the PANI particles. Here the same effect was occurred in the case of blanks on AF which was filled with the PEDOT.
Electrochemical deposition of EDOT on the AF/PEDOT(C)
AF/PEDOT(C) was used as working electrode in a three electrodes cell as shown in Figure 8 and coated electrochemically with PEDOT. Resulting composite was called as AF/PEDOT(C)/PEDOT(E). For these experiments, working electrode AF/PEDOT(C) obtained by using 0.3 M FeCl3 having 2 cm2 length attached to a copper plate having 2 mm2 surface area by clamping (Figure 8). The counter electrode was Pt wire and the reference electrode was a silver wire. Three electrodes consisting electrochemical system and Cu tape/AF/PEDOT(C) as a working electrode.
Electrochemical measurements were done in a mixture of ACN:PC with the ratio (9.5:0.5) that contains 0.1 M LiClO4 in the range of −0.7 V–1.85 V, by applying 10 cycles at a scan rate of 50 mV/s. Results obtained during electropolymerization and characterization of the AF/PEDOT(C)/PEDOT(E) were given in the following sections.
Electrochemical coating of AF/PEDOT(C) with PEDOT
In order to increase the conductivity of AF/PEDOT (C, 0.03 M EDOT) and improve redox behavior, it was coated with PEDOT in the presence of 0.01 M and 0.03 M EDOT, in the solution of ACN: PC (9.5: 0.5) containing 0.1 M LiClO4 by potentiodynamic method, at a scan rate of 50 mV.s−1.
In order to investigate the redox behavior of AF/PEDOT(C), the first cycle was obtained in the absence of EDOT (Figure 9) and further cycling was continued in the presence of 0.01 M EDOT. As the number of cycles increases, the peak potential shifted from 0.5 V to 0.8 V and current intensities increased. This result showed the growing of PEDOT on the AF/PEDOT (C) electrode surface. CV of AF/PEDOT (C, 0.03 M EDOT) in ACN:PC (9.5:0.5) solution containing 0.01 M EDOT, 0.1 M LiClO4 with a scan rate of 50 mV s−1.
The polymerization was repeated by using 0.03 M EDOT and as it can be seen, higher current intensities were observed than the case of 0.01 M EDOT which resulted better electroactivity of PEDOT film on the AF/PEDOT(C) (Figure 10(a)). Comparison of CV’s obtained during PG’s on AF/PEDOT(C, 0.03) (0.01 M EDOT) and AF/PEDOT(C, 0.03) (0.03 M EDOT) (a) CV’s of resulting AF/ PEDOT(C)/PEDOT(E,0.01) and AF/PEDOT(C)/PEDOT(E, 0.03) (b) in ACN:PC (9.5:0.5) solution containing 0.1 M LiClO4 with a scan rate of 100 mVs−1.
Comparison of the CV’s of AF/PEDOT(C)/PEDOT(E) obtained at 0.01 M and 0.03 M EDOT in monomer-free electrolyte were given in Figure 10(b).
An irreversibly redox behavior of AF/PEDOT(C)/PEDOT(E) in CVs might be due to the insulating property of AF which had been observed similarly in the literature. 58 While a rectangular shape was observed for CV of PEDOT when it was coated on SWCNT, the redox behavior of PEDOT coated jute was irreversible.
Since the current intensities were higher during polymer growth and redox behavior of AF/PEDOT(C)/PEDOT(E) was better in monomer free solution, 0.03 M EDOT was selected as optimum EDOT concentration for electropolymerization.
Effect of oxidant concentration used during the chemical polymerization on the Redox behavior of AF/PEDOT(C)/PEDOT(E)
The conductivity of the chemically obtained composite will affect the electropolymerization reaction and the properties of the obtained composite. For this reason, the effect of FeCI3 concentrations used to obtain AF/PEDOT(C) on the redox behaviors of the resulting AF/PEDOT(C)/PEDOT(E) was investigated by comparing the CVs obtained at each FeCI3 concentration (Figure 11). During electrochemical polymerization of AF/PEDOT(C), 0.03 M EDOT was used in ACN:PC (9.5:0.5) containing 0.1 M LiClO4. As it can be seen, increase in FeCI3 concentration, current intensity of the AF/PEDOT(C)/PEDOT(E) increased up to 0.9 M FeCI3 used to obtained AF/PEDOT(C) and further increase in oxidant concentration might cause the degradation or over oxidation of the PEDOT coating and electroactivity diminished. It seems AF/PEDOT(C) obtained by using 0.9 M FeCI3 was optimum composite to use as working electrode for electropolymerization. The effect of different FeCl3 concentrations (0.3, 0.6, 0.75, 0.9, and 1.2 M) for different AF/PEDOT (C, [EDOT] = 0.01 M), on the CVs of AF/PEDOT(C)/PEDOT(E) in ACN:PC (9.5:0.5) containing 0.1 M LiClO4 at a scan rate of 100 mV.s−1.
EIS Results
Effect of monomer and oxidant concentrations on the capacitive behavior of AF/PEDOT(C)/PEDOT(E) was investigated by EIS measurements to determine the charge-transfer resistance as indicated by the diameter of the semicircle in the Nyquist plot 40 and results were given below.
Effect of monomer concentration
The effect of EDOT concentrations (0.01, 0.02 and 0.03 M) used during the coating of AF/PEDOT(C) with PEDOT, on the capacitive property of AF/PEDOT(C)/PEDOT (E, 0.01 M EDOT) was tested by EIS measurements in the frequency range of 10 Hz - 1 MHz. Nyquist graphs obtained from these measurements at open circuit potential were compared (Figure 12). The effect of EDOT concentrations used during chemical polymerization on the Nyquist graphs of AF/PEDOT (C; 0.01 M EDOT)/PEDOT (E; 0.01 M EDOT), AF/PEDOT (C; 0.02 M EDOT))/PEDOT (E; 0.01 M EDOT), and AF/PEDOT (C; 0.03 M EDOT))/PEDOT (E; 0.01 M EDOT).
As it can be seen, concentration of EDOT can effectively increase the impedance values and the AF/PEDOT (C, 0.03 M)/PEDOT (E, 0.01 M) has the higher impedance values than the other composites obtained other concentrations (C, 0.01 M and C, 0.02 M). This might be due to the result of the positive PEDOT sites with a higher amount in the doped polymer, during chemical polymerization in the case of 0.03 M, which resulted increase in the charge transfer resistance of the resulted coating.
Effect of oxidant concentration
The effect of FeCI3 concentrations used when obtaining AF/PEDOT(C) on the capacitive properties of the resulting AF/PEDOT(C)/PEDOT(E) was determined by comparing the Nyquist plots at open circuit potential of the composites obtained at each concentration (Figure 13). Inset was given an enlarged view of the AF/PEDOT(C)/PEDOT(E) at high frequency. The effect of FeCl3 concentrations on the Nyquist graphs of AF/PEDOT(C)/PEDOT (E; 0.01 M EDOT).
The bulk-resistive properties of the electrolyte and electrode (Rs), can be obtained by the intersection of the curve with the high frequency region of the x-axis and Rs values of all composites were close to each other. In the case of AF/PEDOT(C)/PEDOT(E) obtain at 0.9 M FeCI3, the diameter of the semicircle in the medium frequency which represents the interfacial charge transfer resistance (Rct) was found the smallest. This might be due to better electroactivity of this composite as shown in Figure 11.
At low frequencies region, low frequency specific capacitance (CLF) value give important information on the capacitive behavior of composite. For this reason, CLF values of AF/PEDOT(C)/PEDOT(E) composites were calculated by using the equation (2) and changes of the CLF values of AF/PEDOT(C)/PEDOT(E) composites obtained by using different FeCI3 concentrations with frequency was given in Figure 14. CLF values decrease as the frequency increases, as expected according to equation (2). The highest CLF values are obtained when 0.9 M FeCl3 is used in the frequency range of 1–100 Hz. Between 100 Hz and 10,000 Hz, the highest CLF values are obtained for the case where 0.75 M FeCl3 is used to obtained AF/PEDOT(C). Changes of the CLF values of AF/PEDOT(C)/PEDOT(E) composites obtained by using different FeCI3 concentrations with frequency.
The Csp values of AF/PEDOT(C)/PEDOT(E) composites obtained by using different FeCI3 concentrations.

By evaluating conductivity, CV, and EIS data, the optimum condition was chosen as AF/PEDOT (C; 0.03 M EDOT and 0.9 M FeCI3)/PEDOT (E; 0.01 M EDOT) and suggested an easy way to prepare conductive AF-templated materials with controlled electrical properties.
Conclusion
This study describes optimized method for obtaining of conductive polymer and natural fiber composites. Of particular interest and novelty is use of these sustainable national crop wastes and especially AF within the composite in several engineering applications. (1) For this purpose, conductive composites based on NF/CP, namely AF/PEDOT(C), AF/PPy(C), AF/PCz(C), LF/PEDOT(C), LF/PPy(C), LF/PCz(C), BF/PEDOT(C), BF/PPy(C) and BF/PCz(C) were obtained successfully by oxidative chemical polymerization and conductivities were compared. (2) Since PEDOT is the polymer with the highest conductivity among these CPs, AF/PEDOT(C), LF/PEDOT(C), and BF/PEDOT(C) have higher conductivities than the others and the highest conductivity belongs to AF/PEDOT(C) (12.8 S/cm) among these NF/PEDOT(C)s. When the conductivities of the NF/CP composites in the literature were compared, it was seen that the AF/PEDOT composite had improved conductivity value with the advantage of obtaining the multi-step method. (3) To obtain AF/PEDOT(C), FeCl3 was found the suitable oxidant. (4) AF/PEDOT(C) was characterized by FT-IR, thermal, SEM and mechanical measurements. (5) Further investigation on the effect of monomer and oxidant concentrations was continued with AF/PEDOT(C) and optimum concentrations were determined as 0.03 M EDOT and 0.6 M FeCl3 for chemical polymerization and resulting composite was called as AF/PEDOT (C, 0.03). (6) Polymerization of EDOT by electrochemical method was continued with AF/PEDOT (C, 0.03) and the effect of FeCI3 concentrations used during chemical polymerization and monomer concentration used during electrochemical polymerization on the electroactivity of resulting AF/PEDOT (C, 0.03)/PEDOT(E) were investigated. (7) Detailed characterization of AF/PEDOT(C)/PEDOT(E) showed that, to use this composite as a capacitor, one should use AF/PEDOT (C, 0.03)/PEDOT (0,03) starting from AF/PEDOT (C, 0.03) obtained with 0.9 M FeCl3 and electropolymerized in 0.03 M EDOT by applying 10 cycles. (8) Although adding PEDOT to the natural AF fiber slightly reduces flexibility, it strengthens the composite. (9) Obtained results will be useful to enhance mechanical and conductivity properties of NF to novel AF/PEDOT(C)/PEDOT(E) composite and to convert the AF waste to valuable AF/PEDOT which is suitable conductive composite for several electronic and charge storage applications.
Footnotes
Acknowledgements
The authors would like to thank ‘Istanbul Technical University fund for their financial support (Project number: 40255).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Istanbul Technical University fund, Scientific Research Project (Project number: 40255).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



