Abstract
The electrospinning of nanofibers (NFs) from 2-hydroxypropyl β-cyclodextrin supramolecular inclusion complexes (150% HPβ-CD, w/v) with vitamin B5 (pantothenic acid [PA]) was achieved without using any carrier polymeric matrix. The presence of PA and the formation of the HPβ-CD/PA inclusion complex within the NFs structure were confirmed by SEM, FTIR, XRD, and TGA analysis. SEM images depicted the bead-free uniform NFs and confirmed the incorporation of HPβ-CD/PA NFs did not alter the fiber morphology having an average fiber diameter of 512 ± 24 nm, 328 ± 18 nm, and 150 ± 19 nm, respectively. Fourier transform infrared (FTIR) spectrum indicated inclusion complex from the shifting of the peaks of each component in HPβ-CD/PA NFs, whereas XRD pattern revealed that HPβ-CD/PA NFs was achieved due to the formation of a new amorphous structure. TGA studies showed that the stability of PA after HPβ-CD encapsulation was improved. Molecular docking was used to simulate the positions and interactions of the binding sites of the HPβ-CD/PA inclusion complex. The phase solubility test showed enhanced solubility of PA due to the inclusion complexation; in addition, the stoichiometry of HPβ-CD/PA was determined to be 1:1. The release of PA from HPβ-CD/PA NFs prevented the colonization of Escherichia coli (5.0 ± 0.3%) and Staphylococcus aureus (2.0 ± 0.5%) bacteria to a great extent, as observed in the antibacterial activity results. The cell viability of HCT-116 cells treated with 100 μg/mL of HPβ-CD/PA NFs was registered at 97.5 ± 2.1%. It was observed that HPβ-CD/PA NFs had higher anticancer activity compared to pure PA and HPβ-CD due to the solubility increase. In brief, our results suggested that polymer-free HPβ-CD/PA inclusion complex NFs could have potential applications in food, pharmaceuticals, and healthcare thanks to its efficient antibacterial and anticancer activities.
Introduction
The electrospinning of nanofibers is becoming a promising approach for the encapsulation of active agents such as drugs and food additives. The electrospinning technique has received tremendous attention recently due to its versatility for producing multifunctional nanofibers (NFs) from a variety of polymeric and inorganic materials. 1 The unique characteristics of electrospun nanofibers such as high surface-to-volume ratio, controllable fiber diameter, and morphologies (core-shell, hollow, and porous) can be obtained by changing the process parameters. Nanofibers have been used in purification, catalysis, packing, pharmaceutical, and energy applications. 2 In general, polymeric materials are used to obtain electrospun fibrous materials because long polymeric chains provide certain chain entanglements and overlapping for the fiber formation during the electrospinning process. Hence, the electrospinning of NFs purely from small molecules presents a real challenge. In addition, nano-bio materials for tissue regeneration, drug delivery, and tissue-engineered scaffolds could be produced by electrospinning, given the morphological similarities between electrospun NFs and the extracellular matrix. 3 Moreover, recent reports have shown that electrospinning and electrospun nanofibrous materials could be applicable in food, food packaging, and the agricultural industries.4,5 In addition, NFs can be functionalized with different compounds including drugs, antioxidant and antibacterial agents, flavors, and fragrances. 6 The electrospinning process using organic solvents significantly restricts the use of these electrospun products in medical care, pharmaceutical drugs, food, and nutrition packaging, as most polymers are soluble in organic solvents that pose air pollution. Therefore, the use of water-soluble polymers is a choice to avoid such organic solvent issues by using water as a solvent for electrospinning. Water-soluble and bio-renewable materials such as starch-based oligosaccharides (i.e., cyclodextrins [CDs]) could be alternative materials to produce bio-based nanofibrous materials by using the electrospinning technique. The electrospinning of nanofibers from small molecules and cyclic oligosaccharides such as CDs is quite a challenge when compared to polymeric systems due to the lack of chain entanglement and overlapping.6,7
Pantothenic acid (PA), also called vitamin B5 (VB5), is a water-soluble B vitamin and therefore an essential nutrient.8,9 This water-soluble vitamin acts as an antioxidant and plays a very important role in the decomposition of carbohydrate, fat, and proteins. 10 It plays a key role in numerous physiological responses and thus its deficiency often leads to various diseases or disorders. In particular, a wide variety of studies have shown the benefit of VB5 for the skin. Pantothenic acid is commonly used in cosmetics to promote skin moisturizing, anti-inflammation, and hair growth. It is present in relevant amounts in the liver and kidneys. Although the impact of PA on cell physiology is still unclear, its effects in cosmetics lead to the expectation that PA reduces intracellular oxidative stress. 11 Moreover, PA supplementation increases rates of proliferation and protein synthesis in cultured fibroblasts, and PA stimulates blastocyst production in cultures of one-cell hamster embryos. 12 On taking more than the recommended daily intake (more than 5 mg) of PA, several side effects are observed. These results demonstrate that proliferating cells could have an increased demand for PA and raise the possibility that cells meet this increased demand by increasing rates of PA uptake.
2-hydroxypropyl-β-cyclodextrin (HPβ-CD) is a well-known complexing agent, capable of improving aqueous solubility and increasing the bioavailability of many therapeutic agents. 13 Such complexation within the cavity of cyclodextrin derivatives has also been reported to decrease toxicity and side effects, increase stability, and can control the release of certain therapeutic agents in drug delivery systems.14–18 Enhanced thermal sensitivity and the potential to mask undesirable drug odors and tastes were also shown in the inclusion formulations. Another essential property of CDs is that they are non-toxic, allowing them to be used in the food, cosmetics, and personal care industries.19,20 In the context of the pharmaceutical industry, the solubility-related problems of some drugs remain among the most challenging issues of the drug formulation, where CDs can enhance solubility, dissolution rate, chemical stability, and bioavailability.21,22 In particular, a chemically modified CD-like HPβ-CD is more suitable for the solubilization of hydrophobic drugs because of its better aqueous solubility compared to native CDs. 23 Our particular interest is in the electrospinning of nanofiber using CDs since it allows us to make nanofibers with specified functionalities and physicochemical features. 24 CDs were also used to make nanofibers that dissolve quickly. Moreover, CDs are advantageous over other small molecules that are being used to produce nanofibers by electrospinning since CDs are capable of forming CDs inclusion complexes with various compounds. Electrostatic interactions, van der Waals interactions, and hydrogen bonding are the driving forces of inclusion complex formation. The formation of self-assembly and aggregates in highly concentrated solutions of CDs via intermolecular hydrogen bonding enables the production of nanofibers from CDs solutions. 2-hydroxypropyl-β-cyclodextrin is regarded as one of the supramolecular assemblies that have been widely used in the electrospinning method.24,25 Pantothenic acid plays an important role in maintaining biological function, which is widely applied in the food, pharmaceutical, and cosmetic industries. There are only a few reports in the literature that the encapsulation of CDs into nanofibers for controlling the drug molecule. We observed that the fundamental condition for electrospinning CDs without employing a carrier polymeric matrix is the self-assembly and self-aggregation of these supramolecular CDs molecules in their highly concentrated solutions. One of the ways that we achieve this goal is incorporating the HPβ-CD/PA inclusion complex of active agents into electrospun polymeric nanofibers. As a result, we used an electrospinning approach to make HPβ-CD/PA nanofibers without a polymer matrix in this study. So, HPβ-CD/PA nanofibers without using a polymer matrix would be quite advantageous due to the non-toxic nature of HPβ-CD with the ability to form HPβ-CD/PA inclusion complex which offers great thermal stability, enhanced water solubility, and superior antibacterial or antioxidant properties.
In this contribution, we have formed inclusion complexes of PA with HPβ-CD and free-standing HPβ-CD/PA nanofibrous web via electrospinning without using polymeric carrier matrix (Figure 9). The stoichiometry of the HPβ-CD/PA inclusion complex was found to be at a 1:1 ratio by phase solubility and molecular docking studies. The uniform and bead-free morphology of the HPβ-CD/PA inclusion complex nanofibers was observed by SEM imaging. Further chemical, structural, and thermal characterizations of HPβ-CD/PA inclusion complex nanofibers were performed by UV–Vis, FTIR, XRD, and TGA. It was confirmed that the formation of an inclusion complex between HPβ-CD and PA was effectively achieved and that the efficacy of the complexation was higher for HPβ-CD/PA inclusion complex nanofibers compared to the pure PA drug. The antibacterial activity of HPβ-CD/PA inclusion complex nanofibers against Gram-negative (Escherichia coli) and Gram-positive (Staphylococcus aureus) bacteria was tested using the colony counting method. Finally, the results of anticancer tests showed that HPβ-CD/PA inclusion complex nanofibers have better cancer cell activity than pure PA against HCT-116 cells. In short, this study provides a useful approach for the molecular encapsulation of PA within HPβ-CD inclusion complex nanofibers by electrospinning, in which fast-dissolving and highly water-soluble PA has higher-temperature stability along with antibacterial and anticancer activities were achieved for HPβ-CD/PA inclusion complex nanofibers.
Experimental section
Materials and reagents
2-Hydroxypropyl-β-cyclodextrin (HPβ-CD) (98%, MW = 1541.5, DS = 7) and DL-pantothenic acid hemi-calcium salt (1.0 mg/mL; 90:10 methanol:water) were purchased from Sigma-Aldrich (India) and used without further purification. All chemicals used were of analytical grade. The water used as solvent was obtained from the Millipore water system.
Instruments
The absorption spectra of the PA and HPβ-CD/PA complexes were recorded with a spectrophotometer (UV-2600). The morphology of the NFs was observed with SEM (JEOLJSM-5600LV, Japan) after samples were sputter-coated with gold. By examining approximately 100 fiber diameters of each specimen, the average fiber diameter of the HPβ-CD/PA NFs was determined by using ImageJ software. The Fourier transform infrared (FTIR) spectra were obtained on Nicolet iS10 Fourier transform infrared spectrometer with a scanning range of 4000–400 cm−1. The XRD pattern of the HPβ-CD/PA complexes was performed using a PANalytical X Pert powder diffractometer using Cu Ka radiation over a 2-h range from 10 to 60°C to determine the amorphous structure of the samples. The thermal stability of samples was investigated by thermogravimetric analysis (TGA; TA Q500; TA Instruments, New Castle, DE).
Preparation of HPβ-CD/PA inclusion complex and electrospinning of NFs
The formation of inclusion complexes of HPβ-CD with PA was achieved by using a 1:1 M ratio of HPβ-CD:PA. For HPβ-CD/PA, PA and HPβ-CD (50%, 100%, and 150%; w/v) were dissolved in water separately and then PA solution was drop-wised added into HPβ-CD solution slowly. As the solution was cooled down and stirred overnight at room temperature, a white, highly turbid HPβ-CD/PA solution was obtained. As a control sample, the homogeneous and clear aqueous solution of HPβ-CD was prepared by dissolving HPβ-CD (150%; w/v) in water by stirring for 30 min at 60°C; thereafter, it was cooled down to room temperature before electrospinning. For electrospinning, HPβ-CD solution was loaded into a syringe having a metallic needle with 0.45 mm inner diameter, and it was positioned onto the syringe pump (Model: SP 101IZ, WPI) to control the flow of the solution and the electrospinning parameters were optimized as follows: applied voltage, tip-collector distance, and solution a voltage at 15 kV, distance 10 cm, and flow rate 1 mL/h. The electrospun NFs were accumulated via a piece of aluminum foil on a grounding stationary-covered cylindrical metal collector. At the relative humidity maintained at 25°C with 18%, all studies were designed in a closed plexiglass box. The solutions were started to be prepared from HPβ-CD (150%; w/v) concentration and they were increased to the optimized level until the bead-free uniform NFs were produced.
Phase solubility study
Phase solubility was studied as described by previous literature.
26
In short, excessive PA (0.25 g) was added to 30 mL of an aqueous solution of HPβ-CD concentration ranging from 0.05 to 2.5 mM. The solutions were further mixed using a magnetic stirrer at 25°C for 48 h to ensure equilibrium, and then the solutions were passed through a 0.45-μm filter to remove undissolved solids. The concentration of residual PA in the supernatants was measured with a spectrophotometer (UV-2600). In addition to the solubility diagram obtained according to equations (1) and (2), the apparent stability constant (Ks) and the complexation efficiency (CE) were determined as follows
Molecular docking
The most probable structure of the HPβ-CD/PA inclusion complex is determined also by molecular docking studies using the PatchDock and FireDock servers. Default parameter PatchDock and FireDock server were set as clustering root mean square deviation (RMSD) at 4.0 and docked poses were analyzed by LigPlot Plus for generating 2D schematic representations. The input files are initially optimized and uploaded to the server in PDB format. PatchDock and FireDock use different parameter sets for complex type field parameters to automatically determine the complementarity determining regions of the ligand/receptor molecule. The PatchDock and FireDock server gave several possible docked models for the most probable structure based on the energetic parameters; geometric shape complementarity score, approximate interface area size, and atomic contact energy of the HPβ-CD/PA inclusion complex. The 3D structural data of HPβ-CD and PA is obtained from crystallographic databases that were generated using UCSF-Chimera 1.8.1 (https://www.cgl.ucsf.edu/chimera).
Antibacterial properties
The antibacterial properties of the PA, HPβ-CD and HPβ-CD/PA NFs were evaluated against Escherichia coli (E. coli, ATCC 10536) and Staphylococcus aureus (S. aureus, ATCC 25923) bacteria by using the colony counting method. Before use, the bacteria were cultured independently on a nutrient broth for 24 h at 37°C. Afterward, each bacterial sample (1×106 CFU/mL) was taken in a test tube separately containing nutrient broth and UV sterilized NFs (15 mg/mL) and then incubated at 37°C for 24 h. From this culture, 100 μL of the sample was uniformly swabbed on the nutrient agar plates using a sterile cotton swab and the plates were incubated for 24 h at 37°C. The bacteria without treatment with NFs served as the control and the pure PA treatment was used to compare the activities. All experiments were carried out in triplicate. The antibacterial activity of the HPβ-CD/PA NFs was defined as follows
In vitro cytotoxicity and cellular activity
Cytotoxicity of the samples was analyzed using (3-(4,5-dimethylthiazol-2yl) -2,5-diphenyl tetrazolium bromide (MTT). Briefly, HCT-116 cells were seeded at an initial density of 1×106 cells/microwell in 200 μL flat-bottomed microplates and incubated at 37°C in a humidified atmosphere containing 5% CO2. After 24 h of incubation, the cells were treated with different concentrations of PA, HPβ-CD and HPβ-CD/PA NFs (20–100 μg/mL) for 24 h at 37°C. The volume of PA treated in free form and the NFs complexes of HPβ-CD/PA are equal. The freshly prepared MTT solution (5 mg/mL) is added into each well and the incubation is extended for 4 h. The formation of purple-color formazan crystals was solubilized by adding 100 μl of DMSO. Finally, the absorbance was measured at 470 nm using a UV–Vis spectrophotometer (Thermo Scientific, Vantaa, Finland). For the morphology examination, the HCT-116 cells were incubated with 100 μg/mL of HPβ-CD/PA NFs for 24 h under 5% of CO2 condition. Later, the cells were washed with PBS solution and stained using two fluorescence stains DAPI and PI. The occurrence of morphological changes in the HCT-116 cells was visualized under a NIKON Eclipse 80i fluorescent microscope (Nikon Corporation, Tokyo, Japan).
Statistical analysis
The quantitative data are expressed as mean ± standard error of the mean of three independent experiments. One-way ANOVA testing was carried out to evaluate statistical significance. A Bonferroni t-test analysis was used to validate the ANOVA test. The p-value of ≤ 0.05 was considered statistically significant.
Results and discussions
Optical properties of HPβ-CD/PA NFs
The formation of the inclusion complex of PA with HPβ-CD was confirmed by UV–Vis spectroscopy and phase solubility to determine the stoichiometry of the complexes (Figure 1). The NFs containing HPβ-CD/PA wet easier and the structure disintegrates dissolving the substance quickly. The enhancement in the solubility of PA with HPβ-CD was confirmed by a dissolution test.
27
The UV–Vis spectroscopy of PA, HPβ-CD50%/PA, HPβ-CD100%/PA, and HPβ-CD150%/PA dissolved in water is shown in Figure 1(a). HPβ-CD/PA allows the PA drug to be dispersed in the medium which facilitates its dissolution. Pantothenic acid spectra show a peak at 255 nm while β-CD did not present any peak at this area. The wavelength shift indicated the structural change in PA and the formation of an inclusion complex.
28
As shown in Figure 1(a), at the same PA concentration, the higher the concentration of the HPβ-CD aqueous solution, the stronger the maximum absorption peak at 240 nm, accompanied by a significant blue-shifting of 15 nm. Although the same amount of PA was used for each sample, the solubility of PA has increased much more in HPβ-CD150%/PA. However, in the matter of all the complexes, as the concentration is increased, the intensity of the peak is also increased. The blue-shifting and hyperchromicity indicated that the PA was embedded in the HPβ-CD nanocavity. Its solubility increases dramatically as the drug molecules interact with HPβ-CD, where HPβ-CD/PA NFs are normally more lipophilic than the drug molecules.
29
Additionally, the presence of HPβ-CD may lower the required dose of an active molecule by improving its solubility. It has been reported that the formation of inclusion complexes could enhance the absorption of guest molecules.
30
Consequently, HPβ-CD150%/PA could be used as an efficient drug delivery system for electrospinning purposes.
31
(a) UV–Vis absorption spectra of PA and different ratios of (HPβ-CD; 50%, 100%, and 150%) HPβ-CD/PA NFs inclusion complex and (b) phase-solubility diagram for the HPβ-CD/PA system.
Phase solubility studies are very critical to understand the stoichiometry of the inclusion complexation. Here, HPβ-CD/PA solutions having different HPβ-CD concentrations were stirred for 48 h to reach the complexation equilibrium. The phase solubility graph indicates the solubility trend of PA against increasing HPβ-CD concentrations from 0.05 to 2.0 mM (Figure 1(b)). The diagram obtained by the inclusion complex with HPβ-CD seemed to be AL-type, as established by the Higuchi and Connors model,
26
suggesting the occurrence of complexes with stoichiometry 1:1. The linear host-guest correlation is characteristic of AL-type solubility. In a dilute system, this is expected to be the dominating structure, but CDs are known to not exclusively form 1:1 inclusion complexes but also non-inclusion complexes. However, when the slope of the AL isotherm is greater than unity, higher-order complexes are assumed to be involved in the solubilization. The phase solubility diagram also enabled the calculation of the stability constants (Ks) and complexation efficiency (CE) of the inclusion complex formed between PA and HPβ-CD.
32
In these experiments, the slope was calculated at 0.9, confirming the formation of 1: 1 complex (HPβ-CD/PA). The stability constant, Ks and CE were found at 214.46
Morphology analysis of HPβ-CD/PA NFs
2-hydroxypropyl-β-cyclodextrin (HPβ-CD) makes hydrophobic drug molecules water-soluble by forming inclusion complexation; therefore, the electrospinning of HPβ-CD/PA systems can be performed in water without the need of using toxic organic solvents.
33
On the contrary, in this study, we have performed the electrospinning of HPβ-CD/PA NFs from an aqueous solution of HPβ-CD/PA inclusion complex displays SEM images and average fiber diameter along with fiber diameter distributions (Figure 2). All of the NFs exhibited a randomly aligned morphology without porous regions or beads. We prepared a 1:1 Representative SEM image of NFs obtained from (a) HPβ-CD50%/PA, (b) HPβ-CD100%/PA, and (c) HPβ-CD150%/PA inclusion complex. Fiber diameter distributions with average fiber diameter of the NFs obtained from solutions of (d) HPβ-CD50%/PA, (e) HPβ-CD100%/PA, and (f) HPβ-CD150%/PA inclusion complex calculated from SEM images (n = 100).
The nanofibers variations in HPβ-CD50%/PA, HPβ-CD100%/PA, and HPβ-CD150%/PA diameters are due to differences in conductivity and viscosity of the HPβ-CD solutions at 50%, 100%, and 150% (w/v). 40 Therefore, the viscosity of the solution has a significant effect on the morphology of the fibers formed by electrospinning. For instance, the lower conductivity and higher viscosity result in less stretching of the electrified jet that forms thicker fibers during the electrospinning process. 41 Moreover, HPβ-CD150%/PA NFs was thinner than HPβ-CD50%/PA NFs and HPβ-CD100%/PA NFs possibly due to the higher conductivity and slightly lower viscosity of the parent solution. 42 In addition, the interference of the inclusion complex concentration of β-cyclodextrin and cinnamon essential oil or zein containing eucalyptus essential oil/cyclodextrin inclusion complex on the diameter of the poly (lactic acid) fibers and found that, as the inclusion complex concentration increased, the mean diameter increased as well.43,44 In this study, HPβ-CD150%/PA samples were in the form of self-standing and flexible webs which make them applicable for oral drug delivery as fast-dissolving systems.
Structural and thermal properties of HPβ-CD/PA NFs
The structural and thermal stability characterizations of PA, HPβ-CD, and HPβ-CD/PA NFs were carried out by using FTIR, XRD, and TGA techniques (Figures 3–5). The FTIR spectrum analysis of PA, HPβ-CD, and HPβ-CD/PA NFs is shown in Figure 3. Fourier transform infrared analysis confirms the presence of hydroxyl and amide bonds in the monomer and the PA structures. The presence of -NH, -OH, C-O, and C-N stretching to 3425 cm−1, 3334 cm−1, 1050 cm−1, and 1127 cm−1, respectively, suggests the availability of polar groups for absorption.
45
The asymmetric and symmetrical stretching of the CH bond of the group is at 2968 cm−1 and 2911 cm−1, respectively. The vibrational stretching at 1647 cm−1 is attributed to the C-O signal of the amide bond. The band at 2923 cm−1 is assigned to asymmetric C–H stretching. The FTIR spectrum of HPβ-CD shows a broad stretching band of hydroxyl groups on the primary and secondary faces of the cavity, while the vibrations of the C-C/C–O stretching and the antisymmetric stretching of the C–O–C glycosidic bridge appear between 1020 and 1150 cm−1.
46
The broad band with a transmission peak was at 3395 cm−1 corresponding to the O–H extension. The peak at 2929 cm−1 represents C–H stretching vibration and 1649 cm−1 due to the H–O–H bending. We also observed small shifts for PA peaks in the FTIR spectra of HPβ-CD/PA NFs, for instance, -OH, -NH, C–O, C–H, and C–N absorption peaks shifted to 3402, 3295, 1046, 2968, and 1128 cm−1, respectively. So, the FTIR study suggested the presence of host-guest interactions between the HPβ-CD and PA in the HPβ-CD/PA NFs samples. FTIR spectra of PA, HPβ-CD, and HPβ-CD/PA NFs inclusion complex. Powder X-ray diffraction pattern of PA, HPβ-CD, and HPβ-CD/PA NFs inclusion complex. Thermal analysis of PA, HPβ-CD, and HPβ-CD/PA NFs inclusion complex.
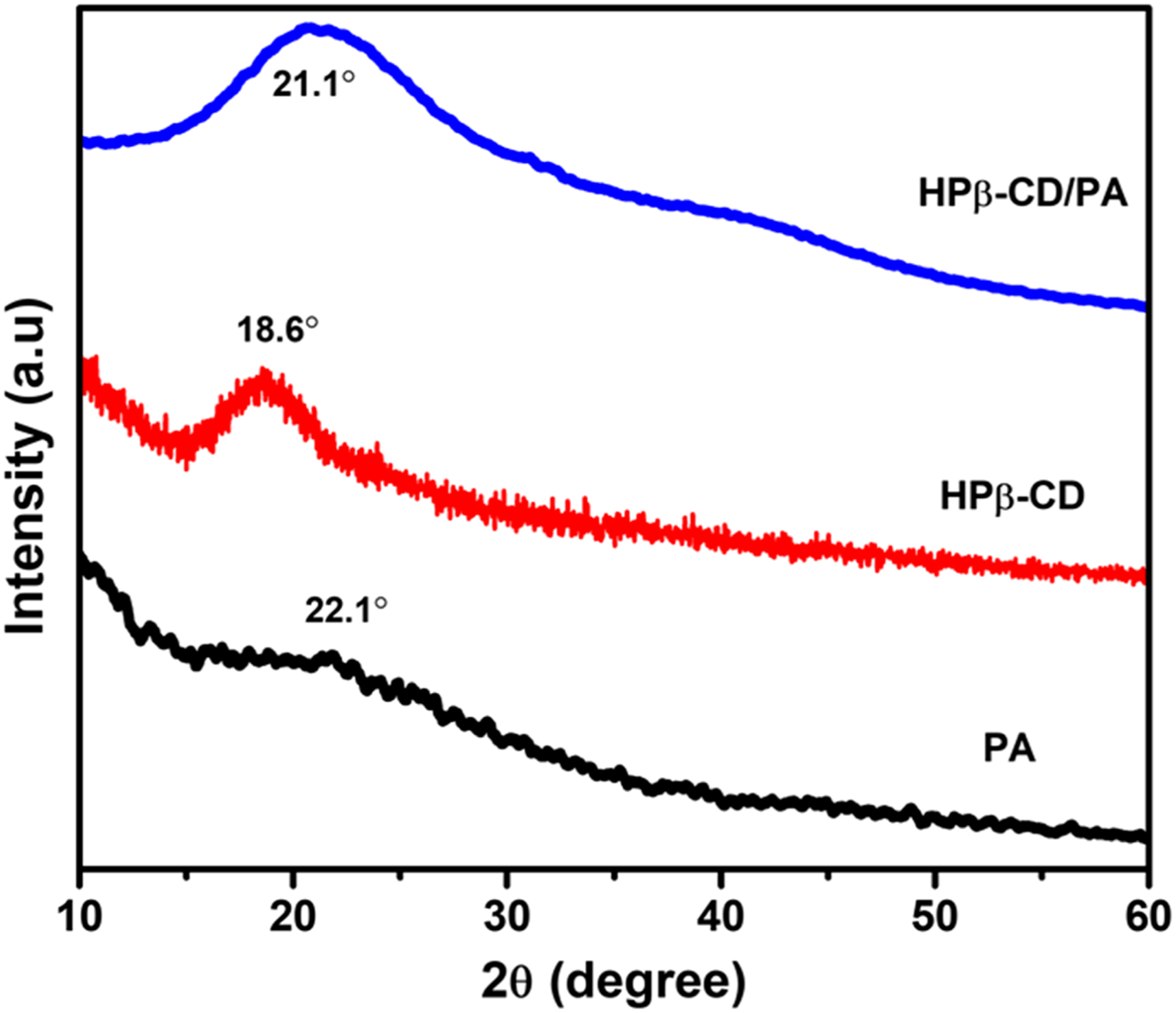
The XRD studies were performed for PA, HPβ-CD, and HPβ-CD/PA NFs, and the resulting diffraction patterns are displayed in Figure 4. XRD pattern of PA showed an amorphous structure, which is indicated by a broad featureless or sharp-free peak at 2θ = 22.1°. 47 On the other hand, pristine HPβ-CD displayed a broad halo pattern in the range of 2θ = 18.6° confirming their amorphous nature. 46 In contrast, the HPβ-CD/PA NFs has shown a broad halo pattern range of 2θ = 21.1°, similar to pristine HPβ-CD indicating the complete inclusion complexation and amorphization of PA in case of HPβ-CD/PA NFs. This result is the proof inclusion complexation between PA and HPβ-CD, suggesting that PA molecule when being included in HPβ-CD cavity. Moreover, amorphization was one of the necessary conditions for the rapid dissolution of drugs. Hence, the shifting of a PA diffraction peak in HPβ-CD/PA NFs samples strongly suggests that PA is encapsulated as an inclusion complex within the HPβ-CD nanofiber matrix after electrospinning.
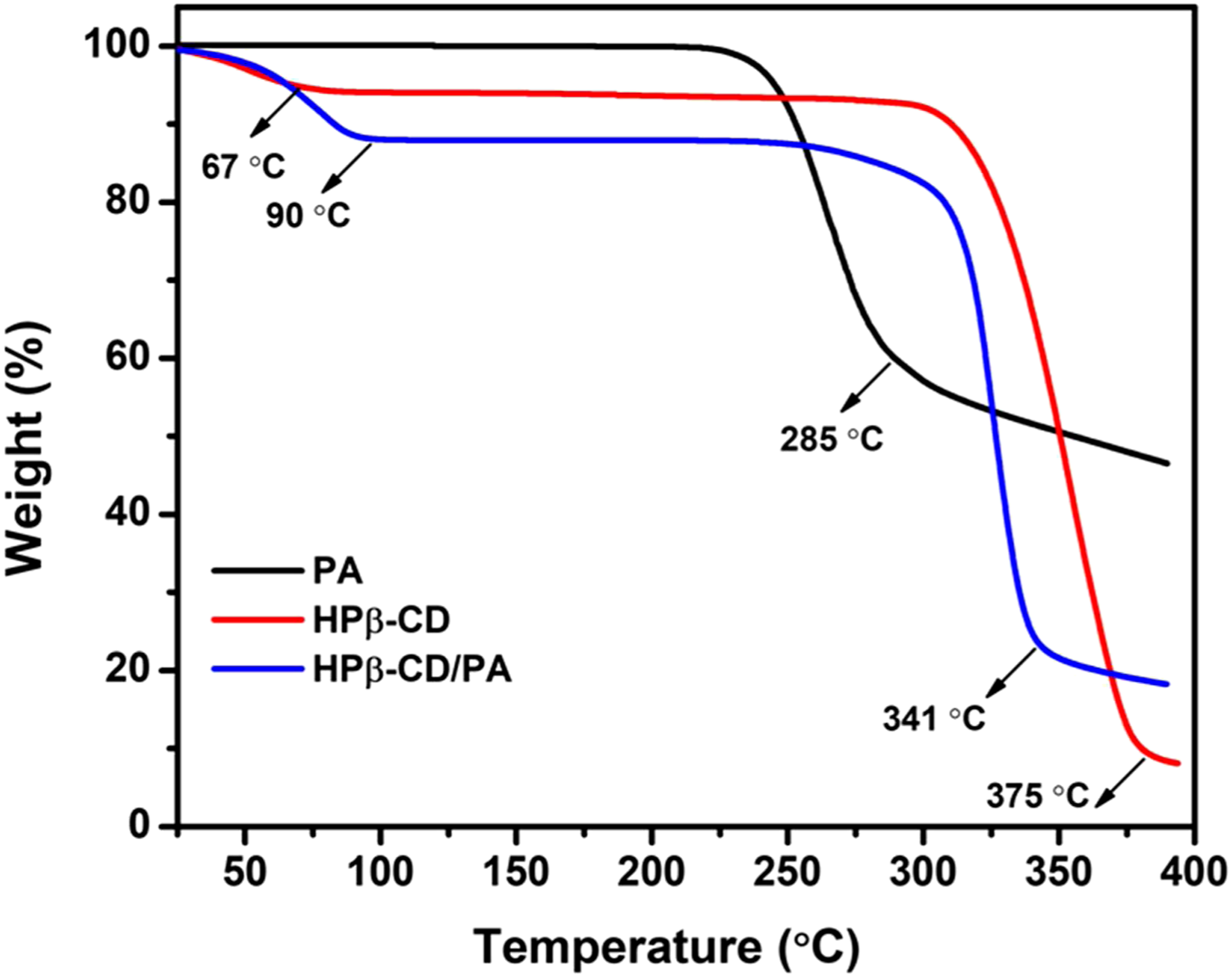
The thermal stability of PA, HPβ-CD, and HPβ-CD/PA NFs was determined by thermogravimetric analysis (TGA) as shown in Figure 5. The thermal evaporation/degradation of the PA started at 285°C. The weight losses at 67°C and 375°C correspond to the water loss and major thermal degradation of HPβ-CD, respectively. For the HPβ-CD/PA NFs, two stages of weight losses were observed between 25°C and 400°C. On the other hand, HPβ-CD/PA NFs have a significant change in the weight loss having at 320°C and belong to the evaporation/degradation of PA and HPβ-CD. 47 For HPβ-CD/PA NFs, the initial weight loss at 9°C is due to water loss. Since the degradation step of PA and HPβ-CD corresponded in the thermograms, we could not observe a distinct weight loss. The significance shifted to the higher temperature (341°C) suggesting a complexation between PA and HPβ-CD. That means thermal degradation of HPβ-CD/PA NFs occurred at high-temperature range compared to pure HPβ-CD (375°C) confirming the inclusion of complex formation between PA and HPβ-CD molecules. The PA amount in HPβ-CD/PA NFs was calculated as 17% (w/v, concerning HPβ-CD) that is correlated with the 1:1 M ratio complexation and proved the preservation of PA during the electrospinning.
Molecular docking studies
Molecular docking is a powerful approach, which involves docking a bioactive compound in the potential binding site of macromolecular targets. The concordant results have been obtained in this case suggesting the competence of PatchDock as a good docking server. Followed by this the protein to ligand docking solutions obtained from the PatchDock server was further refined by FireDock which rendered additional refinements to docking complexes.48,49 The best-docked conformation is judged based on optimal geometric score, interface area size, and desolvation energy generated by the complex. FireDock server analysis sorts out the refined complex structures based on energy functions like global energy, atomic contact energy, the contribution of the hydrogen bond, attractive and repulsive VdW forces, and so on.
50
The 3D structure of PA, HPβ-CD, HPβ-CD/PA obtained from crystallographic databases is shown in Figure 6. The best docked three models of HPβ-CD/PA 1:1 structure (Figure 6 and Table 1) with the highest score (2498, 2126, and 1934), interface area size (271.90, 222.60, and 210.80 Å2), atomic contact energy (−207.34, −163.08, and −124.30 kcal/mol), global energy (−33.72, −25.84, and −21.21 kcal/mol), attractive VdW force (−13.65, −11.71, and −9.23 kcal/mol), repulsive VdW force (3.57, 4.87, and 1.96 kcal/mol), and ACE (−10.27, −8.77, and −7.75 kcal/mol) of the complex is the highly probable and energetically favorable model and it is in good correlation with results obtained through experimental methods.51,52 Molecular docking studies on PA, HPβ-CD, and the different docked models of HPβ-CD/PA inclusion complexes with stick and sphere (CPK) representations. Computed using PatchDock and FireDock servers’ score of the top 10 docked models of HPβ-CD/PA inclusion complex. aGeometric shape complementarity score. bApproximate interface area size of the complex. cAtomic contact energy. dIndicating binding energy of the solution. eRepresenting contribution of the van der Waals forces to the global binding energy. fACE shows the contribution of the atomic contact energy (ACE) to the global binding energy.

Antibacterial activity of HPβ-CD/PA NFs
The antibacterial activity of PA and HPβ-CD/PA NFs against E. coli and S. aureus was tested by using the viable cell-counting method. As shown in Figure 7, compared to the control experiment, the E. coli and S. aureus bacteria which were incubated by PA and HPβ-CD/PA NFs presented lower cell viability. It seems the inclusion complex of HPβ-CD/PA NFs had strong antibacterial activity than pure PA against both the tested bacteria.53–55 Although the bacterial viability was decreased for PA nanofibers, almost 32 ± 1.7% were live, the HPβ-CD/PA NFs have excellent bactericidal properties against E. coli (5.0 ± 0.3%). Upon treatment of S. aureus with PA and HPβ-CD/PA NFs, 41 ± 2.8% and 2.0 ± 0.5% of the cells were live, respectively, in the inclusion complex of HPβ-CD/PA NFs had strong antibacterial activity than the pure PA against both model bacteria. Growth inhibition rate (%) and typical images of E. coli and S. aureus colonies treated by control, PA, and HPβ-CD/PA NFs at 37°C (n = 3).
The increased antibacterial activity of PA is caused by the inclusion complex mechanism that increases the solubility and therefore provided an efficient release of the hydrophobic agent in the agar medium and also confirmed by the phase solubility studies. As the quote above, inclusion complex formation with HPβ-CD could increase into contact with microorganisms when it was dispersed in aqueous solutions leading to enhanced antimicrobial activity at lower concentrations of encapsulated compounds.56–61 As the primitive sites for antimicrobial action were found at the cell membrane and inside the cytoplasm, HPβ-CD may enhance the accession of PA to these regions by improving the water solubility of PA.
In vitro cytotoxicity and cellular uptake analysis
Cytotoxicity study is an important indicator to assess the activity of carriers and drugs on tumor cells. The viability of HCT-116 cells against different concentrations (20–100 μg/mL) of PA, HPβ-CD, and HPβ-CD/PA NFs was evaluated as a function of 24 h incubation time (Figure 8(a)). Figure 8(a) reveals that the viability of HCT-116 cells treated with 20–100 μg/mL of pure PA, HPβ-CD, and HPβ-CD/PA NFs for 24 h was ranged from 97.5 ± 3.3% to 15.3 ± 1.2% and the activities were found to be in a dose-dependent manner. Of these, the HPβ-CD/PA NFs inclusion complex exhibited had an excellent cytotoxic effect on the HCT-116 cells than the pure PA and HPβ-CD.62,63 The results might be interpreted as the immediate release of pure PA and the delayed and sustained characteristics of the inclusion complex. The viabilities of HCT-116 cells treated with 100 μg/mL of pure PA, HPβ-CD, and the HPβ-CD/PA NFs inclusion complex was recorded at 61.8 ± 1.9%, 27.4 ± 0.6%, and 97.5 ± 2.1%, respectively, which may due to the specific binding of PA with the surface of cancer cells. Furthermore, the cell uptake and intracellular distribution by HCT-116 cells of HPβ-CD/PA NFs inclusion complex (100 μg/mL) were measured by fluorescence confocal microscope. The cell uptake image of HPβ-CD/PA NFs inclusion complex with obvious blue and red fluorescence using DAPI and PI staining was shown in Figure 8(b). This phenomenon indicated that the HP_x3b2;-CD/PA NFs inclusion complex was more easily taken up by cells, which may be caused by the specific binding between the complex and cancer cell surface.64,65 These results were consistent with the results of the obvious in vitro cell cytotoxicity assay. It is concluded that the cyclodextrin inclusion complex drug delivery could improve the biocompatibility of the drug. As a result, cell cytotoxicity and inclusion complex uptake studies revealed that HPβ-CD/PA NFs drug deliveries exhibited promising anti-cancer drug release and can be suggested for human colon cancer treatment.
66
According to the above characterizations, HPβ-CD/PA NFs inclusion complex was synthesized and showed much better chemical and biological properties. To our best knowledge, it is the first time to synthesis HPβ-CD/PA NFs inclusion complex and targeted modification was also carried out. (a) Cell viability of PA, HPβ-CD, and the inclusion complex HPβ-CD/PA (20–100 μg/mL) on HCT-116 cells after 24 h. Data are presented as mean ± standard deviation (n=3). (b) Bright-field, DAPI and PI-staining, and merged images of HCT-116 cell lines after incubation for 24 h with inclusion complex of HPβ-CD/PA at 100 μg/mL (scale: 100 μm). (a) 3D structure of pantothenic acid (PA), hydroxypropyl β-cyclodextrin (HPβ-CD) molecules, and HPβ-CD/PA inclusion complex and (b) schematic representation of the electrospinning of nanofiber from HPβ-CD/PA solution with antibacterial activity.

Conclusions
In this study, the nanofibers from HPβ-CD/PA system were achieved, although it is quite a challenge to obtain nanofibers from these without a polymer matrix. In vitro dissolution tests demonstrated that HPβ-CD/PA exhibited a desirable drug solubility enhancement profile for a 1:1 M ratio by UV–Vis absorption and phase solubility studies. The electrospinning of HPβ-CD/PA nanofibers resulted in bead-free and uniform nanofibers having a diameter of 150 ± 19 nm, as confirmed by SEM imaging. The FTIR, XRD, and TGA analyses proved the host-guest inclusion complex formation between PA and HPβ-CD in the HPβ-CD/PA nanofibers. Furthermore, molecular docking studies performed using PatchDock and FireDock methods also revealed that the stoichiometry of the HPβ-CD/PA was 1:1. Therefore, the results of molecular docking are in good agreement with the experimental data. We have observed a more efficient antibacterial effect of HPβ-CD/PA nanofibers against E. coli and S. aureus compared to pure PA. The in vitro cell cytotoxicity tests demonstrated excellent cellular uptake of the HPβ-CD/PA nanofibers inclusion complex. All these results suggested a great potential of HPβ-CD/PA NFs inclusion complex, due to the non-toxic nature of HPβ-CD containing other types of bioactive agents would be applicable in biomedical technology area such as wound healing and drug delivery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (2021R1A2B5B02002436 and 2021R1F1A1061566). The authors are grateful to the RUSA scheme Phase 2.0 grant [F-24-51/ 2014–U, Policy (TNMulti-Gen), Dept of Edn. Govt. of India. Dt. 09.10.2018] for their financial support.



