Abstract
The resistance of natural fibers toward flames is essential for their use in engineering applications. Hence, the present research focused on the preparation of a bio-flame-retardant liquid (BFL) to enrich the sustainability of natural fibers (kenaf fibers (KF), animal (WF), and cocoon (SF)) towards flame by employing a low cost, bio-waste chitosan and phosphoric acid (PA) through a chemical approach. Scanning electron microscopy, Fourier-transform infrared spectroscopy, thermogravimetric analysis, UL-94 testing, and microcalorimetry were conducted to evaluate the essential characteristics of the treated BFL on various natural fibers. The BFL altered the surface chemistry and exhibited remarkable fire-extinguishing properties (V-0 flammability rating in the UL-94 test and suppression of peak heat release rate of approximately 78%), as well as enhanced thermal stability (49 wt.% residue at 700°C) for the natural fibers without significant differences among them. Furthermore, the treated fibers were reinforced individually into vinyl ester (VE) matrix and manufactured respective composites, namely, VE_TKF, VE_TWF, and VE_TSF. The experimental results concluded that the BFL-treated fiber effectively improved the flame resistance (30% reduction in peak heat release rate) of VE composites. The findings of this study can be used to introduce flame retardancy in natural fibers to enable their use in the present and next-generation engineering applications.
Introduction
Natural fiber-reinforced composites are widely used in construction, automobile, aerospace, and energy applications due to properties, such as abundance, sustainability, low cost, specific weight, and acoustic insulation.1–3 For instance, in March 2020, Polestar, a Swedish electric performance car brand owned by Volvo Cars, replaced interior parts with flax fiber composites in the model Precept, and the French boat company, IDBmarine, launched a yacht at the end of 2019 whose deck was made from flax fiber-reinforced composite. However, unlike synthetic fibers, natural fibers are inherently flammable, thermally degradable, and burn readily, limiting their applications.
Hence, with the growing interest in Natural fiber-reinforced composites, satisfactory flame retardancy of the composite material has become increasingly critical to comply with safety requirements. There are numerous methods for enhancing the flame-retardant (FR) performance of Natural fiber-reinforced composites, especially in the case of matrices, such as grafting, coating, and the incorporation of additives. 4 After grafting, FR components considerably alter the modified materials’ morphology and physical properties, that is, the glass transition temperature and density. 5 Weathering and adhesion to substrates are the main drawbacks of external coating systems, both of which incur additional costs related to applying top and primer coatings. 6 The incorporation of FR additives leads to deteriorated mechanical properties of composites owing to the low compatibility between the matrix and additives. In addition, FR particles can induce a stress concentration and deteriorate the interfacial bonding between the fiber and matrix, resulting in a loss of strength of the composite. 7 Different fiber types and specifications can result in significant functional differences, affecting the performance and cost of the material. The fiber characteristics determine the principal mechanical properties, such as Young’s modulus, tensile strength, and stiffness. Therefore, desirable properties of NFRCs can be achieved by modifying the fiber to overcome the limitations mentioned above.
There are several common techniques such as plasma treatment, 8 sol-gel treatment, 9 UV irradiation, gamma, or E-beam irradiation, 10 and layer-by-layer assembly 11 which have been used to improve the FR properties of natural fibers. However, these methods are disadvantageous in terms of time consumption and additional equipment requirements. One-step dip-coating fiber treatment is facile, economical, controllable, and deposit the flame retardants onto the fibers more uniformly. 12
Over the past few years, interest in halogen-free FR coatings has increased because halogenated additives generate smoke and cause combustion, harming human health and the environment. 13 Therefore, the synthesis of bio-based flame retardants is a promising route to enhance FR performance and mitigate environmental hazards. Moreover, potential green FR materials, such as lignin, 14 protein, 15 cardanol 16 clay, 17 and chitosan, 18 have attracted wide attention. Chitosan is the second most abundant bio-based polysaccharide composed of amino-containing 2-deoxyglucose and is available as a bio-waste product. 19 The presence of nitrogen and multiple hydroxyl groups (an important chemical element for fire suppression) makes chitosan a promising eco-friendly char-forming compound in an intumescent system to enhance the flame retardancy of natural fiber composites. 20 Although chitosan alone has no competitive significance on flame retardancy with commercial flame-retardants, it is the best carbon agent/source for an intumescent flame retardant system that significantly works for polymer composites by increasing the char yield. On top of that, chitosan can be utilized as an auspicious substrate to synthesize effective flame-retardants.
In this study, a bio-flame-retardant liquid (BFL) was synthesized by utilizing chitosan and phosphoric acid (PA) to treat natural fibers and analyzed by Fourier-transform infrared (FTIR) spectroscopy for structural formation. One-step dip-coating was performed to treat three types of natural fiber (kenaf fibers (KF), wool fibers (WF), and silk fibers (SF)) (Figure S3) with the BFL and examine their spectral, morphological (scanning electron microscopy–energy-dispersive X-ray spectroscopy (SEM–EDX), thermal (thermogravimetric analysis (TGA)), FR (horizontal burning test (HBT), and micro-combustion calorimetry (MCC)) properties. Furthermore, the treated fibers were reinforced into a vinyl ester (VE) resin, and composites were prepared via vacuum-assisted resin transfer molding (VARTM). The composites were subjected to TGA and flame retardancy tests (HBT and cone calorimeter). This work offers an economical, sustainable, and practical approach to increasing the flame retardancy of natural fiber polymer composites. The obtained results of this research suggest a few research potentials considering environmental, thermal, and flame retardancy. The treated fabric is the best for commercial applications, and reinforcement for polymer composites can be applied to outdoor engineering applications, including automobiles, furniture, temperature-sensitive packaging, etc.
Materials and methods
Materials
Chitosan (viscosity: 50–800 mPa/s, molecular weight: ∼600,000, degree of deacetylation: 80%–95%) was purchased from Kwangjin Chemical Co Ltd, South Korea. PA, glacial acetic acid (99.7%), and sodium hydroxide (beads, 98%) were purchased from Samchun Pure Chemical Co Ltd Plain woven KF (density 1.4 g/cm3 and mean diameter of fiber 31 μm) was provided by SungChang Co Ltd, Korea. Merino knitting wool threads (density was 1.31 g/cm3 and fiber diameter was 17 μm) were purchased from a conventional domestic market. Waste and tangled SF (produced from degummed cultivated Bombyx mori mulberry silk, the density of approximately 1.3 g/cm3 and average diameter of fiber was 11 μm) were collected from an Indian silk mill. VE resin (viscosity: 150 cps and specific gravity: 1.03), and its hardener methyl ethyl ketone peroxide (MEKP) and promoter cobalt naphthalate (CN), and Teflon sheets were purchased from CCP Composites, Korea. Deionized (DI) water was used for all experiments. All materials and solvents were used without further purification.
Preparation of the bio-flame-retardant liquid
5% of chitosan solution was prepared using 2% acetic acid under magnetic stirrer (600 r/min) (Misung Scientific Co Ltd, Korea) at 60°C. Stirring was continued for 12 h until the solution became yellow, after which 5% PA was added, and stirring was further continued for 3 h. The resulting BFL solution can be used for further experiments.
Fiber surface treatment
The lignocellulosic fibers (kenaf fibers) (300 × 300 mm) were initially treated with 2 wt.% NaOH for 30 min at room temperature (∼25°C) to prepare it for further FR treatment by removing the impurities and partially removing the weakly bound lignin.
3
The three types of fibers (lignocellulosic, animal, and cocoon) were maintained in a vacuum oven at 60°C. The fibers were then soaked in the BFL for 30 min, dried for 24 h at room temperature, and dried in a vacuum oven for 6 h at 40°C. The nature of treated fabrics is almost identical, with less flexibility than before and the weight of the treated fibers increased around 7% compared with untreated fibers. The overall schematic of the treatment process is shown in Scheme 1. Schematic of natural fibers treatment with the bio-flame-retardant liquid.
Composite manufacturing
The VARTM process is used to manufacture the composites, as discussed in a recently published article. 2 Initially, a Teflon sheet was attached to the aluminum mold for easy release of the cured composites. Silane tape was attached around the perimeter based on composite size (300 mm × 300 mm). The inlet and outlet hoses were attached at the center opposite to each other for uniform resin distribution. The required amount of fibers (3 fabric layers and the fiber % almost 30±3 wt.) was placed in the mold and covered with a peel ply, a flow net, and finally, a vacuum bag. The outlet pipe was connected to a vacuum pump and maintained at a constant pressure of approximately 0.03 MPa. The inlet pipe was placed in the prepared resin reservoir (the following percentage was used to manufacture the composites: VE resin:MEKP:CN = 100:1:1). After the completion of resin infusion, the fabricated composite was pre-drying at ambient temperature for 24 h and post-drying at 60°C for 6 h in a curing oven. The fiber volume fraction and composites thickness information are presented in the supplemental section Table S1 and the digital pictures of VE_TKF, VE_TWF, and VE_TSF composites panels are shown in Figure S1. A similar procedure was followed for the fabrication of the pristine panel. A composite cutting machine was used to prepare the specimens for the required tests according to specified standards.
Characterization and testing
FTIR spectroscopy: Spectral analysis was performed using the FT/IR-6300 spectrometer (JASCO International Co Ltd, Japan) in the wavenumber range of 400–4000 cm−1.
SEM–EDX analysis: The surface morphology was studied using a low-voltage scanning electron microscopy (LV-SEM) apparatus (TESCAN, LYRA 3 XM, Czech Republic) at an accelerated voltage of 5–30 kV coupled with an energy-dispersive X-ray spectroscopy analyzer (EDX, D8 Discover, Bruker Corp, USA). All samples were sputter-coated with gold using an automated fine coater (JEOL JFC-1600).
TGA: A thermogravimetric analyzer (STA 6000, PerkinElmer, USA) was used to examine the thermal degradation behavior in the temperature range of 30–700°C at a heating rate of 20°C/min under a nitrogen atmosphere according to ASTM E1131 standards. The mass of the KF, TKF, WF, TWF, SF, and TSF are 10.1, 10.2, 10, 9.9, 10.1, and 10 mg, respectively, was used for the analysis.
MCC: MCC was carried out by following ASTM D7309 standards in an FAA Micro Calorimeter (Federal Aviation Administration, FTT Fire Testing Technology Ltd, UK (Model No. 11,311)), and the obtained results were used to calculate the heat release rate (HRR) and total heat release rate (THR). The mass of the KF, TKF, WF, TWF, SF, and TSF are 7.73, 7.98, 7.3, 7.11, 10.72, and 10.42 mg, respectively, was used for the analysis.
UL-94 test: The fire test was carried out according to ASTM D3801 standards using a UL 94 test chamber (Fire Testing Technology, UK) based on recent literature. 21 The size of the samples was 130×13×3 mm. A 20 mm yellow-tipped flame of a Bunsen burner was used to illuminate the specimens.
Cone calorimeter test: Cone calorimeter tests were used to estimate flame retardancy according to ASTM E1354. Tests were performed in FTT Dual Cone Calorimeter (Fire Testing Technology ltd, United Kingdom). The sides and bottom of the specimens with dimensions of 100 mm × 100 mm × 3 mm were wrapped with aluminum foil and horizontally exposed to a heat flux of 50 kW/m2.
Results and discussion
Fiber properties
The surface features of the modified natural fibers were confirmed by spectral, morphological, thermal, and flame analyses and the obtained results are discussed in the following sections.
Infrared spectroscopy analysis
Figure 1(a) shows the effect of homogeneous hydrolysis and phosphorylation between chitosan and PA. The spectral peaks of base compound chitosan were exhibited at 1637, 1549, 1462, 1415, 1376, 1154, 1073, and 1023 cm−1 corresponding to C=O stretching, NH2 bending, –CH bending in side chain, –CH2OH bending, C–N amide symmetric deformation, C–O–C stretching, and free amino group at C2, respectively.
22
Similarly, phosphoric acid showed spectral peaks at 1634, 1171, and 1001 cm−1 corresponding to H2O stretching vibrations, in-plane P–O–H deformation vibration and P=O stretching, and PO stretching, respectively.
23
Spectral analysis of (a) BFL and (b) BFL-treated natural fibers. BFL: bio-flame-retardant liquid.
In the case of the phosphorylated chitosan, BFL exhibited almost all peaks of chitosan and phosphoric acid. In addition, there are few new peaks at 1389, 1263, and 943 cm−1, corresponding to stretching vibrations phosphate, and N-H moiety of chitosan, which may represent the formation of hydrogen bonds between the amino group of chitosan and phosphate group of PA. One hydrogen atom of the amino group is substituted with the phosphate group, resulting in the formation of a new P–O bond. 24
The spectral peaks of the untreated (KF, WF, and SF) and corresponding BFL-treated fibers (TKF, TWF, and TSF) are shown in Figure 1(b). The TKF spectrum exhibited the spectral peaks at 1236–1238 (C–O stretching), 1314 (CH2 stretching for cellulose), 1424 (CH2 bending for cellulose), 1461 (asymmetric bending in CH3 for lignin), 1641 (C=O stretching of the acetyl group or ester linkage of the carboxylic acid group), 1707 (hemicellulose in C=O stretching), 2916 (C–H stretching), and 3200–3400 cm−1 (O–H stretching vibration of cellulose). The peaks at 981 and 1374 cm−1 indicated the presence of the BFL. The appearance of new peaks at 1125 and 1529 cm−1 confirmed the intermolecular interactions between O–H and PO32−, proving the FR properties of the prepared BFL. 25
The TWF spectrum showed the following spectral peaks: 1021 (S–O vibration), 1226 (in-phase combination of C–N stretching and N–H in-plane bending), 1448 (free carboxyl group), 1532 (N–H band for amide II), 1641 (C=O stretching vibration for amide I), 2919 (carboxyl group), 3057 (hydroxyl group), and 3265 cm−1 (amide A band). 26 A new peak was observed at 1155 cm−1, confirming the formation of a peptide bond between the N–H of phosphorylated chitosan and amino acids in WF. Thus, this interaction may also be responsible for the FR properties.
TSF exhibited several new peaks at 971, 1092, 1149, and 1457 cm−1, attributed to the displaced BFL after treatment with silk. Furthermore, the peaks for β-sheets occurred at 1621 (amide I), 1448 (amide II), and 1228 cm−1 (amide III), and hydrogen bonds were formed. Amino acids, such as serine (1064 cm−1), valine (1410 cm−1), and tyrosine (1515 cm−1) established peptide bonds, ensuring strong interaction between the phosphorylated chitosan and SF, affording significant FR properties.
27
Scheme 2 shows the possible chemical interaction mechanisms and molecular interactions between BFL and KF/WF/SF based on the FTIR spectra. Proposed synthesis mechanism of the molecular interaction between BFL and natural fibers. (C.1) Acid hydrolysis of chitosan using acetic acid, (C.2) phosphorylation of acetylated chitosan, (C.3) peptide bond formation between silk/wool and the BFL, (C.4) hydrogen bond formation in the β-sheet, and (C.5) covalent bonding between the BFL and kenaf fibers. BFL: bio-flame-retardant liquid.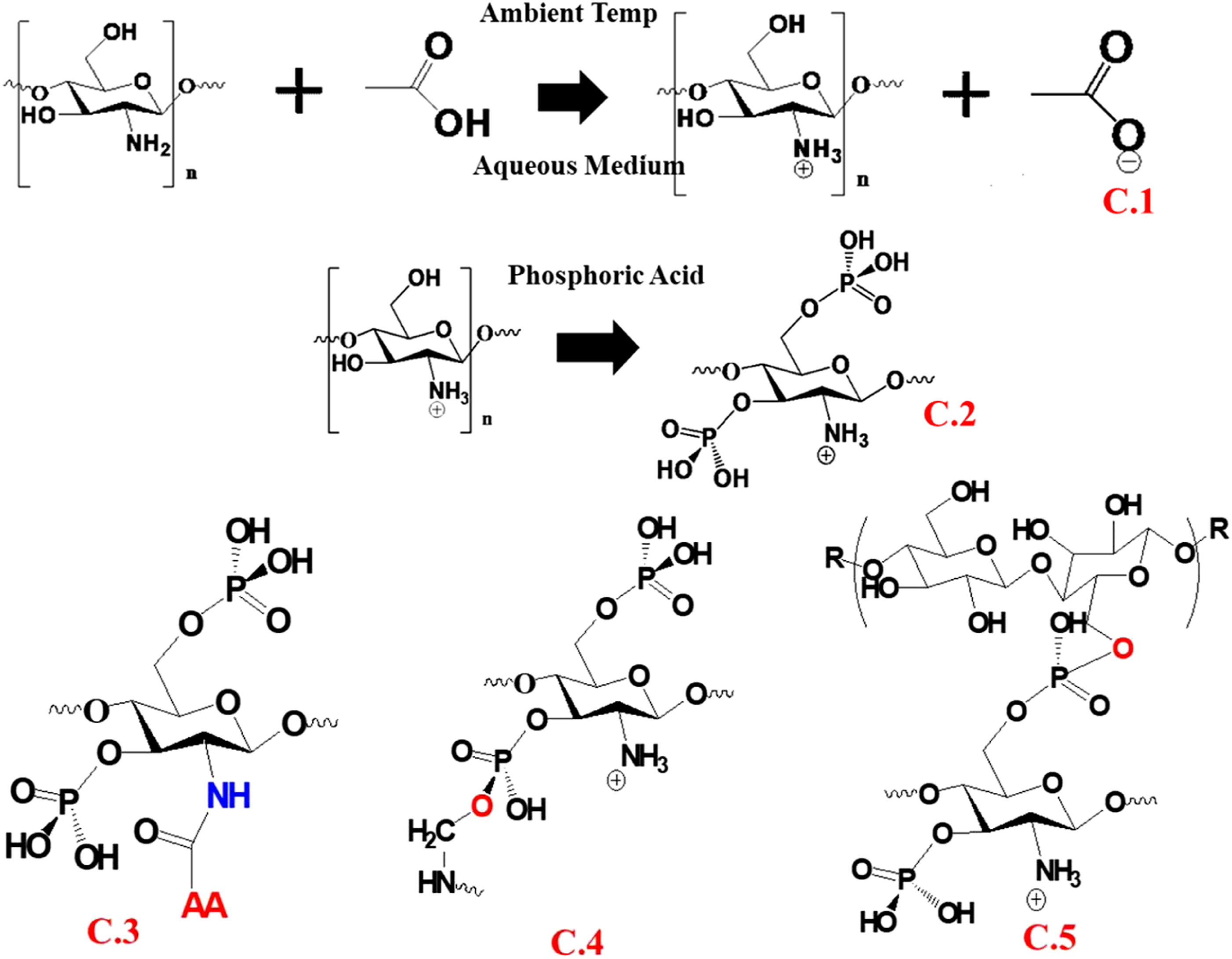
Surface morphology
The surface micrographs (×2500 magnification) of untreated and BFL-treated natural fibers are shown in Figure 2. The untreated KF exhibited a rough surface with distinct grooves between single fiber bundles, whereas the morphology of TKF was altered entirely, as observed from the distinct grooves coated by the treated compound. In WF, cuticles were observed on the fiber surface. However, TWF exhibited a coating on the fiber without any structural changes, rendering it potentially suitable as a reinforcement. A reinforcement can improve the mechanical strength of the composite by enhancing the bonding interaction with the matrix. Similarly, the morphology of raw SF revealed fibroin as a smooth single fiber with sericin acting as a binding agent that sticks to fibroin during spinning.
28
Typically, sericin is removed via degumming during manufacturing. However, waste SF was used in this study, and sericin was partially removed, as revealed in Figure 2. In contrast, TSF possessed the same smooth fibroin structure, but the grooves between the fibers were occupied due to the treatment. Scanning electron micrographs, energy-dispersive X-ray profiles, and elemental mapping results for treated and untreated natural fibers.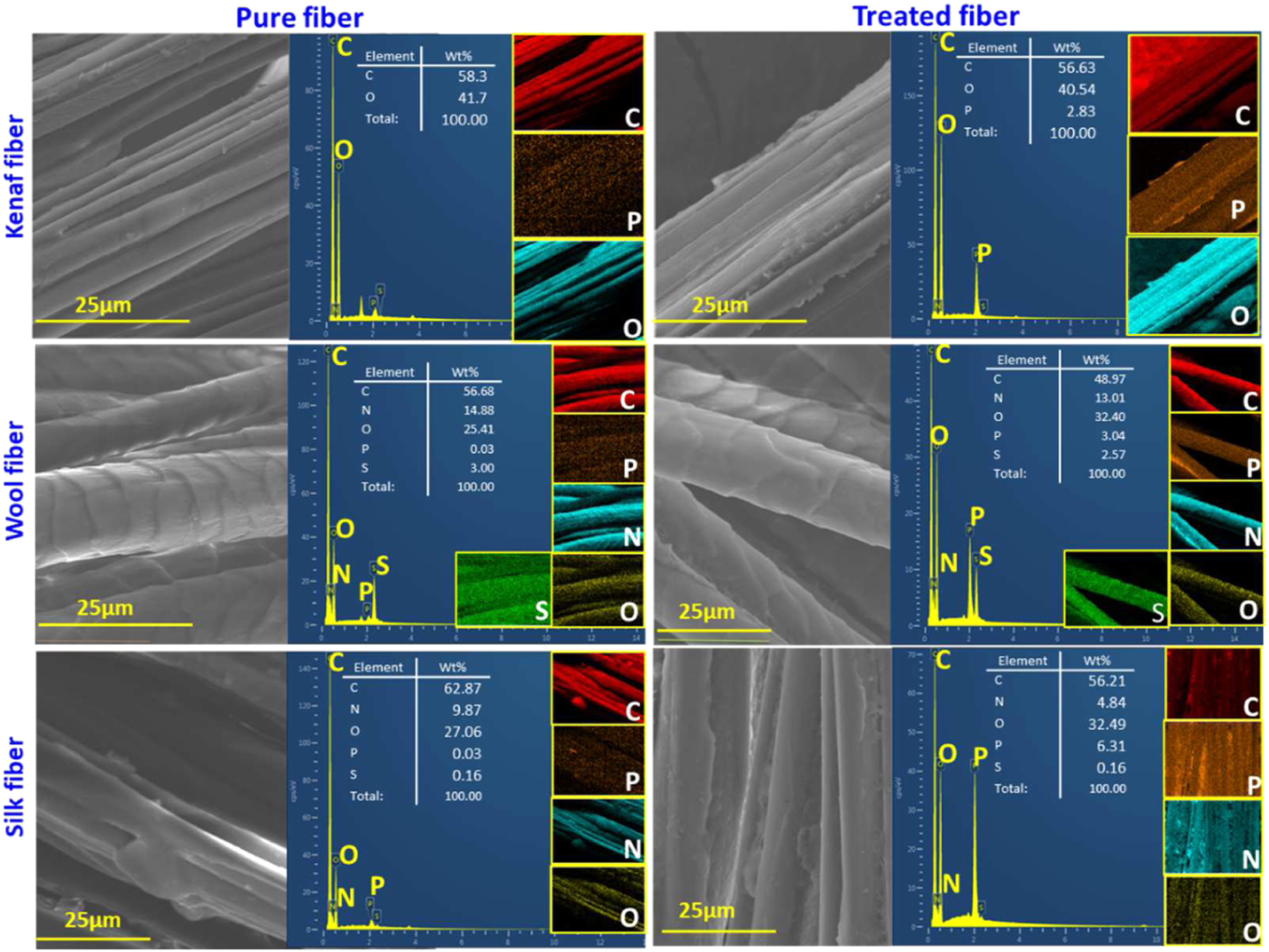
The EDX mapping was performed to study the uniformity of the BFL treatment on the fibers. The respective EDX profiles and data (wt%) for the main detected elements are presented in Figure 2. The profile for the treated natural fibers exhibited peaks for phosphorus (P), nitrogen (N), carbon (C), and oxygen (O), which are predominant in wool and silk because protein fibers inherently consist of these elements. Furthermore, protein fibers can absorb more BFL owing to hygroscopicity. 29
Thermal analysis
TGA of the untreated and BFL-treated fibers was performed under a nitrogen atmosphere, and the results are shown in Figure 3 Thermogravimetric analysis curves of untreated and bio-flame-retardant liquid-treated natural fibers (in N2 atmosphere).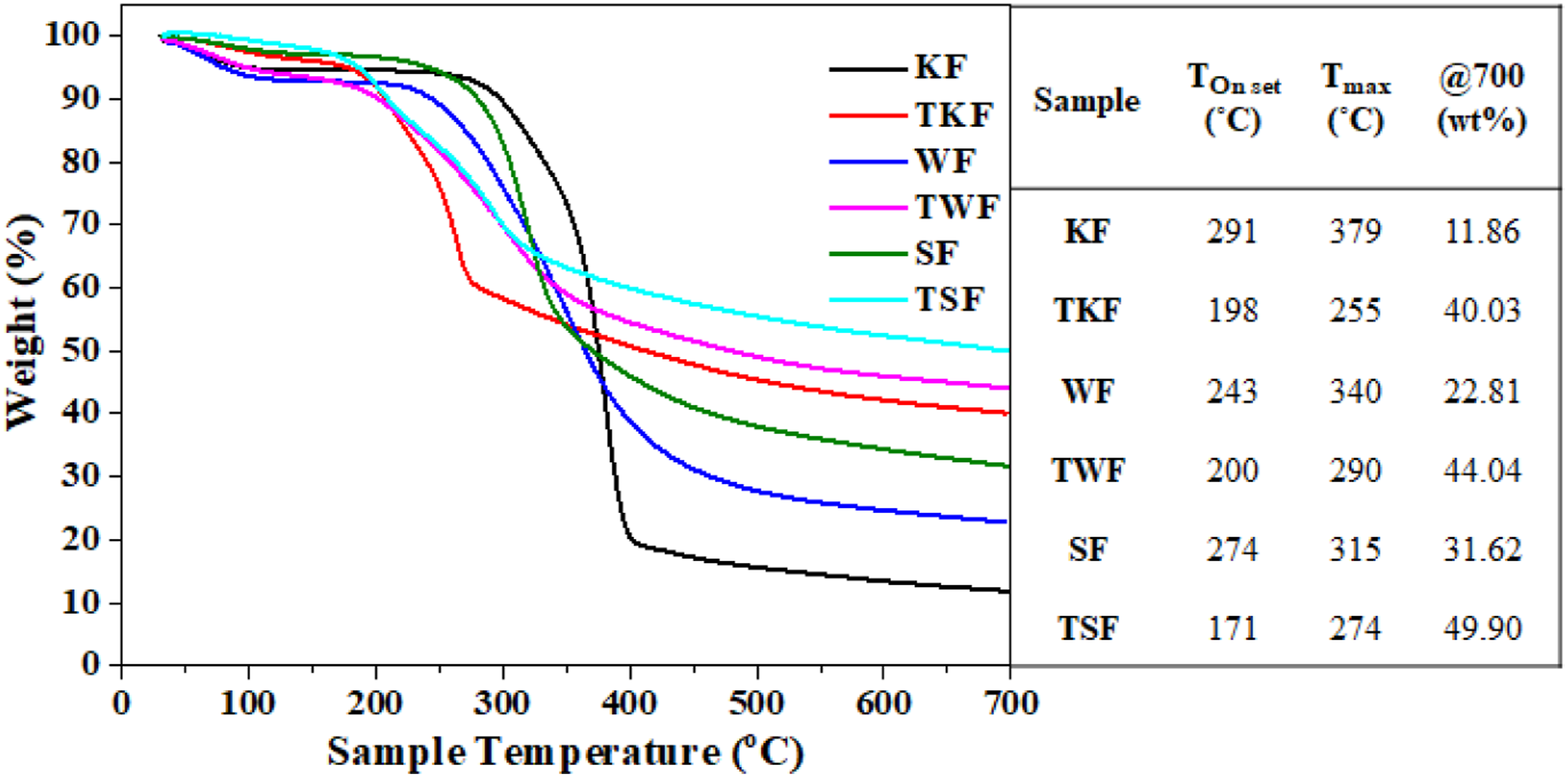
However, BFL treatment predominantly influences the Tonset of natural fibers and accelerates degradation to temperatures between 171 and 340°C. This may be due to the presence of PA, which tends to decompose at earlier stages. Further, the chitosan polymer chain breaks and forms phosphorous-containing char, which acts as a protective layer that significantly increases the thermal stability of the fibers. 18 Moreover, phosphorous-containing compounds interact with KF and form a shielding barrier for mass loss and heat transfer. 31 In addition, all the samples reinforced with treated fibers showed similar decomposition behavior, even at high temperatures. The mass-loss rate decreased considerably for all treated fibers. Consequently, the weight retentions of TKF (40.03%), TWF (44.04%), and TSF (49.90%) increased by 238%, 93.1%, and 57.8%, respectively, compared with the untreated fibers.
Fire analysis
The effect of BFL treatment on the flammability of natural fibers was determined by carrying out the UL-94 test using a 20 mm yellow-tipped Bunsen burner flame. The results of the test, as well as the digital images before and after the test, are shown in Figure 4(a). Due to the flammable nature of KF, WF, and SF, the specimens burned during the test. Moreover, the KF fabric showed an afterglow (103 s) after removing from the flame. At the same time, protein-based fibers exhibited dripping. Dripping is not typical of SF and WF; however, the char density is not sufficient to adhere to a relatively thin fiber bundle. The treated fibers exhibited excellent self-extinguishing ability and reached V-0 ranking in the UL-94 testing standard. TKF, TWF, and TSF exhibited distinct FR properties because PA promotes the formation of a thermally stable char layer, which inhibits the transfer of heat and oxygen (Figure 4(b)). At the same time, the nitrogen produced by chitosan dilutes the combustible gases and decreases the influence of fire.
32
(a) Digital images of vertical burning test (VBT) test and (b) VBT results for untreated and treated natural fibers after 10 s of flame exposure (in air atmosphere).
Micro-combustion calorimetry analysis
The peak heat release rate (pHRR) and THR of the untreated and BFL-treated fibers were measured by MCC analysis, and the obtained data are shown in Figure 5 Heat release rate curves of untreated and bio-flame-retardant liquid-treated natural fibers (in O2+N2 atmosphere).
Treated-fiber-reinforced vinyl ester composite
Thermal analysis
The degradation behavior and thermal stability of the untreated and BFL-treated natural fibers-reinforced VE composites were studied by TGA under a nitrogen atmosphere (Figure 6(a) and Table 1). The TGA curves show the different decomposition patterns of the samples. All samples exhibited very low weight loss in the range of approximately 30–300°C due to the loss of trapped moisture. Table 1 presents the values of Tonset (onset degradation temperature), Tmax (maximum degradation temperature), and residual weight at 700°C for different samples. The final residue content at 700°C, Tonset, and Tmax of VE were 3.6%, 394°C, and 453°C, respectively, owing to disintegration by the elimination of hydroxyl, CO2, as well as aliphatic (such as CH2) and aromatic carbon chains. However, the natural fiber-reinforced composites exhibited different degradation patterns. (a) Thermogravimetric curves (in N2 atmosphere) and (b) burning time and rate of VE, untreated, and treated-natural fibers-reinforced VE composites (in air atmosphere). UL 94 test results of vinyl ester, untreated and treated natural fiber-reinforced VE composites (in air atmosphere).

The TOnset and Tmax of VE_KF occurred at 366°C and 443°C, respectively, due to the decomposition of natural fiber constituents followed by the pyrolysis of the matrix, with a final residue content of 9%. However, VE_TKF underwent two distinct degradation stages in the temperature range between 180–270°C and 380–470°C. The total mass loss in the first stage accounted for ∼15%, at which PA phosphorylates the C (6) of the fiber and chitosan, promoting its dehydration and aromatic char formation. This char becomes thermally stable and acts as a barrier that shields the unburned composite material from heat. In the second stage, the previously formed char reduces the mass of volatile fragments by retaining carbon and hydrogen in the condensed phase. Furthermore, the resin degradation of VE_TKF was slower than that of VE_KF. This occurred because of the presence of thermally insulating bonds (P–O–C) and chitosan and PA moieties, which increased the thermal stability of the composite. 19 The final residue content increased from 9% for VE_KF to 18.7% for VE_TKF.
The gradual thermal decomposition of VE_WF at temperatures between 264 and 398°C was due to the rupture of the helical structure and cysteine disulfide bond of wool fibers. Subsequently, the VE matrix decomposed in one sharp step at 398°C. The total residue percentage at 700°C was 8.27%. However, the degradation of VE_TWF shifted to lower temperatures because the absorbed PA promoted the degradation of WF and chitosan and increased the speed of char formation between 210 and 420°C. This led to the formation of a significantly higher amount of final residue at 700°C (22.9 vs 8.27%, representing an increase of 176.9%). 37
The initial thermal decomposition of VE_SF occurred between 300 and 350°C, resulting in the cleavage of the peptide bonds and pyrolysis of the side chain groups in the amino acid, yielding char. The subsequent and main degradation occurred in the temperature range between 409 and 477°C with Tonset and Tmax values of 409 and 439°C, respectively, via the decomposition of the VE matrix and main SF chain. In contrast, the degradation of VE_TSF shifted to a lower temperature than that of VE_SF (from 300 to 285°C) owing to PA, which dehydrates both silk and chitosan. This dehydration promotes the formation of a thermally stable and dense char (14.5 and 23.4% for VE_SF and VE_TSF, respectively), providing adequate protection from the negative influence of temperature. 38
Flammability analysis
UL 94
The horizontal flame test was performed to determine the effect of the BFL-treated fibers on the flammability properties of the VE composites. The obtained data is presented in Figure 6(b) and Table 1. VE exhibited the highest burning rate (26.9 mm/min), indicating its flammable nature; however, VE_KF, VE_WF, and VE_SF showed burning rates of 19.5, 20.6, and 13.7 mm/min, respectively. Furthermore, VE_KF and VE_WF exhibited afterglow phenomena due to the release of combustion gases. The flame retardancy of the treated fiber-incorporated VE composites drastically increased, irrespective of the fiber. The combustion velocity decreased by 20% for both VE_TKF and VE_TWF, with 15.6 and 16.4 mm/min values, respectively. Predominant flame suppression and fire-extinguishing behavior were observed for the VE_TSF composite. In addition, the BFL treatment suppressed the afterglow phenomena in the KF- and SF-reinforced composites. As mentioned earlier, composites reinforced with SF (VE_SF and VE_TSF) present better FR performance than other composites. This may be due to the high protein content of SF compared to that of other fibers and the amount of absorbed BFL that contributes to the FR performance of the VE composite.
Cone calorimeter test
The inherent properties of the untreated and BFL-treated fiber-reinforced VE composites were investigated by forced flaming using a cone calorimeter, a widely used approach for assessing the fire behavior of polymer materials. The various flame retardancy parameters, such as pHRR, THR, mass loss rate (MLR), and maximum average heat rate emission (MAHRE), are presented in Figure 7 and Table 2. It is evident from Figure 7(a) that the pHRR values for the VE and VE_KF composite were the highest, at 871 and 861 kW/m2, respectively, confirming the low effectiveness of KF toward improving the FR properties of the composites. However, the pHRR of VE_TKF was 566 kW/m2, which is approximately 34% lower than that of VE_KF. The pHRR values of VE_WF and VE_SF were 552 and 512 kW/m2, respectively. The corresponding pHRR values for the treated fiber-reinforced composite were reduced to 461 and 388 kW/m2 for VE_TWF and VE_TSF, respectively. (a) HRR and (b) total heat release rate (THR) curves of VE, untreated, and treated-natural fibers-reinforced VE composites (In air atmosphere). Thermogravimetric and cone calorimeter test results of VE, and untreated and treated natural fibers-reinforced VE composites.

Figure 7(b) shows the THR curves of the VE, VE_KF, VE_TKF, VE_WF, VE_TWF, VE_SF, and VE_TSF composites with values of 139.9, 93.8, 70.1, 113.0, 78.8, 104.0, and 81.8 MJ/m2, respectively. Thus, composites reinforced with treated fibers yielded lower THRs during combustion. The MAHRE values of the VE_KF, VE_WF, and VE_SF composites were 379, 446, and 332 kW/m2, whereas those of VE_TKF, VE_TWF, and VE_TSF were 347, 370, and 293 kW/m2, respectively (Table 2). The values for the treated fiber-reinforced composite were lower by 8, 17, and 12% than those of the untreated analogs. This reduction in the MAHRE was due to the release of nonflammable gases from the BFL treatment, which reduced the volume of combustible volatile substances.
In addition, Table 2 reveals that the MLR of composites reinforced with untreated fiber is higher than that of the treated ones. The MLRs of VE_KF, VE_WF, and VE_SF were 34.0, 27.2, and 20.6 g/s.m2, respectively, whereas those of VE_TKF, VE_TWF, and VE_TSF were 28.2, 20.8, and 18.9 g/s.m2 with a corresponding reduction of 17, 24, and 8%, respectively. These trends may occur due to the formation of dense char during burning, which could effectively protect the undamaged material from flame propagation, thereby mitigating its volatility. Overall, the pHRR and mass loss of BFL-treated fibers-reinforced VE composites showing lower than treated fibers alone, the reason might be domination of VE resin, fiber volume fraction and removal of treated material during manufacturing process.
Char analysis
The SEM images of the residual char of the specimens after the HBT are shown in Figure 8. The char of VE is observed to be brittle and porous with low effectiveness in resisting fire spread. The SEM image of VE_WF shows a similar trend, wherein the char is brittle and uneven with holes, and fiber residue is observed. The char residues of VE_KF and VE_SF feature almost the same appearance, predominantly comprising fiber residues. The residues of these two samples are considerably less potent in resisting the spread of fire than VE_WF owing to the afterglow effect that occurs during burning. The SEM images of VE_TKF, VE_TWF, and VE_TSF exhibit considerably denser and intumescent solid char morphology, which acts as an effective barrier between the fire and unburned material. Scanning electron micrographs of untreated and bio-flame-retardant liquid-treated fiber composites and char microimages of respective composites after the horizontal burning test (air atmosphere).
Possible flame-retardant mechanism
The possible FR mechanisms of BFL-treated natural fibers based on literature, as well as TGA and LV-SEM, results are shown in Scheme 3. First, PA, ammonia, and water are released from the BFL during the initial stages of combustion.
39
Second, PA initiates the esterification of fiber constituents and chitosan, resulting in the formation of a phosphate ester that might react further with fiber or chitosan. This generates noncombustible and continuous residues that effectively protect the unburned material from the fire.
40
In addition, noncombustible gases, such as H2O, CO2, NH3, and SO2 (only for WF and SF), are produced from treated fibers, which decrease the concentration of flammable volatiles and improve the FR properties of natural fibers.
35
Plausible flame-retardant mechanism of the bio-flame-retardant liquid-treated natural fibers composite system.
Conclusions
The bio-flame retardant liquid (BFL) was successfully synthesized by utilizing low-cost chitosan and phosphoric acid. The BFL treatment significantly induced the flammability properties of various natural fibers, including KF, WF, and SF. The Spectral (FTIR) and morphological (SEM–EDX) analyses demonstrated the effective interaction and uniformity of BFL treatment on natural fibers. The thermal stability increased 237.5%, 93.1%, and 57.8% compared with untreated natural fibers, and similarly, the flame retardancy enhance 77.6%, 50.2%, and 73.4%. Furthermore, the treated fiber reinforcement into VE composites significantly improved the flame resistance. The treatment could effectively enhance the thermal stability of the VE composites by forming a dense char residue at 700°C, with weight retentions reaching up to 18.7%, 22.9%, and 23.4% for VE_KF, VE_WF, and VE_TSF composite, respectively. Similarly, the treated fiber reinforcement induced the flammability properties of VE composites by significantly suppressing the pHRR 34%, 16%, and 24% due to the presence of the BFL, which works in condensed and gaseous phases by providing stable char and an inert atmosphere. Moreover, the char residue microanalysis further supported the flame retardancy of treated-fiber-reinforced VE composites, revealing that the BFL promotes the formation of a dense char that arrests flame propagation. Overall, this investigation is an innovative and economical approach to obtain significant FR properties for natural fibers, thereby serving as the optimum reinforcement for FR polymer composites.
Supplemental Material
sj-pdf-1-jit-10.1177_15280837221079273 – Supplemental Material for Preparation of a novel bio-flame-retardant liquid for flame retardancy of natural fibers and their composites
Supplemental Material, sj-pdf-1-jit-10.1177_15280837221079273 for Preparation of a novel bio-flame-retardant liquid for flame retardancy of natural fibers and their composites by Li Maksym, M N Prabhakar and Jung-il Song in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science Education (grant numbers 2018R1A6A1A03024509 and 2021R1A2B5B03002355).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
