Abstract
Coconut oil (CO) is a naturally derived bio-oil which exhibits specific characteristics such as biocompatibility and antibacterial activity. In this work, the biological properties of poly(caprolactone)/gelatin (PCL/Gel) nanofibers are improved using CO encapsulation. This bio-oil was added to the PCL/Gel polymer solution with different concentrations (5–40%). Nanofibers were crosslinked using glutaraldehyde vapor. Different types of characterization techniques such as SEM, FTIR, DSC, tensile measurements, water contact angle, and water vapor permeability were used to study the chemical, physical, thermal, and morphological properties of resultant nanofibers. Results showed an average diameter of 300–370 nm for as-spun nanofibers, which increased to 360–470 nm after the crosslinking reaction. The presence of CO was confirmed using FTIR and DSC experiments. Moreover, results indicated that the presence of CO increases the hydrophilicity and water vapor permeability of nanofibers, which are desirable for their final application. Biological tests, such as antibacterial activity, cell viability, and cell morphology tests were performed to evaluate the possible application of the produced nanofibers for wound healing applications. Results indicated that the crosslinked PCL/Gel nanofibers containing 20% CO exhibited the highest cell compatibility and antibacterial activity against gram-positive (S. aureus) and gram-negative (E. coli) bacteria.
Introduction
Skin is the most extensive organ in the human body and is known as the first protective system against external threats. Skin can be damaged because of many different accidents or illnesses such as burning, scratching, and diabetes [1]. Wound healing is a long and time consuming process because of the different cell types that must be regenerated [2]. Recently, improvements in tissue engineering scaffolds have brought us alternatives to regenerate organ functionality [3]. The ideal scaffold for skin regeneration should exhibit several characteristics, such as biocompatibility, antibacterial properties, and cell attachment ability [4]. Besides, this scaffold should be similar to the extracellular matrix (ECM) and mimic their chemical, mechanical, and biological properties [1]. It is widely reported that nanofibers are great candidates for simulating the ECM properties. As a result, there are many research articles on nanofibers application in tissue engineering, drug delivery, and wound dressing because of their excellent properties, such as perfect permeability, high specific surface area, and small pore size [5,6]. Electrospinning has been the most popular technique for nanofiber production due to its simple, cost-effective, and versatile procedure [7]. This structure has been used for drug, herbal extract, growth factor, and cell encapsulation [8,9].
Nanofibrous scaffolds can be produced from natural biopolymers like gelatin, collagen, sericin, and hyaluronic acid, or synthetic biopolymers such as poly(caprolactone), poly(vinyl chloride), poly(acrylic acid), and poly(acrylamide) [10]. Gelatin is a natural biopolymer that is derived from collagen and exhibits beneficial properties for scaffold fabrication such as hydrophilicity, biocompatibility, biodegradability, inexpensiveness, and availability. Gelatin also improves cell attachment and proliferation when used in wound dressing applications [11]. Although this biopolymer has excellent biological properties, it possesses weak mechanical stability. Therefore, a combination of this natural biopolymer with another synthetic biopolymer will provide both biological features and mechanical stability to the final construct [12]. Poly(caprolactone) (PCL) is a semi-crystalline, linear aliphatic synthetic biopolymer, widely used in biomedical applications such as tissue engineering [13], drug delivery [5], and wound dressing [14] due to its biocompatibility, biodegradability, and mechanical properties [1]. It is reported that the combination of PCL and gelatin could provide all the fundamental properties of an ideal scaffold, but also, these constructs showed excellent in vivo results [15]. Chong et al. reported that antibacterial activity is an essential factor in skin tissue engineering. Therefore, they produced tetracycline encapsulated PCL/Gel nanofibers with excellent antibacterial activity [16]. Naseri Nosar et al. produced PCL/gelatin nanofibers and reported that the addition of the cerium oxide improves hydrophilicity, mechanical and antibacterial properties, and cell growth. Cerium oxide reduces the number of reactive oxygen species (ROS), leading to lesser cell death in the wound environment [17]. Herbal extracts are also loaded into PCL/Gel nanofiber. For instance, Ramalingam et al. have developed Gymnema sylvestre extract loaded hybrid PCL/Gel nanofibers. Their product showed better hydrophilicity, cell viability, and antibacterial activity [18]. Natural oil encapsulation is another approach to introduce specific characteristics to the tissue engineering scaffolds. A timeline summary of oil encapsulated scaffolds for biomedical applications is given in Table 1.
Timeline summary of oil loaded scaffolds for biomedical applications.

Coconut oil (CO) is a long fatty acid with antibacterial, antioxidant, antifungal, and anti-inflammatory properties [33]. It is composed of several materials such as lauric acid (antimicrobial and anti-acne), caprylic acid, oleic acid, stearic acid, and capric acid (antifungal and anti-inflammatory). Besides, CO is useful for Alzheimer’s disease, controlling blood sugar level, weight loss, and wound healing [34]. According to earlier studies, CO can help collagen binding and fibroblast proliferation [35]. Evangelista et al. used CO to heal skin eczema in children and observed that about 46% of patients were treated after eight weeks [36]. In another research, improved thermal stability, antimicrobial properties, and cell growth were observed for CO loaded gellan gum film [22]. In this research, CO loaded PCL/gelatin blend nanofibers are fabricated using the single nozzle electrospinning method. We believe the biological properties of nanofibers, such as antibacterial activity and biocompatibility, can be enhanced by CO encapsulation. Nanofibers containing different amounts of CO content were produced, and its effect on different properties of nanofibers was investigated. We hypothesize this newly developed nanofibrous scaffold could be used as a potential skin wound dressing.
Experimental
Materials
Polycaprolactone (MW = 70 kDa) and Glutaraldehyde were purchased from Sigma-Aldrich. Gelatin, acetic acid, potassium bromide, disodium hydrogen phosphate, and sodium hydrogen phosphate were obtained from Merck. CO was extracted directly from coconut fruit using an OPM 240 oil extractor and used without any purification. Tween 80/Polypropylene glycol (50/50) surfactant was provided from Goldaro Company.
Production of CO loaded PCL/gel nanofiber
First, 0.75 g gelatin and PCL were separately dissolved in 5 ml acetic acid aqueous solution (90% v/v) and mixed using a magnetic stirrer for 4 and 12 hours, respectively. These solutions were then mixed, and CO was directly added to the polymer solution at different contents (5-40% w/w based on the weight of polymer). Since Tween 80 has been used widely in biomedical applications [37,38], this surfactant (0.48-1.6% w/v) was added to the polymer-CO system for emulsion stabilization. The emulsion mixture was then transferred into a syringe (needle gauge 19), and the electrospinning process was performed according to the protocol reported by Heidari et al. [39] at 15 kV voltage, 1 ml/hour feed rate, 15 cm nozzle to the collector distance, 25°C and 65% relative humidity for 5 hours. A flat aluminum foil (thickness 1 mm) was used for collecting the produced nanofibers. Scaffolds made from gelatin are usually crosslinked to prevent their dissolution in the aquatic media. Therefore, produced nanofibers were crosslinked in a desiccator containing 10 ml of 25% glutaraldehyde solution for 24 hours. The white color of nanofiber membranes turned into yellow when reacted with glutaraldehyde. Crosslinked samples were finally kept in a fume hood for five days to remove the excess unreacted glutaraldehyde molecules.
Characterization
Polymer solution properties
The viscosity of CO loaded polymer solutions was measured using a Brookfield viscometer (Model DV-II Pro, USA) at 20 rpm rotational speed and 25°C. Surface tension was investigated using a Kruss K100 tensiometer at 25°C. This experiment was performed based on Wilhelmy plate method [40].
Scanning electron microscope (SEM)
Nanofiber morphology was examined using scanning electron microscopy (SEM) before and after the crosslinking process. A 1 cm2 sample was cut from each nanofiber membrane and mounted on an SEM sample holder using a conductive carbon tape. Specimens were then installed in a Bal-tec apparatus and sputter-coated with a thin layer of gold. SEM images were recorded via a Philips XL30 electron microscope at 15 kV accelerating voltage. The average diameter and pore size dimension of prepared nanofibrous membranes were determined using Digimizer and ImageJ softwares.
Fourier transform infrared spectroscopy (FTIR)
Nanofiber samples before and after crosslinking were ground into small pieces using a ceramic porcelain pestle and mortar. They were then mixed with potassium bromide salt under infrared radiation. The resultant mixture was converted into a solid tablet under pressure. These tablets were transferred into a Thermo Nicolet NEXUS 670 apparatus, and FTIR spectra were recorded at a wavenumber range of 400–4000 cm−1.
Thermal analysis
The thermal behavior was studied using the differential scanning calorimetry (DSC) technique. About 5 mg sample was detached from the electrospinning collector and placed in a sealed DSC pan. The pan was then transferred into a DSC 2010 TA instrument and heated from −20°C to 100°C at a 10°C/min heating rate. Samples were then kept at 100°C for 10 minutes to erase the polymer’s thermal history. In the end, they were cooled down to −20°C at 10°C/min cooling rate. All the thermal studies were performed under a nitrogen atmosphere.
Tensile properties
Mechanical properties of nanofiber membranes (before and after crosslinking) were tested using an Instron 5566 Universal Testing Machine. Nanofibers membranes were cut into 0.5 × 3 cm2 strips. The test was repeated 6 times for different sites of each nanofiber membrane. Stress-strain curves were recorded at a 10 mm/min crosshead speed and ambient condition (25°C, 65% relative humidity). Finally, tensile strength, tensile stress, and young’s modulus were extracted from stress-strain curves.
Water contact angle measurement
The water contact angle was measured to investigate the hydrophilicity of webs before and after crosslinking. Samples were mounted on a smooth holder, and a water droplet (≈5 µl) was placed on the nanofiber membrane’s surface. A PC conjugated Sony camera (model SSC-DC318P) recorded all these events. Images were captured at the first moment of the water droplet injection, and the contact angle was measured using Digimizer image analyzing software. This experiment was repeated five times for each specimen.
Water vapor permeability measurement
This test was performed according to the ASTM E96-00 standard test method. Nanofiber membranes were cut into 2 × 2 cm2 specimens, and their thicknesses were measured. They were mounted on glass bottles with 1.3 cm diameter and containing 7 ml of distilled water. Also, the control sample was a glass bottle without any covering. Then, all glass bottles were weighed and placed in a desiccator containing 20 ml of saturated ammonium sulfate solution, and the experiment was performed at 37°C and 65% relative humidity. Finally, bottles were weighed after 24, 48, and 72 hours and the water vapor transport (WVT) at each time point and water vapor permeability (WVP) was calculated according to equations (1) and (2) [41,42]. This test was repeated three times for each sample

Biological properties
In-vitro degradation
PCL/Gel and PCL/Gel/CO 40% before and after crosslinking were selected for in vitro degradation evaluations. Samples were cut into 3 × 3 cm2 pieces, then immersed in 10 ml PBS solution (pH = 7.4). They were then incubated in a 37°C water bath for 1, 14, and 28 days. The degradation media (PBS solution) was refreshed every week. Samples were collected at each time point, rinsed several times with distilled water to remove absorbed PBS solution. Samples were finally dried at room temperature overnight, and SEM images were recorded for morphological studies.
Antibacterial properties
Antibacterial activity of webs was tested quantitatively according to the ASTM 2149-01 standard method. This experiment was performed based on the broth dilution method and against gram-negative (Escherichia Coli) and gram-positive (Staphylococcus Aureus) bacteria. Briefly, nanofiber membranes were cut into 1 × 1 cm2, exposed to UV light for 15 min, and placed in sterilized tubes, including bacterial suspension (4.0 × 103 cfu/ml) followed by incubation at 37°C for 16 hours. They were then plated on Mueller-Hinton agar and incubated at 37°C for 18 hours. Subsequently, antibacterial activity was calculated using the number of colonies that grew on each plate (equation (3)) [43]
MTT assay (extraction method)
The viability of 3T3 fibroblast cells on nanofiber membranes was exanimated by colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay according to the ISO 10993-5 standard test method. Nanofiber membranes were first sterilized via UV light exposure for 2 hours, followed by submersion in 70% ethanol for 30 min. Then they were cut into 6 cm2 specimens, and 1 ml cell culture medium was added and incubated for 1, 3, and 7 days at 37°C, 95% relative humidity, and 5% CO2. At each time point, 90 µl of the incubated cell culture medium and 10 µl fetal bovine serum (FBS) was added to a 96 well cell culture plate. Then, 100 µl cell culture media containing 5000 3T3 fibroblast cells was added to each well and incubated at 37°C, 95% relative humidity, and 5% CO2 for 24 hours. The old media was then replaced with 100 µl of 0.5 mg/ml MTT solution. After incubating for 30 min, each plate's contents were substituted with 200 µl dimethyl sulfoxide (DMSO). Finally, the number of viable cells was recorded by an Eliza Reader at 570 nm. The fresh cell culture media without nanofiber was used as the control sample. The cell viability percentage calculated using equation (4) [44]
Cell morphology
Nanofiber membranes were cut into 1 × 1 cm2 squares and washed several times to remove the excess glutaraldehyde. Then, they were sterilized via UV light exposure and washing with 70% ethanol for 30 min. Samples were placed in a 12 well cell culture plate, followed by adding 4 ml of 4 × 104 3T3 fibroblast cells. Cell culture plate incubated for three days, at 37°C, 95% relative humidity, and 5% CO2. The media was then removed, and cells were fixed with the addition of 4 ml 2.5% glutaraldehyde aqueous solution. After 2 hours, the glutaraldehyde solution was removed, and samples washed successively with 50%, 60%, 70%, 80%, 90%, and 100% ethanol. Samples were dried in an oven at 37°C, and SEM images of cells on nanofiber membranes were recorded.
Statistical analysis
Average values and standard deviations are presented as (mean ± s.d.) for all reported data. Statistical analyses were performed using SPSS software. One-way ANOVA has been used for statistical analysis, and the Tukey post hoc test was used to identify groups with significant differences. Results with a p-value of less than 0.05 were considered significant.
Results and discussion
Nanofiber characterization
Polymer solution properties
Electrospinnability and the diameter of resultant nanofiber are correlated to the viscosity of polymer solution. The viscosity of polymer solutions containing different amounts of CO is reported in Figure 1. The viscosity was decreased with increasing the amount of CO. This phenomenon could happen because of the nonhomogeneous medium after CO addition, which is dispersed as suspended oil droplets in the solution. This led to reduced entanglements and viscosity drops from 200 cP to 175 cP when only 10% of CO was added to the polymer solution. At higher CO contents, the viscosity decreases at a slower rate. We believe the number of oil droplets remains almost constant after a critical point, and bigger droplets will be formed. Moreover, CO's viscosity was 55 cP [45], which is lower than PCL/Gel polymer solution, and this could be another reason for reduced viscosity at higher CO loading contents, and the solution wants to reach an equilibrium between two fluids’ viscosity. These findings were in agreement with earlier research by Figueroa-Lopez et al. that reported a decrease in viscosity of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) solution by adding the essential oil [46].
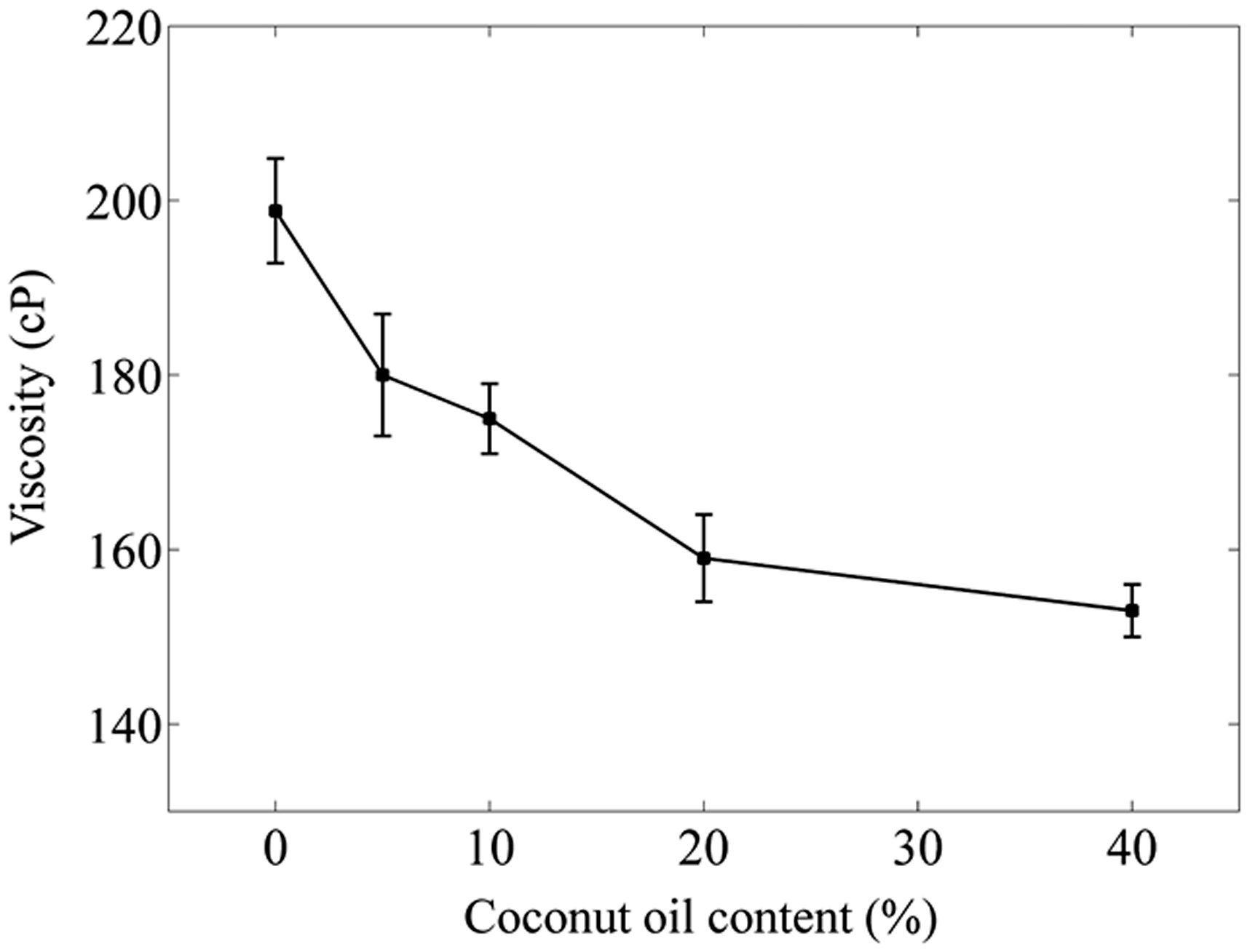
The viscosity of PCL/Gel solutions containing different amount of CO.
Surface tension is the other polymeric solution property that plays an essential role in the electrospinning process. This factor has been measured for different samples, and its results are presented in Table 1. Obtained results for PCL and gelatin polymer solutions were in agreement with earlier reports. Gomes et al. reported surface tensions of 34 and 33 mN/m for gelatin and PCL dissolved in acetic acid, respectively [47]. Also, Salles et al. measured a surface tension of 30–40 mN/m for gelatin polymer solution depending on the pH of the acetic acid aqueous solution [48]. The surface tension measured for CO was close to an earlier article by Chumpitaz et al. that reported a surface tension of 32.8 mN/m for pristine CO [49]. Results also indicate a decreasing trend with increasing CO contents. It should be noted that higher oil content requires a higher amount of surfactant to obtain a stable polymer-CO emulsion. Therefore, in accordance with Sahlan et al. findings, the decrease in surface tension happens due to a higher surfactant in solution [50]. Rafiq et al. also observed the same behavior with the addition of clove oil, lavender oil, and cinnamon oil into the PVA/alginate solution [51].
Morphology of nanofibers
SEM images of nanofibers were captured for morphological evaluations, and their results are shown in Figure 2 and Table 2. Obtained results indicated that the electrospinning process is adjusted appropriately, and produced nanofibers are uniform, smooth, and without any beads. The average diameter decreases with the addition of CO. This phenomenon can be supported by reduced solution viscosity and surface tension. The decrease in viscosity, or in other words, lesser polymer chain entanglements, facilitate polymer solution sliding and results in finer nanofibers. Also, in earlier studies, researchers have pointed out that CO increases the solution conductivity, and therefore stronger elongation forces will be applied to the electrospun polymer, and finer nanofibers will be formed [27].

SEM images of nanofibers before (top) and after crosslinking (bottom) for pure PCL/Gel (a) and PCL/Gel nanofibers loaded with 5% (b), 10% (c), 20% (d), and 40% (e) CO.
Surface tensions (mean ± SD) for different samples based on the Wilhelmy plate method.

SEM images of crosslinked nanofibers suggested that the reaction time is appropriate because the nanofibrous structure has been preserved. The average diameter of crosslinked nanofibers increased due to swelled nanofibers and the interconnection of adjacent fibers during this reaction. Moreover, the average pore size (between the electrospun fibers) obtained from image analysis results (Table 2) shows that the presence of CO does not significantly affect the porosity while crosslinking reaction has decreased the pore sizes, which could be attributed to the swelling phenomenon.
FTIR spectroscopy
FTIR spectroscopy was used to confirm the presence of CO in nanofibrous webs. Spectra of CO, pure PCL/Gel nanofibers, and 20% loaded nanofiber sample are shown in Figure 3. Characteristic peaks of PCL were observed at 2927 cm−1 (asymmetric C–H stretching sp3), 2859 cm−1 (symmetric C–H stretching sp3), 1724 cm−1 (C=O stretching ester bond), 1251 cm−1 (C–O and C–C stretching), 1238 cm−1 (asymmetric C–O–C stretching), and 1173 cm−1 (symmetric C–O–C stretching) [1]. Functional groups of gelatin were detected at 3251 cm−1 (N–H stretching of amide), 1653 cm−1 (C=O stretching of amide I), 1521 cm−1 (N–H bending of amide II), and at 669 cm−1 (N–H out-of-plane bending) [11]. The FTIR spectrum of CO exhibited several characteristic peaks at 2922-2853 cm−1 (C–H stretch sp3), 1743 cm−1 (C=O stretching ester), 1471 cm−1 (C–H out-of-plane bending), and 1151 cm−1 (C–O stretching ester) [27]. According to FTIR results, the C=O stretching peaks in PCL/Gel and CO spectra were detected at 1724 cm−1 and 1743 cm−1, respectively, while this peak was broader in the FTIR spectrum of the CO loaded nanofiber with a maximum ranging from 1724 cm−1 to 1743 cm−1. Additionally, the observed shoulder peak at 2954 cm−1 of CO was observed in the oil loaded sample's FTIR spectra. Thus, FTIR results confirmed the presence of CO in the nanofiber structure.

FTIR spectra of CO, pure and 20% CO loaded PCL/Gel nanofibers(top), and PCL/Gel nanofibers before and after crosslinking (bottom).
The FTIR spectrum of crosslinked PCL/Gel nanofiber is reported in Figure 3. Spectra were normalized to the peak at 1187 cm−1, which is related to the C–O stretch and remains intact through the crosslinking reaction. The aldehyde functional groups of glutaraldehyde react with amine functional groups of gelatin during the crosslinking reaction. This reaction results in the addition of imine functional groups. Thus, the decrease in amine bond’s intensity at 3251 cm−1 and increased intensity of the imide bond’s peak at 1653 cm−1 overlapped with C=O stretching of amide shows the successful crosslinking reaction [52].
Thermal analysis
Differential scanning calorimetry (DSC) was performed on pure PCL/Gel, and 40% CO loaded samples to evaluate the effect of oil on the thermal behavior of nanofibers. Diagrams of the first heating and cooling cycles from −15°C to 80°C are shown in Figure 4. The PCL/Gel sample showed melting and crystallization points at 60°C and 29°C, respectively [53]. The gelatin portion in this sample does not show any phase changes, and these peaks are attributed to the melting and crystallization points of PCL. The CO sample showed a melting point at 25°C and a freezing point staring at 3°C with two exotherm maximums at 0°C and −6°C. These maximums are attributed to the difference in crystallization points of molecules with different molecular weight or structure [54]. In the CO loaded sample, two peaks were added to DSC diagrams, one melting point at 25°C (heating cycle) and a crystallization point at about 3°C (cooling cycle) [55]. These peaks are related to the melting and freezing points of CO. Also, CO has decreased melting and crystallization temperatures of PCL to 59°C and 25°C, respectively.

DSC diagrams for the first heating and cooling cycle of CO, PCL/Gel, and PCL/Gel/CO 20% nanofiber samples.
Moreover, nanofiber's melting enthalpy decreased from 32 J/g to 28 J/g for CO loaded PCL/Gel nanofibers. These observations show the formation of a semi-interpenetrated system. The melting enthalpy of CO loaded PCL/Gel at 25°C was about 15 J/g; according to the melting enthalpy of CO (125 J/g), it can be concluded that 12% CO was encapsulated into nanofibers. Therefore encapsulation efficiency in this study was about 60%.
Tensile properties
The mechanical property of the nanofiber membrane is an important parameter in medical applications. A suitable wound dressing should be flexible and, at the same time, should exhibit good strength. The modulus and tensile strength of scaffolds are recommended to be close to the skin modulus (60–70 Mpa) and strength (17–21 Mpa) [56]. In this study, the mechanical properties of CO loaded PCL/Gel nanofibers before and after crosslinking are investigated.
According to Table 3, the modulus and tensile strength are increased when CO was added to nanofibers with concentration up to 10%, but for higher oil contents (20% and 40%), the trend was vice versa. These results were similar to Mani et al. findings; they reported an increase in the tensile strength of vegetable oil loaded polyurethane nanofibers. This phenomenon happens due to reduced nanofibers' diameter, leading to more fibers in the web’s cross-section per unit area. Thus, webs can resist more robust forces [57]. Additionally, because of the elongation forces during the electrospinning process, polymer chains are highly aligned in finer nanofibers. This means more polymer chains are parallel to the nanofiber’s axis, which improves their tensile properties. Unnithan et al. also reported an increase in mechanical properties of Emu oil loaded polyurethane nanofibers. They suggested that oil adhesion properties and the hydrogen binding between polyurethane and oil prevent tearing of polymer chains and result in increased mechanical properties [58]. Zamani et al. reported the same result for PCL/PEG/Emu-oil system [28].
The average diameter and pore size (mean ± SD) of PCL/Gel nanofibers containing different amounts of CO.

On the other hand, the presence of oil droplets in the nanofibers at higher CO contents (20% and 40%) might act as stress concentration zones and reduce the tensile strength. The other reason could be attributed to the formation of the semi-interpenetrated system, which can decrease polymeric systems' mechanical properties. Obtained results were analogous to Jaganathan et al., findings on electrospun corn, neem, and olive oil loaded polyurethane webs [30,59]. Although we expected an increase in the elongation at break in CO loaded samples due to the plasticizing effect, no relationship was observed.
The ANOVA also showed that the crosslinking has significantly increased the modulus and tensile strength of nanofiber membranes (p < 0.05). This event happens due to the creation of a network of interconnected polymers by covalent bonding.
Water contact angle
The contact angle between the water droplet and the surface of the web is a method for investigating nanofibers' hydrophilic nature. Materials with contacts angle of 0–30°, 30–90°, and above 90° are respectively classified as hydrophilic, semi-hydrophilic, and hydrophobic [60]. Results obtained from water contact angle measurements are presented in Figure 5 and Figure S1. It can be observed that the water contact angle decreases significantly (p < 0.05) with the addition of CO. Post-hoc statistical analysis showed a significant difference between PCL/Gel nanofibers containing 10%, 20%, and 40% CO with pure and 5% oil loaded samples (p < 0.05). This phenomenon can happen due to higher contents of carboxylic acid groups or surfactant alignment. We believe the hydrophobic tail of the surfactant is located inside the oil droplets. At the same time, its hydrophilic head is arranged outside them, so at higher oil content, more hydrophilic sites are available, which improves the hydrophilicity. At lower oil contents (PCL/Gel/5% oil), the oil droplets tend to locate inside the nanofibers, and this could be the reason why the contact angle did not change for this sample. The exciting part of these results is that there was no significant change in hydrophilicity in 10–40% CO loaded samples, suggesting that the number of oil droplets remains almost constant over a critical CO loading (10%), but they became more massive in size. These results were in agreement with Banner et al. findings on CO coated gelatin nanofibers [61].

The water contact angle for as-spun and crosslinked PCL/Gel nanofibers containing different amounts of CO (*p < 0.05).
After crosslinking, the water contact angle has slightly increased. This phenomenon happens due to the amine functional group's consumption during the crosslinking reaction, which reduces the web's hydrophilic nature. It should also be noted that the water contact angle is highly sensitive to porosity, surface roughness, and diameter of nanofibers. So, this experiment was replicated on PCL/Gel films to omit these factors. Interestingly, the results showed the same trend.
Water vapor permeability
The average amount of evaporated water from normal skin is 204 ± 12 gm−2, while this factor varies from 279 ± 26 gm−2 to 5138 ± 202 gm−2 per day for wounded skin [62]. That is why an appropriate wound dressing must transfer water vapor and absorb wound exudates [63]. In this research, water vapor permeability of as-spun and crosslinked PCL/Gel and crosslinked nanofibers containing 40% oil content was investigated, which its results are shown in Figure 6. According to the results, the water vapor transport increases over time for all samples. Also, comparing with the control sample, the WTR value is not altered significantly for nanofibrous samples, which proves their excellent water vapor transport. The calculated WVP for control, PCL/Gel, crosslinked PCL/Gel, and crosslinked 40% CO loaded PCL/Gel are respectively 1220, 1163, 1150, and 1207 g/m2 per day that is comparable with commercial wound dressings such as Bioprocess, Biobrane II and Op-Site (426–2047 g/m2 per day) [62]. It can be observed that the permeability is slightly decreased after the crosslinking reaction, which can be related to their smaller pore size. On the other hand, the addition of CO has increased the WVP. These results suggest that the addition of the CO can help improve the absorption of wound exudates and enhance the vapor permeability of nanofibrous membranes. Ma et al. studied the vapor transport of tissue engineering sponges for wound dressing; their reported values for the control sample were similar to the obtained results in this article, so it can be concluded that this experiment is performed precisely [41]. Additionally, nanofibrous samples showed higher vapor transport compared to the sponges, which is beneficial for wound healing applications.

Water vapor permeability results for as spun PCL/Gel and crosslinked PCL/Gel and PCL/Gel/40% CO nanofibers.
Biological properties
Biodegradability
The morphology of as-spun and crosslinked nanofibers at different time points are shown in Figure 7. According to the SEM images, the degradation rate is slowed down in crosslinked samples. PCL portion in nanofibers degrades in a long time, but gelatin is water soluble and leaves the nanofiber structure at the early time points. After crosslinking, gelatin’s water solubility drops dramatically, and this could be the main reason for the slower degradation rate for crosslinked nanofibers. Also, the time had a significant effect on the extent of degradation. Images of the degraded sample after 28 days show that PCL/Gel nanofibers almost have lost their initial morphology, fibers are broken, pores are almost closed, and the structure is not appropriate for cell attachment. While the nanofibrous structure is preserved in the crosslinked PCL/Gel sample. Results also showed that the biodegradability of nanofiber membranes increases in CO loaded samples due to their higher hydrophilicity. Although the degradation rate was faster for the crosslinked sample containing CO, the nanofiber structure is preserved even on the 28th day. Thus, cells can attach, spread, and proliferate on these samples at long term usages.

Morphologies of PCL/Gel and PCL/Gel/CO nanofiber samples after 1, 14, and 28 days of degradation in PBS solution at 37°C.
Antibacterial properties
Tables 4 and 5 demonstrates the antibacterial activity of crosslinked CO loaded PCL/Gel nanofibers against gram-positive (Staphylococcus Aureus) and gram-negative (Escherichia Coli) bacteria. Samples showed suitable antibacterial properties against both bacteria. This property was augmented with increasing oil content, and the sample containing 20% oil exhibited the highest antibacterial activity. The presence of lauric acid in the composition of CO could be the main reason for the antibacterial and anti-inflammatory activity of this material. At higher CO loadings number of free -COOH groups on the surface of the matrix increases, which is associated with bacterial inhibition. In high CO content samples, these functional groups are blocked by steric effects associated with the high content of hydrophobic regions in the materials, therefore not favoring bacterial inhibition. Although nanofiber samples were more effective against Staphylococcus Aureus, they exhibited lesser activity against Escherichia coli [64]. This could happen because of the impenetrable wall, inner cell membrane, and hydrophobic property of gram-negative bacteria that makes them more resistant to antibiotics and herbal extracts [65].
Tensile properties of PCL/Gel nanofibers containing different amounts of CO.

Antibacterial activities (mean ± s.d.) of crosslinked CO loaded PCL/Gel nanofibers against gram-positive and negative bacteria.

Cell viability
MTT assay was performed on crosslinked nanofiber samples, and results are illustrated in Figure 8. Statistical analysis showed that the cell viability is significantly increased on the 3rd and 7th days. Therefore, it can be concluded that the suggested construct is not toxic, and cells can grow on nanofibers over time. Results also indicated that on the first day of cell culture, only PCL/Gel, and PCL/Gel/40% oil samples showed low cell viability. From our perspective, this phenomenon may happen because of crossing the critical CO concentration for cell viability. Since CO is an antibacterial substance, it may decrease cell viability at high concentrations. On the third day, cell viability increased in all samples, but the PCL/Gel sample still showed the least viability. Finally, on the seventh day, there was no significant difference between the cell viability of nanofibers and the control sample. These results suggest that CO has increased cell growth and, in some cases, showed better cell viability compared to the control sample. Scaffolds containing 10%–20% CO provide a better environment for fibroblast cell attachment and proliferation. Besides, these samples allow better diffusion of nutrients and oxygen (according to WVT analysis). However, high concentrations of oil limit these processes, which alters cellular viability. It should also be noted that CO is capable of tissue repair because of its long fatty acid. The results obtained in this article are in agreement with earlier reports by Amna et al. and Unnithan et al., which reported an increase in cell viability of olive oil and Emu-oil loaded polyurethane nanofibers, respectively [20,58]. Jaganathan co-workers also showed that Ylang Ylang oil could raise fibroblast cell proliferation on polyurethane nanofibers [57]. Moreover, obtained results state that the glutaraldehyde vapor phase crosslinking reaction does not affect gelatin biocompatibility [66,67].
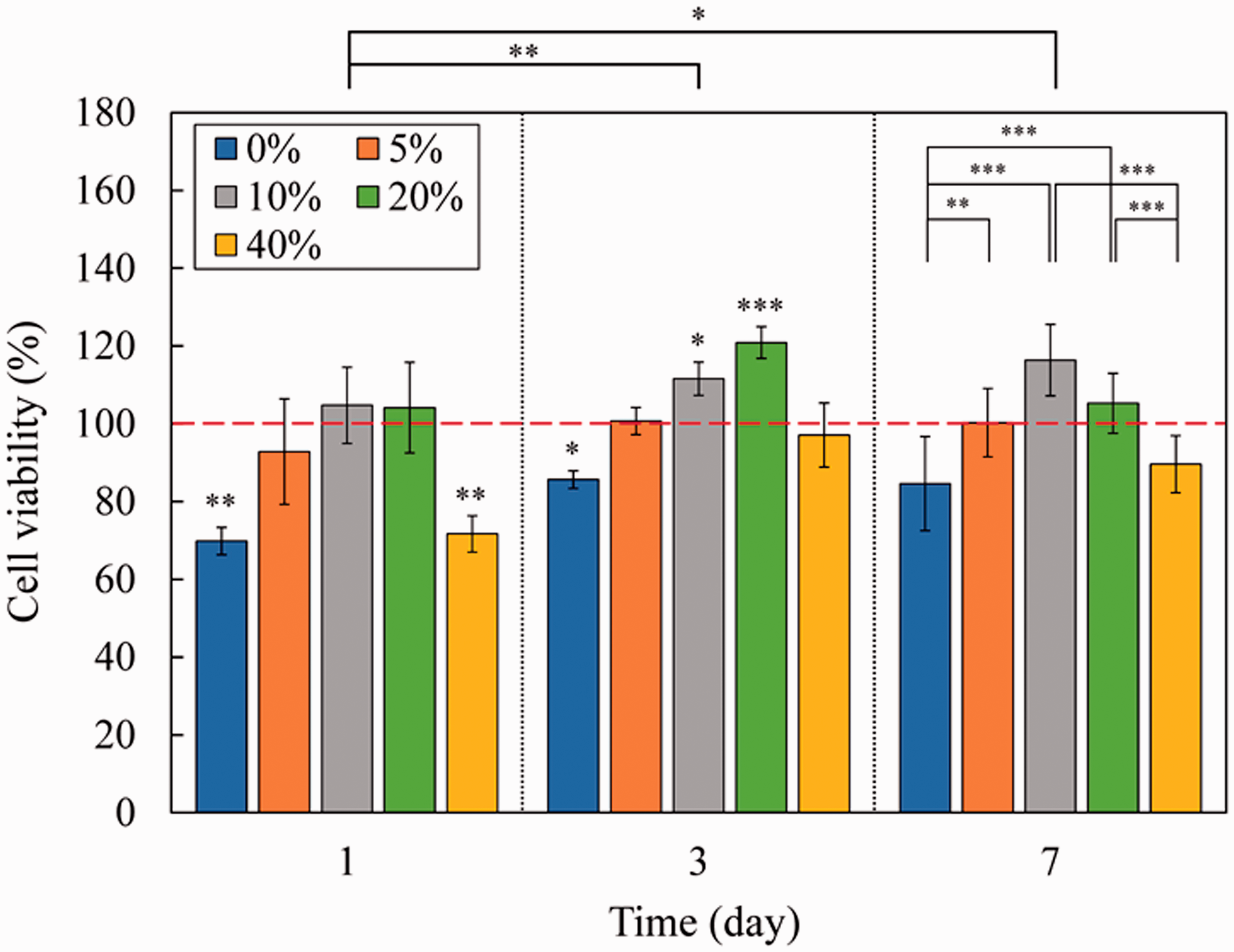
Cell viability results obtained from MTT assay for crosslinked PCL/Gel nanofibers containing different amounts of COs (*p < 0.05, **p < 0.01, ***p < 0.001). Significantly different groups from the control sample (red dashed line) at different days of culture are given on the top of bars. Also, statistical analysis between samples and days of cell culture is shown on the top right corner and top of the chart, respectively.
Cell morphology
SEM images of cultured fibroblast 3T3 cells were used to evaluate the attachment of cells on nanofibers (Figure 9). The results showed that cells are grown and covered the surface of nanofibers. However, the best cell growth was observed in nanofibers containing 10% and 20% CO. This behavior could be related to the higher hydrophilic nature of these samples, which provides a better environment for cell attachment, growth, and proliferation. The other reason for this observation is the reduction of nanofiber diameter in samples containing a higher amount of CO, which stimulated the cell attachment. Interestingly, cells on the sample with the highest oil content did not spread. In the thermal analysis section, it was observed that CO melts at 25°C. Because most of this sample was composed of oil, this oil leaves the nanofiber structure quickly and may alter cell attachment. While in 20% and 10% samples, the oil droplets can be supported by nanofibers' polymeric matrix, which lessens their release. The cell morphology results are in agreement with the MTT assay. We suggest the 20% CO loaded sample as the best candidate for cell attachment. These findings were in agreement with earlier findings. It has been reported that oils enhanced cell attachment because of their long fatty acid [58]. Jalilzadeh-Tabrizi et al. claimed the Emu-oil in PCL/PEG nanofibers could improve the ADSCs cell adhesion [68]. Zarghami et al. observed the proper morphology of fibroblast cell attachment on PEO/Chitosan/PCL/Olive oil [24].

SEM images of cultured 3T3 fibroblast cells on crosslinked PCL/Gel nanofibers with different amounts of CO.
Conclusion
In this research, CO loaded PCL/Gel nanofibers were successfully fabricated for the first time. Nanofibers were then cross-linked by glutaraldehyde vapor to enhance their mechanical strength and reduce their water solubility. The presence of CO and surfactant reduced the viscosity, surface tension, and diameter of the nanofibers. SEM images of as-spun nanofibers showed a uniform, smooth, and beadless morphology. The initial morphology was preserved, but nanofibers were swelled in crosslinked nanofiber membranes. The FTIR and DSC results respectively, confirmed the crosslinking process and the presence of CO into nanofibers. The hydrophilicity and mechanical properties were augmented by incorporating the surfactant and CO, respectively. The hydrophilicity decreased, but modulus increased in crosslinked samples. The water vapor permeability increased in CO loaded samples demonstrating that electrospun membranes are excellent candidates for wound dressing application. Webs were also observed to be biodegradable, and CO accelerated the degradation of samples with increasing hydrophilicity. However, the structure of nanofibers is preserved after 28 days. Although the sample containing 10% CO had a better mechanical property, nanofiber membranes loaded with 20% CO exhibited superior antibacterial behaviors. Besides, cell culture and MTT assay results showed that 3T3 fibroblast cells attached and proliferated better on 20% CO loaded nanofiber membranes. Therefore, 20% CO loaded nanofibers can be suggested as the optimal sample for wound healing applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
