Abstract
In this study, the production of hybrid scaffolds containing keratin/polyvinyl alcohol (Ker/PVA) and poly (Ɛ-caproactone) (PCL) for application in skin regeneration has been investigated. In the first step, the extraction of keratin from wool fibers was done, and then electrospinning process was used to fabricate hybrid nanofibers. Ker/PVA blend solution was extruded from one syringe and poly(ε-caprolactone) solution from the other one and hybrid nanofibers were gathered onto a rotary drum collector. The effect of different ratios of Ker/PVA (30:70), (50:50), and (70:30) with PCL were studied on the morphology, hydrophilicity and mechanical features of the scaffolds. The Ker/PVA (50:50)-PCL nanofibers presented an appropriate modulus and mechanical strength similar to the natural skin. These scaffolds possessed high levels of hydrophilicity, which improved cell-scaffold adhesion and increased the growth and proliferation of fibroblast cells. Keratin based nanofibers demonstrated antibacterial characteristic against Gram-negative Escherichia coli and Gram-positive Staphylococcus aureus.
Introduction
In the past few years, nanotechnology has created tremendous advances in the world of science and technology [1]. Nowadays, nanofibrous scaffolds have been shown to possess high potential for tissue formation because of their excellent features such as small diameter, high surface area, high flexibility, interconnected porous structure, closely resembling the naturally occurring characteristics of the extracellular matrix (ECM), and special mechanical properties [2]. Nanofibers from synthetic polymers such as polyglycolide, poly(L-lactic acid) (PLA), poly(glycolideco-lactide) (PLGA), poly(Ɛ-caprolactone) (PCL), and poly (dioxanone) have been fabricated for application in regeneration of different tissues [3,4]. PCL as a biodegradable and biocompatible polyester has been used in tissue engineering, extensively. It is a thermoplastic polymer with good stability, proper processability, low tensile strength and high elongation at breakage [5,6]. However, its poor hydrophilicity and slow degradation limit its application in many fields. These aspects of PCL were improved by blending PCL with natural polymers such as gelatin, collagen, gum tragacanth and chitosan [7–10]. Shalumon et al. produced nanofibrous scaffolds from PCL and chitosan and showed that the presence of chitosan contributes significantly to the improvement of the hydrophilic property of scaffolds [8]. Recently, there has been great interest in the production of natural polymer-based nanofibers because of their proven biocompatibility, higher hydrophilicity, low-to-no cytotoxicity, quicker degradation, as well as enhancement of cell adhesion and proliferation [11]. Keratin is one of the most plentiful proteins in nature and in the human body which can be applied in different fields of biomedical category because of its biorenewable nature, biofunctionality, biodegradability and biocompatibility by accelerating migration, adhesion, and proliferation of the cells [12]. This polymer is the main ingredient of major component of hair, feathers, wool, nails and horns of mammals, reptiles and birds [13]. Wool keratin contains high amount of cystine, which accounts for 7–20% of the total amino acids compared to other proteins (e.g., fibroin and gelatin) [14]. Because of low molecular weight and brittle property of keratin, this polymer is difficultly electrospun into nanofibers [15]. However, less attempts have been done to produce keratin-based nanofibers by electrospinning compared to other natural polymers such as gelatin. Nanofibers containing poly (hydroxyl butylate-cohydroxy valerate) (PHBV)/keratin were produced but the final structure of nanofibers contained many beads because of the broad molecular weight distribution and low dissolvability of keratin [16]. Most of keratin based nanofibers, such as keratin/PVA and keratin/PEO, need chemical crosslinking such as using glutaraldehyde to preserve their structural integrity in aqueous medium and enhance mechanical strength [17], but most of such crosslinking chemicals are cytotoxic, which is a major problem in biomedical applications. To overcome this problem, synthetic polymers such as PCL or PLA have been blended with keratin [18,19], but there are no reports for the production of hybrid nanofibers from keratin and PCL. In current study, for first time in literature, hybrid nanofibers from keratin/poly (vinyl alcohol) (Ker/PVA) and PCL by double nozzle electrospinning technique were fabricated. For this aim, polymeric solutions of Ker/PVA and PCL were injected from different syringes. Chemical and mechanical properties of fabricated nanofibers were evaluated. The cell adhesion ability and cytocompatibility of different blend ratios of Ker/PVA with PCL were examined on fibroblast cells through fluorescent staining (DAPI) and MTS assay. Moreover, morphology of fibroblast cells and their integration with fabricated scaffolds were evaluated by scanning electron microscopy. These hybrid nanofibers maintained their integrity and fibrous form in water media for a period of 27 days without using any chemical crosslinking agent.
Materials and methods
Materials
PCL (Mw 80 KDa) was prepared from Sigma-Aldrich; Glacial acetic acid was purchased from Merck Co. Poly (vinyl alcohol) (Mw = 72 kDa, Hy = 99.5%) was bought from MERK, Co.
Wool keratin was extracted from Iranian Afshari sheep. Briefly, 250 g of washed wool was placed in 5 L extracted solution containing urea (9 M), Sodium dodecyl sulfate, and 2-mercaptoethanol, then shaking process was done at 50°C for 2 days. After that, the wool was stirred with high speed in industrial mixer for 3 hours. The resulting mixture was filtered using the stainless-steel mesh with large and fine pores, respectively. Finally, the keratin powder was obtained throughout spray drying of the filtrated solution.
Preparation of electrospun hybrid nanofibers
In this research, for polymeric solution production, formulations containing Ker/PVA with different blend ratios of 70/30, 50/50 and 30/70 with final concentration of 12 wt% were prepared. Distilled water was used as solvent to dissolve Ker/PVA. Then PCL was dissolved in acetic acid 90% at concentration of 15 wt%. Hybrid nanofibrous substrate was formed by two nozzles. For this, the Ker/PVA nanofibers from one syringe and PCL solution from another one were collected onto rotating drum with rotation speed of 250 rpm, simultaneously. Spinning distance and voltage were set 140 mm and 15 Kv, respectively. The most important step in preparing hybrid nanofibers is to match the feed rate of two separate polymer components in the final substrate. In this paper we tried to have equal amount (50:50) from Ker/PVA and PCL in the fabricated structure. For this the feed rate of Ker/PVA and PCL were set at 1 mL/h and 0.8 mL/h, respectively. The test was carried out under constant temperature and humidity condition of 25°C and 65% humidity. Figure 1 exhibits the total process of all hybrid nanofiber production process.

The total process of all hybrid nanofiber production process.
Characterization
The surface morphology of the nanofibers was assessed by field-emission scanning electron microscope (FE-SEM, TESCAN MIRA3-XMU, Czech and FEI Nova Nano SEM 450, USA). The average diameter of the nanofibers was measured and calculated from 100 random points chosen from the SEM images, using image analysis software (Image J, National Institute of Health, Bethesda, MD). Fourier transform infrared spectroscopy (FTIR) analysis (Nicolet Magna-IR 560) was applied to investigate the structural changes using KBr method for fabricated structures. In order to report the hydrophilic/hydrophobic characteristics of the Ker/PVA-PCL nanofibers, the contact angle of nanofibers was calculated by the video contact angle instrument (KRÜSS G10, Germany). For this, about 1 µ L of distilled water was pipetted on to the rectangular shape (20 mm × 20 mm) mats and temporal images of the droplet were taken from droplets. The contact angles were reported by the sessile drop approximation of the inbuilt software of the instrument. These experiments were performed at room temperature and was repeated three times for each sample.
Mechanical properties
The tensile properties of the nanofibers were analyzed using Instron (model 5566 made in U.K.) using a 10-N load cell under a cross-head speed of 10 mm/min according to the ASTM standard D638 [20]. The nanofibers were cut into a size of 30 × 10 mm2 and the thicknesses of the samples were measured by a micrometer. Five replications of each formulation were performed and average values of the measurements were used.
Degradation behavior
The in vitro degradation test was done by the immersion of dry fabricated hybrid samples (50 × 15 mm2) in 10 mL of SBF solution (pH 7.4) at 37°C, during 27 days. At pre-determined periods, the samples were removed from the degradation medium, washed with distilled water, were placed in an oven and dried at 37°C until a constant mass was achieved, to evaluate the weight loss. The degradation medium was replaced weekly. The degradation was reported by calculating the weight loss (WL), as follows:
To study the morphology of samples after degradation test, the fabricated samples were immersed in PBS (pH of 7.4) and incubated for a period of 27 days at 37°C. At each specific time point, the nanofibers were washed with water and subsequently dried in vacuum oven for 48 h. The morphology changes were reported by SEM.
Antibacterial assay
For antibacterial test, disc diffusion method was applied and the antibacterial characteristic of Ker/PVA (30:70)-PCL, Ker/PVA (50:50)-PCL and Ker/PVA (70:30)-PCL nanofiber was analyzed according to SN 195,920 against Escherichia coli (ATCC 25,922) as Gram positive bacteria and Staphylococcus aureus (ATCC 6538) as Gram negative bacteria. Briefly, the agar surface was inoculated by using a swab dipped in the bacterial cell suspension adjusted to a turbidity of 0.5 McFarland standard units and then allowed to dry for approximately 5 min. After placement of sterilized nanofibrous structures (1*2 cm2), the susceptibility plates were incubated at 35 °C for 16–18 h. Inhibition zones around the discs were reported by images [22].
Cell culture and cytocompatibility test
Evaluation of cellular behavior of L929 cells on Ker/PVA-PCL hybrid nanofiber
The fabricated nanofibers placed on the plate’s wells at appropriate sizes, re-suspended in PBS and then UV irradiation was done to sterilize them, for 20 min. L929 cell line was obtained from Pasteur Institute (Tehran, Iran). Cells were maintained in RPMI 1640 (Gibco-Invitrogen) with 10% fetal bovine serum (Gibco-Invitrogen) and 1% penicillin/streptomycin (Sigma-Aldrich). The cells were incubated at 37°C with 5% CO2. The cells’ medium was changed every two days. When the cells reached more than 80% of confluency, they were “passaged” and seeded on the non-coated 24-well plates and sterilized nanofibrous coated plates for cell behavior assays as following:
Cell attachment
Cell adhesion on hybrid nanofibers was evaluated by DAPI staining. L929 cells were cultured on 24 well plates (20,000 cells/well) coated with sterilized Ker/PVA (30/70)-PCL, Ker/PVA (50/50)-PCL, Ker/PVA (30/70)-PCL, PCL nanofibers and non-coated plate as control group. After 12 hours, cell washing was done with PBS and fixation was performed with 4% formaldehyde. The nuclei were marked with 40-6-diamidino-2-phenylindole (DAPI) after permeabilization with 0.5% Triton X-100 and then observed by a fluorescence microscope.
Cell proliferation
Cell proliferation on Ker/PVA (30/70)-PCL, Ker/PVA (50/50)-PCL, Ker/PVA (30/70)-PCL and PCL nanofibers was assessed by MTS- assay according to ISO standard 10993–5:2009 for the biological evaluation of medical devices. Briefly, L929 cells (5000 cells/well) were seeded on 96 well plates. After 48 hours, the number of viable cells was determined by the CellTiter 96 Aqueous One Solution Cell Proliferation Assay (ProMega). Briefly, after washing cells with PBS, 100 µl of fresh RPMI-1640 culture medium supplemented with 20 µl of MTS solution were added to each well/, incubated for 2 h at 37c. ELISA reader recorded the Optical density (OD) at 490 nm. Survival fraction was calculated as follows (equation (1)) [23,24]
Also for evaluation of scaffold morphology effects on cell growth, cells viability on keratin film coated plate was compared with keratin-nanofibers coated plates. For this, wells of the plate were coated with aqueous solution of keratin (3 mg/ml), air-dried and then heated in 80 C° for 20 min. Sterilization of keratin film was done same as nanofibers.
Cell morphology
Scanning electron microscopy (SEM) was used to evaluate the morphology and integration of cells on the Ker/PVA–PCL nanofibers. After 72 hours of culture, cells were fixed using 3% glutaraldehyde for 20 min and subsequently dehydrated using a series of ethanol solution (50%, 70%, 95% and 100%) for 10 min. After air drying, the samples were sputter-coated with gold at a voltage of 15 kV under argon gas and pictured by a Field Emission Scanning Electron Microscopy (FE-SEM) (TESCAN MIRA3-XMU, Czech and FEI Nova NanoSEM 450, USA).
Statistical analysis
The differences among three groups and multiple comparison evaluated by Anova and Tukey tests, respectively. T-test was used to analysis differences between two groups. Differences less than 0.05 (P-value < 0.05) were considered statistically significant.
Results
Morphology of nanofibers
The electrospinning method appeared stable and successful for the samples at a Ker/PVA total concentration of 12 wt%. The SEM images of PCL and prepared hybrid electrospun nanofibers at different ratios of Ker/PVA with PCL are displayed in Figure 2. The average diameter for PCL, Ker/PVA (30:70)-PCL, Ker/PVA (50:50)-PCL and Ker/PVA (70:30)-PCL was about 560 ± 33, 345 ± 22, 231 ± 29 and 163 ± 36, respectively. The SEM images of fibers exhibited a porous, fibrous, and randomly oriented structure for all of the fibers. Increasing keratin in final formulation, resulted in reduction in fiber diameter and spindle like morphologies in the structure. Li et al showed that when the content of keratin in spinning solution increases, the average diameter decreases. This could be explained by the reduction in the viscosity and the increase in the conductivity of spinning solution with increasing keratin content [15].

Morphology of nanofibers, (a) PCL, (b) Ker/PVA (30:70)-PCL, (c) Ker/PVA (50:50)-PCL, (d) Ker/PVA (70:30)-PCL nanofibers.
Contact angle measurements
Contact angle of samples depends on chemical composition and their topographic pattern. This characteristic reflects the hydrophilicity or hydrophobicity nature of nanofibrous substrates. Figure 3 demonstrates the results of contact angle measurements of different samples for three different tests. Compared with the hydrophobicity of PCL nanofiber membranes (91°) in other papers [25], incorporating PVA or Ker to the composition of nanofibers decreased contact angle amount. The presence of keratin which contains high amounts of amino and carboxylic groups could improve the polar groups, which results in the hydrophilic and rougher surface of the electrospun mats [19]. PVA has a semi-crystalline structure with high hydrophilic property due to the presence of hydroxyl groups in its structure and ability of forming hydrogen bonding with water molecule. Contact angle for Ker/PVA (30:70)-PCL, Ker/PVA (50:50)-PCL and Ker/PVA (70:30)-PCL was about 45 ± 1°, 50 ± 4° and 60 ± 4°, respectively (Figure 4). These results revealed that the surface hydrophilicity of nanofibrous membranes increased significantly with the more loading of PVA (P ≤ 0.05). Higher contact angle was observed for the Ker/PVA (70:30)-PCL nanofibers.

Contact angle measurements for (a) PCL nanofibers, (b) Ker/PVA (30:70)-PCL, (c) Ker/PVA (50:50)-PCL, (d) Ker/PVA (70:30)-PCL.

Contact angle of different samples (*p-value < 0.05).
Mechanical properties of nanofibers
The mechanical properties of electrospun nanofibers is a main factor in their application when implanted in injured part, which largely depends on the choice of material, morphology of nanofibers, as well as the polymeric solution properties [26]. PCL is usually used as the matrix for wound dressing application because of its elastomeric and highly flexible nature [27]. Due to its semi-crystalline and hydrophobic nature, PCL has mechanical properties suitable for a variety of applications [28]. The elastomeric structure of PCL nanofibers and the presence of methyl group in the chemical structure of this polymer led to a higher toughness and flexibility compared to other samples (strain at break 59%) [26]. The reported tensile strength, modulus and strain observed for PCL nanofibers in Figure 5 were about 1.62 ± 1.2 MPa, 2.5 ± 1.7 MPa and 59.58 ± 4%, respectively. In the case of Ker/PVA-PCL nanofibers, it is evident that with increasing the amount of this polymer in the composition from 30 to 50 wt%, the tensile strength increased significantly (P-value < 0.05) from 2.18 ± 0.8 to 3.59 ± 0.3 MPa (Figure 6). It can be due to the presence of hard keratin in the formulation. There are two main types of keratin. Alpha-keratin, the keratin present in the mammalian skeleton, and beta-keratin, which is stronger in mechanical properties than alpha-keratin. By further increasing the amount of keratin to about 70 wt%, the strength decreases slightly to about 2.65 ± 0.4. The reason for this may be due to the morphology of the formed fibers or the inappropriate dispersion of the keratin polymer with the polyvinyl alcohol polymer in Ker/PVA (70:30)-PCL. Also, the difference of strength between Ker/PVA (30:70)-PCL and Ker/PVA (70:30)-PCL is not significant. The results demonstrated that, the presence of keratin and its increase in the composition reduced the strain significantly (Figure 6) so that the strain in PCL, (30:70) Ker/PVA-PCL, (50:50) Ker/PVA -PCL and (70:30) Ker/PVA-PCL was reported to be 59.58 ± 4, 48.22 ± 1, 44.8 ± 2 and 25.99 ± 6%, respectively. The reason for this behavior can be related to the increasing beta keratin in the final structure. Also the reported modulus values for PCL, (30:70) Ker/PVA-PCL, (50:50) Ker/PVA-PCL and (70:30) Ker/PVA-PCL were about 2.5 ± 1.7, 5.8 ± 2, 17.8 ± 2 and 21 ± 3.1 MPa, respectively, indicating that increasing keratin content improves the resistance of the structures significantly (P-value < 0.05) in front of deformation (Figure 6). Li et al. reported in their work that the tensile strength of Ker/PVA nanofibers increases with the addition of keratin [29]. In other research, tensile strength of Ker/PVA nanofibers was higher than tensile strength of pure PVA fibers [30]. The enhancement in tensile strength was attributed to better compatibility and strong interactions between higher keratin amount and polyvinyl alcohol [8]. Also, at higher amounts of keratin, the amount of beta keratin in the structure increases [31]. Mechanical Properties of the desired specimen for use as a skin scaffold should be within the mechanical properties of normal skin. For normal skin the strain is in the range of 35%–115% and the young modulus is about 15-150 MPa [32]. The sample Ker/PVA (50:50)-PCL with strain about 44% and modulus about 17 MPa was considered to be the optimum sample in terms of mechanical properties.
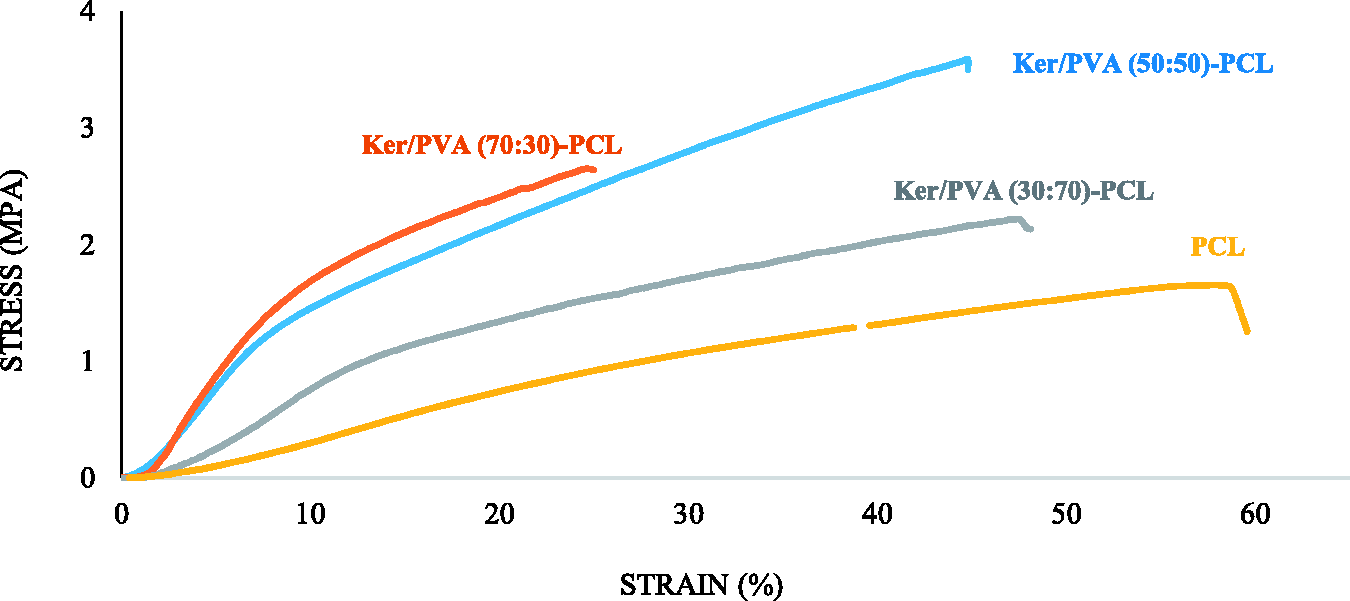
Mechanical properties of nanofibers.

Tensile strength (a), strain (b) and modulus (c) of fabricated samples for 5, The experiments were repeated for five times for each sample (*P-value < 0.05).
FTIR results
FTIR analyze was utilized to study the interaction of the functional groups of polymers in nanofiber structure (Figure 7). Electrospun PVA nanofibers represent peaks at about 3383, 2925, 1734, 1098 and 850 cm−1, which belong to the OH, CH2, C–O, C–O and C–C groups, respectively (Figure 5(a)). In the case of keratin polymer, the peaks at about 1633, 1500 and 1380 cm−1 demonstrate the amide groups of types 1, 2, and 3. The strong peak observed at 3409 cm−1 is assigned to the N-H and the hydroxyl groups. The peak observed at 1024 cm−1 indicates the vibrational tensile band of S-O. The peak of the carboxyl group is also visible in the region 2700-3128 cm−1 (Figure 7(a)) [33]. PCL nanofibers exhibit prominent peaks at 2934, 2861, 1727, 1294, 1240, and 1164 cm−1, indicating asymmetric CH2-stretching, symmetric CH2- stretching, carbonyl groups, CC and C-O stretching, C-O-C symmetric and asymmetric stretching (Figure 5(a)) [34]. In the case of Ker/PVA nanofibers, by adding polyvinyl alcohol to keratin, the peaks at 3409 and 3383 cm−1 related to the Ker and PVA structures have moved to a lower wave number of about 3320 cm−1 which may be related to the formation of hydrogen bonds between keratin and polyvinyl alcohol. He et al. reported the interaction between feather keratin and PVA nanofibers by FTIR test. Their results showed existence of O–H group in its structure. When the FK was added to the PVA solution, new peaks were created that belong to the C=O and N–H functional groups. They concluded that the major interaction between FK and PVA fibers was related to the formation of hydrogen bonds, which improved the electrospinning capability of Ker/PVA solution [35]. In Ker/PVA-PCL nanofibers, main peaks of all three polymers, such as the presence of carbonyl group in the PCL structure, the amide groups, as well as the presence of the carboxyl group in the keratin structure are visible (Figure 7(b)).
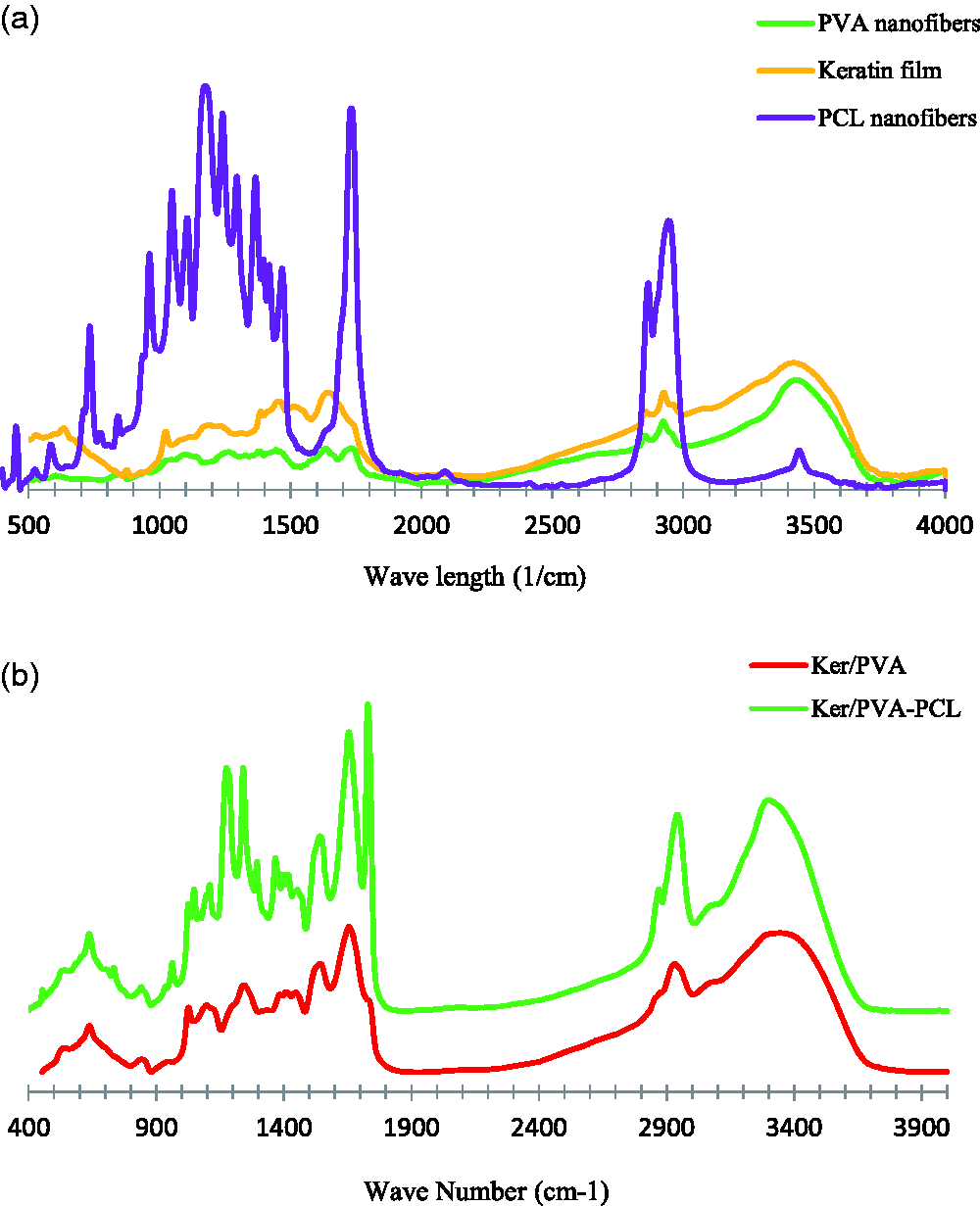
FTIR analysis of different samples.
Biodegradation test
To perform this test, hybrid nanofibers containing PCL with different ratios of Ker/PVA 30:70, 50:50 and 70:30 were analyzed. According to the biodegradability results (Figure 8), the highest rate of degradation was visible on the first day, but from the end of the first day to the twenty-seventh day, the degradation continued at a lower rate. The results indicate that the weight loss increased with increasing percentage of polyvinyl alcohol at Ker/PVA (30:70-PCL), due to the superhydrophilic properties of polyvinyl alcohol polymer compared to natural keratin polymer. After passing 27 days of degradation study, more cracks are visible in the SEM images of Ker/PVA (30:70-PCL) sample in Figure 9. The lower contact angle of Ker/PVA (30:70)-PCL could explain this higher weight reduction and created cracks in the SEM image compared to other samples, too. In the diagram corresponding to Ker/PVA (70:30)-PCL combination, less weight loss was observed, SEM images of degradation study confirms this subject, too. In the case of the Ker/PVA (50:50)-PCL blend, this amount of weight loss is more balanced.

Weight loss of samples.

SEM images from degradation test (a) Ker/PVA (30:70)-PCL, (b) Ker/PVA (50:50)-PCL, (c) Ker/PVA (70:30)-PCL.
Cell adhesion and proliferation enhancement on keratin based nanofiber
Over the past decade, the use of nanofibers with synthetic polymer in their structure has been considered as a promising scaffolds in tissue engineering due to their high similarity to the structure and topography of the natural ECM [36]. Nevertheless, alongside with physical properties, presence of proper biochemical context on substrate that influence cell attachment and function should be addressed [37]. However, nanofibers with synthetic polymer in their structure have enough mechanical strength and stability in water media, but their hydrophobic properties and lack of cell adhesion ligands limits their utilization in tissue regeneration. Based on the recent studies, fabrication hybrid nanofibers based on synthetic polymers and natural hydrophilic component such as native protein, create substrate that provide proper ligand sites to increase cell attachment as well as retaining of the substrate mechanical strength [38].
Nowadays, wool keratin type I microfilament as an inexpensive ECM-like protein has gained interest by many researches in preparing appropriate substrates for fibroblasts and different cancer cell lines due to availability, easy extraction and reconstruction capability [23,39]. In present study, cell adhesion on Ker/PVA-PCL nanofibers with different blend ratios of Ker/PVA was evaluated by DAPI staining (Figure 10). Fluorescent microscopy images of stained nucleus showed increased cell adhesion in structures with higher keratin content, so that highest cell attachment was occurred on Ker/PVA (70:30)-PCL nanofiber.

Fluorescent microscopy images of cell nucleus stained with DAPI after 12 hours of culture on Ker/PVA-PCL nanofiber with different concentration of keratin.
Recent studies showed cell attachment and proliferation on keratin coated plate was higher than non- coated plate [40–42]. Keratin molecules contain receptor-binding motifs such as LDV and RGD that can interact with cell surface receptors, especially the integrin family [30]. Based on the recent studies these molecular interactions can mediate cell adhesion, promote cell-matrix and cell-cell interactions that consequently activate the cell-signaling pathway and affect cellular behaviors such as cell proliferation and differentiation [10,16,17]. As shown in Figure 11, cell proliferation increased significantly on keratin based-nanofiber and keratin film (P < 0.05).

Morphology of L929 cells cultured on different substrates and non-coated plate after 48 hours of culture (Inverted microscopy images, 10X).
As expected, MTS assay results demonstrated presence of keratin lead to the progressive increase in the cell proliferation (Figure 12). The cell viability on Ker/PVA (30:70)-PCL, Ker/PVA (50:50) -PCL and Ker/PVA (70:30)-PCL was 1.18, 1.27 and 1.44 times higher than control group, respectively. Besides, physical characteristics of electrospun nanofibers can cause conformational changes of protein ligands that may affect their accessibility and interaction with cell integrins [43,44]. In this regard, higher cell viability and proliferation in the keratin based nanofibers compared to keratin film samples might contribute to the more effective cell integrin–keratin motives interactions and therefore keratin based nanofibers has become a high potential substrate.

Survivals fraction of L929 cells (Optical density) cultured on different substrates (PCL, Ker/PVA (30:70)-PCL, Ker/PVA (50:50)-PCL, Ker/PVA (70:30)-PCL nanofibers and keratin film than non- coated plate after 2 days of culture, (*P-value < 0.05).
In agreement with cytotoxicity assay, Fe-SEM images showed rapid expansion and growth of cells on the keratin based- nanofibers after 72 hours of culture (Figure 13). L929 cells not only completely covered surface of Ker/PVA (70: 30)-PCL nanofiber, but also three-dimensional growths can be seen on some places of substrate. Ker/PVA (50:50)-PCL as optimum sample exhibited proper cell attachment and proliferation.

Fe-SEM images of L929 cells on keratin based- nanofiber 72 hours after culture.
Antibacterial analysis
Antibacterial test was performed using E. coli as gram-negative and S. aureus as gram positive bacteria on Ker/PVA (30:70)-PCL, Ker/PVA (50:50)-PCL and Ker/PVA (70:30)-PCL nanofibers. The results of the antibacterial analysis in Figure 14 shows that the samples with higher amount of keratin are resistant to bacterial growth and the inhibition zone is visible for Ker/PVA (50:50)-PCL and Ker/PVA (70:30)-PCL samples against both bacteria. The antibacterial property of electrospun nanofibers originates from the peculiar secondary structure of the protein [45].
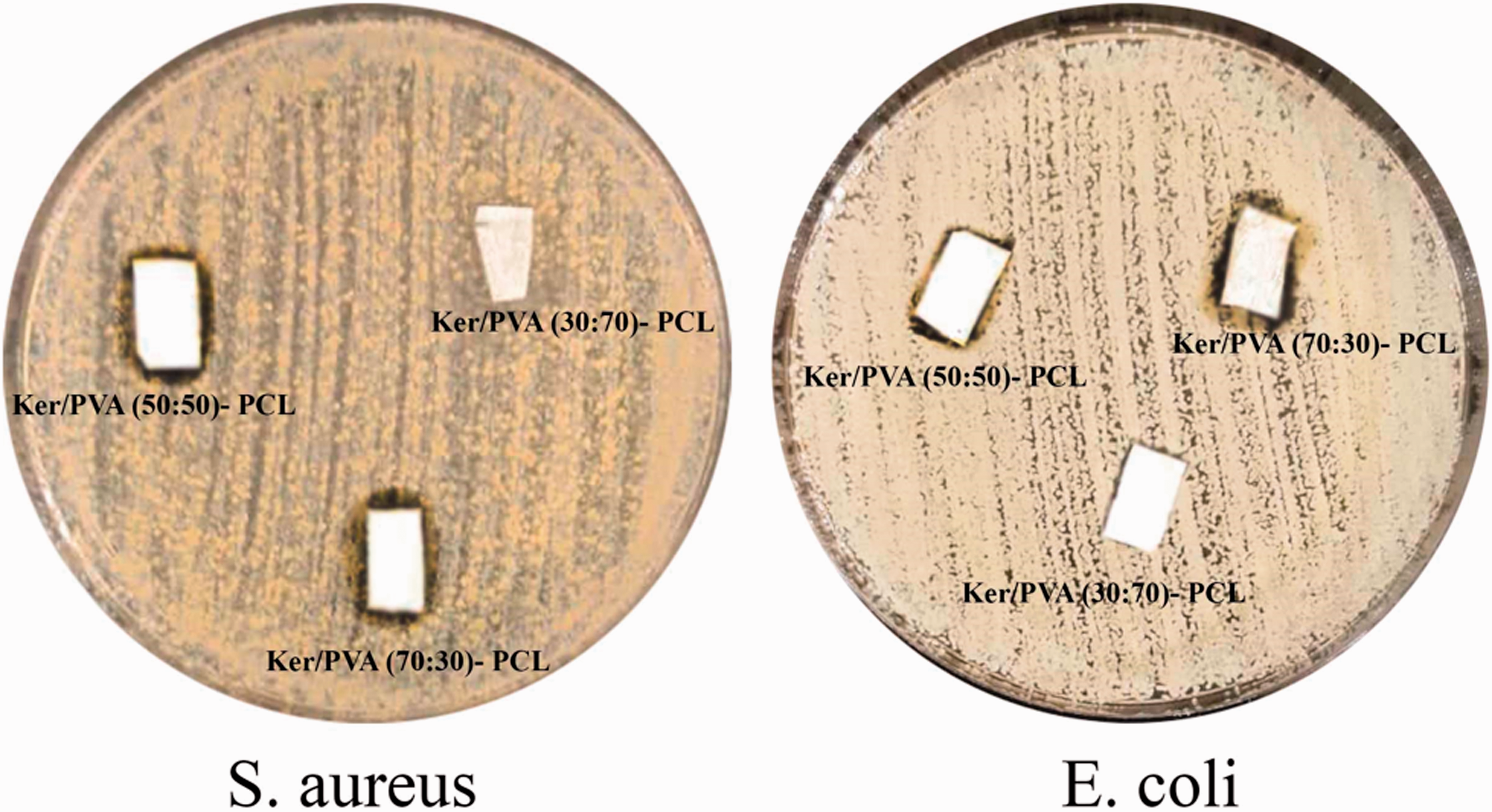
Antibacterial activity of samples against S. aureus and E. coli bacteria.
Conclusion
The efficacy of biological scaffolds is strongly dependent on the choice of proper polymers for their designing. The use of natural wool keratin is a great field of interest in this area because of its biocompatibility, bioactivity, ease of availability and fiber protein structure. In present study, after extraction of keratin, we fabricated keratin-based nanofibers by using double nozzle electrospinning method from Ker/PVA and PCL solutions, instead of using cytotoxic chemical crosslink. Our results showed Ker/PVA (50:50)-PCL nanofibers had an appropriate modulus and mechanical strength that can simulate the modulus, elasticity and mechanical strength of the skin. Increasing PVA amount in the formulation of fibers increased the hydrophilicity of them and Ker/PVA (50:50)-PCL structure exhibited contact angle about 50 ± 4°. The fabricated scaffolds did not have any cytotoxicity effects on fibroblast cells. Interactions of keratin protein in Ker/PVA-PCL nanofibers with cell integrins not only increased cell adhesion but also enhanced cell proliferation up to 40% on Ker-PLA (70:30)-PCL nanofiber. It can be claimed that the Ker/PVA-PCL hybrid scaffold has excellent biological property that can be used as wound dressing. The produced hybrid keratin based nanofibers were resistant to bacterial growth.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


