Abstract
Harmful portion of ultraviolet (UV) radiation is one of the significant physical carcinogen in our natural environment. The damage caused by UV exposure to our body is cumulative and builds up over the years. UV protective textiles have been used to protect the wearer from harmful UV radiation. UV blocking effect of textile depends on various parameters
Keywords
Introduction
Traditional clothing has light weight, less physical stress, improved breathability, improved comfort, however, it has a reduced protection against pathogen and external environmental hazards. Technological advancement can produce textiles with added functionalities. Few examples of functional textiles include flame retardant textiles, antibacterial textiles, UV protective textiles, super hydrophobic textiles, nanofibers web by electrospinning, thermo-regulating textiles, medical textiles, self-cleaning textile, industrial textiles etc [1–5]. Surface wettability of textiles is a very important issue in order to create super hydrophobic textiles with functionalities like self-cleaning, waterproofness etc. Various materials can be used to create the super hydrophobic surface such as silica nanoparticles immobilized by polyvinyl alcohol and further modified by using polydimethylsiloxane (PDMS) [6], PDMS and ZnO coating [7], plant polyphenol metal complex on the surface of an electrospun polyimide (PI) nanofibrous membrane followed by modification with PDMS [8], hollow structured SnO2 nanoparticles [9] etc.
Personal protective clothing protects the wearer from various environmental/industrial hazards
Ultraviolet (UV) radiation from sunlight is one of the significant physical carcinogen in our natural environment [20,35]. Mainly, residual UVB radiation that reaches to earth surface can still be absorbed by proteins and DNA, and is sufficient to kill unprotected cells, because, UV exposure can form reactive oxygen or radical species [20]. DNA with an absorption maximum of 260 nm, is a major epidermal chromophore [36]. Therefore, both UV-A (280–315 nm of wavelength) and UV-B (315–400 nm of wavelength) can induce structural damage to DNA. We know that UV rays in sunlight is an important factor for the synthesis of vitamin D [37]. However, excessive exposure of unprotected skin to UV radiation can be hazardous which accelerates the aging of skin and induce other deleterious effects on human skin e.g. skin cancer [38,39]. The degree of risk for hazardous effect of UV rays on human skin depends on the intensity of UV rays, duration of exposure and whether the skin was protected or not [40]. Damage caused by the UV exposure is cumulative and builds up over the years [41]. Apart from the solar UV radiation, artificial UVR sources such as xenon arc or germicidal lamp, UVR emission from certain welding arc are significant. Therefore, UV protective textiles are also essential in these artificial significant UVR hazard industrial work areas [42]. Apart from these, UV protective textiles can be used for other industrial applications such as aerospace, automobile etc [43]. UV protective textiles can manufactured by using various approaches such as selection of fibers/yarns, weaving parameters, coating and textile finishing approaches, etc [44–47]. Recent research study revealed that metal oxide nanomaterials and some of the carbon based nanomaterials have excellent UV blocking properties due to their unique macromolecular structure and properties [32,33,48–52]. The objective of this review article is to present an overview on UV blocking properties of various nanomaterials, their UV blocking mechanism, incorporation of nanomaterials in the textile matrices and the durability for the UV blocking property of treated textiles.
A brief overview of nanomaterials for textile applications
Nanomaterials has at least one dimension up to 100 nm. Depending on the number of dimension not confined to the nanoscale (i.e. up to 100 nm), nanomaterials can be categorized into four classes
Nanomaterials can be used in functional textiles for various purposes such as water repellant, antistatic property, wrinkle resistance, strength enhancement, UV blocking, antibacterial, wearable electronics etc [56]. Self-assembly coating of CsxWO3 nanostructured material on cotton fabric can provide multifunctional properties
UV protective textiles
Clothing covers an extensive surface area of our skin which is permanently exposed to environmental stresses including UV radiation. However, all types of clothing are not effective to prevent environmental hazards. When, UV radiation falls on the clothing surface, it can be divided into mainly three parts
Kocic et al. reported UV blocking property of five knitted fabrics such as pure hemp, pure cotton, pure viscose, blend of hemp/cotton and blend of hemp/viscose. Pure cotton yarn knitted fabric showed highest UV protection factor (UPF) (118.7) followed by blend of hemp/cotton (45.1) yarn knitted fabrics, whereas, pure hemp knitted fabrics showed lowest UPF (24.4) despite having high UV radiation absorption capacity of hemp fiber [72]. Shabbir et al. studied the UV blocking properties of wool fabrics which has been dyed by natural dye (marigold). Undyed fabric has an UPF of 6.8 which is un-ratable, with 5% marigold extract, an UPF has been increased to 35 which is rated as very good. UPF of 20% marigold treated fabric has been increased to more than 50 which is rated as excellent. With increasing natural dyes on the wool, the UPF has been increased due to the more energy dissipation throughout absorbed molecules on the textile surface [73]. Yadav et al. reported UV protective property of linen fabrics by using natural dyes. UV protection of fabrics increases with increasing color value due to the combined effect of mordant and dye molecules. Optimum sample showed an UPF of around 45 which considered as excellent [74]. UV blocking property of woolen fabric which has been dyed with natural dye extracted from grape pomace showed a significant improvement of UPF from 5 for undyed fabric to 30 for dyed fabric without using any mordant. However, application of alum or ferrous sulphate mordant has been increased the UPF for more than 50 which is rated as excellent as per AS/NZS 4399:1996 [75]. UV resistance of clothing can be improved by incorporating UV blocking agent into the textile matrix [76,77]. UV absorber materials with an absorption spectrum of elevated absorption especially in the wavelengths corresponding to the UV-B zone can be considered as an appropriate material to develop UV protective textiles [78].
Nanomaterials for UV protective textiles
Nanomaterials with a large specific surface area can increase the effectiveness of UV blocking property with a very low concentration as compared with its micro particles counterpart [79]. UV radiation blocking effect of nanomaterial treated textile can be measured by UV protection factor. UV protection factor of textile is defined as follows [47]: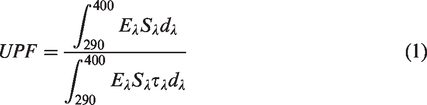
UV protection factor of textiles treated with various nanomaterials.

MWCNT: multiwalled carbon nanotube; PS: polystyrene; ATRP: atom transfer radical polymerization; HBP: hyper branched polymer; PU: polyurethane; NR: Not reported.
aMaterials treated with 30 mmol/L of sodium stearate.
Various standards have been reported in the literature for the measurement of UV protective property of textiles. Australian New Zealand standard has been defined the UV protection factor (UPF) as the ratio of UV transmittance in the wavelength range of 290–400 nm. According to the AS/NZS 4399:1996 method, UV protection factor of textiles can be rated as good, very good or excellent with an UPF of 12 to 24, 25 to 39 or above 40, respectively. The spectra of treated textiles composed from 280–400 nm through software which automates the identification of UPF [20,28,83,89]. European Standard EN 13758-2 defined UV protection factor as the ratio of UV transmittance under the wavelength range of 280 – 400 nm [91,95]. UPF rating as per EN 13758-2 are good, very good or excellent with an UPF of 20–29, 30–40 or above 40, respectively [72]. American Association of Textile Chemists and Colorists test method (AATCC 183–2014) defined UV protection factor as the ratio of UV transmittance under the wavelength range of 280 – 400 nm. According AATCC 183–2014 method, an UPF of 15–24, 25–39 or over 40 referred as good, very good or excellent, respectively. UPF of 40 means, 1/40 or 2.5% of UV radiation has been penetrate through the textile [99,100].
ZnO nanomaterials for UV protective textiles
Nanostructured thin ZnO film on textiles surface demonstrated excellent UV blocking property [101]. Bleached cotton fabrics which has been treated with 2% ZnO nanoparticle (particle size 40 nm) in acrylic binder showed 75% of UV blocking. Nano ZnO with an increased surface area and uniformly distributed particles on the fabric surface can enhanced the UV blocking property of coated fabrics [79]. Transmission of UV radiation through the nanostructure material treated textiles decreases with increasing of metal oxide nanomaterial in the textile matrix [80]. UV absorption study of ZnO nanorod (10–50 nm dia and 300–500 nm length) treated textile has been revealed high UPF as compared with UPF of less than 15 (non-ratable) for untreated fabric. Excellent UV blocking property of ZnO nanorod treated fabric is due to the crystallinity and better electronic property of nanostructured material [102]. UV blocking property of cotton fabric which has been treated with nanostructure hybrid ZnO and polystyrene (PS) revealed that UPF of more than 900 as compared with 4.9 for untreated fabric, 42.2 for pure PS nanoparticle treated fabric, 86.6 for nanostructure ZnO coating and 143.9 for PS blended with ZnO coating. Ultrahigh UV blocking property of hybrid ZnO nanoparticle coated fabric was due to the extraordinary electronic property of ZnO core compounded with phenyl group on the polymer shell. Pure polystyrene nanoparticle with phenyl groups in its structure can improve the UV blocking property of textile materials [81]. Wang et al. reported UV blocking properties ZnO treated hollow cotton fiber fabrics. Nanostructured ZnO (∼20 nm) has been attached on textiles surface through in situ growth of ZnO on grafted polymer brushes by electroless deposition technique. Functionalized fabrics has been showed durable UV blocking properties. More than 250 UPF has been achieved for the treated textiles with 1 hr deposition time due to the excellent UV blocking property of nanostructured ZnO [82]. Plasma treatment of cotton fabric can increase the uptake of ZnO nanoparticle and this can subsequently enhanced the UV blocking property of treated textiles. Scanning electron microscopic images revealed much higher concentration of ZnO nanoparticles on plasma treated cotton fabric [103,104]. Mai et al. reported UV protective property of cotton fabric which has been treated with nano ZnO by using coating with polyvinylsilsesquioxane polymer. UPF of coated fabric increases from 58 to 158 with increasing ZnO nanoparticle in polymer solution from 0.3 to 3 g/100 mL. Excellent UV blocking property of ZnO nanoparticle treated fabric is due to the scattering effect and electronic property of ZnO nanoparticle [105]. Veluswamy et al. reported UV blocking property of cotton fabrics which were treated with nanostructured ZnO and ZnO-Ag & ZnO-Sb composites. Treated fabrics showed much better UV blocking property as compared with untreated fabric (UPF 6.7). ZnO treated fabric has an UPF of around 64, whereas, ZnO-Ag treated fabric has displayed slightly higher UPF ∼68. Composite of ZnO-Sb treated fabric showed much higher UPF (∼84) as compared with only ZnO treated fabric [106]. Mousa and Khairy reported liquid precipitation method to prepare zinc oxide nanoparticle in three different media such as water, methanol and ethylene glycol. Nature of solvent media can influence the morphology of nanoparticles on its interaction with growing crystals. Depending on the type of solvent media various structures of ZnO nanoparticles have been formed
TiO2 nanomaterials for UV protective textiles
Nanostructure TiO2 has been widely used as an UV blocking agent for the manufacturing of UV protective textiles [108]. Nano TiO2 treated fabric revealed that transmission of UV has been completely cutoff in the UV-B region and up to 340 nm in the UV-A region. UV blocking mechanism of nanostructure materials can be correlated with band gap energy. Band gap energy of nano TiO2 lies in the UV region of solar spectrum [109]. TiO2 nanoparticle can be applied on cotton fabrics by sol-gel method in order to prepare UV protective textiles. Undyed control sample showed an UPF of 8.9 whereas nanoparticle treated fabric has been displayed an excellent UV protective (UPF +50) property. Rutile phase of nanoparticle has been worked as a better UV blocker than anatase phase due to the narrow band gap of rutile phase of TiO2 [110]. Ahmad et al. reported UV protective cotton fabrics which have been treated with reactive blue dye, C. I. Reactive Blue 21 (RB -21) containing TiO2 nano sol by pad-dry-cure method. Treated fabrics showed an excellent UV blocking property with an UPF of more than 100 as compared with untreated cotton fabric with an UPF of 6.9 which is un-ratable. Excellent UV blocking property of TiO2 nano sol treated fabrics is due to the extraordinary absorption capacity of UV rays by nanomaterial. Nanosol treated fabrics showed negligible transmission of UVB (280–315 nm) (<1%) as compared with ∼30% transmission of UVB (280–315 nm) for pristine fabric (Figure 1). Treated fabrics can also allow negligible UVA (315–400 nm) transmission up to 350 nm of wavelength as compared with pristine fabric. Further, nano sol treated UV blocking textile possesses excellent laundering capability even after five washing (each washing for 40 min at room temperature in absence of any detergent) [111].

UV transmission spectra of cotton fabrics which have been treated with various concentration of reactive blue dye containing TiO2 nano sol. Significant amount of both UVB (280–315 nm) and UVA (315–400 nm) have been transmitted through the pristine fabrics. While, all nano sol coated fabric shows negligible transmittance of UVB (280–315 nm) and cut down of majority of UVA (315–400 nm) radiation transmittance in the reported wavelength range. (Reproduced from Reference [111] © 2019 The Royal Society of Chemistry, Open access).
Yang et al. reported UV blocking properties of silk fabrics which were treated with nano TiO2 coating by atomic layer deposition (ALD) which is a low temperature vacuum deposition method. In a typical ALD process, at first titanium (IV) isopropoxide (TIP) has been purged in the reaction chamber and chemisorb onto the surface of silk fabrics. Afterward, nitrogen has been purged in order to remove the residual reagent. Then, water has been introduced into the chamber for the reaction of TIP with silk fabrics. Finally, nitrogen has been used as purged gas again. UPF of treated silk fabric was around 50 which is excellent, as compared with untreated fabric [91]. Khan et al. compared the UV protective effect of cotton fabric which has been treated with ZnO and TiO2 nanoparticles. Nanoparticles has been applied on textile materials by pad-dry-cure technique using binder. TiO2 containing formulation provide better UV protection than ZnO. With 30 g/L of TiO2 in formulation solution has been provided UPF of 147.7 and with 30 g/L of ZnO nanoparticle in the formulation has been provided UPF of 35.7. Whereas, untreated fabric showed an UPF of 5.7 which is un-ratable. Excellent UV blocking property of TiO2 nanoparticle treated fabric is due to the scattering effect along with reflection and absorption by nanoparticles [112].
Carbon nanotube for UV protective textiles
Multiwalled carbon nanotube (MWCNT) showed an excellent UV blocking property which could be incorporated into the textile matrix by coating with polymer solution. UPF of coated cotton fabric with only polyurethane (PU) solution was 46 (excellent, >40) as compared with untreated fabric for which UPF was 5.6 (un-ratable). UV transmission of coated fabrics with MWCNT was almost zero which has been indicated that the MWCNT containing polymer coated fabrics could be able to protect the wearer from both UVA and UVB rays. UV protection factor of treated fabric has been increased with increase of MWCNT concentration in PU solution. UPF rating of coated fabric with 1.00 wt% of MWCNT containing polymer was 174 and for the coated fabric containing 2.5 wt% of MWCNT was 421. A very high UPF rating for carbon nanotube containing PU coating was due to the excellent UV-blocking characteristics of MWCNT. Experimental result revealed that UV absorption in the range of 200–700 nm has been increased with the increase of MWCNT in the coating solution [51]. Nasouri reported influence of multi walled carbon nanotube and nano ZnO on UV blocking properties of cellulose acetate electrospun nanofiber web. Thickness of mat has highest influence on UPF followed by ZnO concentration and MWCNT has lowest influence on UV blocking property of mat. Highest UPF of 180 has been obtained with 4.9 wt% nano ZnO, 1.1 wt% MWCNT and mat thickness of 79.5 µm [99].
Other nanomaterials for UV protective textiles
Emam and Abdelhameed reported UV blocking property of cotton and silk fabrics which have been treated with nano MIL (Ti, In) metal organic framework (MOF). UV protection factor of treated fabrics were linearly proportional with MIL-MOF and metal content. Incorporation of greater than or equal to 10.4 g of MIL-MOF/Kg on textile surface was sufficient to achieve excellent UV blocking property. Around 40% MIL-MOFs has been washed up during the first five washing cycles, however, washed fabrics showed good UV blocking rate (UPF 26.7–36.2). MOFs were attached on the textiles surface by in situ synthesis process [113]. Tian et al. reported UV blocking property of cotton fabric treated with graphene oxide and chitosan by a layer-by-layer technique. They have observed remarkable increase of UPF which depends on number of cycles for treatment. With one cycle of treatment, UPF has been increased to 88.9 as compared to 9.4 for the untreated cotton fabric. With further increase of treatment cycle to 10 cycles, the UPF has been increased to 452 which is nearly 40-folds increment as compared with controlled sample [98].
Silva et al. reported UV blocking property of gold nano particle containing chitosan coating on soybean protein based knitted rib fabrics. Pure soybean knitted fabric presents an UPF of around 7 and gold nanoparticle containing fabric showed an UPF of around 14 due to the low concentration gold nanoparticle immobilization on soybean fabric. Chitosan treated fabric displayed UPF of around 62, while, gold nanoparticle containing chitosan coating showed an UPF around 289 (nearly 40 fold as compared with pristine fabric) with negligible transmission of UVA and UVB. Gold nanoparticle containing chitosan coated fabric retains excellent UV blocking property even after several washing cycles [114]. Zuo et al. reported UV blocking cotton fabrics which were treated with boron and nitrogen co-doped carbon dot (BN-CD). BN-CD has been attached on the textiles surface by impregnation and drying method with 2% poly (vinyl alcohol). UPF of BN-CD treated fabrics without PVA and with PVA were 28.6 and 38.6, respectively [115]. Shabbir and Mohammad reported UV blocking property of woolen fabrics which were treated with silver nanoparticles. Silver nanoparticles were synthesized by using three plant extract such as
Dhineshbabu and Bose reported extraordinary UV blocking cotton fabrics which were treated with polyurethane solution containing iron titanate (FeTiO3) nanoparticles. Nanoparticles were derived naturally from the ilmenite sand. X-ray diffraction peaks of pristine cotton fabrics and only polyurethane coated fabrics shows (Figure 2) similar results due to the amorphous nature of the polyurethane. Whereas, iron titanate (FT) containing polyurethane coating shows typical diffraction peaks at 27, 48.5, and 67.3° due to the attachment of FT nanoparticles on cotton fabric (Figure 2) [116]. UPF of pristine cotton fabric was 12, and blocked around 75% of UVA and UVB. While, polyurethane coated fabric displayed an UPF of 467, and blocked around 85% of UVA and around 94% of UVB. Moreover, iron titanate nanoparticle containing polyurethane coating showed an UPF of 1218 and allowed negligible transmission of both UVA and UVB through the fabric. Dramatic increase of UPF for FT nanoparticle containing fabric was due to the strong UV absorption capacity of nanoparticle [116].

X-ray diffraction pattern of uncoated cotton (UC) fabric, coated cotton fabric with polyurethane (PUC) and coated cotton fabric with iron titanate containing polyurethane (FTPC). Pristine fabric and PU coated fabrics showed similar diffraction peak for cotton due to the amorphous nature of polyurethane. While, FTPC containing PU coated fabrics showed typical additional diffraction peaks for iron titanate. (Reproduced with permission from Reference [116] © 2018 American Chemical Society, Link: https://pubs.acs.org/doi/10.1021/acsomega.8b00822.).
Vilchez-Maldonado et al. reported UV protective textiles treated with ethylcellulose nanoparticles which have been synthesized by emulsion techniques with two different concentrations
Durability of UV blocking properties for nanoparticle treated textiles
Durability of UV blocking properties of nanomaterial treated textiles is an important issue from the repeated applications point of view. Wang et al. reported wash fastness of nanostructured ZnO treated hollow cotton fiber textile. Nano ZnO particles were attached on the inner wall of the fiber which provides good wash fastness properties (washing performance was investigated as per Technical Manual of the AATCC at 49°C). UPF of treated textiles has been decreased for the first 5 washing cycles, afterwards, UPF remained almost constant up to 50 washing cycles. ZnO attached on the outer surface of fiber have been quickly washed out during the first 5 cycles of washing due to the friction force [82]. Durability of UV protective cotton fabric treated with boron and nitrogen co-doped carbon dot nanostructured materials has been improved with poly (vinyl alcohol). UPF of 28.6 has been reduced to 19.8 after 20 laundering cycles due to no covalent bond formation between BN-CD and cotton for the BN-CD treated fabrics applied by impregnation drying method. Whereas, UPF of 38.6 has been reduced to 30.2 after 20 laundering cycles, when, the nanostructured materials have been applied by using PVA, and PVA coating protects the BN-CD [115]. Chen et al reported durability of UV blocking properties for nano structured TiO2 treated silk fabrics. Treated textile has maintained excellent UPF value after 10 accelerated laundering cycles. UPF of TiO2 treated textiles was 51.8 (800 cycles of atomic layer deposition) before laundering and after 10 cycles of laundering it had a UPF value of 50.6 [95]. Durability of UPF value for the herbal nanoparticle extracted from Aloe vera, treated cotton fabric has been studied by Subramani et al. Nanoparticles were applied on the textiles surface by coating with chitosan. UPF of washed fabric has been slightly decreased (7–10%) for fifth and tenth washing cycles due to the removal of coating materials [96]. In an another study, Subramani et al. reported similar result for the UPF after washing, when the fabric has been treated with nanoparticle extracted from natural extract such as Azadirachta indica (neem) [97]. Ibrahim et al. reported marginal decrease of UPF for the nanoparticle treated cotton/PET blend fabric after 15 washes due to the removal of unfixed nanoparticles from the fabric surface [83]. Vı´lchez-Maldonado et al. reported durability of ethylcellulose nanoparticles treated UV protective cotton fabrics. Qualitative evaluation of functional nanoparticles attached with treated fabrics have been evaluated by scanning electron microscopic images. Amount of nanoparticle attached with fabrics has been decreased with increasing number of washes. The interaction between nanoparticles and treated fabrics were weak physical interactions such as hydrogen-bonding and van der Waal forces. Therefore, entanglement between polymer chain of ethylcellulose and treated fabrics has been played an important role for the adhesion of nanoparticles and treated fabrics. Morphological study of nanoparticles after washing process revealed that morphology of nanoparticles did not changed significantly as the washing process does not solubilized the nanoparticles [117]. Kathirvelu et al. reported excellent durability of UV blocking effect for the ZnO nanoparticles treated polyester/cotton blend knitted and woven fabrics even after 25 cycles of washing due to the excellent bonding between nanoparticles and textile surface even without binder [119].
Mechanism of nanoparticle based UV blocking property
When UV rays fall on textile surface, it has three parts such as transmission, reflection/scattering and absorption. In order to make the UV blocking effect on textile material, it should not allow transmission of UV rays, and this can be achieved by reflection/scattering and absorption of UV ray by textiles materials. UV blocking effect of various nanomaterials treated textiles can be of different mechanisms such as absorption, reflection and/or conversion of UV energy to other form of energy e.g. heat energy. In order to act as an effective protection against harmful part of UV radiation, UV blocking nanoparticles must transformed the absorbed energy into less harmful energy. UV blocking capability of nanomaterials depends on their photochemical mechanism of light absorption [120]. The main mechanism of nanomaterials as an UV blocker acts on selective absorption of harmful part of UV radiation and its dissipation as thermal energy [78]. UV blocking mechanism of textiles treated by nanomaterials consist of two components such as absorption and scattering. Lu et al reported preparation of ZnO nanoparticles by two steps: synthesis of ZnO precursor via homogeneous precipitation and calcination of precursor at different temperature. UV blocking property of ZnO nanoparticle has been increased with increasing calcination temperature possibly due to the conversion of more precursor to ZnO nanoparticle, and this is beneficial for more UV rays absorption [81]. Excellent UV shielding property of nanoparticles is due to the high refractive index and UV absorption capacity of this class of material [30]. The UV blocking mechanism of metal oxide nanoparticles and carbon nanotube can be attributed due to the excellent electronic properties of these nanoparticles [110,121]. Solar UV radiation can be absorbed by excitation of electrons from the valance band to the conduction band. For example, TiO2 and ZnO nanoparticle have bandgap energies corresponding to wavelengths of 365 nm, 380 nm, respectively. UV light below these wavelength has sufficient energy to excite electrons, subsequently be absorbed by nanoparticles [122]. Light with wavelength longer than band gap wavelength of nanomaterials will not be absorbed [123].
The proposed mechanism of UV blocking property of nano ZnO is due to its excellent absorption, refraction and/or scattering of UV rays. Efficiency of UV blocking property of nanomaterial treated fabric depends on particle size, shape and concentration of nanomaterials on the treated textiles [124]. Chen et al. reported UV blocking property of cotton fabric treated with various concentration of TiO2 nanoparticles. Robust UV protective fabrics have been developed by nano TiO2 containing polyvinylsilsesquioxane (PVS) coating on cotton fabrics. Nano TiO2 particles were attached with fabric by Ti – O – Si bond after curing through surface condensation crosslinking reaction among hydroxyl groups of cotton fabric with PVS polymer and TiO2 nanoparticles. UPF of treated and washed (washing condition: 300 rpm magnetic stirring for 30 min duration at 40°C with 5 g/L detergent) cotton fabrics has been significantly improved from about 39.4 with 0.5% (w/v) TiO2 nano particle containing solution to 121.5 with 5% (w/v) TiO2 nano particle containing solution. Significant increase of UPF with increasing TiO2 nano particle concentration was due to their scattering and absorption capacity of UV rays [125]. UV protection capability of nano structured TiO2 is due to the strong absorption and high refraction of nanostructed TiO2 to UV rays [33,91]. Nano structured TiO2 can absorb UV energy that matches or exeeds its band gap energy [95]. TiO2 nanomaterials can absorb the light completely with an energy higher than its bandgap energy (
Nahrawy et al. reported UV protective Kevlar which has been prepared by in situ sol-gel technique of copper lithium silicate (CLS) dopepd by zirconium (Zr) nanomaterial. CLS has formed different energy level, π the low energy level and π* the high energy level. When, CLS treated textile were exposed to UV radiation, the shared electron transfer between π–π*, and this offered increased chance for more electron to absorbed the wide range of UV rays. The chance of transfer has been incerased with increasing Zr dopping in the complex [89]. Rehan et al. reported UV protective effect of viscose fiber by in situ synthesis of Ag/Ag2CO3 or Ag/Ag3PO4 on viscose fiber. Excellent UV protective effect of nanomaterial was due to the large refractive index of Ag nanoparticles leading to very effective UV scattering [92]. Metal nanoparticles can reduced the transmission of UV ray through treated textiles [126,127]. UV transmission through silver nanoparticle treated textile has been decreased significantly as compared with untreated textiles (Figure 3) due to the reflection by silver nanoparticles [128]. Noorian et al. reported in situ synthesis of Cu2O/ZnO on textile surface and this can create synergistic effect of UV protection property of treated textiles [121].

UV transmission through untreated and silver nanoparticles treated textiles. Untreated cotton fabric do not prevent transmittance of UVA due to its porous nature, and untreated woolen textile allow significant transmittance of UVA through the textile. Whereas, silver nanoparticles treated both cotton and woolen textiles showed dramatic reduction of UVA transmittance due to the excellent absorption capacity of Ag nanoparticle. (Reproduced with permission from Reference [128] © 2015 Elsevier Ltd.).
Excellent UV blocking property of carbon based nanomaterial can be correlated with its some of the extraordinary properties [129]. For example, carbon nanotube (CNT) has very good thermal property. Typical thermal conductivity of carbon nanotube is around 6000 W·m−1·K−1 [130]. Ultra violet energies of UV-A and UV-B (280 – 400 nm) has significantly above the fundamental band gap of the electronic states of CNT. When, CNTs are exposed to light energy higher than its band gaps, the energy of photons will be absorbed by the electrons and it will generate the heat. Due to its excellent thermal conductivity heat will be readily dissipated from UV-irradiate CNTs [51]. UV blocking property of herbal extract from Aloe Vera is due to the presence of active photochemical component that effectively blocks the UV-B radiation. High surface area with a particle size of around 40 nm may also provide optical polarization effect [96]. Few chemical groups e.g. phenolic group in organic inorganic nanohybrid can dissipated the absorb UV energy, thus enhanced the UV blocking capability [81,123].
Synthesis of nanomaterials and incorporation of nanomaterials in textiles matrix
UV blocking nanomaterials can be incorporated into the textile matrix by several approaches

Schematic showing ZnO-PS hybrid nanocoating on cotton fabrics. (a) ZnO-PS nano-sphere containing carboxylic surface functional groups, (b) attachment of ZnO-PS hybrid nano sphere on cotton by esterification reaction of –OH groups of cotton and –COOH groups of ZnO-PS hybrid nano-sphere. (Reproduced with permission from Reference [81] © 2006 Elsevier Inc.)

Scanning electron microscopic image of ZnO nano powder (a); transmission electron microscopic image of polystyrene nanoparticle (b); and ZnO-PS nanohybrids at different magnifications (c–e). Microscopic images shows spherical shape of hybrid nanosphere with smooth surface morphology. (Reproduced with permission from Reference [81] © 2006 Elsevier Inc.).

Schematic showing functionalization of carbon nanotube with carboxylic functional groups, and attachment of –COOH functionalized CNT on cotton fiber by esterification reaction of –OH functional groups of cotton and –COOH functional groups of functionalized CNT.
Carbon nanotube (CNT) based UV blocking agent can also be incorporated into the textile matrix by coating with a polymer solution. However, uniform dispersion of CNT in the polymer solution is a significant challenge due to the inert surface property of CNT. Surface of CNT can be modified with several approaches in order to improve the dispersion of carbon nanotube in polymer solution, such as (i) aromatic molecules can be appended to nanotubes by using π-π interactions, (ii) polymer molecules can be wrapped on CNT surface by non-covalent interactions (including π-π interactions, van der Waals forces and charge-transfer interactions), (iii) covalent attachment of molecules to carbon nanotube surface [133]. Theoretical studies suggested that functionalization of CNTs do not greatly alters its strength [134]. Apart from the functionalization, ultrasonic energy can also be used to uniformly short time period dispersion of untreated CNTs in the polymer solution [135]. CNT dispersion in polyurethane (PU) solution with different concentration of nanomaterial in N,N’-dimethyl formamide solvent can be prepared by ultra-sonication stirring. Then, CNT dispersed PU can be coated on cotton fabric by knife over roller machine. Figure 7 shows the scanning electron microscopic images of uncoated and CNT dispersed PU coated fabrics with two different concentration of multi walled carbon nanotube [51].

Scanning electron microscopic images of: (a) uncoated cotton fabric; (b) coated fabric with 0.25 wt% of CNT dispersed PU and (c) coated fabric with 2.5 wt% of CNT dispersed PU. Uncoated fabric shows porous surface structure. While, inter fiber pores were filled up by polyurethane coating. (Reproduced with permission from Reference [51] © 2006 Wiley Periodicals, Inc.).
Nanomaterials used for the development of UV protective textiles can be synthesized by using various synthesis approaches such as sol-gel method, chemical varpor deposition, emulsion method etc. Ran et al. reported development of UV protective cotton fabrics by CuO/BiVO4 (BVO) nanocomposite photo catalyst immobilized by polydopamine (PDA) templating. CuO/BiVO4 photocatalyst has been synthesized by impregnation calcination method. In order to prepare the CuO/BVO on cotton surface, at first, cotton fabric has been coated with PDA film and then, cotton fabrics with PDA templated were placed into the CuO/BVO suspension with oscillation for 24 h, washed with deionized water to remove the unreacted material and dried at 60°C. The whole process is schematically shown in Figure 8 [90]. UV blocking treatment on cotton fabrics can be carried out by thin film of sol-gel processed titania. Sol-gel process formed transparent film by nanostructured metal oxide on various textile substrate. In this process a colloidal oxide network as sol state has been formed, which subsequently formed gels upon drying [109]. Strong adhesion of titania nanoparticle on cotton fabrics is due to the dehydration reaction between –OH group of cotton and –OH group of titania. Due to the strong adhesion of nanoparticles on textile which sustained the UV blocking property of treated fabrics even after 20 home laundering [136]. Nahrawy et al. reported synthesis of copper lithium silicate (CLS)/zirconium nanoparticle by using sol-gel technique for the in-situ coating on Kevlar. Inorganic sol gel process has been formed nanogel with three dimensional network structure on Kevlar surface. ZrO2 nanoparticles has been used for the doping of complex CLS network structure [89].

Schematic showing preparation of CuO/BVO treated cotton fabrics. Nanocomposite film has been formed on the cotton fabric by polymer pre-treated fabric with subsequent CuO/BVO nanoparticle treatment. (Reproduced with permission from Reference [90] © 2019 Elsevier B. V.).
Other methods for the incorporation nanomaterials on the textile matrix are in situ polymerization, melt compounding and spinning etc. By using polycondensation polymerization method, polyethylene terephthalate (PET)/nano TiO2 nanocomposite can be prepared. In order to introduce functional groups on nano TiO2 surface, TiO2 could be treated with a coupling agent. Afterward, functionalized nanoparticle can be dispersed in the monomer of PET, e.g. ethylene glycol (EG). Finally, PET/TiO2 nanocomposite can be obtained by polycondensation reaction of TiO2 dispersed EG and terephthalic acid (TPA) [80]. Erdem et al. reported incorporation of nano TiO2 in the polypropylene (PP) filament. Nano TiO2 and PP have been mixed by melt compounding, afterwards, melt spinning has been carried out by single screw extruder with a spinneret having 40 orifices. Subsequently spun filaments has been drawn with 2.5 draw ratio at 115°C in order to prepare the nanocomposite fiber [88].
Conclusions
The potential risks of solar UV radiation to the human health is relevant due to the high concentration of UV radiation reaching on the earth surface due to the adverse weather changes. Unique features of nanomaterials provided a great potential to develop UV protective textiles. Interaction between ultra violet rays and nanomaterials treated textile surfaces has been summarized in this review paper. This review paper presented wide ranges of nanomaterials as UV blocking agents to fabricate UV protective textiles. Mechanism of UV blocking effect for various nanomaterials are described. Various methods for the incorporation of UV blocking nanomaterials in the textile matrix are discussed. Taking into account the subject matter discussed in this review article, it is evident that emerging material like nanomaterials as UV blocking agent for textiles is a research field with good future prospects. However, several challenges of nanomaterials for further development of UV protective textiles can be summarized as follows: (a) We are working on a multidimensional problem on UV protective textiles, and that researchers must work on the interaction between the parameters and the impact factors of each parameter. (b) Novel nanomaterials especially environmental friendly nanomaterials with improve UV protective properties. (c) Nanomaterials incorporated UV protective textiles with other functionalities in order to form multifunctional textiles. (d) Novel application techniques to incorporate nanomaterial in the textile matrix in order to develop UV protective textiles at the same time to un-alter other functional properties of textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
