Abstract
In recent years, several studies have been published on the synthesis of carbon nanomaterials—such as carbon nanospheres, carbon nanoframes, carbon nanorods, carbon nanoblocks, and carbon octadecahedrons—in concentrated salt systems. These novel carbon nanomaterials reported in the literature have attracted widespread interest. The fabrication approach described in these studies leverages the interaction between surfactants and concentrated salts to produce carbon nanomaterials. Unlike traditional synthetic routes, the assembly route of carbon nanomaterials in concentrated salt systems is characterized by the controllable construction of diverse carbon nanostructures. As an extension of conventional carbon nanomaterial manufacturing methods, this strategy holds significant development potential in the field of carbon nanomaterial production. Based on existing literature reports on concentrated salt systems, this review summarizes the theory and practice of carbon nanomaterial formation in such systems, along with the formation’s underlying mechanisms, advantages, and future development trends.
1. Introduction
The discovery of C60 in 1985 [1] broadened the understanding of carbon allotropes, thereby opening up new possibilities for elemental carbon. Six years later, Japanese physicist Sumio Iijima became the first to successfully synthesize carbon nanotubes via the arc discharge method [2], which once again demonstrated the rich morphological diversity of carbon allotropes. Since then, approaches for synthesizing carbon nanomaterials have proliferated. In 1997, Young and colleagues achieved the batch synthesis of single‐walled carbon nanotubes using laser evaporation [3]. In the 21st century, chemical vapor deposition (CVD) began to be widely studied and applied to the synthesis of carbon nanotubes [4]. To this day, the CVD method retains its unique advantages in the preparation of carbon nanomaterials, and accordingly, carbon nanotubes produced via CVD exhibit outstanding properties [5, 6]. Following this, the template method emerged as a primary technique for fabricating carbon nanomaterials [5], securing a pivotal role in nanotechnology research. In the ongoing exploration of carbon nanomaterial synthesis, novel methods such as electrospinning and hydrothermal synthesis [7, 8] have been gradually developed, becoming integral to the field. Among these, catalytic cleavage [9] has risen to prominence due to its low‐cost and high‐efficiency advantages.
While the aforementioned methods have significantly advanced the field of carbon nanomaterials, their limitations cannot be overlooked. For instance, the arc discharge method not only exhibits high energy consumption but the elevated temperatures also induce numerous defects in the resulting carbon nanomaterials. Additionally, its high production cost renders it unsuitable for large‐scale industrialization. Similarly, the laser evaporation method suffers from costly equipment, low efficiency, and a tendency toward adhesion. Although the CVD method offers more pronounced advantages, it still incurs substantial gas consumption.
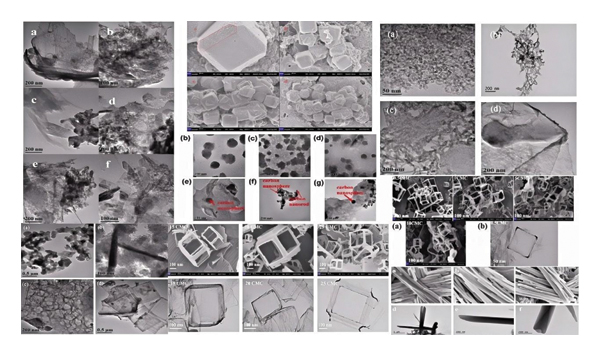
The literature has reported the synthesis of carbon nanomaterials in concentrated salt systems, which systems leverage the interaction between surfactants and concentrated salts and employ in‐situ carbonization at elevated temperatures to fabricate carbon nanomaterials [10–17]. When preparing carbon nanomaterials using this approach, the carbonization temperature is maintained below the melting point of the inorganic salt, and the carbonization duration typically lasts 2 hours. As shown in Figure 1, the morphology of carbon nanomaterials prepared using a concentrated salt system. Notably, the concentrated salt can be recovered and reused after the synthesis process. The advent of this method offers a novel strategy for carbon nanomaterial synthesis, sparking extensive interest in the scientific community.
2. Practice Under the New System
Typically, studies on the aggregation states and self‐assembly of surfactants are conducted under aqueous solution conditions [18–22]; some researchers have also carried out relevant investigations in dilute salt solutions and even seawater systems (a natural complex dilute salt system). The results indicate that the presence of dilute salts reduces the critical micelle concentration (CMC) of surfactants [23–28]. Further, researchers have explored the properties of surfactants in seawater systems to clarify the influence of complex salt components [29–32]. However, basic research on surfactant‐related properties in concentrated salt systems remains scarce. Against this backdrop, the synthesis of carbon nanomaterials in concentrated salt systems represents a bold exploration of the interactions between concentrated salts and surfactants.
Based on existing literature reports, there are two approaches to leveraging the interactions between surfactants and concentrated salts in such systems: one involves directly adding inorganic salt solids to a surfactant solution of specific concentration [17]; the other utilizes evaporative crystallization and shear flow to induce such interactions [10–16]. By adopting these approaches and using different types of surfactants as carbon sources with various concentrated salt systems, researchers have synthesized a range of carbon nanomaterials with novel morphologies. Specific parameters (e.g., surfactant type, salt concentration, and product morphology) are provided in Table 1.
Carbon nanomaterials prepared by the interaction of different concentrated salts and carbon sources.
In the reported literature, inorganic salts used in concentrated salt systems are characterized by high solubility and stability at elevated temperatures. Cationic surfactants, anionic surfactants, nonionic surfactants, and even polymers (e.g., sodium hyaluronate) can all serve as carbon sources for carbonization in concentrated salt systems. Moreover, the same type of surfactant can form carbon nanomaterials with distinct morphologies in different concentrated salt systems—a phenomenon that highlights the regulatory role of salt systems in product morphology.
This offers multiple carbon source options for the future synthesis of carbon nanomaterials, enabling the formation of a wide range of carbon nanomaterial morphologies by leveraging the relevant properties of salts and the diversity of carbon sources. Consequently, this method holds substantial promise for the synthesis of carbon nanomaterials.
Existing literature suggests that current research in concentrated salt systems primarily focuses on modulating the morphology and size of carbon nanomaterials. Unlike carbon nanomaterial synthesis via the CVD method [33], synthesis in concentrated salt systems offers simpler and more convenient operation, along with lower energy consumption. Furthermore, the concentrated salt can be recovered and reused afterward, preventing secondary environmental pollution. In addition, the synthesis of novel carbon nanomaterials in concentrated salt systems indirectly confirms the crystal structures formed by inorganic salts during the crystallization process, offering new insights and methodologies for investigating inorganic salt crystal formations. In Figure 2, the formation mechanism of carbon nanostructures is illustrated.

Explanation of carbon nanoformation mechanism [14].
Although a variety of carbon nanomaterials with distinct morphologies have been synthesized in concentrated salt systems, there remains a lack of intentional design in the early stages of synthesis. As‐prepared carbon nanomaterials have not been studied in terms of their applications in the fields of energy storage and conversion.
3. Discussion
The aggregation and self‐assembly of carbon sources in concentrated salt systems are key research areas that require in‐depth investigation. Specifically, the aggregation of single organic molecules and the self‐assembly of multiple organic molecules in such systems need further exploration to enhance the controllability of interactions between initial organic molecules and concentrated salt systems. Notably, investigating the crystallization behavior and interactions of concentrated salts in organic molecular solutions is crucial for better regulating the aggregation and self‐assembly states of organic molecules in these systems; this, in turn, enables more precise control over the morphology of carbon nanomaterials. The exploration of novel self‐assembly methods is a critical direction for advancing concentrated salt system research. According to existing reports, self‐assembly methods based on surfactants (as carbon sources) and concentrated salt systems remain relatively limited. To mitigate phase separation between surfactant solutions and inorganic salts, most current approaches rely on shear flow‐induced self‐assembly with concentrated salts. Given these limitations, future development of concentrated salt systems should also focus on advancing self‐assembly methods—for instance, the freeze‐drying method could be explored, as it may enable gentler, more uniform self‐assembly between surfactants and concentrated salts. Interactions between different types of concentrated salts and carbon sources are a core research focus for advancing concentrated salt systems. In existing reports on such systems, the employed concentrated salts are characterized by high solubility and thermal stability, with chloride ions as their sole anions. These salts interact favorably with surfactants, enabling the formation of carbon nanostructures with diverse morphologies under high‐temperature conditions. To expand the scope of concentrated salt systems, future research should further explore interactions involving multivalent anions/cations, as well as those between slightly soluble, thermally stable inorganic salts and carbon sources. Such investigations will facilitate more precise regulation of the morphology and size of carbon nanomaterials. Interactions between concentrated salts and carbon nanostructures are currently limited to two roles in carbon nanomaterial synthesis: “coating agents” and “partitioning agents.” For the future development of concentrated salt systems, exploration can be directed toward new types of concentrated salts that act not only as “coating agents” and “partitioning agents” but also as “activators.” Ideally, the resulting carbon nanostructures would possess both nanoscale dimensions and the characteristic properties of activated carbon—a combination that would expand the application scope of such materials. Exploring other applications of concentrated salt systems is a promising direction for expanding their utility. The utility of these systems can be extended to the synthesis of other inorganic nanomaterials by leveraging their “partitioning agent” properties. For instance, by utilizing the interaction between soluble concentrated salts (e.g., sodium chloride) and insoluble salts (e.g., nanoscale magnesium carbonate), nanoscale magnesium oxide can be prepared: first, we calcine insoluble nanoscale magnesium carbonate in a sodium chloride system at high temperature and then dissolve the sodium chloride. Similarly, inorganic materials like nanoscale magnesium hydroxide can also be synthesized in soluble concentrated salt systems. In these synthesis processes, soluble concentrated salts act as “dispersing agents” to effectively inhibit the agglomeration of nanoscale magnesium oxide and magnesium hydroxide.
The successful synthesis of carbon nanospheres, carbon nanorods, carbon nanoframes, and carbon octahedra in concentrated salt systems offers a novel approach for carbon nanomaterial preparation. This method holds significant implications for both the development of new carbon nanomaterials and salt lake recycling research. Moving forward, strengthening research on the front‐end design of synthesis and subsequent applications will enhance the prospects for carbon nanomaterial development in concentrated salt systems—thereby providing a potential and feasible strategy for the future industrial production of carbon nanomaterials and other inorganic nanomaterials.
Data Availability Statement
Research data are not shared.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
No funding was received for this research.
